Abstract
This study investigated the effect of bark extract of Bathysa cuspidata on hepatic oxidative damage and blood glucose kinetics in rats exposed to paraquat. Wistar rats were exposed to a single dose of paraquat (30 mg/kg, i.p.) and treated with an ethanolic extract of Bathysa cuspidata (200 and 400 mg/kg). Analyses were conducted of liver edema, blood glucose, serum transaminases, alkaline phosphatase, collagen, malondialdehyde, catalase, superoxide dismutase, and histomorphometry. In the animals intoxicated with paraquat and treated with 400 mg/kg of extract, edema, hypertrophy of the nucleus of hepatocytes, serum transaminases, alkaline phosphatase, and malondialdehyde levels decreased significantly (p < .05). Catalase and superoxide dismutase activity, the number of hepatocytes per unit volume, and the glycogen proportion were maintained. The increase followed by progressive reduction of blood glucose observed in paraquat-exposed groups was significantly attenuated in the group treated with 400 mg/kg of extract (p < .05). Phytochemical analysis of the extract revealed the presence of flavonoids, proanthocyanidins, and phenolic compounds. The bark extract of Bathysa cuspidata was able to inhibit large variations in blood glucose and reduce hepatic damage in rats exposed to paraquat. This finding suggests a contribution of the extract in reducing lipid peroxidation and the morphofunctional damage to the liver parenchyma.
Introduction
Liver disorders are commonly reported in cases of poisoning by agricultural chemicals, including paraquat (PQ) (Giri et al. 1979; Igarashi et al. 1999; Igarashi et al. 2001; Konstantinova and Russanov 1999). Paraquat (1,1′-dimethyl-4,4′-bipyridinium dichloride) is a quaternary nitrogen herbicide widely used around the world (Chang et al. 2009). Its high toxicity to humans and animals when absorbed through ingestion, skin contact, or inhalation has been extensively demonstrated (Chang et al. 2009; Dasta 1978; Suntres 2002). The toxic effect of PQ is related to its ability to induce oxidative stress, leading to the generation of reactive oxygen species (ROS) through redox cycling by microsomal NADPH-cytochrome P-450 reductase (Bismuth et al. 1990). The increased ROS production induces oxidation of lipids and proteins from the cell membrane, leading to morphological and functional changes in several organs and eventually to death (Fukushima et al. 2002; Nwabisi and Nwanze 1997; Sittipunt 2005; Suntres 2002). As a large percentage of liver disorders occur as a result of exposure to chemical compounds capable of inducing oxidative stress, models of PQ-induced liver injury have been useful in investigating plants with hepatoprotective potential (Elbling et al. 2005; Igarashi et al. 2001; Tieppo et al. 2006).
Bathysa cuspidata (A. St. Hil.) Hook f. belongs to the Rubiaceae family. The infusion of its stem bark is used in the traditional medicine of South America for the prevention and treatment of stomach and liver disorders (Leite 2008). Studies with the ethanolic extract and the dichloromethane fraction of B. cuspidata have shown antinociceptive and anti-inflammatory effects in albino mice that were previously enterally treated. In addition, in vitro toxicity tests revealed no mutagenic effects from the ethanolic extracts of the leaves and bark of this species (experiment conducted by our research group, unpublished data). Use of the B. cuspidata extract to treat liver disorders is believed to be associated with a potential antioxidant effect, an issue that is still poorly understood and requires further investigation.
Considering that liver lesions resulting from PQ exposure occur through the induction of oxidative stress and the activation of an inflammatory response, it is not unrealistic to assume that compounds with antioxidant and anti-inflammatory action may potentially have a beneficial effect in reducing acute liver injury and mortality in PQ poisoning. To clarify its relevance, efficacy, and possible mechanism of action, the objective of this study was to investigate the effect of bark extract of Bathysa cuspidata on hepatic oxidative damage and blood glucose kinetics in rats exposed to PQ.
Materials and Methods
Chemicals
Paraquat (composition: 1,1′-dimethyl-4,4′-bipyridinium dichloride ion [200 g/L] and inert ingredients [876 g/L]) was purchased from the manufacturer Syngenta (Basel, Switzerland). Thiobarbituric acid, hydrogen peroxide, sodium phosphate, formaldehyde, glutaraldehyde, 4',6-diamidino-2-phenylindole (DAPI), Coomassie blue, carmine, and hematoxylin and eosin were purchased from Sigma Chemical Co. (St. Louis, MO, USA). Aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), and γ-glutamyl transferase (GGT) biochemical kits were purchased from Human in Vitro Diagnostics (Minas Gerais, Brazil).
Plant Extraction and Phytochemical Analysis
Samples of B. cuspidata were collected in a biome of Brazilian Atlantic forest in the state of Minas Gerais, Brazil (20°43’00,0”S and 42°29’10,8”W, 1,200 meters above sea level) and adequately documented in the herbarium of the Federal University of Viçosa under registration VIC 21559. Stem bark samples were separated; dried for forty-eight hours at room temperature in a darkened, well-ventilated room; pulverized in a knife mill; and stored. The powdered stem bark of B. cuspidata (500 g) was exhaustively extracted by percolation with ethanol 95%. The extract was concentrated under vacuum at 50°C using a rotary evaporator and then lyophilized until complete removal of the solvent was achieved, yielding an ethanolic extract (139 g). Phytochemical analysis of the extract was performed on chromatography plates coated with silica gel GF 254 (Merck, Darmstadt, Germany) using different mobile phases and detection reagents in accordance with the protocol described by Wagner and Bladts (2009). The preparation of B. cuspidata was analyzed for the presence of phytochemicals by qualitative chemical analysis previously described (Nunes 2008; Wagner and Bladts 2009).
Animals and Their Care
Male Wistar rats weighing 364 ± 37 g were obtained from the main animal laboratory of the Federal University of Viçosa (Brazil) and kept under controlled temperature conditions (21 ± 2°C) with relative air humidity of 60% to 70% and fourteen hours of light daily (5 a.m. –7 p.m.). The animals had free access to rat chow and water. All experiments were conducted in accordance with the Brazilian College of Animal Experimentation and approved by the Animal Research Ethics Committee of the Federal University of Viçosa, Brazil (approval protocol 064/2010).
Paraquat Exposure and Treatment Groups
Paraquat was dissolved in 0.5 mL of 0.9% NaCl saline solution (10 mg/mL) and injected intraperitoneally (i.p.) in a single dose of 30 mg/kg of body weight (BW). The ethanolic extract of the stem bark of B. cuspidata (BCE) was resuspended in 700 µL (w/v) of vehicle (VE) dimethylsulfoxide (DMSO) and administered to the animals by gavage. Treatment with this extract at the doses of 200 mg/kg (BCE 200) per day and 400 mg/kg BW (BCE 400) per day began two days before the application of PQ and continued to be administered until two days after application of the herbicide. The same protocol used for the groups receiving PQ was followed in the control groups, which received BCE 400, DMSO (VE), or saline 0.9% (700 µL) alone. Sixty rats were randomized into 6 treatment groups with ten animals in each group, as follows: (1) SAL (control), saline solution (NaCl 0.9%); (2) VE (vehicle control), DMSO; (3) BCE 400 (extract control), BCE 400 mg/kg; (4) PQ/VE, PQ + DMSO; (5) PQ/BCE 200: PQ + BCE 200 mg/kg; and (6) PQ/BCE 400: PQ + BCE 400 mg/kg.
