Abstract
The innate immune response is known to modify hepatocellular injury induced by toxicants. To assess the role of IL-10, a component of the innate immune response, in toxicant-induced injury of biliary epithelium, wild-type (WT) and IL-10 knockout mice (KO) were given a single toxic dose (50 mg/kg) of α-napthylisothiocyanate (ANIT) and assessed at twenty-four–hour intervals for four days following treatment. Clinical signs of toxicity were greater in WT mice. Unexpectedly, over the course of the study, there was a consistent tendency for ANIT-treated IL-10 KO mice to have less hepatocellular injury than WT mice. However, changes in the biliary epithelium differed in that there was more histologic evidence of inflammation and necrosis on days 2 and 3, respectively, in ANIT-treated IL-10 KO mice compared with WT mice. Proliferation of biliary epithelium and hepatocytes was greater and/or occurred earlier in the ANIT-treated IL-10 KO mice compared with the ANIT-treated WT mice, suggesting a greater reparative response was needed for recovery after toxicant injury in the IL-10 KO mice. Overall, our data suggest that IL-10 KO mice have less hepatocellular injury than WT mice following a toxic dose of ANIT and that biliary epithelial injury is accentuated in the KO mice.
Keywords
Introduction
Mechanistic investigations of acute, toxicant-induced hepatocellular injury have focused on metabolic activation leading to reactive moieties that form covalent bonds with cellular proteins and other macromolecules, a process that leads, directly or indirectly, to hepatocyte injury and activation of pathways and then to apoptosis or necrosis (Zimmerman 1999). In addition, the interplay between proinflammatory and anti-inflammatory components of the innate immune system can modulate the outcome of drug-induced liver injury (Gunawan and Kaplowitz 2007). The innate immune system provides the host’s immediate nonspecific responses to cell injury or infection, in contrast to the adaptive immune system, which requires specific antigen exposure and is slower to respond than the innate immune system (Bourdi et al. 2002; Bourdi et al. 2007; Cover et al. 2006; Ju et al. 2002; Michael et al. 1999). Components of the complement system and cytokines are the main soluble factors of the innate immune system. The main cellular components include macrophages, neutrophils and other granulocytes, and natural killer cells. Activation of the innate immune system leads to the release of a wide variety of cytokines that orchestrate the host response to injury. Using acetaminophen as a model toxicant, studies have demonstrated that acute hepatocellular injury can be modified by the innate immune responses, which determine the progression and severity of hepatotoxicity (Bourdi et al. 2002; Bourdi et al. 2007; Cover et al. 2006; Ju et al. 2002; Liu and Kaplowitz 2006; Michael et al. 1999). Despite the new findings regarding hepatocytes, little is known about the role of the innate immune system in toxicant-induced injury of the biliary epithelium.
The role of IL-10 in chemically induced hepatic injury has been of particular interest because of its anti-inflammatory and immuosuppressive actions. Increased hepatic IL-10 synthesis is part of a normal response to toxic doses of acetaminophen (Bourdi et al. 2002; Dambach et al. 2006), whereas acute hepatocellular necrosis is significantly increased in acetaminophen-treated, IL-10 knockout (KO) mice (Bourdi et al. 2002). Similarly, cycloheximide hepatic toxicity is increased when IL-10 is neutralized (Kumagai et al. 2009). IL-10 is produced by macrophages (including Kupffer cells), dendritic cells, B-lymphocytes, hepatocytes, and subsets of T-lymphocytes (Louis et al. 2003; Moore et al. 2001; Stordeur and Goldman 1998). Its role is, primarily, to inhibit inflammation via direct effects on monocytes/macrophages that influence T-lymphocyte activation (Couper et al. 2008). Specifically, IL-10 inhibits major histocompatibility (MHC) class II and co-stimulatory molecule B7-1/B7-2 (CD80/CD86) expression on monocytes/macrophages and limits production of proinflammatory cytokines including IL-1, IL-8, IL-12, IFN-γ, and TNF-α (Moore et al. 2001; Rajasingh et al. 2006) and chemokines, as well as nitric oxide (NO) and reactive oxygen intermediates (Stordeur and Goldman 1998; Moore et al. 2001). In addition, IL-10 has been shown to reduce neutrophilic infiltration following concanavalin A administration in murine liver (Louis, Le Moine, Peny, Quertinmont et al. 1997). IL-10 has a hepatoprotective role in various chemically mediated, as well as macrophage- and T-lymphocyte-mediated, rodent models of acute hepatic injury (Louis et al. 2003; Louis, Le Moine, Peny, Gulbis et al. 1997; Louis, Le Moine, Peny, Quertinmont et al. 1997). In view of these broad effects of IL-10 on hepatocellular injury, we evaluated the effects of IL-10 deficiency on chemically induced biliary injury.
In this study, we compared the responses of IL-10 knockout (KO) and wild-type (WT) mice with a classic biliary toxin, α-napthylisothiocyanate (ANIT). ANIT a model hepatobiliary toxicant, causes intrahepatic biliary injury in mice that is morphologically similar to drug-induced cholangitic cholestasis in humans (Blazka et al. 1996; Schaffner et al. 1973; Ungar and Popp 1976). In rats and mice, histologic evidence of hepatocellular and biliary epithelial injury is typically observed over a period of hours, with overt necrosis in both cell types seen by twenty-four hours following oral administration of ANIT (Glaser et al. 2006; Goldfarb et al. 1962; Kodali et al. 2006; Ungar and Popp 1976). Inflammation of the gall bladder in mice occurs approximately twenty-four hours later (Ungar and Popp 1976). Periportal to multifocal hepatocellular necrosis, caused by direct toxicity of ANIT (Jean et al. 1998; Orsler et al. 1999), neutrophils (Dahm et al. 1991; Hill and Roth 1998), or possibly associated with cholestasis and bile salt retention within hepatocytes, is also produced (Lock et al. 1982; Orsler et al. 1999). Following a single dose of ANIT, necrotic cell debris is usually cleared by seventy-two hours, and bile flow is returned to normal by ninety-six hours (Kodali et al. 2006; Kossor et al. 1993).
Materials and Methods
Animals
Forty male C57BL/6J mice (WT) and forty male IL-10 KO (B6.129P2-
Groups of male WT (Groups 1 and 2) and KO (Groups 3 and 4) mice (20/group) were randomized using Provantis NT Client Server Oracle (CSOR), and the body weight was balanced across groups. Mice were given a single dose of ANIT (batch no. 08420DD, Sigma Aldrich Chemical, St. Louis, MO) at doses of 0 (vehicle; Groups 1 and 3) or 50 mg/kg (Groups 2 and 4) by oral gavage. α-Napthylisothiocyanate was formulated as a solution in corn oil and administered to mice at a dose volume of 10 mL/kg on day 1. Mice from all treatment groups were necropsied in groups of five on days 2, 3, 4, and 5.
