Abstract
Nerve fiber teasing is a sensitive technique utilized in diagnostic neuropathology practice, laboratory research, and animal toxicity studies for characterizing changes in single myelinated nerve fibers over extended distances. In animal toxicity studies, a nerve portion (approximately 10 mm in length) is stained with Sudan black for 24 to 48 hours and then transferred into a drop of viscous medium (eg, glycerin) mounted on an adhesive-coated glass slide, positioning it such that the proximodistal orientation is known. Individual fibers are removed using fine forceps while the sample is viewed under a stereomicroscope. In general, lesions can be identified during teasing, but more detailed characterization and photodocumentation is undertaken once nerve fibers have been dried and coverslipped. Nerve fiber teasing is particularly useful for distinguishing early stages of axonal degeneration (which presents as ovoid fiber fragments in the midinternodal region) from segmental demyelination (which presents as loss of original myelin segments and their replacement by thinner, shorter segments in the absence of axonal damage). The slow, laborious nature of nerve fiber teasing dictates that the technique will be employed on a few samples as an auxiliary method to better define the pathogenesis of nerve lesions first identified by conventional histopathologic assessment.
Introduction
Nerve fiber teasing is a well-recognized technique for evaluating the structure of myelinated nerves. Initial methodology was developed in the 19th century by pioneers of neuroanatomy and neuropathology such as Albert Gombault, Albert von Kölliker, Santiago Ramon y Cajal, and Robert Remak. More recently, many investigators have described a number of protocol refinements for the technique 1 –6 as well as provided insights regarding interpretation of lesions in isolated nerve fibers. 4,7 –9
Teased fiber preparations are a preferred means for exploring lesion initiation and progression during mechanistic neurotoxicity investigations due to the exceptional microarchitectural detail that can be observed in axons and myelin segments of isolated nerve fibers. In particular, evaluation of isolated nerve fibers is the most sensitive means for differentiating early axonal degeneration from early segmental demyelination and is quite useful for evaluating remyelination. Therefore, nerve fiber teasing is a key technique for discriminating the actual peripheral nerve target of a toxic agent, knowledge of which is essential to define the prognosis for recovery. The current article is designed to acquaint pathologists, toxicologists, and regulatory scientists with the basic principles needed to perform the teased fiber technique and interpret changes observed in the resulting isolated fibers during the course of animal toxicity studies.
Methods
The methodological details described in this article concentrate primarily on protocols relevant to nerve fiber teasing in animal toxicity studies. A cursory introduction is given to options for nerve fiber teasing in other biomedical science settings.
Sample Selection, Fixation, and Study Design in Different Settings for Nerve Fiber Teasing
The primary scenarios in which teased fiber preparations are produced on a regular basis are diagnostic neuropathology practice, independent research laboratories focused on fundamental neurobiological research, and animal toxicity studies. Protocols for nerve fiber teasing differ among these settings. Variations include the sample site, fixation technique, and use of additional diagnostic techniques.
Diagnostic Neuropathology Practice
Nerve fiber teasing in medical or veterinary diagnosis is done as a primary procedure to establish the category of neuropathy that exists in a patient. The sample is a surgically procured biopsy from a distal nerve (typically the sensory-only sural nerve in humans 10 but a mixed nerve in animals 11 ) that is immediately preserved by immersion in medical-grade glutaraldehyde (MGG; usually 2% to 2.5% in 0.1 M cacodylate or phosphate buffer, pH 7.4) for 2 to 4 hours at room temperature (RT). The fixed nerve is subsequently impregnated with osmium tetroxide (1% in 0.1 M buffer, pH 7.4) for 1 to 4 hours at RT to help stabilize myelin lipids, after which tissues are shifted to buffer. Whole mount immunohistochemical (IHC) labeling of cell type-specific markers using a chromogen to detect antibody binding is an alternative approach to visualizing fiber features. 12 Specimens then are allocated to nerve fiber teasing and/or processed routinely into hard plastic resin for ultrastructural evaluation.
For a given case, nerve fiber teasing typically is undertaken for a small portion (5-10 mm) of a much larger biopsy sample (5 cm). 13 The extended length of the entire biopsy is desirable since shorter specimens are less likely to produce a definitive diagnosis but will still result in a postoperative sensory deficit. Incubation in collagenase 14 (eg, 300 U/mL in physiological saline for 1 hour at 37°C) is helpful in digesting epineurial and perineurial connective tissue to facilitate extraction of single fibers following extended periods of fixation 15 (eg, for referral biopsies shipped through the mail). Evaluation of teased nerves usually is performed on 100 13 to 300 15 isolated fibers. In most cases, the prosector selects visibly affected fibers (ie, nonrandom sampling). Where necessary, comparisons to the expected features of normal fibers depend on reference to representative images in nerve pathology atlases (depicting lesions in human-derived samples) 16 –18 and, over time, the diagnostic neuropathologist’s own experience.