Euthanasia and Biometric Analysis
Twenty-four hours following the end of treatment, the animals were anesthetized with ketamine (10 mg/kg BW, i.p.) and xylazine (2 mg/kg BW, i.p.). Next, a laparotomy was done and the liver was removed intact and weighed using analytical scales. Two sections of the liver (median lobe) of each animal were quickly removed, one of which was frozen in liquid nitrogen (−196oC), and the other immersed in Karnovsky’s fixative solution.
To investigate differences in BW in the animals in the different groups, the rats were weighed at the beginning and at the end of the experiment. Liver edema was assessed by the ratio between organ weight (OW) and BW and expressed as a liver somatic index (LSI), where LSI = OW × 100/BW. After removal of the fragments for biochemical and histological analysis, the water content per unit weight of the tissue (mL/g) was measured in the remaining liver tissue. This parameter was determined after drying the liver at 60°C for ninety-six hours using the relation (wet weight – dry weight) × 103/ wet weight. Because the specific gravity (σ) of water is 1.0, water volume is obtained by volume = weight/σ, or, simply, volume (103 mm3) = weight (g) (Weibel 1979).
Biochemical Analysis
Blood glucose kinetics was analyzed in all groups every six hours during the first twenty-four hours after PQ administration and at the moment of euthanasia (ninety-six hours) using a glucose oxidase method (OneTouch Ultra, Johnson & Johnson, Milpitas, CA, USA). The blood used in this analysis was collected by tail puncture.
Aliquots of blood were collected and centrifuged at 3,000 rpm for fifteen minutes, and the serum was immediately used in biochemical measurements of AST, ALT, ALP and GGT. Serum samples were processed in accordance with the instructions supplied by the manufacturers of the diagnostic kits (Human in Vitro Diagnostics).
For analysis of tissue malondialdehyde (MDA), an end product of lipid peroxidation, an aliquot of frozen liver (100 mg) was homogenized in phosphate-buffered saline, the homogenate was reacted with thiobarbituric acid, and the formation of thiobarbituric acid–reactive substance was monitored at 535 nm, as described previously (Gutteridge and Halliwel 1990). Catalase (CAT) activity was evaluated according to the method described by Aebi (1984) by measuring the rate of decomposition of H2O2, and superoxide dismutase (SOD) activity was estimated by a xanthine oxidase method based on the production of H2O2 and the reduction of nitroblue tetrazolium (Sarban et al. 2005). Total protein levels in liver tissue were measured using the Bradford method (Bradford 1976).
For all animals in both groups, the collagen content in the liver tissue was determined using a spectophotometric method previously described (López-De León and Rojkind 1985).
Stereology and Kariometry
Liver fragments of all animals from each group were placed in Karnovsky’s solution for twenty-four hours, then dehydrated in ethanol and embedded in paraffin. Sections 4 µm thick were cut with a Multicut 2045 rotary microtome (Reichert-Jung, Germany), stained with the best carmine method for glycogen and DAPI to DNA labeling (Masotti et al. 1982) and mounted using Entellan mounting medium (Merck, Frankfurt, Germany). To avoid repeat analysis of cells, sections were evaluated in semi-series, using 1 in every 5 sections. The slides were visualized and the images captured using a light microscope (Olympus BX-60, Tokyo, Japan) connected to a digital camera (Olympus QColor-3, Tokyo, Japan). Sections stained with DAPI were analyzed using the epifluorescence mode on the same microscope. Sixty fields from each carmine method (20× objective) and DAPI (40× objective) were randomly chosen, and a total of 4.37 × 106 µm2 and 1.41 × 106 µm2 liver areas, respectively, were analyzed.
For the stereological analysis, a test system of 300 points was used in a standard test area (At) of 73 × 103 µm2. In sections stained using the carmine method, volume density (Vv) was estimated by counting the points situated over the hepatocytes [cell] and glycogen-containing cytoplasmic inclusions [glyc] using the following formula: Vv= PP [structure] / PT where PP is the number of points situated over the structure in question and PT is the total number of points in the test. In sections stained with DAPI, hepatocyte volume density per unit volume (Nv[h]) was measured. Nv[h] (cells/µm3) was calculated using the ratio Nv[h] = Qn / At × t, where Qn is the number of nuclear profiles situated on the “lookup plane” in the test area (At) and t is the thickness of the sections (Mandarim-de-Lacerda 2003). Nuclei with a distance of less than the calculated geometric mean axis of the nuclei (9 µm) between them were considered to belong to the same cell and were counted only once. The volume of 50 hepatocyte nuclei stained with DAPI for each animal was determined according to Sala et al. (1994). All histomorphometric analyses were performed using the Image Pro-Plus 4.5 image analysis software (Media Cybernetics, Silver Spring, MD, USA).
Statistical Analysis
Results are expressed as measures of central trend, means, and standard deviations (mean ± SD). The data were submitted to unifactorial one-way analysis of variance followed by the Tukey post hoc test for multiple comparisons. Statistical significance was established at p < .05. All tests were performed using the GraphPad Prism 5.01 statistical software program (Released August 7, 2007, GraphPad Software, Inc, San Diego, CA, USA).
Results
Phytochemical Analysis of B. cuspidata
The phytochemical analysis of the stem bark of the B. cuspidata ethanolic extract showed positive results for alkaloids, coumarins, flavonoids, tannins, and triterpenes. The quantitative analysis indicated the content of total phenolic compounds (58.7 mg/g), proanthocyanidins (39.7 mg/g), and total flavonoids (3.4 mg/g).
Effect of B. cuspidata Extract on Biometric Parameters
As shown in Table 1 , there was no significant difference in the initial and final BW between the groups. The edema indexes (LSI and liver water content) were significantly higher in the PQ/VE and PQ/BCE 200 groups relative to all other groups (p < .05). There were no statistically significant differences in edema indexes between the SAL, VE, BCE 400, and PQ/BCE 400 groups. (Table 2).
Effects of treatment with BCE on biometric parameters of Wistar rats exposed to PQ.