The following were evaluated: clinical observations, body weight, food consumption, clinical chemistry, liver (with gallbladder) weight, macroscopic and microscopic observations, and immunohistochemistry results for liver (dual staining for Ki-67 as a marker of proliferation and cytokeratin [pan-cytokeratin and cytokeratin 19] as a marker for the presence of biliary epithelial cells).
Clinical Chemistry
At necropsy, whole blood was collected from the caudal vena cava of treated and control mice under isoflurane anaesthesia. Serum was separated and analyzed on an Olympus AU640e chemistry analyzer (Olympus America, Melville, NY).
Tissue Collection
Following blood collection under isoflurane anaesthesia, samples of the left lateral and the median lobe of liver including the gall bladder were collected. Samples were immersed in 10% neutral buffered formalin for approximately twenty-four hours and transferred to 70% ethanol. Following fixation, liver sections were processed routinely into paraffin. Six-micron sections were stained with hematoxylin and eosin for microscopic review.
Histology
Livers and gall bladders were evaluated using a subjective scale to score hepatic and biliary injury. Biliary and hepatic lesions were scored as follows: 0, no evidence of abnormality; 1, minimal; 2, mild; 3, moderate; and 4, marked manifestation of the lesion. Biliary necrosis ranged from scattered individual necrotic epithelial cells (scored as 1) to complete absence of viable biliary epithelial cells in interlobular bile ducts (scored as 4). Biliary neutrophilic infiltration varied from one to three neutrophils within or immediately adjacent to the biliary epithelium of an individual bile duct (scored as 1) to more than twenty-five neutrophils within the lumen or epithelium of the bile ducts (scored as 4). Fibroblast proliferation referred to the circumferential proliferation of spindle-shaped cells (possibly myofibroblasts) around bile ducts, and scoring ranged from the presence of one or two cell layers of reactive fibroblasts (scored as 1) to more than six layers of cells (scored as 4). Hepatic necrosis ranged from scattered individual necrotic cells (scored as 1) to confluent areas of hepatocellular necrosis that bridged between lobules (scored as 4).
Immunohistochemistry
Sections of formalin-fixed and paraffin-embedded liver were dual labeled with rabbit polyclonal antibody specific for pan cytokeratin (Abcam, Inc., Cambridge, MA) and rabbit monoclonal antibody specific for Ki-67 (Clone SP6, Lab Vision Corp., Fremont, CA) to identify proliferating biliary epithelial cells and hepatocytes. Ki-67 and pan cytokeratin-labeled slides were pretreated with ER2 (EDTA). Ki-67 slides were labeled with Biocare Rabbit on Rodent HRP (Biocare Medical, Concord, CA), and developed with Leica Bond Max Refine DAB chromagen (Leica Microsystems, Inc., Bannockburn, IL). Pan cytokeratin slides were labeled with Biocare Rabbit on Rodent AP (Biocare Medical), and developed with Leica Bond Max AP Red chromagen (Leica Microsystems, Inc.). Sections were counterstained with hematoxylin to label the nuclei.
A quantitative assessment of immunohistochemical labeling in the liver was performed using the iCyte Laser Scanning Cytometer (CompuCyte Corporation, Cambridge, MA). Proliferation of biliary epithelial cells was assessed by automated quantitation of Ki-67–labeled nuclei in cells that were also labeled with the pancytokeratin antibody (indicative of biliary origin). Hepatocyte proliferation was also assessed using Ki-67 labeling; these cells had a characteristic histologic appearance and were not labeled with cytokeratin markers. The total number of hematoxylin-stained nuclei was determined for each liver section. This population of nuclei was further restricted through the use of gating that was applied to scatter plots of nuclei area versus circularity. This step was done in addition to looking at laser light absorbance levels around each nucleus that was collected using peripheral contours. Hepatocyte nuclei tended to be larger and more round, and they had a moderate level of scatter produced by the hepatocyte cytoplasm in contrast to nuclei of other cell types that were smaller and less round and had little to no cytoplasm. Once gating was applied, region galleries were examined to ensure accurate segmentation of hepatocytes. Multiple test scans were conducted until the optimal placement regions and segmentation of hepatocyte nuclei were achieved. Hepatocytes that were Ki-67 positive were plotted and separated in a similar manner by factoring in the laser absorbance of the Ki-67–positive cells. These DAB-stained, Ki-67–positive cells had higher absorbance levels than the Ki-67–negative, hematoxylin-stained hepatocytes, which were less dense.
Within the bile ducts, Ki-67–positive and –negative nuclei were segmented based on the amount of cytokeratin staining in the peripheral contours. Cells containing cytokeratin were counted as bile duct nuclei. Proliferating cells were separated from nonproliferating cells based on Ki-67 labeling (DAB). With this method, there was the potential for a peribiliary proliferating cell that is closely adjacent to a bile duct to be counted as a duct cell if the peripheral contour contained cytokeratin staining; however, the peripheral contours were set in close proximity to the primary event nucleus to make this a rare occurrence. Gallery images were examined to confirm optimal placement of gated regions in the scatter plots.
A labeling index for hepatocytes and biliary epithelium was developed using the ratios of Ki-67–stained and –unstained biliary epithelium and hepatocytes at each time point.
Statistics
The clinical chemistry and immunohistochemistry (IHC) parameters were first analyzed via 1-way analysis of variance (ANOVA) using Proc GLM in SAS, version 9.1 (SAS Institute, Cary, NC, 2002). The results of the Brown-Forsythe homogeneity of variance (HOV) tests and the plots of the residual versus predicted values were found to be unsatisfactory with respect to homogeneity of variance across treatment groups for a majority of the parameters (Littell et al. 2002). With the exception of two clinical chemistry parameters, a transformation to address the lack of homogeneity of variance between the groups could not be identified. Because of the lack of homogeneity of variance between the groups, all of the clinical chemistry parameters and IHC parameters were then analyzed using methods for unequal variances. The pairwise comparisons were performed via a 1-way Welch ANOVA using Proc GLM in SAS. The Hochberg multiplicity adjustment in Proc Multitest was used. The multiplicity adjustment was applied to the raw
Results
In-life Observations
Overall, WT mice were more severely affected by ANIT than IL-10 KO mice, based on changes in body weights, food consumption, and clinical signs. Although group mean body weights for both WT and IL-10 KO mice given ANIT were decreased relative to the vehicle control of the same strain as early as day 2 (approximately twenty-four hours after treatment), the WT mice showed a greater decrease (0.92-fold and 0.97-fold, for WT and KO, respectively). The nadir for both strains was day 4 (0.76-fold and 0.79-fold for WT and IL-10 KO, respectively). Compared with controls, body weights in treated mice were still reduced on day 5, but they were beginning to show recovery (0.78-fold- and 0.84-fold for WT and KO, respectively). These decreases in body weight were owing to dramatic reductions in food consumption following ANIT administration. Group mean food consumption for the day 1 to day 2 interval for WT and IL-10 KO ANIT-treated mice was 0.38-fold and 0.30-fold, respectively, compared with the vehicle control of the same strain. The nadir in food consumption was over the day 2 to day 3 interval (0.09-fold and 0.10-fold- for WT and KO mice, respectively). Food consumption increased over the next two intervals for both strains, but the increases were less for the WT mice compared with the IL-10 KO. At the final interval, from day 4 to day 5, food consumption in the ANIT-treated IL-10 KO mice was no different than the vehicle-treated IL-10 KO mice, whereas food consumption in the ANIT-treated WT mice was still decreased (0.71-fold) compared with the vehicle-treated WT mice. Eleven of twenty WT mice and eight of twenty IL-10 KO mice given ANIT showed clinical signs of toxicity including ataxia, decreased activity, rough coat, cold to touch, tremors, hunched posture, irregular or slow breathing, closed eyes, and/or loss of skin elasticity. In general, the incidence and severity of the clinical signs were greater in WT mice compared with IL-10 KO mice.
