Abstract
Xenobiotic-induced peripheral nerve damage is a growing concern. Identifying relative risks that a new drug may cause peripheral nerve injury over long periods of administration is gathering importance in the evaluation of animal models. Separating out age-related changes in peripheral nerves of rats caused by compression injury from drug-induced effects has been difficult. Biopsy of the sural nerve is utilized in humans for investigations of peripheral neuropathy, because it is largely removed from the effects of nerve compression. This study used transmission electron microscopy to identify incidental findings in the sural nerves and dorsal root ganglia of aged control rats over time. The goal was to establish a baseline understanding of the range of possible changes that could be noted in controls compared to rats treated with any new investigative drug. In this evaluation, most sural nerve fibers from aged control rats had few ultrastructural abnormalities of pathologic significance. However, glycogenosomes, polyglucosan bodies, swollen mitochondria, autolysosomes, split myelin, Schwann cell processes, and endoneural macrophages with phagocytosed debris (considered an indication of ongoing degenerative changes) were occasionally noted.
Keywords
Introduction
Standardized evaluation of the peripheral nervous system (PNS) during general nonclinical toxicity studies has gained more attention of late with the publication of recommended best practices for PNS assessment. 1 The importance of standardization for PNS toxicity evaluation is driven by the expanding list of xenobiotics that are known or suspected of inducing PNS toxicity. In addition, the clinical use of nerve biopsies of nerves primarily composed of sensory fibers, such as the sural nerve, is sometimes enlisted in order to determine the cause of peripheral neuropathy of unknown etiology. 2,3 Sensory-predominant nerve fibers are used in screening because they are long and thus more vulnerable, and the usual pattern of xenobiotic-induced polyneuropathy occurs first as sensory dysfunction. Finally, in our experience, the use of transmission electron microscopy (TEM) for the assessment of the PNS in nonclinical toxicity studies is a consideration for therapeutic candidates who have a known potential to induce PNS toxicity.
Numerous medications can cause, or are suspected to cause, damage to the PNS. These include medications used to treat heart arrhythmias, 4 bacterial infections, 5,6 retroviral infections, 7 –11 cancer, 12 –14 high blood pressure, 15 autoimmune disorders, 16 epilepsy, 17 high cholesterol, 18,19 alcohol addiction, 20 and vitamin over-/undersupplementation. 21,22 As would be expected, there is no commonality between the structure and/or the mechanism of action between these varied therapeutic agents.
Standard histopathologic evaluation of the PNS in nonclinical toxicity studies typically involves the light microscopic evaluation of a single peripheral nerve, generally the sciatic nerve (a mixed motor/sensory type nerve). If peripheral nerve toxicity is suspected based on the results of short-term nonclinical toxicity studies or known drug class effects, recent best practice recommendations suggest expanding the types of microscopic techniques used to evaluate various components of the PNS, including incorporating TEM to evaluate the potential chemical or drug-induced ultrastructural changes. 1 To date, few ultrastructural studies have reported drug-induced changes in the sural nerve of rats administered xenobiotics over the course of long-term nonclinical toxicity or carcinogenicity studies or their comparison to nerves from age- and sex-matched controls. The purpose of this article is to report the ultrastructural findings observed in the sural nerves of control rats from a 2-year carcinogenicity study. These findings could help separate normal age-related nerve changes from those induced by xenobiotics.