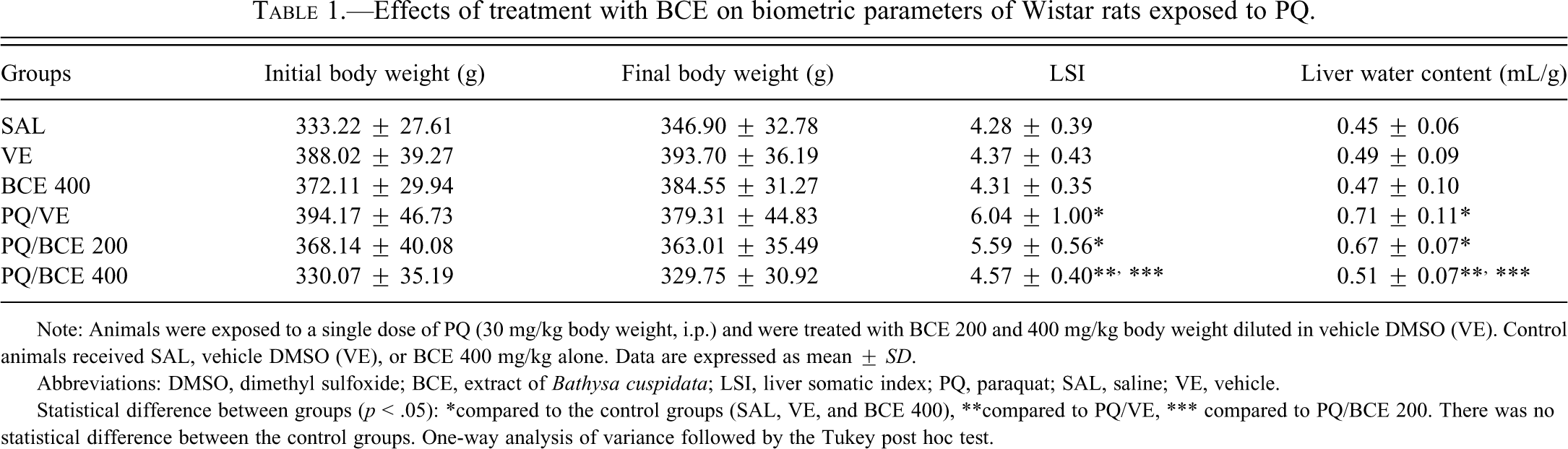
Note: Animals were exposed to a single dose of PQ (30 mg/kg body weight, i.p.) and were treated with BCE 200 and 400 mg/kg body weight diluted in vehicle DMSO (VE). Control animals received SAL, vehicle DMSO (VE), or BCE 400 mg/kg alone. Data are expressed as mean ± SD.
Abbreviations: DMSO, dimethyl sulfoxide; BCE, extract of Bathysa cuspidata; LSI, liver somatic index; PQ, paraquat; SAL, saline; VE, vehicle.
Statistical difference between groups (p < .05): *compared to the control groups (SAL, VE, and BCE 400), **compared to PQ/VE, *** compared to PQ/BCE 200. There was no statistical difference between the control groups. One-way analysis of variance followed by the Tukey post hoc test.
Effects of treatment with BCE on liver function tests and collagen content in liver tissue of Wistar rats exposed to PQ.
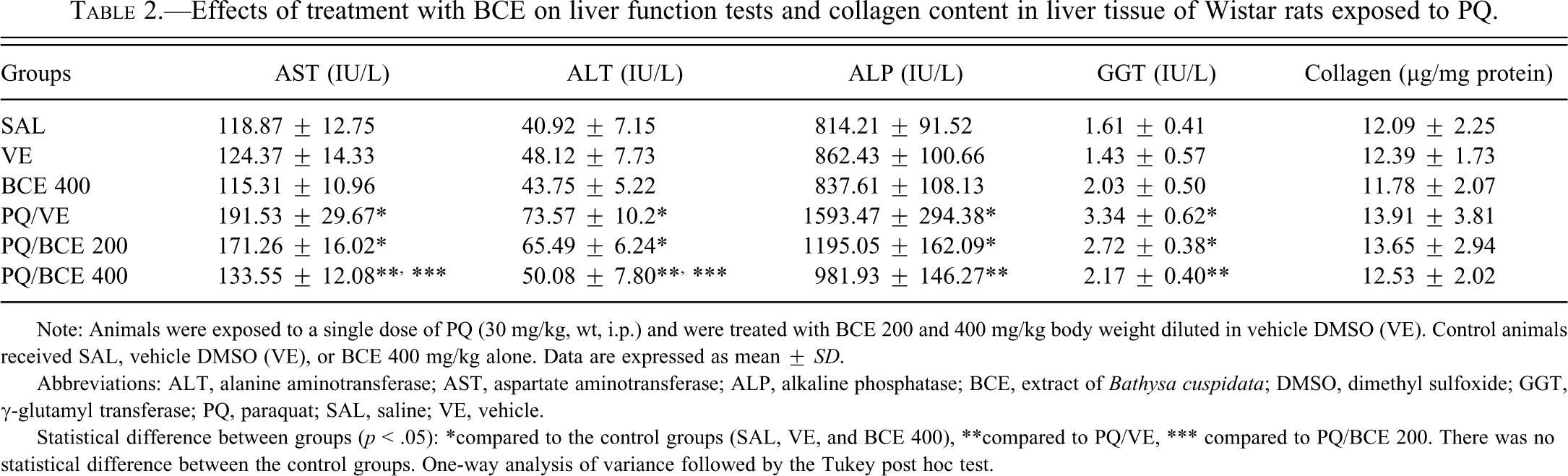
Note: Animals were exposed to a single dose of PQ (30 mg/kg, wt, i.p.) and were treated with BCE 200 and 400 mg/kg body weight diluted in vehicle DMSO (VE). Control animals received SAL, vehicle DMSO (VE), or BCE 400 mg/kg alone. Data are expressed as mean ± SD.
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; BCE, extract of Bathysa cuspidata; DMSO, dimethyl sulfoxide; GGT, γ-glutamyl transferase; PQ, paraquat; SAL, saline; VE, vehicle.
Statistical difference between groups (p < .05): *compared to the control groups (SAL, VE, and BCE 400), **compared to PQ/VE, *** compared to PQ/BCE 200. There was no statistical difference between the control groups. One-way analysis of variance followed by the Tukey post hoc test.
Effect of B. cuspidata Extract on Biochemical Parameters in Serum and Liver Tissue
Serum AST , ALT, ALP and GGT levels were significantly higher in the PQ/VE and PQ/BCE 200 groups compared to the other groups (p < .05). There was no significant difference in ALP and GGT levels in the comparison between PQ/BCE 200 with the PQ/VE and PQ/BCE 400groups. In the latter group, treatment with 400 mg/kg of B. cuspidata extract significantly reduced the PQ-induced increase in serum AST, ALT, ALP, and GGT levels (p < .05). There were no significant differences in collagen content in the liver tissue among all groups.
According to the analysis of oxidative stress, levels of the lipid peroxidation marker malondialdehyde (MDA) were significantly higher in the liver tissue from animals in the PQ/VE and PQ/BCE 200 groups compared to the other groups (p < .05; Figure 1 ).