Organ Weights
Absolute and relative (to body weight) liver weights were similar between WT and IL-10 KO mice (Groups 1, 2, 3, and 4) on day 2. On days 3, 4, and 5, although absolute liver weights did not differ between WT and IL-10 KO mice, the relative liver weights were slightly greater for mice given ANIT (Groups 2 and 4) compared with the respective controls (Groups 1 and 3), but the degree of increase was higher in the WT mice (1.17-fold to 1.36-fold for WT compared with 1.08-fold to 1.30-fold for IL-10 KO). The relative liver weight changes may have been a reflection of the decreased terminal body weights for the ANIT-treated mice.
Clinical Chemistry
The majority of the statistically significant differences between the two ANIT-treated groups and their respective control groups occurred on or after day 3 (Table 1). Treatment of WT mice with ANIT resulted in statistically significant differences in total bilirubin (TBIL) as early as day 2; alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and bile acids as early as day 3; paraoxonase 1 (PON1) as early as day 4; and glutamate dehydrogenase (GLDH) on day 5 compared with the WT vehicle control (Groups 2 versus 1). Treatment of the IL-10 KO mice with ANIT resulted in statistically significant differences based on adjusted
There was no statistically significant difference based on adjusted
Mean Subjective Scores from Histologic Evaluation of ANIT-treated Male C56BL/6 and IL-10 Knockout Mice.
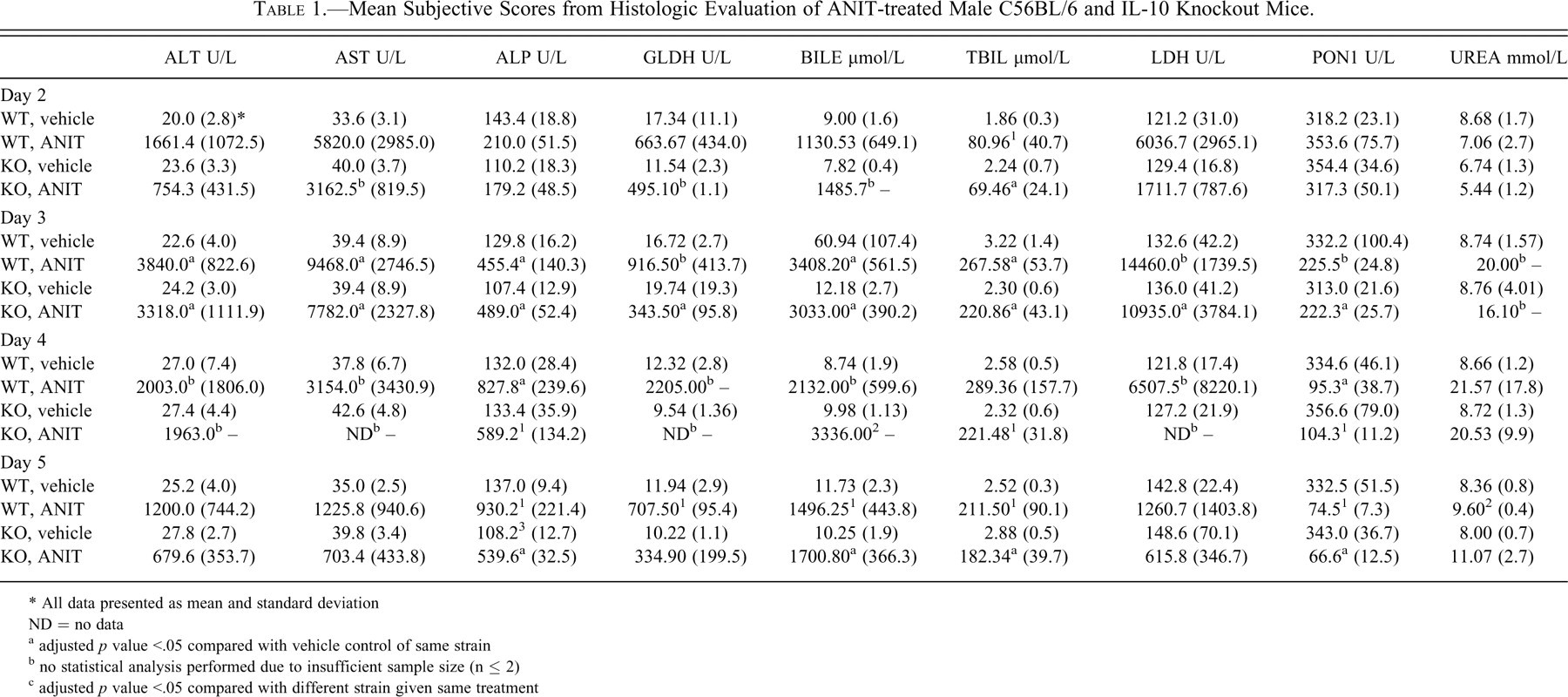
* All data presented as mean and standard deviation ND = no data
a adjusted
b no statistical analysis performed due to insufficient sample size (n ≤ 2)
c adjusted
Histology
The phenotypes of the injury produced by ANIT treatment were similar in both groups of mice. However, based on subjective scoring, there were modest differences in the degree of injury between the two groups. For clarity, only subjective differences of > 0.2 units were interpreted as a significant change.