Materials and Methods
Animal Handling and Treatment
This animal study was conducted at Covance Laboratories, Inc, Madison, Wisconsin. Covance Laboratories is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. All procedures in the protocol were in compliance with applicable Animal Welfare Acts and were approved by the local Institutional Animal Care and Use Committee. Male and female Crl: CD(SD) rats were received from Charles River Laboratories, Raleigh, North Carolina. Animals were acclimated to the test facility for 1 week prior to initiation. At initiation of dosing, animals were 6 to 7 weeks old, and body weights ranged from 137 to 243 g for males and 139 to 218 g for females. Animals were group-housed (up to 3 animals/sex/cage) in polycarbonate cages with hardwood chip bedding. Reverse osmosis-purified water was provided ad libitum. Animals were offered Certified Rodent Diet #2014C (Harlan Laboratories, Inc., Madison, WI) ad libitum, unless fasted for study procedures. Some animals were fed the crushed meal form of Certified Rodent Diet #2014C (Harlan Laboratories, Inc). Environmental controls were set to maintain a temperature range of 20°C to 26°C, a relative humidity range of 30% to 70%, 10 or greater air changes/hour, and a 12-hour light/12-hour dark cycle. The light–dark cycle was interrupted for study-related activities. Animals were given various cage-enrichment devices and dietary enrichment. After an overnight fast, animals were anesthetized with sodium pentobarbital, exsanguinated, and necropsied.
Transmission Electron Microscopy Procedures
Sections of sural nerve (left longitudinal and transverse sections) and dorsal root ganglia (DRG; lumbar—left L4-L6) were collected from 5 male and 5 female rats that were administered 0.9% sodium chloride for injection, USP (sterile saline) once weekly via subcutaneous injection for 82 to 88 weeks. Sural nerves were collected rapidly and sliced into approximately 1-mm cubes and immersed in ½ strength Karnovsky fixative, in 0.1 M phosphate buffer, and held overnight in a refrigerator set to maintain 2°C to 8°C. Tissues were postfixed in 1.0% osmium tetroxide solution for 90 minutes at ambient temperature, dehydrated stepwise, and subsequently embedded in epoxy resin. Up to 6 blocks were prepared from each tissue/animal, sectioned at 1 micron, stained with toluidine blue, and examined by light microscopy to identify representative areas of interest. Based on review of the toluidine blue sections, a minimum of 1 block from each tissue was trimmed for ultrathin sections (approximately 50-70 nm thicknesses), placed onto coated grids, and stained with uranyl acetate followed by Reynold’s lead citrate. Image capture was conducted on a Philips CM 120 STEM (FEI, Hillsboro, Oregon) equipped with a BioSprint side-mounted camera with an AMT Image Capture Engine using version 7.0.0.130 software (Advanced Microscopy Techniques, Corp, Woburn, Massachusetts). All study procedures (including TEM) were Good Laboratory Practice compliant.
Results
Light Microscopy of Sural Nerve
No significant histopathologic findings were noted based on histopathologic evaluation of hematoxylin and eosin–stained sections of sural nerve, sciatic nerve, or dorsal root ganglion from control aged rats. Toluidine blue–stained thin sections of sural nerve exhibited a few microscopic abnormalities. These sural nerve findings included infoldings of myelin or intra-axonal loops of myelin, occasional intra-axonal circular bodies that appeared to disrupt the axoplasm, and regions of myelin separation with/without the appearance of debris within variably sized spaces. There were no histopathologic changes noted in toluidine blue–stained thin sections of dorsal root ganglion.
Myelinated Sural Nerve Fibers: TEM
Normally, myelinated sural nerve axons are wrapped by Schwann cell–derived myelin sheaths composed of tightly packed, concentric layers of plasma membrane, and normal Schwann cells have a distinct basal lamina (see Figure 1). In control rats, age-related changes noted in Schwann cells included Reich (Pi) granules, Elzholz bodies (or Mu granules), and lysosomes containing phagocytized myelin debris. Reich (Pi) granules were characterized by intracytoplasmic electron-dense elongated, polygonal, or spicule-shaped paracrystalline inclusions (see Figure 2A and B). Elzholz Bodies (Mu Granules), which have been described in detail elsewhere, 23 were evident as intracytoplasmic bodies that had the appearance of phagocytosed myelin or a section of myelin just out of the plane of section with the same periodicity of the associated axon (see Figure 3). Finally, lysosomes containing phagocytosed myelin debris and/or lipidic inclusions, often with complete loss of myelin periodicity (see Figure 4), were noted in Schwann cells and interpreted to likely represent an age-related degenerative change as well.