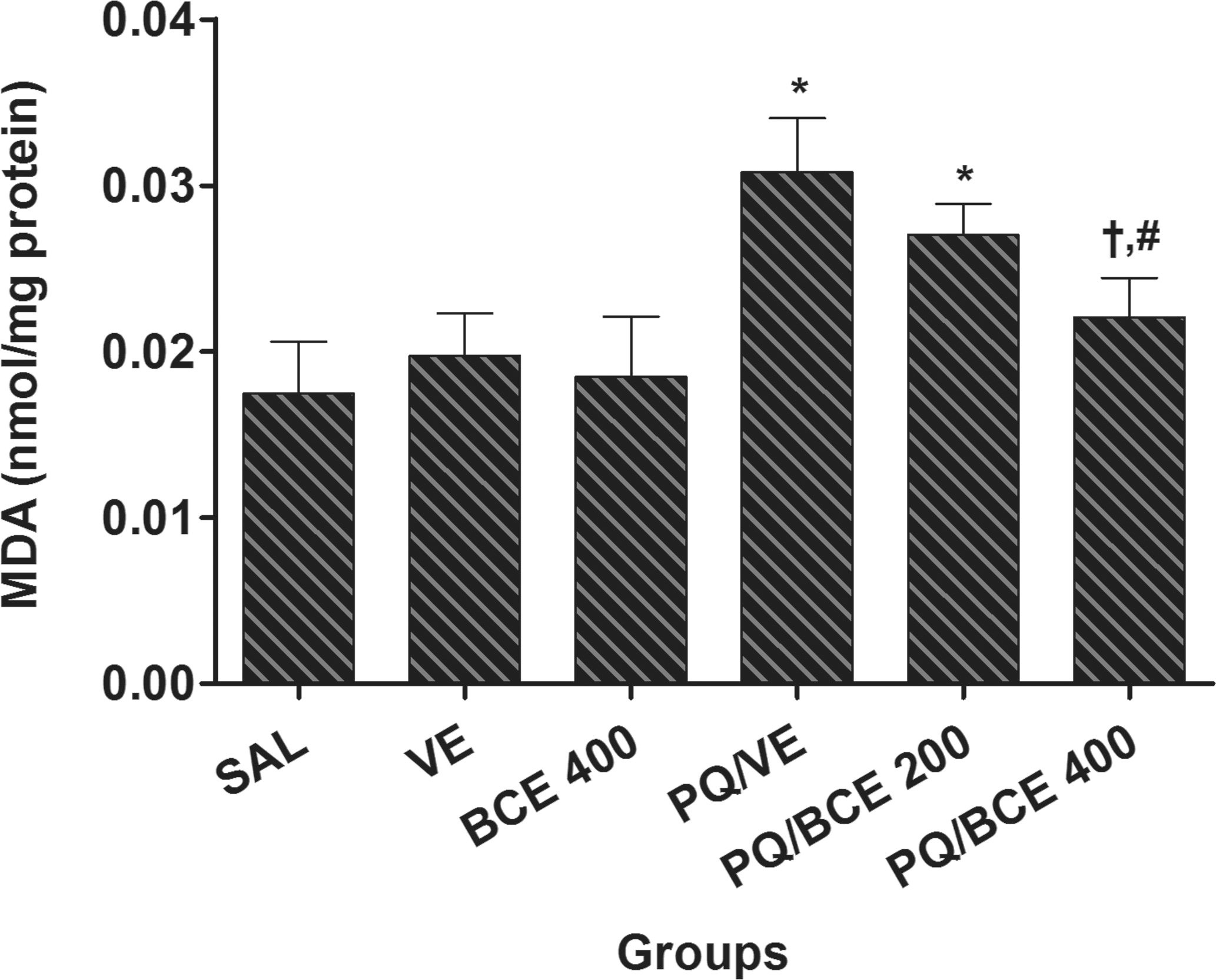
Effect of treatment with bark extract of Bathysa cuspidata (BCE 200 and 400 mg/kg, wt.) on malondialdehyde (MDA) levels in liver tissue of Wistar rats exposed to a single dose of paraquat (PQ, 30 mg/kg, wt, i.p). SAL, saline; VE, vehicle (DMSO). Data are expressed as means ± S.D. *,†,# Statistical difference between groups (p < 0.05); *compared to the control groups (SAL, VE and BCE 400), †compared to PQ/VE, # compared to PQ/BCE 200; there was no statistical difference between the control groups, one way ANOVA followed by Tukey's post hoc test.
Catalase activity in liver tissue was significantly lower in the PQ/VE group compared to the other groups (p < .05) with the exception of the PQ/BCE 200 group (Figure 2A). Significant reductions in the SOD activity were observed in the PQ/VE and PQ/BCE 200 groups compared to the other groups (p < .05; Figure 2B). Malondialdehyde, CAT, and SOD showed no significant differences between the SAL, VE, BCE 400, and PQ/BCE 400 groups.
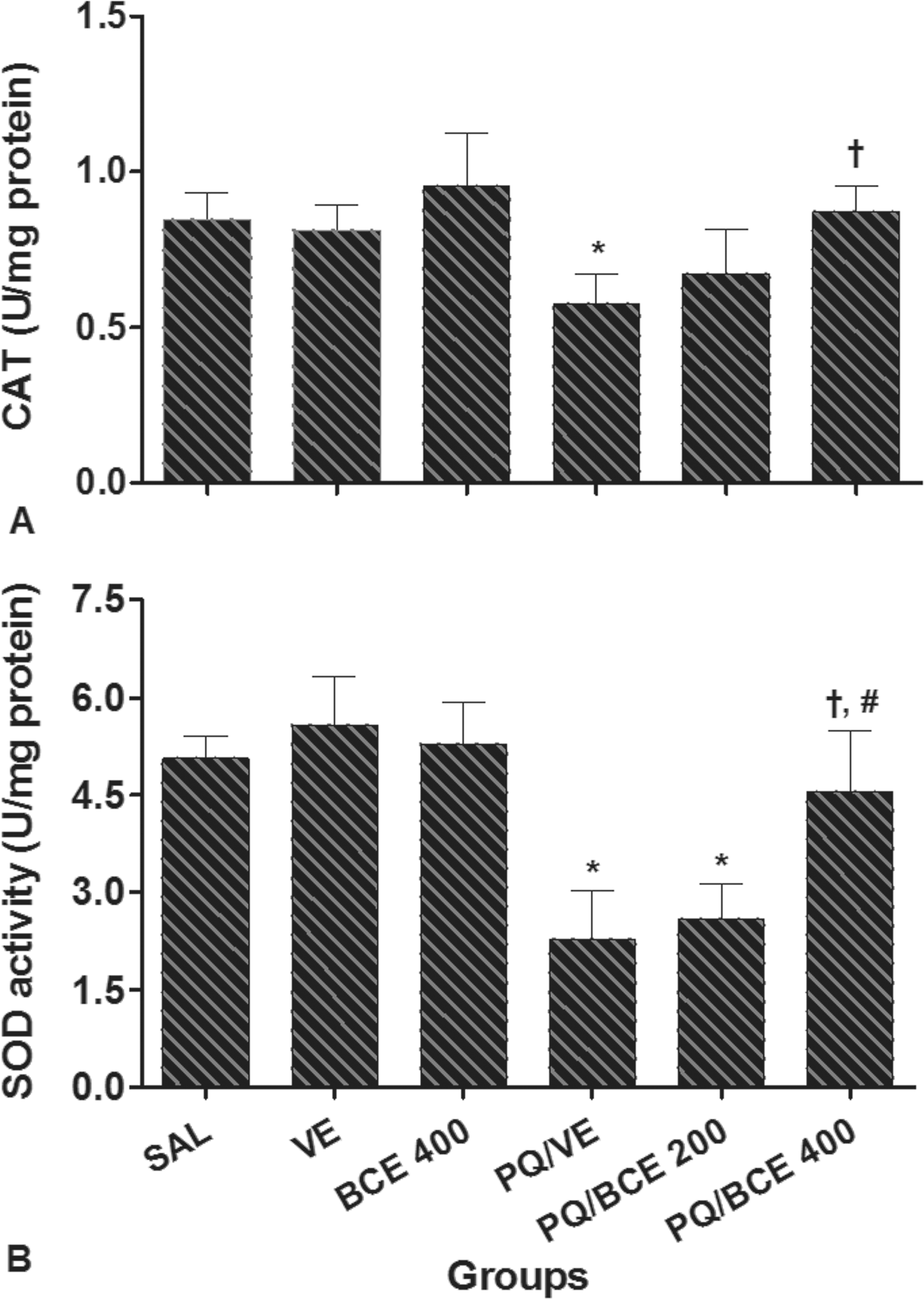
Effect of treatment with bark extract of Bathysa cuspidata (BCE 200 and 400 mg/kg, wt.) on catalase (CAT) and superoxide dismutase (SOD) activity in liver tissue of Wistar rats exposed to a single dose of paraquat (PQ, 30 