Abstract
Although the dog is a common choice among nonrodent species in evaluation of compound safety for regulatory submission, information regarding the incidence of spontaneous or incidental microscopic changes in canine peripheral nerve is limited. A retrospective examination was performed of routine histologic preparations of sciatic nerve from eighty-one control dogs in toxicity studies ranging from ten days to three months in duration. Spontaneous background changes included digestion chambers, foci of vacuolation, nerve fibers circumscribed by proliferating Schwann cells (bands of Büngner), and small foci of myelin aggregation. The latter accounted for 91% of the microscopic changes and were noted in all sections examined. These changes were quantified, and the number per square millimeter of evaluable nerve tissue was determined for each slide. Densities of foci varied among the slides examined; no age- or sex-related trends were apparent. In addition, anatomic features of peripheral nerves including nodes of Ranvier, Schmidt-Lanterman incisures, Renaut bodies, and effects resulting from sectioning plane were noted. By demonstrating the range of effects observed within control animals, these observations provide a basis for recognition of possible compound-related effects in routine nerve preparations from dogs included in toxicity studies.
Introduction
Published descriptions of histopathologic changes in peripheral nerves are largely restricted to accounts describing specialized techniques, such as teased nerve fiber analysis (Braund et al. 1982a; Dorman et al. 2002; Krinke et al. 2000), electron microscopy (Hoover et al. 1981), or thin transverse sections of plastic embedded nerves (Braund et al. 1982b; Jortner 2000). Ideally, these techniques should be applied prospectively, a situation that is often not feasible in toxicology studies designed to screen large numbers of animals and all organ systems. The more specialized evaluations generally are reserved for circumstances of known or suspected neurotoxicity. Toxicologic pathologists routinely assess potentially compound-related effects on peripheral nerve by examination of formalin-fixed, paraffin-embedded, hematoxylin and eosin (H&E)–stained longitudinal sections. It is important to have an understanding of what constitutes “normal” background change in order to recognize what might be compound-related. Reviews of spontaneous microscopic effects in peripheral nerves in the context of toxicity studies have been provided for rats (Cotard-Bartley et al. 1981; Eisenbrandt et al. 1990), but similar information for dogs is lacking. This retrospective review of sciatic nerves is intended to provide an understanding of the range of spontaneous microscopic change among dogs.
Materials and Methods
Control beagle dogs were identified from seventeen toxicologic studies (identified as A through Q) conducted in Abbott Park, Illinois. All dogs were maintained in a facility accredited by the Association for Assessment and Accreditation of Laboratory Care International, and studies were reviewed and approved by the Institutional Animal Care and Use Committee. The eighty-one dogs, forty-four females and thirty-seven males, with ages ranging from 5.6 to 18.0 months (mean 10.9; median 9.7 months), were from two sources, Marshall BioResources, North Rose, New York (thirty-four females, thirty-two males) and Covance Research Products, Inc., Cumberland, Virginia (ten females, five males). Study durations ranged from ten days to three months. One formalin-fixed, paraffin-embedded longitudinal section of sciatic nerve, cut at approximately 5 μm and stained by H&E, was reviewed from each dog. None of the nerve samples was immersed or stored in ethyl alcohol prior to processing. Each slide was scanned at 40× using the Aperio ScanScope XT imaging system (Vista, CA, USA). Images were reviewed and annotated using Aperio ImageScope, version 10.0, and foci of change were counted. Estimation of nerve area per slide was made using linear measurements in Aperio ImageScope and verified by Metamorph software (Molecular Devices, Inc., Sunnyvale, CA, USA). Numbers of foci per unit area were calculated for each slide. Annotated images in Aperio were peer reviewed. Immunohistochemical (IHC) staining was performed on selected paraffin sections against neurofilament (NF; ab24571 Abcam, Cambridge, MA, USA), myelin basic protein (MBP; 24567 Abcam), von Willebrand factor (vWF; A0082 Dako, Carpenteria, CA, USA) and macrophages (calprotectin; ab22506, Abcam) using horseradish peroxidase or alkaline phosphatase polymer detection (Biocare Medical, Concord, CA, USA) on an automated platform (intelliPATH FLX, Biocare Medical). Sections were counterstained with hematoxylin.