On day 2 (twenty-four hours after ANIT treatment), there was uniform marked bile duct necrosis and edema in both ANIT-treated groups affecting essentially every portal tract (Table 2 ). Neutrophil infiltration (neutrophilic cholangitis), typical of ANIT-induced biliary injury, was more prominent in ANIT-treated IL-10 KO mice (Group 4) than the ANIT-treated WT mice (Group 2; Figure 1 ). Biliary hyperplasia was not evident in any group. Inflammation of the gall bladder was similar between ANIT-treated WT and IL-10 KO mice. Hepatocellular necrosis, characterized by distended, pale hepatocytes with wisps of remnant cytoplasm evident, was distributed in a multifocal fashion, although most often adjacent to damaged bile ducts, involving small aggregates of cells, typically about twenty to forty cells in a focus. Hepatocyte necrosis was more extensive in WT mice than in IL-10 KO mice on day 2. Similarly, microvesicular vacuolization was more apparent in WT mice than IL-10 KO mice. Hepatocytes in both groups of ANIT-treated mice lacked glycogen vacuolization likely associated with anorexia-driven glycogen catabolism (Figure 1, panels A and B).
Mean Clinical Pathology Values for ANIT-treated Male C57Bl/6 and IL-10 Knockout Mice.
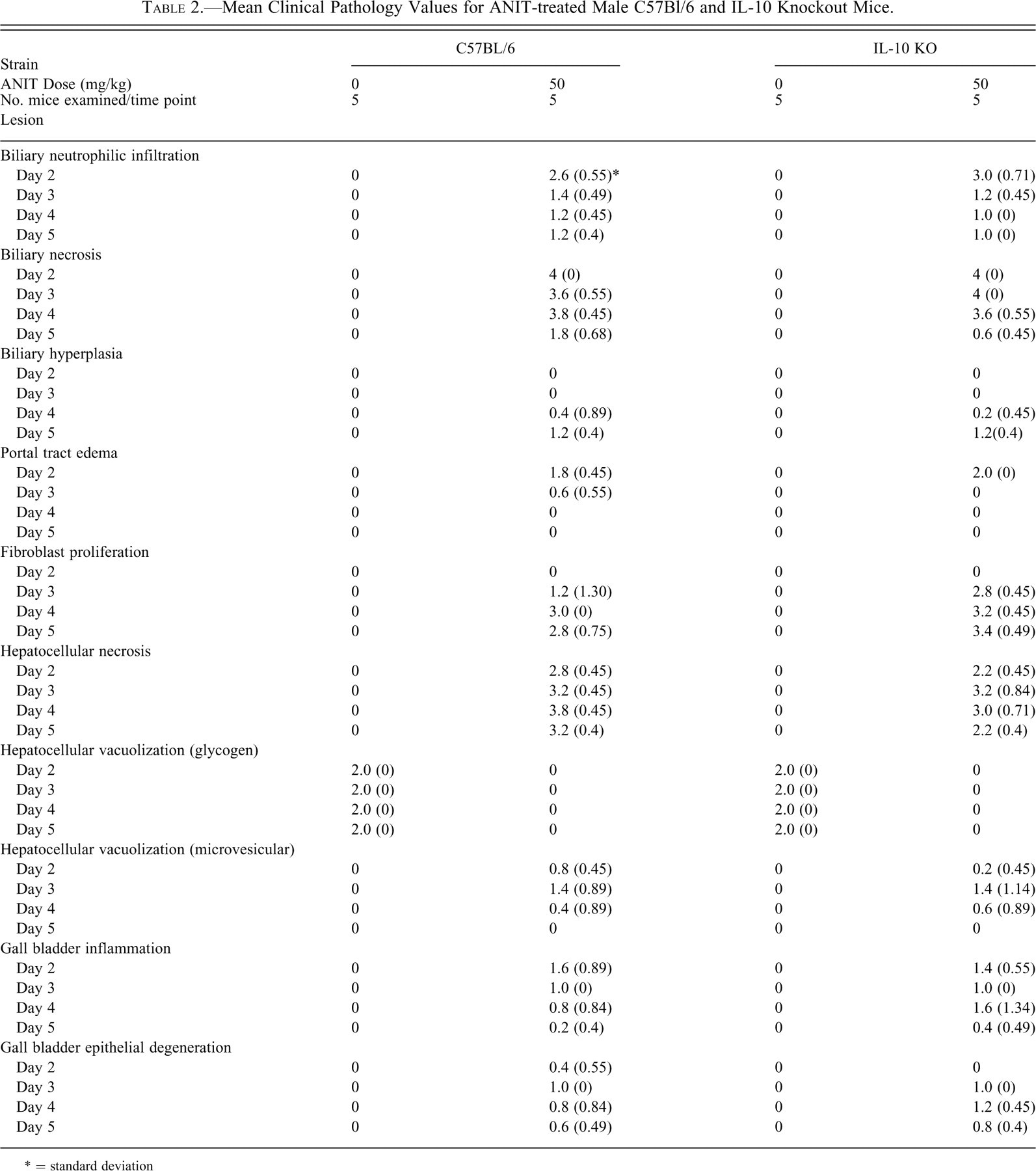
* = standard deviation
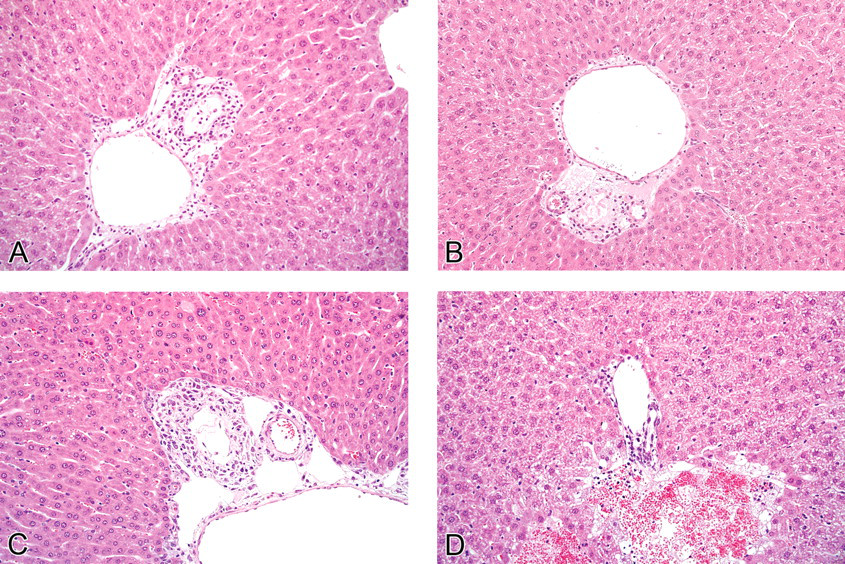
Comparison of α-napthylisothiocyanate (ANIT)–induced biliary lesions in IL-10 knockout (KO) and wild-type mice. On day 2, neutrophil infiltration of bile ducts was more prominent in ANIT-treated IL-10 KO mice (Group 4, panel A) than the ANIT-treated WT mice (Group 2, panel B). On day 3, proliferation of spindle-shaped cells circumferentially surrounding damaged bile ducts were more prominent in the ANIT-treated IL-10 KO mice (Group 4, panel C) than in the ANIT-treated WT mice (Group 2, panel D). Hematoxylin and eosin.