mg/kg, wt, i.p). SAL, saline; VE, vehicle (DMSO). Data are expressed as means ± S.D. *,†, # Statistical difference between groups (p < 0.05); *compared to the control groups (SAL, VE and BCE 400), †compared to PQ/VE, # compared to PQ/BCE 200; there was no statistical difference between the control groups, one way ANOVA followed by Tukey's post hoc test.
Histomorphometric Changes of Liver Tissue
Histomorphometric analysis of the liver tissue showed an increase in the distance between the cell nuclei and a significant reduction in hepatocyte volume density (NV[h]) per unit volume of tissue in the PQ/VE and PQ/BCE 200 groups compared to the other groups. Inversely, the proportion of the histological area occupied by hepatocytes (Vv[cell]) and the nuclear volume (V Nucleus) of the hepatocyte were significantly higher in the PQ/VE and PQ/BCE 200 groups compared to the other groups (Figure 3 and Table 3 ).
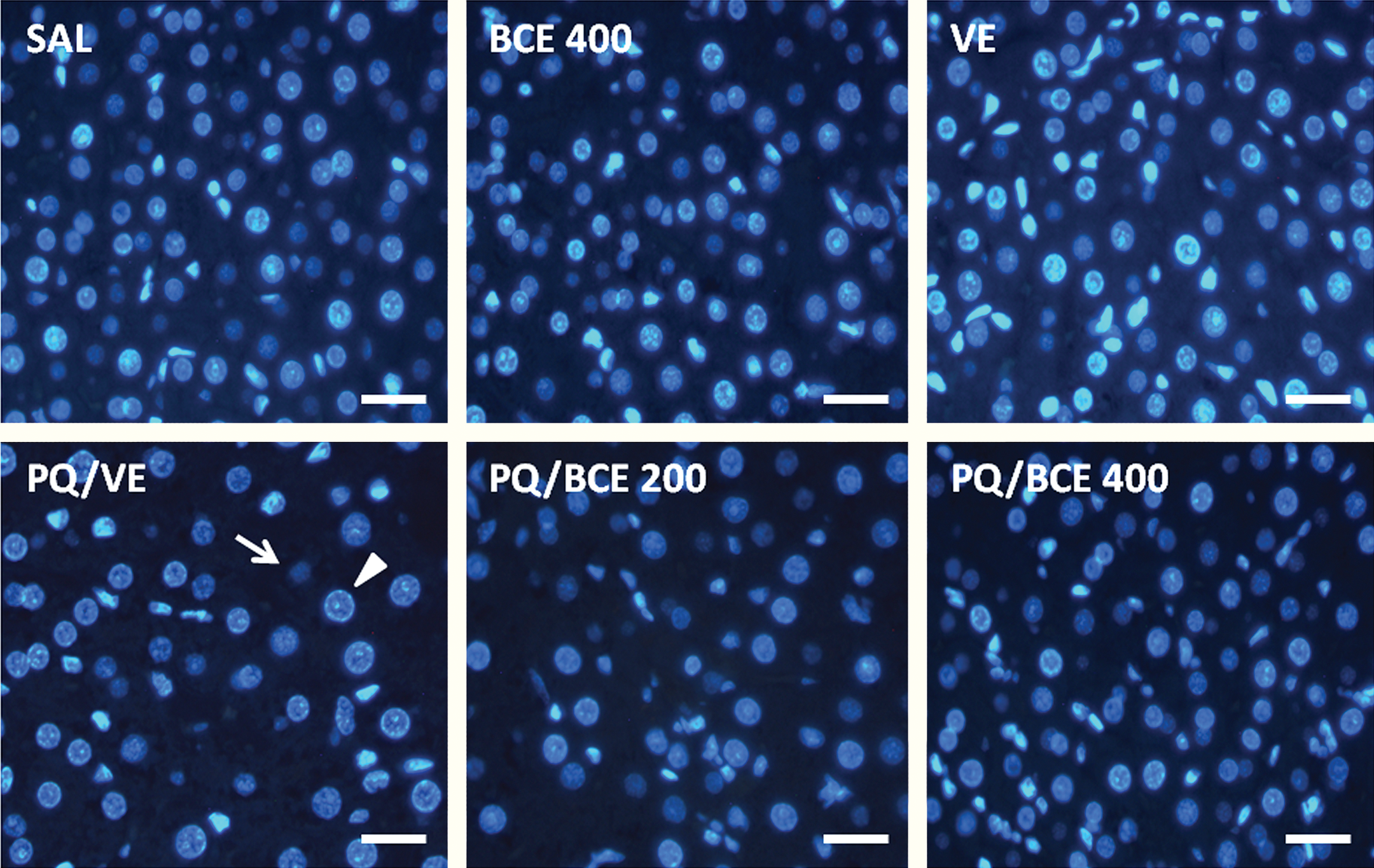
Representative photomicrographs of histological sections of the liver of Wistar rats stained with 4′,6-diamidino-2-phenylindole (DAPI) to DNA and observed under the epifluorescence microscope (bar = 35µm). Hepatocyte nuclear profiles in “look-up and look-down” planes are indicated by arrowhead and arrow in PQ/VE panel, respectively.