Results
The sections of nerve examined ranged from 6 to 43 mm2 (mean: 22 mm2) in evaluable area (Table 1 ). All sections examined had foci of change consisting of one or more of the following: foci of myelin aggregation, vacuolated foci, digestion chambers, and/or Schwann cell proliferation. Numbers of combined foci ranged from 1 to 131 per section (mean: 24.2 per section). Numbers of foci per mm2 of evaluable nerve ranged from 0.1 to 4.1 (mean: 1.1). No sex- or age-related trends in the incidence or density of foci were evident among the dogs studied.
Summary of control dog sciatic nerve findings.
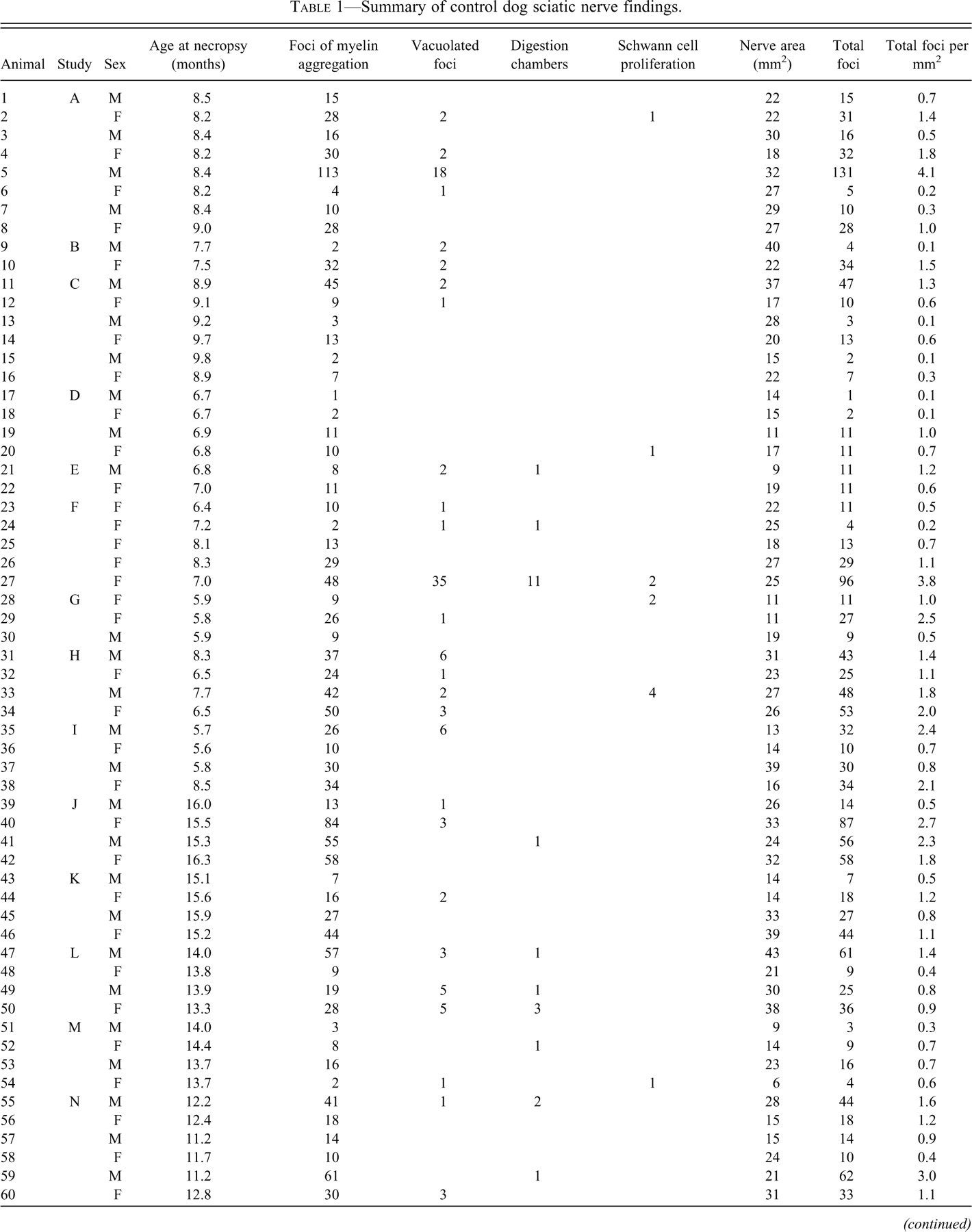
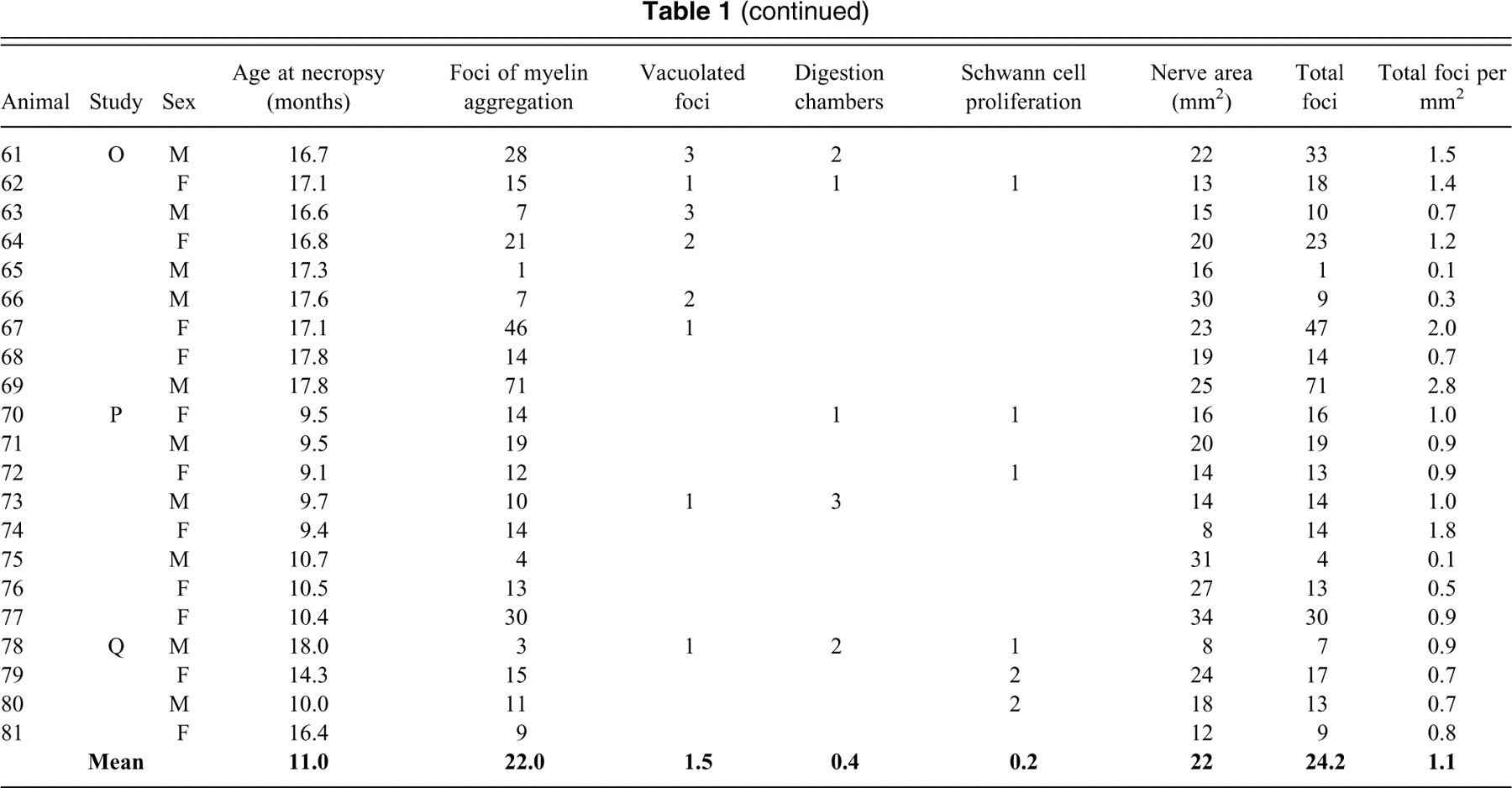
Foci of myelin aggregation (Figure 1A) accounted for 91% of the changes noted and were characterized by eosinophilic hyaline globules (∼5 um diameter) surrounded by a clear space. The predominantly unilocular foci were frequently adjacent to the Schwann cell nucleus and were often surrounded by a narrow rim of basophilic material continuous with the nucleus. These foci were noted in all eighty-one dogs examined.