Independent Research Laboratory
Nerve fiber teasing in basic biomedical research is done as a primary procedure to evaluate fine cellular, functional, and/or molecular features of myelinated axons. The sample is taken from a deeply anesthesitized or freshly euthanized animal (usually a rodent for economic reasons). In most cases, the sciatic nerve is selected as the specimen since this mixed sensorimotor trunk contains the largest number of myelinated fibers in the body, but other sites may be selected if assessment of a sensory-predominant element is desired (eg, the plantar nerve in rodents). 19 Two basic procedures may be used to permit structural evaluations for isolated nerve fibers in this setting.
For the first technique, experiments to assess nerve fiber biology often use IHC endpoints to explore in situ molecular markers, so initial nerve preservation is undertaken by immersion in methanol-free formaldehyde (note 1) (MFF; 4% formaldehyde, pH 7.4) for up to 4 hours at RT or overnight at 4°C. 5 Alternatively, the entire animal may be perfused with fixative 8 to ensure that all portions of the central nervous system and peripheral nervous system (PNS) are well-fixed prior to tissue manipulation, thereby reducing the likelihood of dissection-related artifacts. 20 The fixed but unstained sample then is placed in a glass dish filled with phosphate-buffered saline (PBS; pH 7.4) and teased while being viewed under a stereomicroscope (Figure 1). 5 Isolated fibers are applied to an adhesive-coated glass slide, air-dried, and stored overnight at 4°C (to strengthen the degree of adhesion) before beginning the IHC procedure. In this manner, the isolated fibers finally are stained using antibodies linked to chromogens or fluorophores. 6

Step-by-step procedure for nerve fiber teasing. (A) Nerve portion (fixed but unstained) prior to removal of the epineurium (outer fibrous connective tissue covering). (B) Nerve following partial removal of the epineurium (arrow) to free the nerve fascicles (arrowhead). (C) Stereomicroscopic view showing a partially teased nerve piece. (D) Stereomicroscopic view demonstrating individual isolated nerve fibers oriented in parallel on a standard microscope slide. Specimen: rat sciatic nerve, prepared by overnight fixation in methanol-free formaldehyde at 4°C. (Image and legend adapted from Wen et al 5 by permission of the publisher.)
Second, experiments to assess nerve fiber structure often utilize samples that were stained prior to teasing. The chief options for this purpose are fat-soluble dyes (eg, Sudan black B, as detailed in section “Animal Toxicity Studies”) or metal impregnation (eg, osmium tetroxide, as described in section “Diagnostic Neuropathology Practice”). 8 Fibers stained in this manner have dark-colored, partially or completely opaque myelin sheaths, which masks the appearance of the centrally located axons. Such fibers are acceptable for qualitative evaluation and quantitative measurements but are not well suited for molecular investigations.
Nerve specimens for fundamental neurobiological research generally are 10 mm or greater in length. Teased fiber preparations for this purpose often employ several hundred fibers to maximize the value of quantitative measurements and their subsequent statistical analysis. 8,15 Fibers generally are chosen at random to avoid introducing systematic bias when assessing hypotheses.
Animal Toxicity Studies
Nerve fiber teasing for animal toxicity studies is undertaken as a follow-up technique to characterize the nature of nerve fiber changes first identified during evaluation of routinely processed nerve sections. Given the low throughput possible for the method, nerve fiber teasing generally is performed on a subset of animals (eg, 3-5) only from the high-dose group to find the target cell population affected in early stages of PNS neurotoxicity. The rationale for this design is that all toxicant-exposed animals will develop lesions having the same character, while all contol animals would be expected to have uniformly unaffected fibers. In general, approximately 100 to 200 teased fibers are evaluated per animal. Historically, teased fiber preparations were performed most often in safety testing to register new chemicals since this end point was required by some regulatory agencies (see Table 1); no similar regulatory guidance was promulgated for the development of biomedical products (drugs, biologics, cell and gene therapies, etc). Currently, nerve fiber teasing typically is undertaken infrequently, on an as-needed basis, in investigative toxicology programs as a means for clarifying the pathogenesis, progression, and potential prognosis of nerve lesions.
Regulatory Guidance for Nerve Fiber Teasing in Animal Toxicity Studies.a

aNerve fiber teasing is undertaken in animal toxicity studies only as an auxiliary procedure if lesions are observed during the initial histopathological evaluation of hematoxylin and eosin (H&E)-stained nerve sections.
bIn reorganizing the US Environmental Protection Agency (EPA) in 1998 to create the Office of Chemical Safety and Pollution Prevention (OCSPP), guidance offered in Title 40 was harmonized to create 6 US EPA “Series 870 – Harmonized Health Effects Test Guidelines – Group E: Neurotoxicity Test Guidelines” (https://www.epa.gov/test-guidelines-pesticides-and-toxic-substances/series-870-health-effects-test-guidelines), none of which now specifically mention nerve fiber teasing as a required end point.
Any nerve may be evaluated, but the usual practice in most animal studies is to limit the first evaluation to a large, mixed sensorimotor trunk such as the sciatic nerve or tibial nerve. Nerve fiber teasing may be done on another piece of the same sample that was assessed histopathologically, but teasing also may be directed to the contralateral nerve trunk (as both sides will have been collected and preserved in accordance with recently published “best practice” recommendations for PNS sampling 19 ). Nerve specimens from animals in animal toxicity studies often are quite large since long expanses may be harvested at necropsy.