Effects of treatment with BCE on liver morphometric parameters of Wistar rats exposed to PQ.
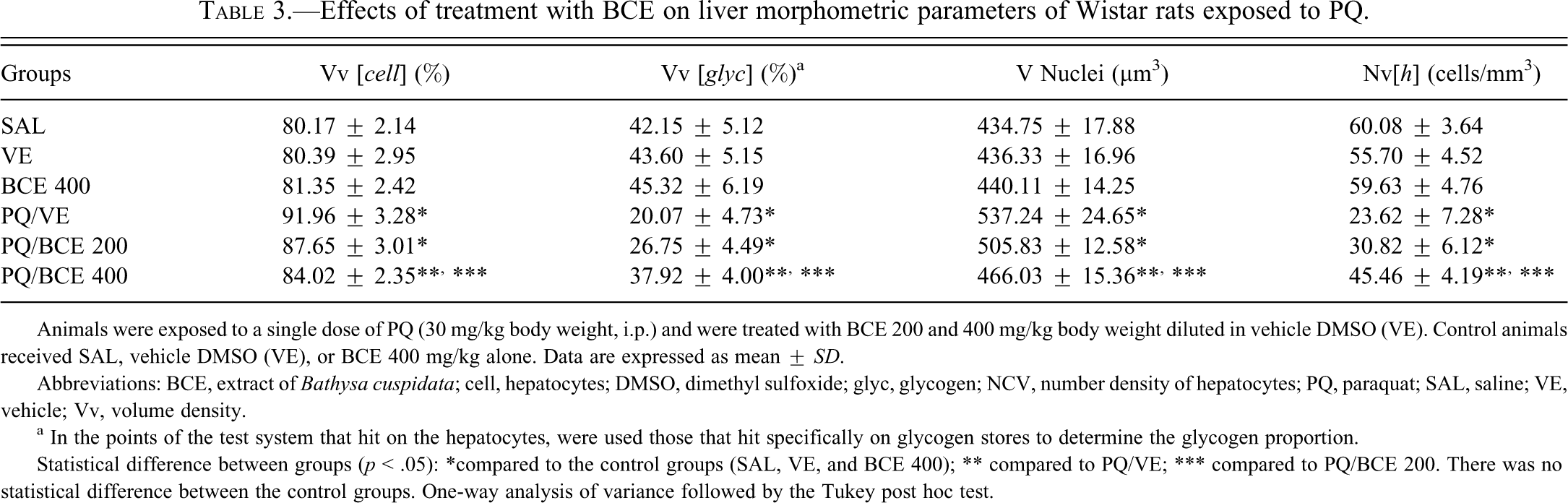
Animals were exposed to a single dose of PQ (30 mg/kg body weight, i.p.) and were treated with BCE 200 and 400 mg/kg body weight diluted in vehicle DMSO (VE). Control animals received SAL, vehicle DMSO (VE), or BCE 400 mg/kg alone. Data are expressed as mean ± SD.
Abbreviations: BCE, extract of Bathysa cuspidata; cell, hepatocytes; DMSO, dimethyl sulfoxide; glyc, glycogen; NCV, number density of hepatocytes; PQ, paraquat; SAL, saline; VE, vehicle; Vv, volume density.
a In the points of the test system that hit on the hepatocytes, were used those that hit specifically on glycogen stores to determine the glycogen proportion.
Statistical difference between groups (p < .05): *compared to the control groups (SAL, VE, and BCE 400); ** compared to PQ/VE; *** compared to PQ/BCE 200. There was no statistical difference between the control groups. One-way analysis of variance followed by the Tukey post hoc test.
Exposure to PQ resulted in significantly increased levels of blood glucose six hours after herbicide administration (Figure 4 ). Treatment with 400 mg/kg of extract inhibited this effect, and the animals receiving this dose showed no significant difference in blood glucose levels in relation to the SAL, VE, and BCE 400 groups. Significant reduction of blood glucose levels was evidenced twenty-four and ninety-six hours post-PQ treatment. At these times, the reduction of blood glucose was significantly attenuated in the PQ/BCE400 group. The PQ/VE and PQ/BCE 200 groups presented a significant reduction in the glycogen-containing cytoplasmic inclusions (Vv[glyc]) relative to the other groups (Figure 5 and Table 3). There were no statistically significant differences in any of the histomorphometric parameters between the SAL, VE, BCE 400, and PQ/BCE 400 groups.
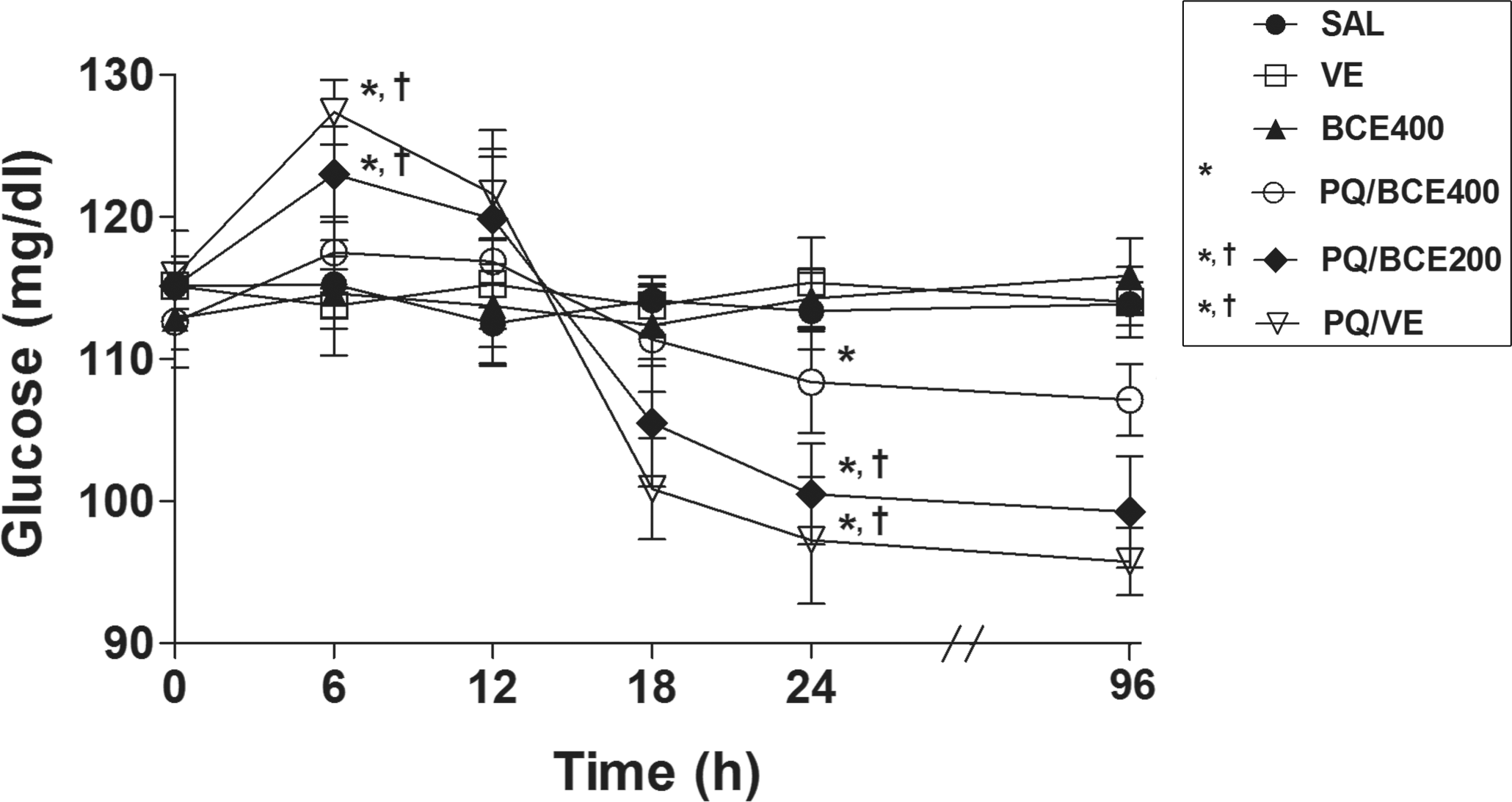
Effect of treatment with bark extract of Bathysa cuspidata (BCE 200 and 400 mg/kg, wt.) on blood glucose levels in Wistar rats exposed to a single dose of paraquat (PQ, 30 mg/kg, wt, i.p). SAL, saline; VE, vehicle (DMSO). Data are expressed as means ± S.D. *,†Statistical difference between groups (p < 0.05); *compared to the control groups (SAL, VE and BCE 400), †compared to PQ/BCE400; there was no statistical difference between the control groups, one way ANOVA followed by Tukey's post hoc test.