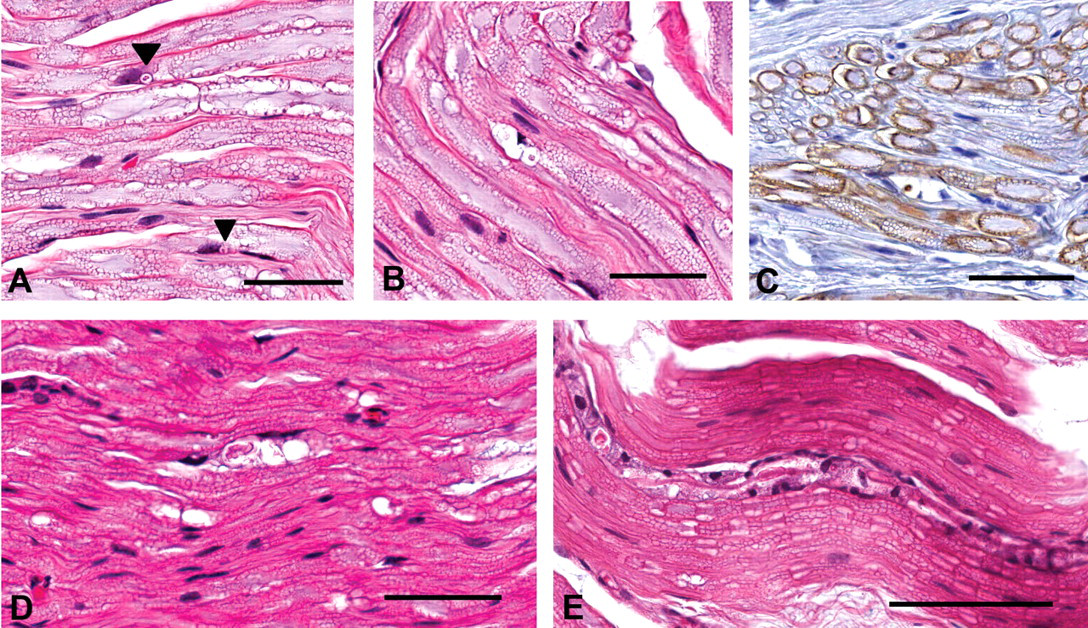
Representative foci of change within canine nerve fibers. (A) Typical foci of myelin aggregation are present (arrowheads), adjacent to one pole of a Schwann cell nucleus, including eosinophilic material within a clear space having a narrow basophilic rim. (B) A vacuolated focus is larger, multilocular, contains less intensely eosinophilic material, and is associated with a distorted cell nucleus. (C) Positive reaction of material with immunohistochemical staining against myeli basic protein confirms the myelin content within a focus. (D) A digestion chamber is a larger, multilocular, linear array of compartmentalized myelin debris. (E) Proliferating Schwann cells surround a degenerate nerve fiber. Hematoxylin and eosin stain (A, B, D, and E). Hematoxylin counterstain (C). Scale bars: 50 μm (A–D), 100 μm (E).
Vacuolated foci (Figure 1B) were less common (6% of all foci noted, occurring in thirty-five of eighty-one dogs) and generally larger than foci of myelin aggregation. The vacuolated foci were multilocular, up to 30 μm in diameter, contained irregularly shaped material with less eosinophilic staining than foci of myelin aggregation, and contained cells having compressed and deformed nuclei. The eosinophilic material within foci was confirmed to contain myelin by immunohistochemical staining against myelin basic protein (Figure 1C).