Nerves in animal studies may have been preserved by either immersion fixaton or by whole-body perfusion fixation. 20 For immersion fixation (eg, for general toxicity studies where neurotoxicity is not anticipated), the standard fixative is neutral buffered 10% formalin (NBF, 4% formaldehyde containing methanol as a stabilizing agent). If perfusion fixation is elected (eg, for studies in which neurotoxicity is known or anticipated), common fixative options include MFF, MGG, or mixtures of MFF and MGG (eg, Karnovsky solution, 2% MFF and 2.5% MGG, or McDowell/Trump solution, 4% MFF and 1% MGG). Nerves slated for teased fiber preparations may have been stored in fresh fixative (NBF) or sometimes buffer (to avoid prolonged exposure to glutaraldehyde, which makes tissue brittle) for weeks to months before detection of nerve changes in routinely processed tissue sections warrants further evaluation of isolated nerve fibers. In general, nerve fiber teasing in the animal toxicity setting is not performed in conjunction with electron microscopy as delayed osmium tetroxide fixation and extended exposure to methanol (in NBF) frequently produce substantial artifacts in myelin sheaths that preclude ultrastructural analysis. However, when transmission electron microscopy is preplanned or optionally envisaged as a part of the analysis, separate samples (ie, contiguous portions of the same nerve) can be harvested from the same animals and processed adequately for both purposes.
Representative Procedure for Nerve Fiber Teasing in Animal Toxicity Studies
The steps described below detail a prototypic nerve fiber teasing method for neuropathology analysis as performed during animal toxicity studies. A nerve portion at least 10 mm (1 cm) in length is separated from a fixed nerve trunk. The piece to be teased is placed in a dish filled with PBS (pH 7.4, at RT). Under a stereomicroscope, any adhering fat and the outer connective tissue sheath (ie, epineurium) are removed using fine forceps (eg, Dumont #5 watchmaker’s forceps). The proximal end of the nerve is identified by application of a small ligature. If desired, the internal connective tissue (ie, perineurium and endoneurium) in the nerve segment may be disrupted using a proteolytic enzyme (eg, Pronase, 1 mg/mL in Tris buffer, pH 7.5 for 2-3minutes at RT). The specimen then is washed briefly in 70% ethanol and stained by immersion in Sudan Black B staining solution for 24 to 48 hours at RT. The staining solution is made by dissolving dye powder (0.1-1.0 g) in 100 mL of boiling solvent (eg, 70% ethanol, isopropyl alcohol, or propylene glycol) according to the manufacturer’s instructions (eg, Sigma-Aldrich cat no. 19,966-4, St. Louis, MO) or other recipe (eg, https://neuromuscular.wustl.edu/pathol/histol/sudan.pdf), with either centrifugation or filtration before use to remove undissolved precipitate. Stained nerves then are briefly rinsed in 70% ethanol before being either transfered to an adhesive-coated (note 2) slide for immediate teasing or moved to a glycerin-filled vial for storage until teasing on a later occasion.
For teasing, the stained nerve sample is applied to the adhesive-coated slide (positioned near the label end) in a drop of glycerin. Alternatives to glycerin include cedar oil and unpolymerized epoxy resin, 20 but these media are more expensive. Fine forceps are used to gently separate individual fibers from the perineurium (Figure 1). All fibers are oriented such that their proximal ends always occupy the same position relative to the label end of the slide. Since the aim of safety studies is to identify and characterize potential tissue responses that would be considered adverse if they were to develop in exposed individuals, the technician specifically selects visibly abnormal fibers for isolation (ie, nonrandom sampling). However, unaffected fibers can be assessed in passing to provide a sense of normal nerve fiber features (internodal segment length, myelin thickness, etc). The proportion of abnormal fibers may be estimated based on the total number of fibers available for evaluation.
When a sufficient number of fibers have been isolated to cover the slide, the slide is moved to an oven overnight for heat (note 3) treatment to more permanently bond the isolated fibers to it. Remaining nerve tissue is retained in glycerin until the teased fibers have been evaluated. When the preparation is satisfactory and the analysis has been completed, the remaining unteased nerve sample may be discarded. If needed to obtain a satisfctory preparation, the stained nerve remnant may be teased again to isolate more nerve fibers.
Results
Isolated nerve fibers typically are assessed qualitatively under a light microscope, but more detailed analysis (sometimes including quantitative measurements) often is performed using reconstructions of the whole lengths of available fibers, including several internodal segments (where an internode is the portion of a myelinated nerve fiber between 2 nodes of Ranvier). Until recently, such reconstructions were produced by tediously assembling serial black-and-white photomicrographs into composite images (Figure 2). Recent instrumentation and software advances in whole slide scanning and digital imaging now permit automated and rapid collation. Common changes seen in teased nerved fibers are shown in Table 2 and illustrated in Figures 3 and 4.