On day 3, bile duct necrosis remained prominent in ANIT-treated mice, but it persisted at a greater severity in the IL-10 KO mice (Group 4) compared with the ANIT-treated WT mice (Group 2; Table 2, Figure 1). The severity of neutrophilic cholangitis in both ANIT-treated groups was reduced compared with day 2 and was similar between the two strains. Portal tract edema was reduced in severity in ANIT-treated WT mice (Group 2) compared with day 2 and fully resolved in the ANIT-treated IL-10 KO mice (Group 4) by day 3. Proliferation of spindle-shaped cells surrounding damaged bile ducts (interpreted as myofibroblasts) was present on day 3 and was considered to be associated with repair of damaged biliary epithelium. These spindle-shaped cells were more prominent in the ANIT-treated IL-10 KO mice (Group 4) than ANIT-treated WT mice (Group 2; Figure 1, panels C and D). Gall bladder epithelial inflammation as well as degeneration and regeneration were similar in both strains of mice. ANIT treated WT and IL-10 KO mice had similar moderate to marked hepatocellular necrosis and microvesicular vacuolization.
On day 4, neutrophilic infiltrate, biliary necrosis, fibroblast proliferation, and biliary hyperplasia in ANIT-treated WT (Group 2) and IL-10 KO (Group 4) mice were similar between the two strains of mice (Table 2). Hepatocyte necrosis was, as on day 2, more severe in ANIT-treated WT mice (Group 2) than the ANIT-treated IL-10 KO mice (Group 4), whereas microvesicular vacuolization was similar in both ANIT-treated groups of mice (Figure 2 ). Gall bladder epithelial degeneration and inflammation was more prominent in the ANIT-treated IL-10 KO mice compared with the WT mice (Figure 2).
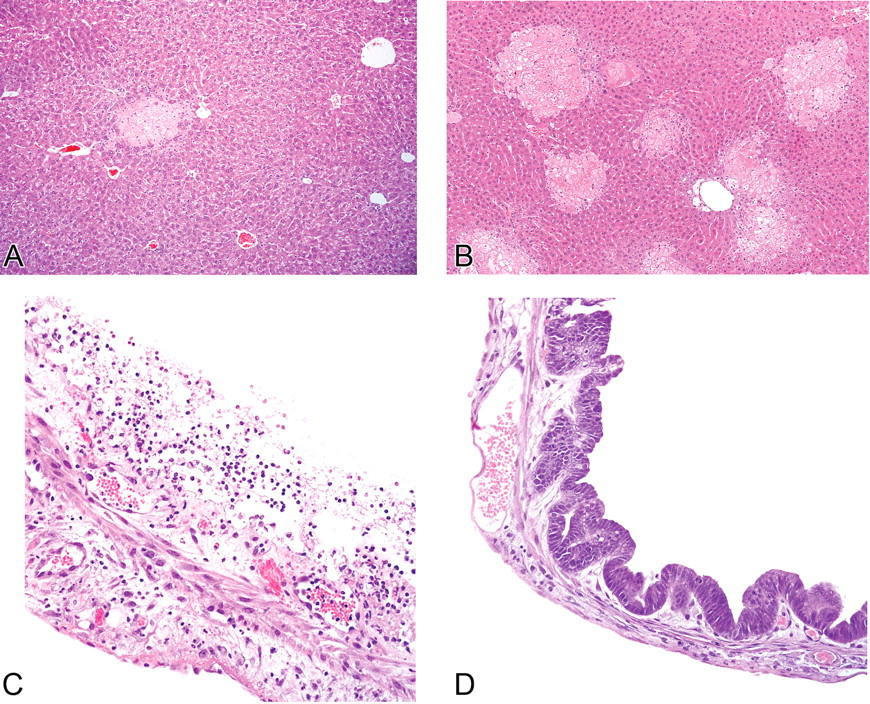
Comparison of hepatocyte and gall bladder injury and biliary proliferation in α-napthylisothiocyanate (ANIT)–treated IL-10 knockout (KO) and wild-type (WT) mice. Hepatocyte necrosis on day 4 was less evident in ANIT-treated IL-10 KO mice (Group 4, panel A) than in ANIT-treated WT mice (Group 2, panel B). On day 4, gall bladder epithelial degeneration and inflammation was more prominent in the ANIT-treated IL-10 KO mice. This image was from the most severely affected animal (panel C) compared with the WT mice (panel D).
By day 5, the pattern in both ANIT-treated groups of mice was one of reduced biliary necrosis and inflammation with persistent hepatocellular necrosis. The neutrophilic infiltrate was minimal in both groups of mice. Biliary necrosis was more prominent in ANIT-treated WT mice (Group 2) compared with ANIT-treated IL-10 KO mice (Group 4) although, in both of these groups of mice, biliary necrosis was much less evident by day 5 compared with the earlier time points. Peribiliary fibroblast proliferation was increased for the ANIT-treated IL-10 KO mice (Group 4) compared with ANIT-treated WT mice (Group 2). Biliary hyperplasia scores were similar between both groups of mice on day 5, and increased from day 4. Gall bladder lesions were similar between both ANIT-treated groups and were reduced compared with day 4. Hepatocellular necrosis persisted until day 5, though to a lesser degree than that seen at day 4. The pattern of more severe hepatocellular necrosis in WT mice persisted and was more prominent in the ANIT-treated WT mice (Group 2) than the ANIT-treated IL-10 KO mice (Group 4). Hepatocellular microvesicular vacuolation was fully resolved by day 5 in both strains.
In summary, biliary necrosis was similar in ANIT-treated WT and IL-10 KO mice, but there was increased neutrophilic cholangitis and moderately less hepatocyte necrosis and injury in IL-10 KO mice on day 2. On day 3, the ANIT-treated IL-10 KO mice had slightly more severe biliary necrosis and appeared to initiate repair of damaged bile ducts (via proliferation of circumferential fibroblasts) to a greater extent than ANIT-treated WT mice. On day 4, hepatocellular necrosis in ANIT-treated WT mice was more severe than in the ANIT-treated IL-10 KO mice, and gall bladder epithelial degeneration and inflammation was more prominent in the IL-10 KO mice compared with the WT mice. By day 5, IL-10 KO mice treated with ANIT had less biliary necrosis and hepatocellular necrosis and more fibroblastic proliferation around damaged bile ducts than WT mice treated with ANIT.
Proliferation Assays
The Ki-67 labelling index for biliary epithelial cells from ANIT-treated WT or IL-10 KO mice did not differ with statistical significance until day 5, although it was increased at various time points (Figures 3 and 4 ). Biliary proliferation in ANIT-treated WT mice (Group 2) was increased 1.74-fold and 6.6-fold on days 4 and 5, respectively, compared with WT control mice. Biliary proliferation in ANIT-treated IL-10 KO mice (Group 4) was increased 7.7-fold compared with IL-10 KO control mice (Group 3) on day 2, did not differ on day 3, and was increased 7.0-fold on day 4. By day 5, ANIT-treated IL-10 KO mice had a statistically significant 12-fold increase in biliary proliferation compared with the IL-10 KO control group (Figure 4 ). There was no significant difference in proliferation between ANIT-treated WT and IL-10 KO strains until day 5, when ANIT-treated IL-10 KO mice had a significant increase (4.2-fold) in biliary epithelial proliferation compared with ANIT-treated WT mice (Group 2).