Digestion chambers (2% of all foci noted, occurring in fifteen of eighty-one dogs) are linear arrays of degenerate material within a nerve fiber, having characteristics of both foci of myelin aggregation and vacuolation (Figure 1D). Whereas most nerve sections contained only one or two affected nerve fibers, one dog had eleven digestion chambers. The chambers ranged from 30 to 150 μm in length and were characterized by variable amounts of aggregated myelin and vacuolated spaces with occasional cells interspersed. Immunohistochemical staining against calprotectin failed to demonstrate macrophages within the degenerate foci (not shown). Digestion chambers were frequently bordered by increased numbers of Schwann cell nuclei.
Arrays of proliferating Schwann cells (bands of Büngner) aligned along a degenerate nerve fiber (within the residual basal lamina generated by the myelinating Schwann cell that served the affected internode) were noted in twelve of eighty-one dogs and made up only 1% of the observed nerve changes (Figure 1E). Schwann cell nuclei within the proliferative zones stained more intensely and had irregularly rounded profiles compared to those associated with adjacent intact nerve fibers.
Normal structures—such as nodes of Ranvier, Schmidt-Lanterman incisures, and Renaut bodies—and plane of section artifacts were frequently observed among the sections examined (Elcock et al. 2001; Ortiz-Hidalgo and Weller 1992; Small et al. 1987; Summers et al. 1995). The loss of lipid from myelin with formalin fixation can result in artifactual vacuoles, which may resemble a degenerative nerve fiber change (Bolon et al. 2008). When nerve fibers bend into the plane of section, the resulting transverse sections can resemble foci of myelin aggregation (Figure 2A).
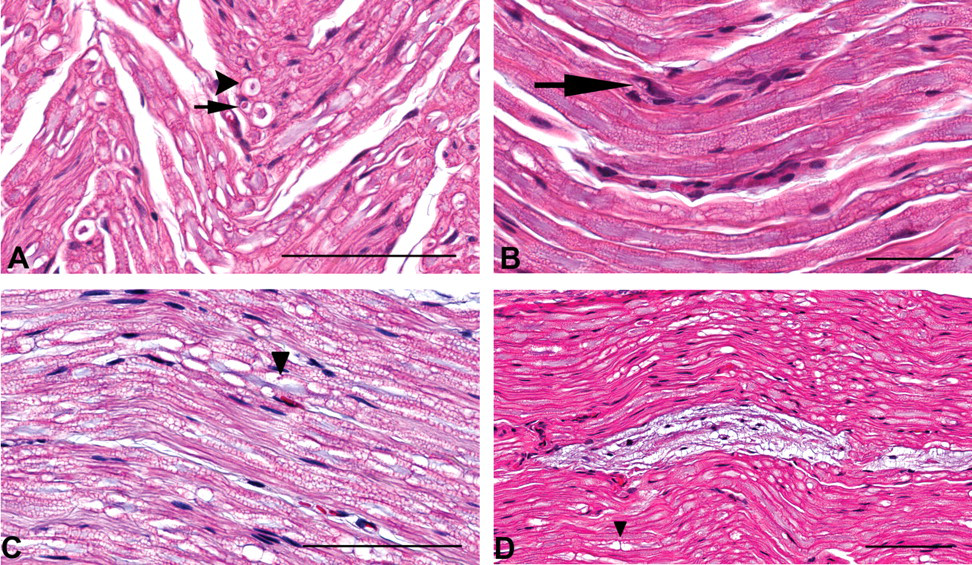
Common incidental or potentially confounding findings in canine nerves. (A) Transverse sections through nerve fibers as they bend into the plane of section can resemble foci of myelin aggregation. Here two such sections (one indicated by the arrowhead) are adjacent to a focus (arrow). (B) A vessel (with luminal erythrocytes evident) in the lower portion of the image is distinguished from an array of proliferating Schwann cells in the upper portion. The arrow denotes a focus of myelin aggregation. (C) Schmidt-Lanterman incisures are normal, longitudinally spaced variations within the coiled myelin layers surrounding an axon. The symmetrical pattern of ovoid clear spaces with eosinophilic round inclusions (arrowhead) represents the expanded spaces between myelin layers and sites of tightly packed myelin associated with these structures. (D) A prominent Renaut body is positioned between nerve fibers. The arrowhead denotes a node of Ranvier. Hematoxylin and eosin stain. Scale bars (A–C): 50 μm; 100 μm (D).