Normal nerve fiber structure. Characteristic features are uniformly thick myelin (dark sheaths); pale nodes (of Ranvier) separating adjacent myelin segments (blue vertical arrowheads); myelin (Schmidt-Lanterman) incisures (at perpendicular dashed lines); and Schwann cell nuclei (red angled arrowheads) at the mid-internodal position on the periphery of nerve fibers. Specimen: sciatic nerve of adult rat. Preparation: immersion fixation in hypotonic 3.7% formalin, Sudan black B stain. (Image reproduced and legend adapted from Krinke et al 4 by permission of the publisher.)
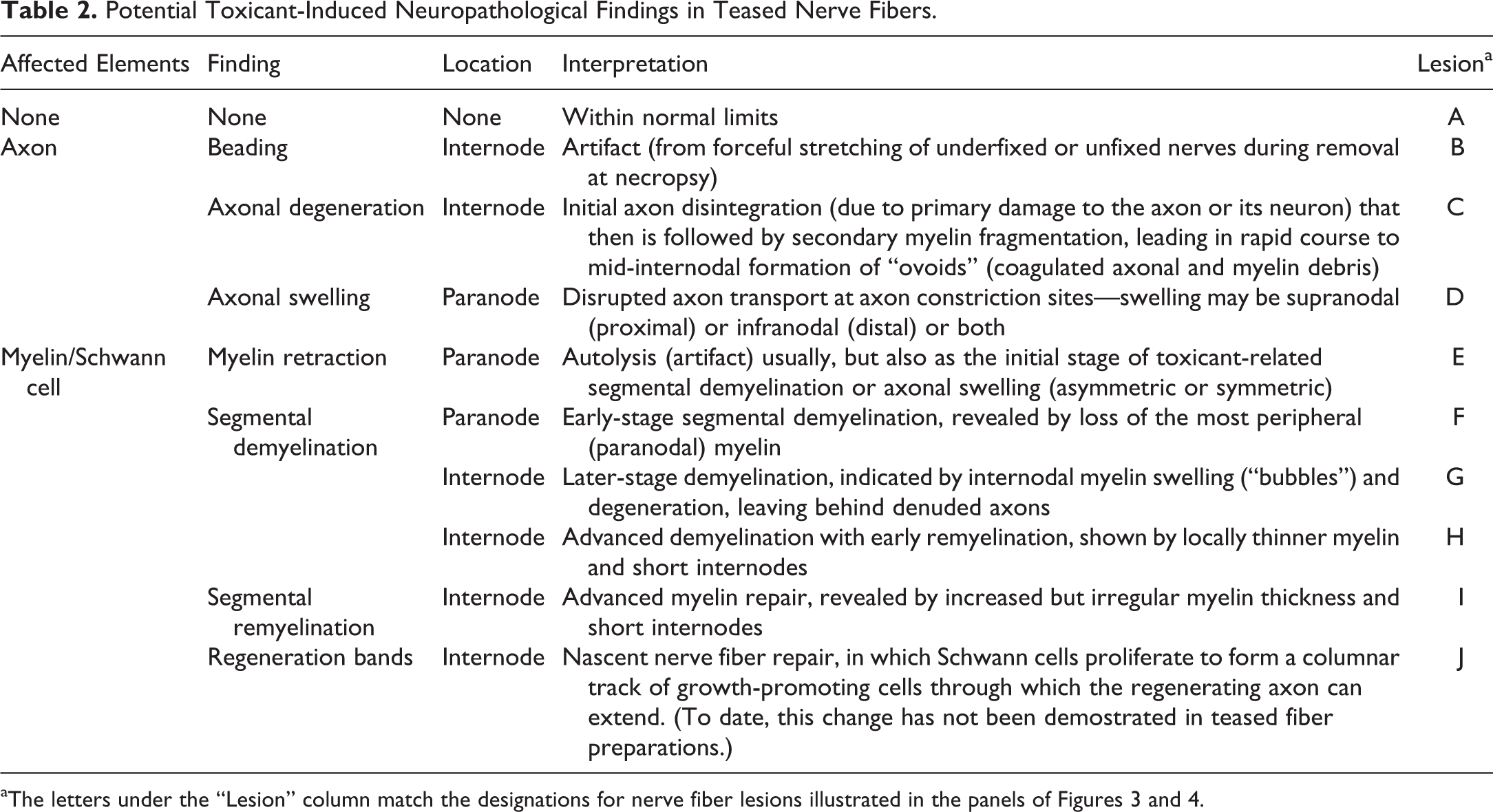
Potential Toxicant-Induced Neuropathological Findings in Teased Nerve Fibers.