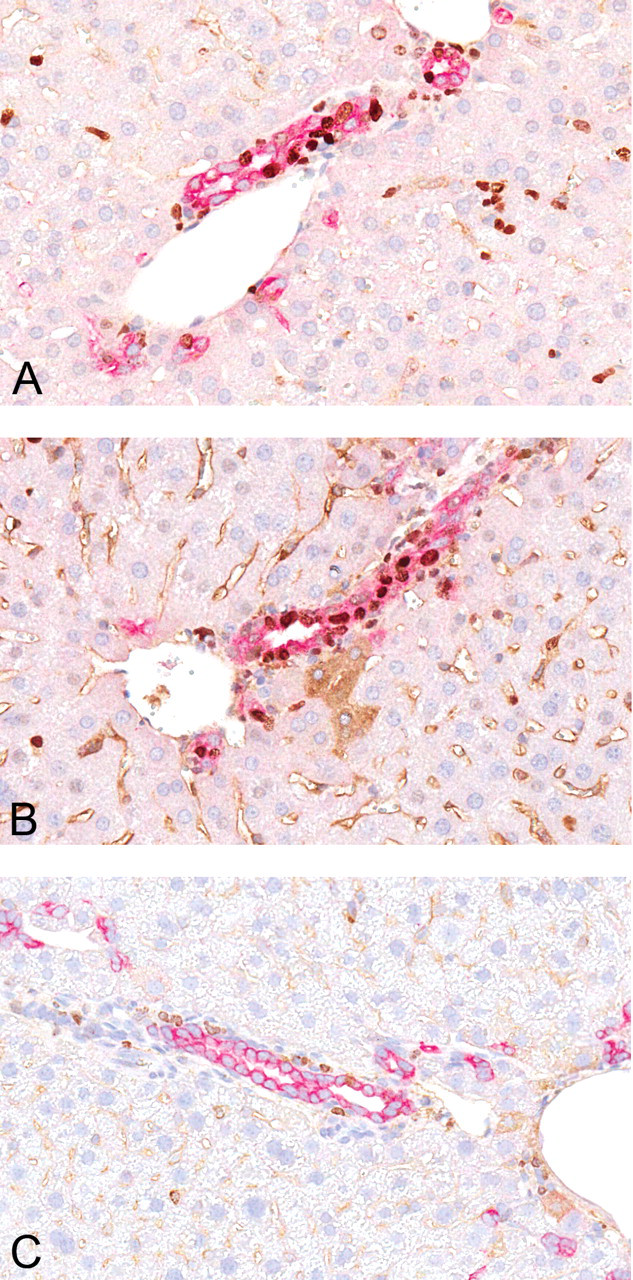
On day 5, α-napthylisothiocyanate–treated wild-type (WT) mice (panel A) had a smaller magnitude increase in biliary proliferation measured by Ki-67 labeling compared with the α-napthylisothiocyanate–treated IL-10 knockout mice (panel B). Biliary proliferation was uniformly low in untreated IL-10 knockout and WT mice. An untreated WT mouse is shown in panel C. Biliary epithelial cells labeled immunohistochemically with anti-pancytokeratin antibodies with a red chromagen and cell proliferation detected by Ki-67 labeling with a brown chromagen.
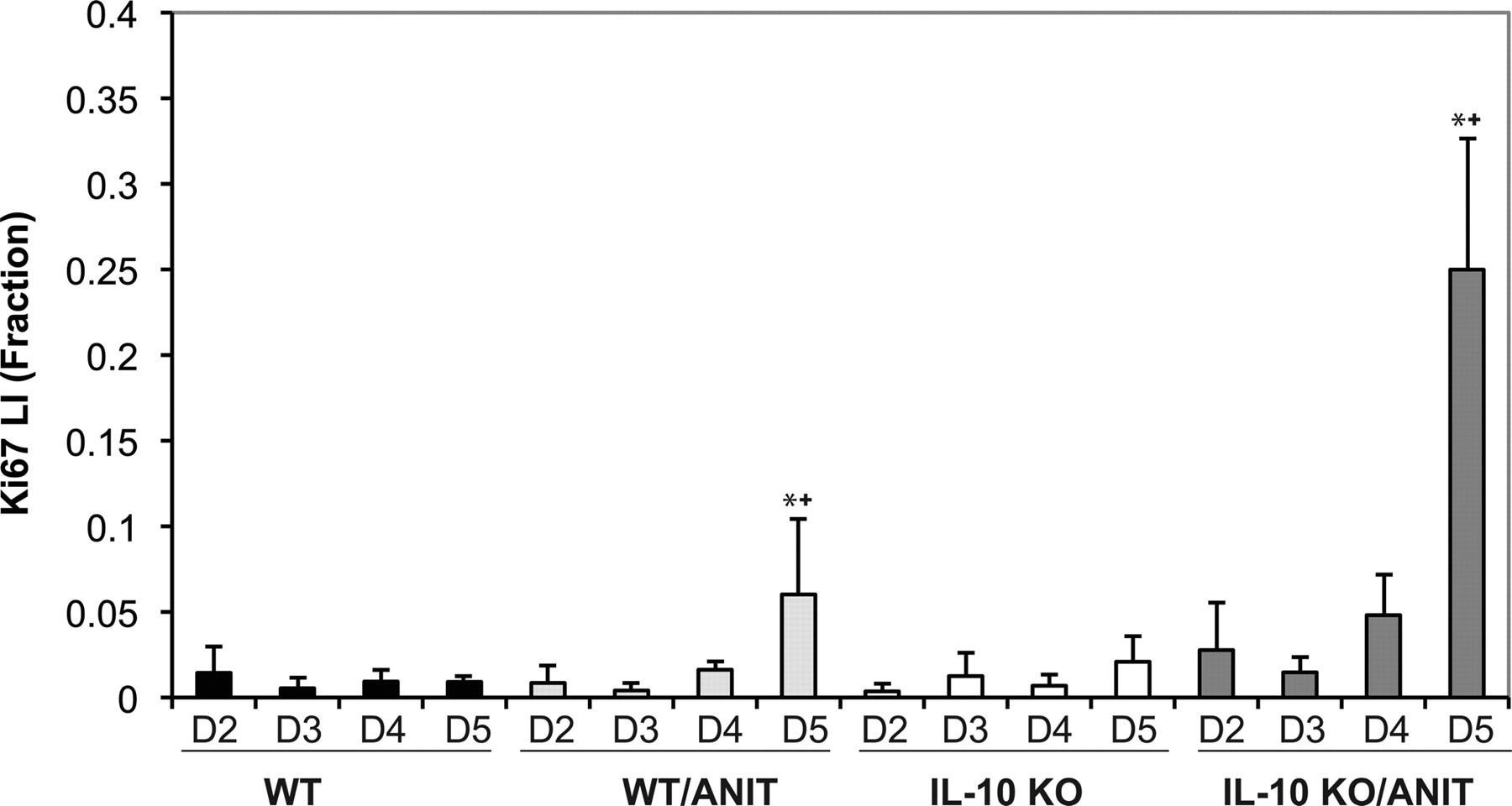
The Ki-67 labeling index for biliary epithelial cells from α-napthylisothiocyanate–treated wild-type (WT) or IL-10 knockout (KO) mice and controls. Labeling did not differ significantly until day 5 for treated WT and IL-10 KO mice compared to their respective controls (*). In addition, on day 5, labeling of bile duct epithelia from treated IL-10 KO mice was increased over the treated WT mice as well (+).