Arrays of regenerating Schwann cells were frequently difficult to distinguish from endoneurial vasculature in the absence of concurrent myelin debris (in the case of Schwann cell proliferation) or the presence of blood cells within the vasculature (Figure 2B). Immunohistochemical staining against von Willebrand factor or CD34 was not beneficial in distinguishing these features because of failure of endoneurial vessels to react robustly with the method, despite adequate results in vasculature within the epineurium (not shown). Bundles of unmyelinated nerve fibers (Remak fibers) accompanied by nonmyelinating Schwann cells can also resemble areas of Schwann cell proliferation, but the nonmyelinating cell nuclei are generally more uniform and less dense, lack the more linearly parallel arrangements of the proliferating Schwann cells, and are not associated with degenerative foci.
Schmidt-Lanterman incisures are normal features of peripheral nerve fibers, representing separations of myelin at the major dense line, forming expanded cytoplasmic spaces between coiled layers of myelin. At certain sectioning planes, they can resemble foci of myelin aggregation (Figure 2C).
Renaut bodies are frequently observed basophilic endoneurial structures composed of collagen fibers, mesenchymal cells, and glycosaminoglycans. Although they are sometimes discrete formations (Figure 2D), the loose, basophilic, sparsely cellular stromal tissue can appear as anastomosing strands throughout the nerve. A node of Ranvier is the junction between two abutting nerve segments (internodes) myelinated by individual Schwann cells. The axon is narrowed at the node, and the ensheathing myelin layers on either side of the node are separated by cytoplasmic expansions, causing adjacent clear bulges (Figure 2D). A tangential or oblique section through a portion of a node can resemble a vacuolated focus.
Discussion
Foci of myelin aggregation, vacuolated foci, digestion chambers, and Schwann cell proliferation are changes of peripheral nerve noted among control dogs. These changes represent a continuum of individual nerve fiber changes distinguished by size and composition. Foci of myelin aggregation accounted for the majority of findings noted in this survey of control dogs and were present in all sections of sciatic nerve examined. The significance of this change is uncertain, but it plausibly represents a process involved in physiologic maintenance of myelin integrity. Whereas the somewhat more conspicuous changes of digestion chambers and proliferating Schwann cells have been associated with Wallerian degeneration and regenerative responses to nerve injury (Bunge 1994; Fernandez-Valle et al. 1995), foci of myelin aggregation have not been described in association with these processes. Perinuclear lamellar rod-shaped bodies within equine Schwann cells have been described ultrastructurally (Force et al. 1986) in the absence of evident nerve fiber degeneration. These inclusions were considered similar to protagon (abbreviated ‘Π’ or ‘pi’) granules of Reich, described as occurring in normal human peripheral nerves, increasing with age, and including metachromatic-staining lysosomal remnants of physiologic myelin metabolism (Tomonaga and Sluga 1970). Similar structures are described in sheep, cattle, goats, and tigers. Reich noted them in dogs in 1907 (Noback 1953), and they have been confirmed ultrastructurally (Illanes et al. 1990). Pi granules are recognized as a component of reactive changes, in contrast to degenerative or proliferative changes, noted in a galactose-induced rat model of diabetic neuropathy (Kalichman et al. 1998). It is possible that canine foci of myelin aggregation apparent on light microscopy are analogous to these ultrastructural manifestations of myelin turnover.
Conclusion
Control purpose-bred beagle dogs can have a range of spontaneous background changes in sciatic nerve sections. To distinguish incidental changes from those related to compound, careful attention to detail and knowledge of characteristics and incidence of spontaneous changes are important.