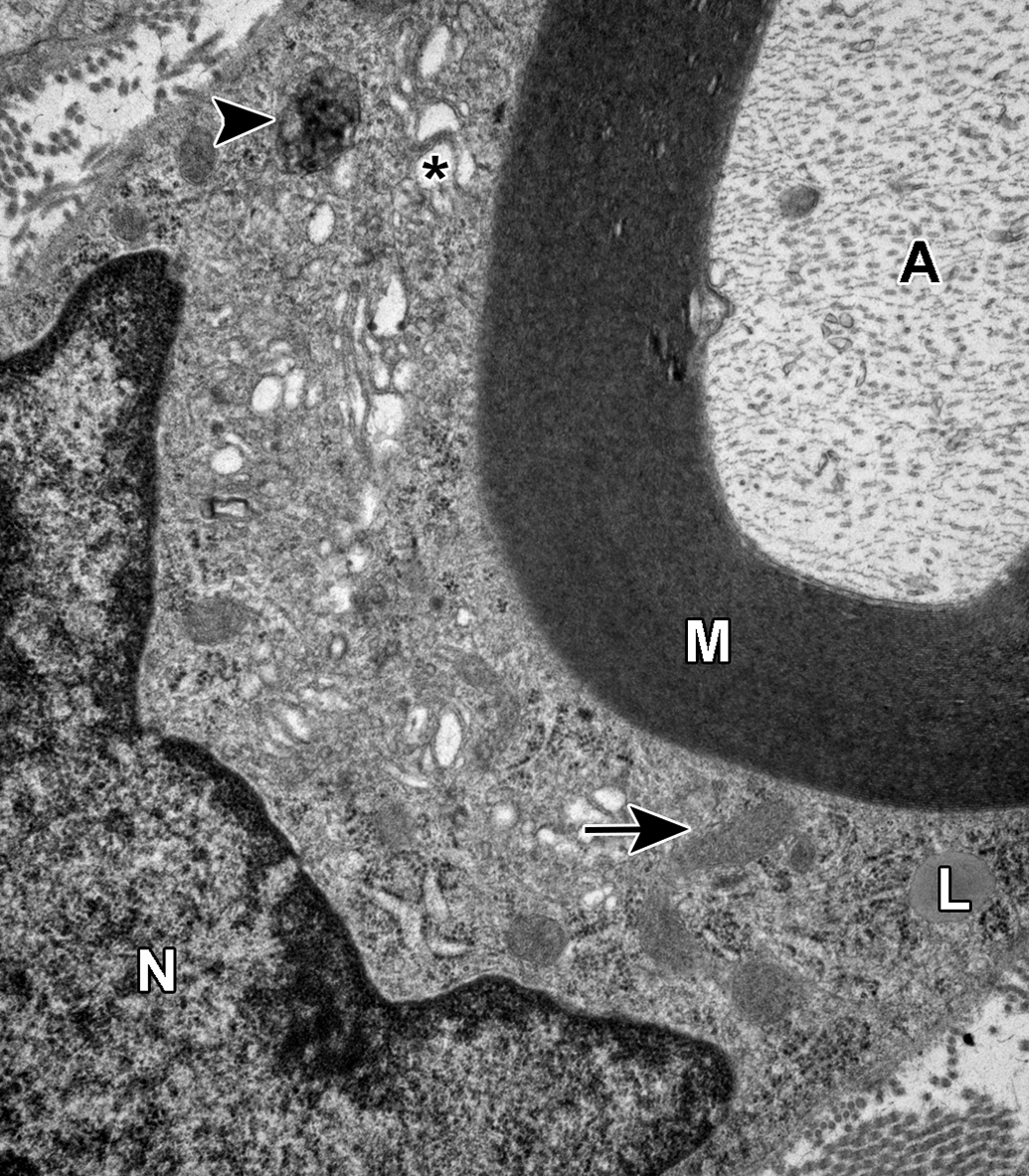
Normal myelinated sural nerve axon and associated Schwann cell. The Schwann cell contains a nucleus (N), cytoplasm containing numerous mitochondria (arrow), a single small lipid droplet (L), and concentric layers of plasma membrane forming the electron-dense myelin sheath (M) investing the axon (A). The Schwann cell endoplasmic reticulum is mildly dilated (*), and a lysosome contains electron-dense material (arrowhead).