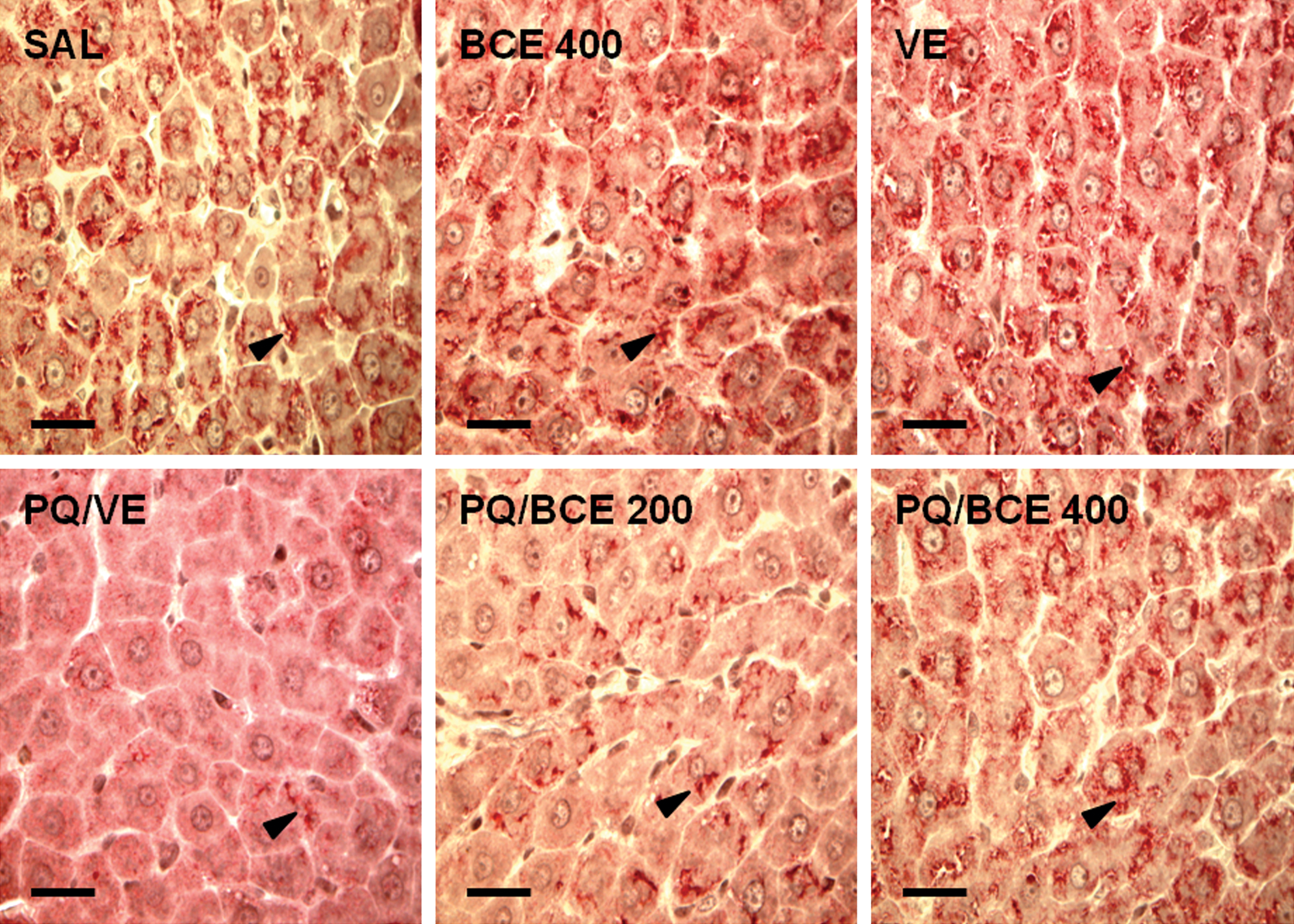
Representative photomicrographs of histological sections of the liver of Wistar rats stained with the carmine method to glycogen and observed under the light microscope (bar = 70µm). Glycogen cytoplasmic inclusions are indicated by the arrowhead.
Discussion
The present study investigated the effect of bark extract of B. cuspidata on PQ-induced acute liver injury in Wistar rats. The experimental model of PQ was previously validated as a model of oxidative liver injury (Igarashi et al. 1999; Igarashi et al. 2001; Konstantinova and Russanov 1999; Tieppo et al. 2006), and the dose administered (30 mg/kg of body weight, i.p.) may be classified as moderate exposure to PQ (60% of the LD50) in rats (LD50 = 50 mg/kg) and high exposure in humans considering the human LD50 of 35 mg/kg (Nwabisi and Nwanze 1997; Sittipunt 2005).