Hepatocyte proliferation was increased in WT and KO mice over the four days following ANIT treatment, though none of the changes was statistically significant (Figure 5 ). There was an eleven- and thirty-one-fold increase in the hepatocellular Ki-67 labeling index for the WT ANIT-treated mice compared with the WT vehicle-treated mice on days 4 and 5, respectively, and a 127-fold and ten-fold increase for the IL-10 KO ANIT-treated mice compared with the IL-10 KO vehicle-treated mice on days 4 and 5, respectively.
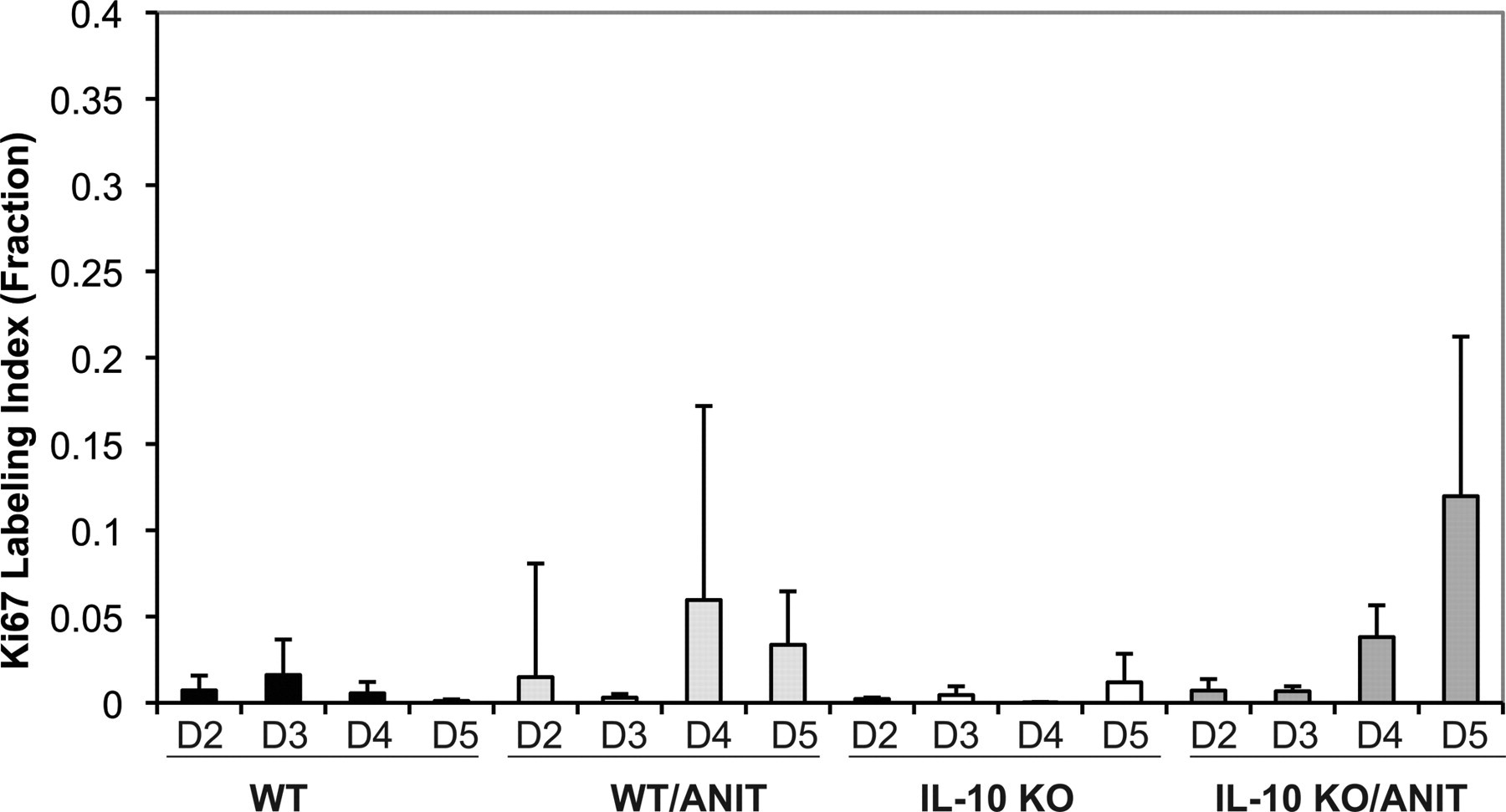
Ki-67 labeling indices for hepatocytes from α-napthylisothiocyanate–treated IL-10 knockout, wild-type and control mice. Hepatocyte proliferation increased in mice during the last two days following α-napthylisothiocyanate treatment, although none of the changes was statistically significant.
Discussion
We investigated the differences in acute toxicity of the well-characterized hepatobiliary toxin ANIT on biliary epithelium and hepatocytes in mice with and without the anti-inflammatory cytokine IL-10. Based on a previous study that showed a significant protective role for IL-10 in acetaminophen-treated mice, exemplified by increased hepatocellular necrosis and injury in IL-10 KO mice (Bourdi et al. 2002), we anticipated an increase in biliary and hepatocellular injury in the ANIT-treated IL-10 KO mice compared with similarly treated WT mice. Surprisingly, WT mice were generally more severely affected than IL-10 KO mice with regard to the general clinical observations, including deteriorating clinical condition, decreased body weight, and decreased food consumption.