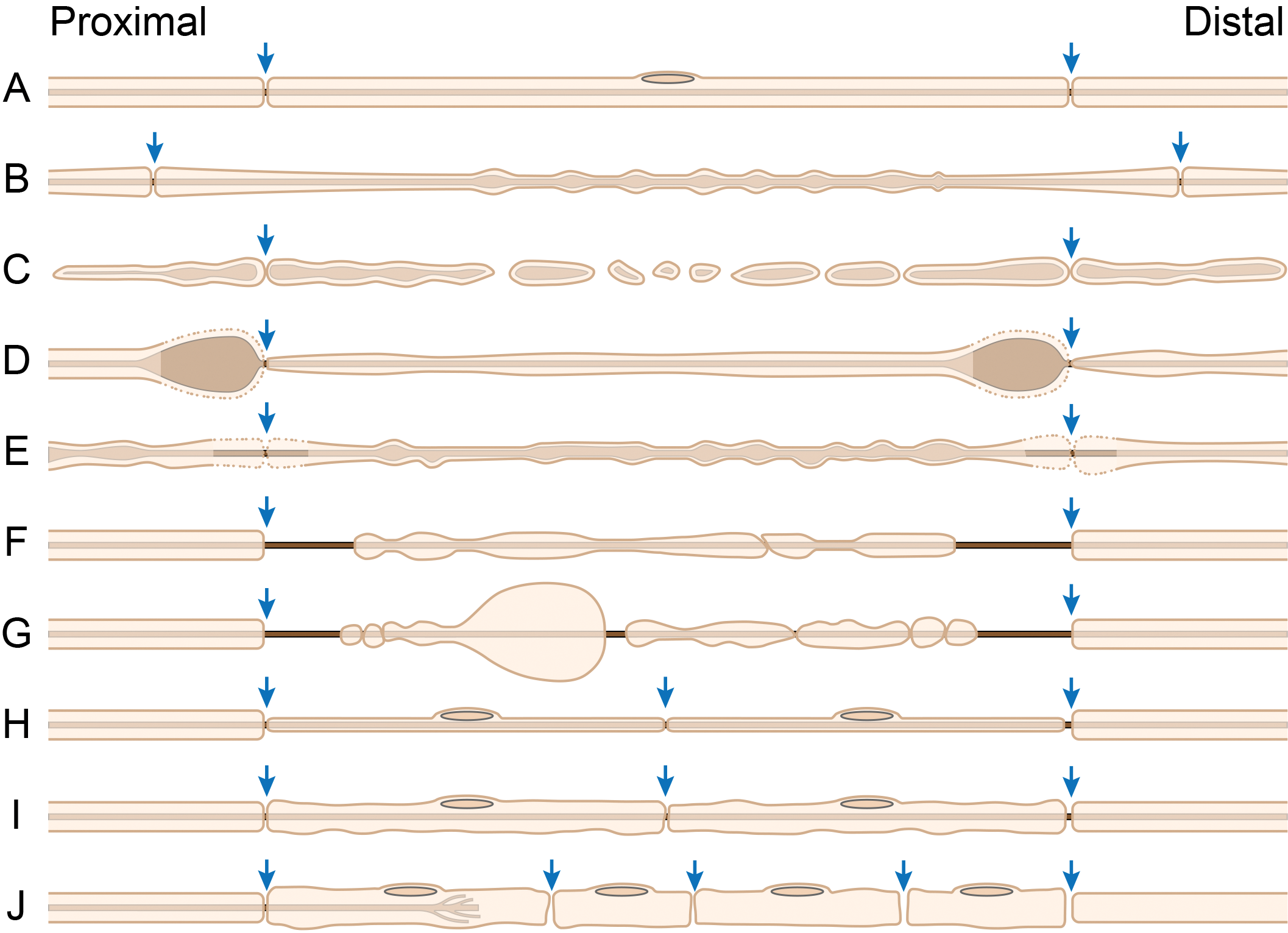
Conceptual diagram of normal and neuropathological features of axons (central dark line), myelin (outer pale tan sheath), and nodes of Ranvier (arrows) in myelinated nerve fibers as they would appear in teased fiber preparations. A, Normal fiber, characterized by long internodes with uniform myelin thickness. A Schwann cell nucleus (pale brown oval on the top margin of the fiber) is depicted in its usual mid-internodal position. B, “Beading” artifact, a consequence of excessively forceful manipulation of an under-fixed fiber during teasing (or the entire nerve at the time of necropsy). C, Axonal degeneration (“Wallerian” type), an early lesion following physical transection in which both the axon and myelin begin disintegrating in the mid-internodal region. D, Axonal swelling, an early indicator of disrupted axon transport (typically due to a molecular logjam caused by chemically induced covalent cross-linking of cytoskeletal proteins). E, Myelin retraction (“naked nodes,” indicated by dotted membranes and pallor of the paranodal myelin), a common artifact due to delayed fixation (ie, autolysis) but which also may be seen as the initial stage of toxicant-related segmental demyelination or with giant axonal swelling. F, Early segmental demyelination, resulting in loss of myelin near paranodes (which are the myelin zones at the farthest distance from the essential perinuclear metabolic machinery of the Schwann cell). G, Later-stage segmental demyelination, leading to myelin sheath swelling (“myelin bubbles”) and fragmentation of internodal myelin in the absence of axon disruption. H, Advanced segmental demyelination with remyelination, characterized by myelin thinning and short internodes. I, Segmental remyelination, shown by restoration of increased but irregular myelin thickness and short internodes. J, Regeneration bands (of Büngner), seen as short columns of plump (activated) Schwann cells forming a conduit through which regenerating axons may grow. So far, the bands of Büngner have been demonstrated in cross and longitudinal histological sections, but not in teased fiber preparations, likely because anaxonal (axon-lacking) Schwann cells do not adhere firmly enough to each other to withstand teasing. The letter identifications for each nerve fiber diagram matches the letter designations given in the “Lesion” column of Table 2. (Image adapted in large part from Schaeppi and Krinke 21 by permission of the Publisher. The diagram was crafted by Mr Tim Vojt, Columbus, OH.)