The redox cycling compound PQ is a bipyridyl herbicide that uses molecular oxygen to initiate production of superoxide anions (
From the biochemical point of view, in acute PQ poisoning, oxidative damage to the vascular components of the liver occurs concomitantly with changes in hepatocyte cell membrane permeability. Traditionally, measurement of serum liver-specific enzyme levels has served as a good indicator of liver damage (Ranawat et al. 2010; Sreelatha et al. 2009). In the present study, elevated serum levels of AST, ALT, GGT, and ALP were found in the PQ/VE group, indicating a reduction and/or loss of functional integrity of the hepatocyte membranes. Although there was a significant reduction in the serum levels of these enzymes in the group treated with the higher dose of the extract (PQ/BCE 400), the same effect was not found in the PQ/BCE 200 group, suggesting a possible dose-dependent effect of the extract.
Down-regulation of the oxidative and inflammatory processes represents an important mechanism for reducing PQ toxicity, which has been investigated in an attempt to find effective approaches to reduce the organ damage related to this herbicide (Dasta 1978; Suntres 2002). The results found in the present study show that PQ poisoning results in a significant increase in MDA concentration and a reduction in CAT and SOD activity in liver tissue, which was inhibited by administration of the B. cuspidata extract in the PQ/BCE 400 group, thus giving strength to the hypothesis that the extract has a dose-dependent effect. These data on MDA levels and on CAT and SOD activity should not be analyzed separately, since they serve as indicators of tissue oxidant/antioxidant balance (Dasta 1978; Fukushima et al. 2002). There is sufficient evidence showing that the metabolites generated through PQ metabolism oxidize the unsaturated fatty acids in the plasma membrane of the hepatocytes and cell organelles, leading to the production of lipid peroxyl radicals (LOO−), alkoxy radicals (RO−), and MDA, which propagate oxidative damage in a cyclic process of lipid peroxidation (Bismuth et al. 1990; Dasta 1978; Konstantinova and Russanov 1999). On the other hand, CAT and SOD act by neutralizing pro-oxidant compounds, thereby reducing liver damage in PQ poisoning (Dasta 1978; Fukushima et al. 2002; Suntres 2002). Interestingly, these enzymes, especially CAT, have been shown to be highly susceptible to PQ-induced oxidative events, a characteristic that is potentially useful for the evaluation and monitoring of PQ poisoning. In this context, vegetable extracts that stimulate or maintain the activity of components of the antioxidant defense system may potentially be beneficial in minimizing liver damage mediated by the oxidative mechanism triggered by PQ. Therefore, it is believed that there may be an association between the results found in the present study and certain components of B. cuspidata extract such as flavonoids, steroids, coumarins, tannins, and triterpenes, compounds with a confirmed antioxidant capacity (Ranawat et al. 2010; Tieppo et al. 2006).
Among the chemical substances identified, quantitative phytochemical analysis showed the predominance of phenolic compounds in the B. cuspidata extract. Previously, two similar studies demonstrated an antioxidant activity of polyphenols from Solanum melongena (Kimura et al. 1999) and Brassica oleraceae (Igarashi et al. 2000) in an experimental model of hepatotoxicity induced by PQ. In these studies, dietary supplementation with 0.15% of polyphenols was sufficient to maintain CAT activity and reduce MDA levels in liver tissue. In addition, polyphenols from S. melongena were able to prevent food intake reduction and weight loss. Similar effects were evidenced using a dietary supplementation with 0.80% of an aqueous extract of Carthamus tinctorius, which presents a high content of phenolic compounds able to suppress oxidative stress caused by PQ in liver tissue (Igarashi et al. 2001). In another study, a 5% aqueous extract of Croton cajucara was effective to inhibit oxidative stress induced by PQ in vitro and in vivo (Tieppo et al. 2006). In this study, the extract increased more than 50% the survival rate of Saccharomyces cerevesiae in culture after PQ treatment. Furthermore, C. cajucara extract decreased levels of MDA and hidroperoxides and increased activities of glutathione peroxidase and CAT in PQ-treated rats, thereby suggesting a marked radical scavenging activity of the extract that the authors attributed to the extract’s phenolic compounds and steroids. Altogether, these studies suggest that polyphenols could be the main phytochemicals related to the down-regulation of oxidative stress in PQ poisoning. Although the molecular mechanism modulated by these compounds remains poorly understood, the up-regulation of CAT or glutathione peroxidase gene expression by different flavonoids has been previously reported (Tieppo et al. 2006). Thus, although other factors cannot be ruled out, a similar effect by components of the B. cuspidata extract could partially explain the present results.
There is evidence of a close correlation between markers of oxidative damage and morphological changes in the liver parenchyma (Dasta 1978; Konstantinova and Russanov 1999; Sreelatha et al. 2009). In addition to the histomorphometric results of the present study, the data on MDA corroborate this finding. Significant pathological changes were found in all the morphological parameters analyzed in the PQ/VE and PQ/BCE 200 groups. On the other hand, the morphological organization of the liver tissue in the animals in the PQ/BCE 400 group was normal, indicating that the 400 mg dose of the extract was sufficient to inhibit the occurrence of histological damage to the liver parenchyma. Events that merit particular attention in the PQ/VE and PQ/BCE 200 groups include the increase in hepatocyte nuclear volume and the reduction in the number of cells per unit volume of liver tissue. These findings indicate that, in addition to water accumulation in the liver tissue, the increase in hepatocyte volume contributed to the increase in the edema index of this organ.
Marked elevation of blood glucose levels was observed within six hours of PQ intoxication, followed by gradual reduction of glucose levels in the subsequent hours. Furthermore, depletion of liver glycogen storage was observed in intoxicated animals. The dose of 400 mg of the extract was sufficient to reduce the variation of blood glucose levels and prevent the PQ-induced depletion of liver glycogen. Although few studies exist in this area, the effect of PQ on glucose metabolism has been described in mammals, including rats (Giri et al. 1979; Giri et al. 1983; Rose et al. 1974). This effect has yet to be fully explained; however, high levels of circulating catecholamines and corticosteroids have been identified in the initial phases of PQ poisoning (twenty-four hours) and are often associated with marked glycogenolysis, gluconeogenesis, and depletion of liver glycogen reserves (Giri et al. 1979; Konstantinova and Russanov 1999; Suntres 2002). It is believed that the high levels of catecholamines and corticosteroids in PQ poisoning are a result of oxidative damage to the cortex and medulla of the adrenal gland (Giri et al. 1979; Rose et al. 1974). Thus, this mechanism would justify the use of antioxidant compounds in the prevention of glucose metabolism disorders related to these hormones in PQ poisoning.
The present study showed that the bark extract of B. cuspidata exerted a hepatoprotective effect against PQ-induced oxidative stress in rats. The results indicated a possible dose-dependent reduction of morphological and functional damage to the liver parenchyma by B. cuspidata extract, which may be associated principally with its inhibitory effects on lipid peroxidation and glucose metabolism dysfunction and maintenance of catalase and superoxide dismutase activity. Further pharmacological evaluations are essential to identify the active components of this extract and fully clarify its mechanism of action, which may be associated with a high potential for the prevention and treatment of acute liver injury.
Footnotes
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article. The authors received no financial support for the research and/or authorship of this article.