Overall, IL-10 KO mice tended to have more biliary epithelial injury than the WT mice. On day 2, neutrophilic infiltrate around the bile ducts was more prominent in the IL-10 KO mice, and biliary necrosis was more evident on day 3 compared with WT mice. However, by day 5, there was less biliary necrosis in the IL-10 KO compared with WT mice. Periductular spindle-cell proliferation, interpreted as part of the repair of previous injury, was more robust in the IL-10 KO mice from day 3 onward. Ki-67 labeling studies demonstrated that the IL-10 KO mice had more biliary epithelial proliferation than WT mice on day 5, in keeping with the evidence of increased numbers of circumferential spindle cells surrounding the damaged ducts and the evidence of greater biliary epithelial necrosis on day 3. Taken together, these observations (the increased biliary epithelial proliferation and spindle-cell proliferation coupled with decreased biliary necrosis on day 5) suggest a greater initial injury, but possibly a more effective response to injury by the IL-10 KO mice. Although the clinical pathology data did not demonstrate a difference between the strains, the lack of significant changes in the clinical pathology analytes may be attributed to the modest differences in the histologic severity of lesions and the relatively small numbers of mice per group. The serum markers are not particularly well characterized for biliary damage in mice, based on the paucity of relevant information in the literature.
Unlike the biliary epithelial effects and contrary to our expectations, over the 5-day course of this study, there was a consistent tendency for WT mice to have a greater degree of hepatocellular injury compared with ANIT-treated IL-10 KO mice based on relative liver weights as well as histopathological assessment. Histologic scoring of hepatocellular necrosis in IL-10 KO mice was less severe at all time points, with the exception of day 3, when the level of injury was equivalent between strains. Consistent with the histologic observations, although not statistically significant, the means of several serum markers of hepatocellular injury such as AST, GLDH, and LDH were also lower on day 2 and day 5 in the IL-10 KO mice. Within twenty-four hours (day 2) of ANIT exposure, hepatocellular microvesicular vacuolization was reduced in IL-10 KO mice compared with WT mice. Microvesicular vacuolization can result from mitochondrial dysfunction (Fromenty and Pessayre 1997), and this lesion is consistent with the published observations that describe mitochondrial structural abnormalities and microvesicular osmophilic material in the cytoplasm of ANIT-treated rodents (Schaffner et al. 1973).
Based on histologic and clinical pathology end points, our data suggest that IL-10 may provide modest protection of the biliary epithelium from ANIT-induced injury. However, IL-10 may not protect hepatocytes, as shown by greater hepatocellular injury in WT mice than in the IL-10 KO mice. This finding is in contrast to the heptoprotective role of IL-10 in acetaminophen toxicity (Bourdi et al. 2002; Bourdi et al. 2007; Ju et al. 2002). It is not clear why hepatocytes from IL-10 KO mice would respond differently to ANIT and acetaminophen toxicity. One possibility is that the mechanism of hepatocellular toxicity of ANIT differs from that of acetaminophen. The precise pathogenesis of acute ANIT-induced biliary injury has not been clearly established. α-Napthylisothiocyanate is thought to diffuse out of the blood and cross the sinusoidal membrane of the hepatocyte owing to its lipophilicity. Within the hepatocyte, it is metabolized by the cytochrome P450 system and conjugated to glutathione (GSH) for elimination into bile, where it can be detected within one hour of oral administration (Jean and Roth 1995). The ANIT-GSH conjugate is likely excreted via the canalicular transporter MRP2, as MRP2 KO mice are protected from ANIT-induced cholestasis (Dietrich et al. 2001). The role of GSH in ANIT excretion is supported by the observation that GSH depletion prevents ANIT accumulation in bile (Jean and Roth 1995). In cultured hepatocytes, ANIT-GSH complexes secreted into the culture medium were unstable at neutral pH, and 95% of the conjugate dissociated to release ANIT within five minutes (Carpenter-Deyo et al. 1991). Roth and co-workers have suggested that a similar dissociation takes place in bile in vivo and then leads to damage of the biliary epithelium (Carpenter-Deyo et al. 1991; Jean et al. 1995; Jean and Roth 1995; Roth and Dahm 1997). Depletion of cellular glutathione and release of cytotoxic and inflammatory mediators, including those that attract neutrophils, have been suggested to contribute to hepatic injury (Dahm et al. 1991; Hill et al. 1999; Roth and Dahm 1997; Roth and Hewett 1990).
Oxidative injury likely plays a large role in ANIT-induced biliary epithelial injury, possibly as a result of one or more possible sulfhydryl-reactive intermediates, accentuated by repeated rounds of secretion and reuptake of the glutathione conjugate (Dietrich et al. 2001; Giles et al. 2002). Hepatocyte injury from ANIT is accentuated by neutrophil infiltration (Kodali et al. 2006), but the exact mechanisms are not well characterized. It is possible the bile acid accumulation in periportal hepatocytes may affect hepatocyte death.
The histologic pattern of injury in acetaminophen is typically centrilobular, reflective of the site of metabolism by the mixed-function oxidases, whereas ANIT produces initial biliary necrosis and inflammation and subsequent scattered hepatocellular necrosis, often centered on the periphery of the portal tracts (Blazka et al. 1996; Goldfarb et al. 1962; Schaffner et al. 1973). Acetaminophen toxicity leads to glutathione depletion and formation of N-acetyl-p-benzoquinoneimine, which can form adducts with cellular proteins and cause downstream events such as mitochondrial dysfunction and production of reactive oxygen species and peroxynitrite (Dambach et al. 2006; Jaeschke and Bajt 2006; Nelson 1990). Neutrophil infiltration appears not to affect hepatocyte injury induced by acetaminophen (Cover et al. 2006; Jaeschke and Liu 2007).The innate immune system, because of its role in the early, nonspecific inflammatory response, may influence toxicant-induced injury via cytokine release from several cell types, such as macrophages and subsets of lymphocytes. Several studies have shown that the innate immune system can alter hepatocellular injury following acetaminophen exposure (Blazka et al. 1996; Ju et al. 2002; Liu and Kaplowitz 2006). However, currently the generally accepted view is that the immune system has a modest modulatory role in hepatocyte injury (Jaeschke et al. 2008). In this study, the effect of the absence of IL-10 was modest, but different, on hepatocytes and biliary epithelium of ANIT-treated mice. α-Napthylisothiocyanate–induced hepatocellular toxicity was modestly reduced, rather than increased, in the IL-10 KO mice, as seen in acetaminophen-treated mice (Bourdi et al. 2002), whereas injury to the biliary tree was accentuated in the IL-10 KO mice. Possibly, differences in the mechanisms of ANIT-induced injury in biliary epithelium and hepatocytes affect the response to the immunomodulatory effects of IL-10. Despite the slightly increased degree of biliary injury in the IL-10 KO mice, these animals appear to respond in a more robust manner to the injury and to resolve the injury more rapidly than the WT mice, as shown by the increased proliferation of hepatocytes, biliary epithelium, and spindle cells and the decreased biliary necrosis noted on day 5.
Footnotes
Acknowledgments
The authors would like to acknowledge the scientific contributions of Dr. Judith Prescott, David Krull, and Leroy Butler to this body of work.