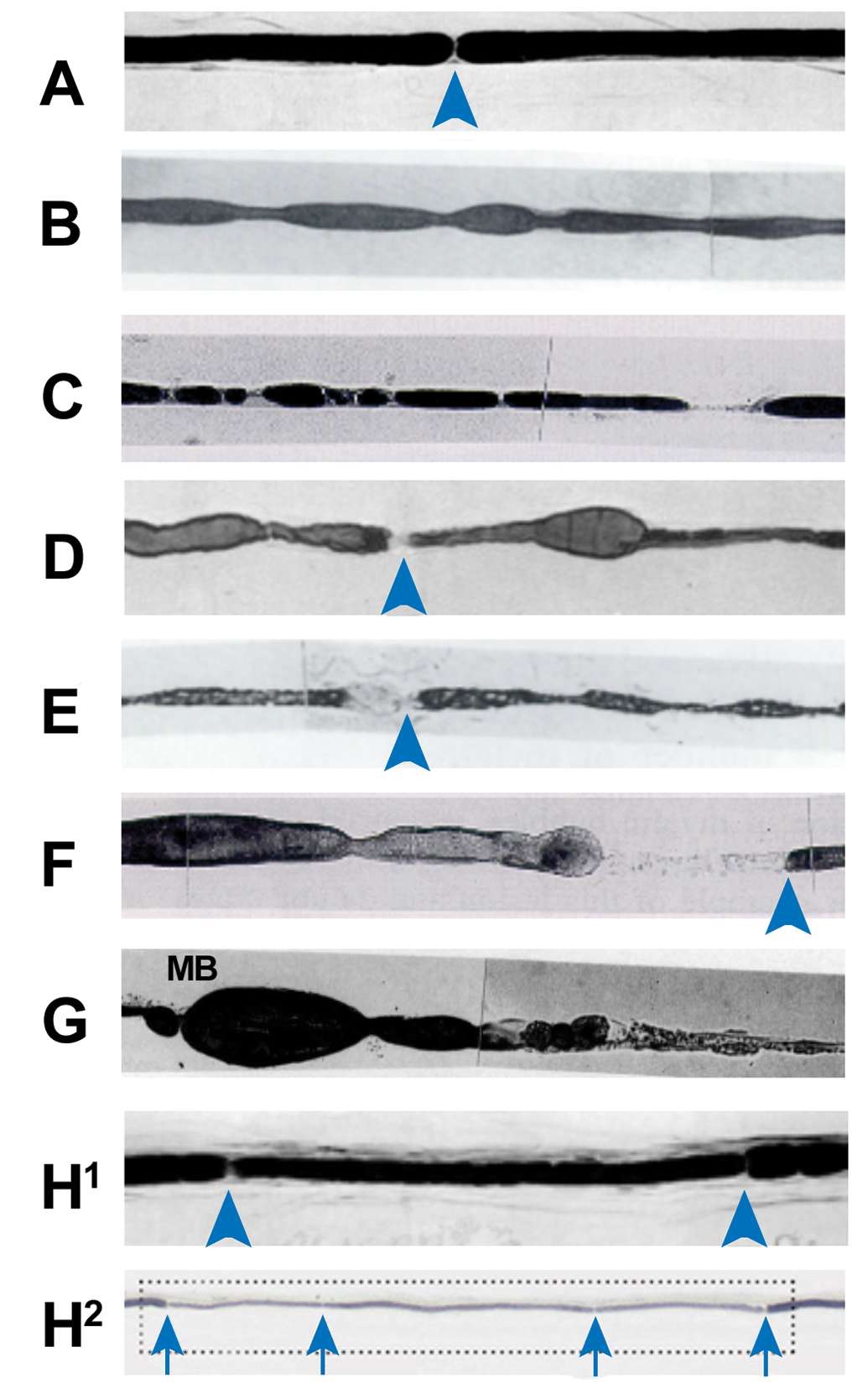
Normal and neuropathological features of stained single myelinated nerve fibers in teased preparations (where the letter identification for each image matches the letter designations given in the “Lesion” column of Table 2). A, Normal fiber, characterized by uniform myelin thickness separated by narrow nodes of Ranvier (arrowhead). (Image reprinted from Braund et al 22 by permission of the Publisher.) B, “Beading” artifact. C, Axonal degeneration (“Wallerian”) in a rat tibial nerve caused by sciatic nerve transaction 36 hours previously. D, Axonal swelling in a rat tibial nerve due to hexane-induced distal polyneuropathy. (Image reprinted from Spencer and Schaumburg 23 by permission of the author and the publisher; the original view has been adjusted minimally by adding an arrowhead to denote the node of Ranvier.) E, Myelin retraction of myelin abutting the node of Ranvier (arrowhead) due to autolysis. F, Early segmental demyelination as an age-related spontaneous lesion in a lumbar ventral spinal nerve root, leading to loss of myelin adjacent to the node of Ranvier (arrowhead). G, Later-stage segmental demyelination (a spontaneous age-related change in rats) in a lumbar ventral nerve root, characterized by myelin sheath swelling (“myelin bubbles” [MB]), fragmentation of internodal myelin (middle), and finally complete myelin loss leaving a naked axon (right). H1, Advanced segmental demyelination with remyelination, here in an intercalated internode from a dog with paraneoplastic polyneuropathy, features thinner myelin relative to adjacent normal myelin segments. Arrowheads indicates nodes of Ranvier. (Image reprinted from Braund et al 22 by permission of the Publisher.) H2, Advanced segmental demyelination with remyelination (inside the dotted box) in a human patient with sensory polyneuropathy showing thin myelin with several short internodes of various lengths. Arrows indicates nodes of Ranvier. (Image reproduced from Therapath Neuropathology, New York, NY; https://www.therapath.com/services/muscle-and-nerve-pathology/nerve-biopsy-education/) In the absence of other image credits, these photomicrographs have been reproduced from Krinke et al 4 by permission of the publisher.
Normal Anatomy of Teased Nerve Fibers
Normal myelinated nerve fibers exhibit a number of characteristic architectural features (Figures 2, 3A, and 4A). Four visible attributes of isolated fibers are: Myelin = the uniformly thick, darkly stained sheath, dervied from Schwann cells (ie, PNS glia), that encompasses the centrally located axon; Myelin (Schmidt-Lanterman) incisures = herringbone-angled, pale lines that span the myelin sheath, more frequently in the middle (ie, midinternodal region) than near the ends (ie, paranodal regions), as a result of cytoplasmic accumulation during the formation of concentric myelin lamellae; Nodes (of Ranvier) = the narrow, clear junctions that separate the paranodal boundaries of adjacent myelin segments (internodes); and Nuclei (of Schwann cells) = eccentric, fusiform, membrane-bound, pale bodies located near the margin at approximately the middle of a myelin segment.
Axons typically are invisible in stained normal myelinated nerve fibers.
The appearance of nerve fibers is influenced by their location and function. Axons of larger caliber (or diameter) traditionally have been thought to possess thicker myelin sheaths and have lengthier internodes. 24 However, internode length varies by location even for axons of similar caliber. 25 -27 Myelin quantity is dictated by internode length, with relatively long internodes possessing slightly thicker sheaths compared to shorter internodes. 28 Longer internodes also have more myelin incisures. 29 Myelin sheath thickness during growth of a nerve is maintained not by increases in Schwann cell number but rather by elongation of internodes over time. 30 The total amount of myelin that must be maintained for spinal nerve roots and proximal nerve trunks appears to be greater than is supported in more distal nerve branches, 28 thereby rendering proximal PNS components more vulnerable to many demyelinating influences. For most nerve fibers, internode length and caliber is relatively uniform over most of the fiber’s length, but occasional fibers from adult control animals in animal toxicity studies have short, thinly myelinated segments of small caliber (Figures 3H and 4H)—known as intercalated internodes—as a result of spontaneous, age-related segmental demyelination and remyelination. 7 To our knowledge, no systematic catalog of nerve fiber structural variations by strain, age, or sex has been compiled for any animal species.
Artifacts in Teased Nerve Fibers
Breakage is the most common artifact related to nerve fiber teasing. Loss of continuity results from fiber trauma during the teasing procedure.
Beading (Figures 3B and 4B) appears as a series of regularly sized and spaced, bulbous ovals dispersed along an intact nerve fiber. This artifact occurs following forceful stretching of unfixed or under-fixed nerve, which typically occurs during dissection rather than teasing. 31 Beading is most obvious in large-caliber fibers but also can occur in unmyelinated fibers, and it is associated with distortion of the axon rather than the myelin sheath. 31 Other stimuli also can elicit this change, so if nerve collection was gentle and fixation was optimal beading cannot be dismissed automatically as an artifact rather than a potential test article-related effect.
Paranodal myelin retraction (Figures 3E and 4E) typically is an artifact related to autolysis. This finding appears as myelin pallor in paranodal regions and represents the onset of demyelination. The reason for this distribution is that paranodal myelin is located the farthest from the perinuclear metabolic machinery of the Schwann cell that contributes to myelin maintenance. Adjacent myelin segments may be affected asymmetrically, such that one paranodal region is pale for an extended distance while that of the adjacent segment is pale only in the immediate vicinity of the node. 4 However, paranodal myelin pallor is also a consequence of genuine toxicant-related demyelination as well as giant axonal swelling, so the relevance of this finding must be assessed carefully in the context of an experiment.
Neuropathologic Lesions in Teased Nerve Fibers
Axonal degeneration
Axonal degeneration manifests first in distal portions of the nerve fiber as rows of ovoids dispersed along an intermittently disrupted fiber (Figures 3C and 4C). Axonal destruction stems from a primary lesion affecting either the axon or its supporting neuron or both. 4 In the vast majority of neurotoxic events (>90%), the nerve fiber disintegration caused by initial axonal degeneration is magnified by secondary myelin degeneration as Schwann cells switch from myelin maintenance to phagocytosis in order to remove the fragmenting nerve fiber. 32 -34 Hematogenous macrophages, however, substantially contribute to removal of myelin debris. 35 This process is called “Wallerian degeneration” if axons are disrupted completely by severe mechanical trauma (nerve crushing or transection) but is termed “Wallerian-like degeneration” if axonal damage results not from physical interruption but rather from derangement of cytoskeletal transport machinery or other cellular organelles by other causes than trauma, including toxicity. Axonal degeneration starts near the site of the injury and proceeds distally 36 even though microtubule dissolution appears to develop simultaneously along the full length of the injured stump. 37
Axonal degeneration appears first as the formation of ovoids in the middle of internodes (ie, near the Schwann cell nucleus; Figures 3C and 4C). Each ovoid represents a composite of coagulated axon fragments and myelin debris. Ovoids are usually delineated by myelin incisures, which become more prominent but do not increase in number. 29 Nerve fibers undergoing repair form regeneration bands (known as bands of Büngner) comprised of rows of Schwann cell nuclei (Figure 3J). Regeneration bands serve as conduits through which regenerating axons may re-grow. The leading edge of the regenerating axon forms approximately 5 to 10 sprouts, of which only 1 will eventually survive to restore axonal connectivity with its target.
The evolution of Wallerian degeneration varies with the nature of the affected nerve fibers and the location of the trauma. Nerve fiber dissolution develops in large-caliber myelinated fibers (diameter, 8.5-11 µm) over approximately 45 hours but manifests in small-caliber myelinated fibers (diameter, 2.5-4 µm) in about 26 hours. 36 Degeneration appears earlier and is more severe when damage occurs in proximal trunks (ie, closer to the location of the nerve cell bodies from which the axons originate). 38 Injury to the very proximal portions of axons (ie, near the axon hillock) tends to produce damage to the nerve cell body followed by permanent loss of the nerve fiber. 39
Axonal swelling
Axonal swelling usually presents as asymmetric paranodal swelling of axons (Figures 3D and 4D). In isolated nerve fibers, expansion of the axon affects the paranodal region proximal (ie, supranodal) to nodes of Ranvier while sparing the distal paranodal (or infranodal) region. These swellings result from accumulation of cytoskeletal elements and organelles that, in toxicant-induced cases, have been shown to be caused by chemically induced cross-linking of proteins. The nerve fiber distal to axonal swelling may undergo Wallerian-like degeneration.
Segmental demyelination
Segmental demyelination results from primary damage to the myelin and/or Schwann cell. The segmental nature likely reflects age-related and/or site-specific differences in vulnerability of Schwann cells. This lesion appears first as multifocal loss of paranodal myelin (Figures 3F and 4F) because the myelin at this location is located the farthest from its source of metabolic support near the center (ie, perinuclear region) of the Schwann cell. Later-stage segmental demyelination leads to myelin sheath swelling (“bubbles”) with lengths of myelin-denuded axon (Figures 3G and 4G). Because the axons remain viable, zones in the throes of advanced segmental demyelination are remyelinated. At first, the restored myelin has shorter internodes (because the tally of myelin segments necessary to cover each denuded axon section is increased relative to the original number) and initially is thin (Figures 3H, 4H1, and 4H2). With time, however, advanced segmental remyelination will yield myelin sheaths of approximately normal or only slightly reduced but generally irregular thickness, with permanently shortened internode lengths (Figure 3I); thus, myelin “repair” does not completely reconstitute the original myelin structure and therefore is likely to yield some degree (however minimal) of functional impairment. Unlike axonal degeneration, which occurs earlier and more severely in distal portions of nerve fibers, segmental demyelination tends to impact proximal nerves (especially spinal nerve roots) since the quantity of myelin to be sustained is greatest in this region. 40
Conclusion
The ability to more easily differentiate early axonal damage from early myelin injury by teased fiber analysis has accorded nerve fiber teasing a place as a suggested method for assessing PNS toxicity in the regulatory setting for certain animal toxicity studies (Table 1). The choice regarding whether or not to initiate nerve fiber teasing for a given study must weigh the advantages and disadvantages of examining isolated nerve fibers vs. routine histopathologic evaluation of intact nerves. The key advantage of teased fiber preparations is that they permit the examination of axon and myelin continuity over extended distances (10 mm or more), which is not possible in routine tissue sections since individual fibers travel in and out of the plane displayed in longitudinal nerve sections. For this reason, teased nerve fibers generally are the best approach for determining which PNS elements (axon vs myelin) represent the target of a neurotoxic agent. Scientific disadvantages of the teased fiber technique are that the method is not useful for all categories of nerve fibers—in general, teasing is easiest for large-diameter myelinated fibers, difficult for small-diameter myelinated fibers, and not feasible at all for unmyelinated fibers—and that the data set may be biased by the technician’s choice of which fibers will be isolated. However, the main disadvantage of nerve fiber teasing is its low throughput (due to the slow, labor-intensive teasing process) relative to conventional histologic preparation and routine microscopic examination. 19 In the final analysis, nerve fiber teasing is a vital tool for characterizing the early evolution of nerve fiber lesions responsible for toxicant-induced peripheral neuropathies.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