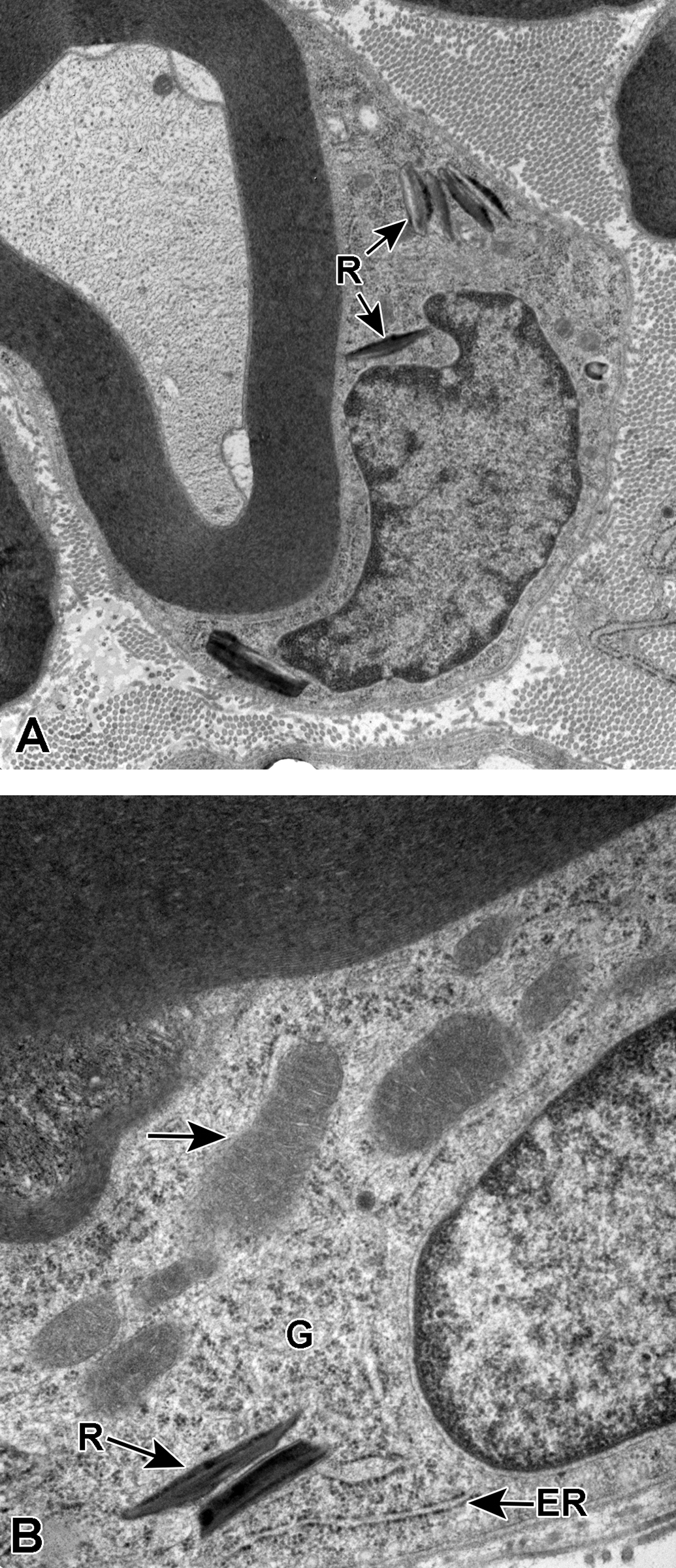
A and B, Myelinated sural nerve axons and associated Schwann cells. In A and B, the Schwann cell cytoplasm contains several Reich (Pi) granules (R). In B, several mitochondria (arrow), glycogen rosettes (G), and endoplasmic reticulum (ER) studded with ribosomes can be seen within the cytoplasm.
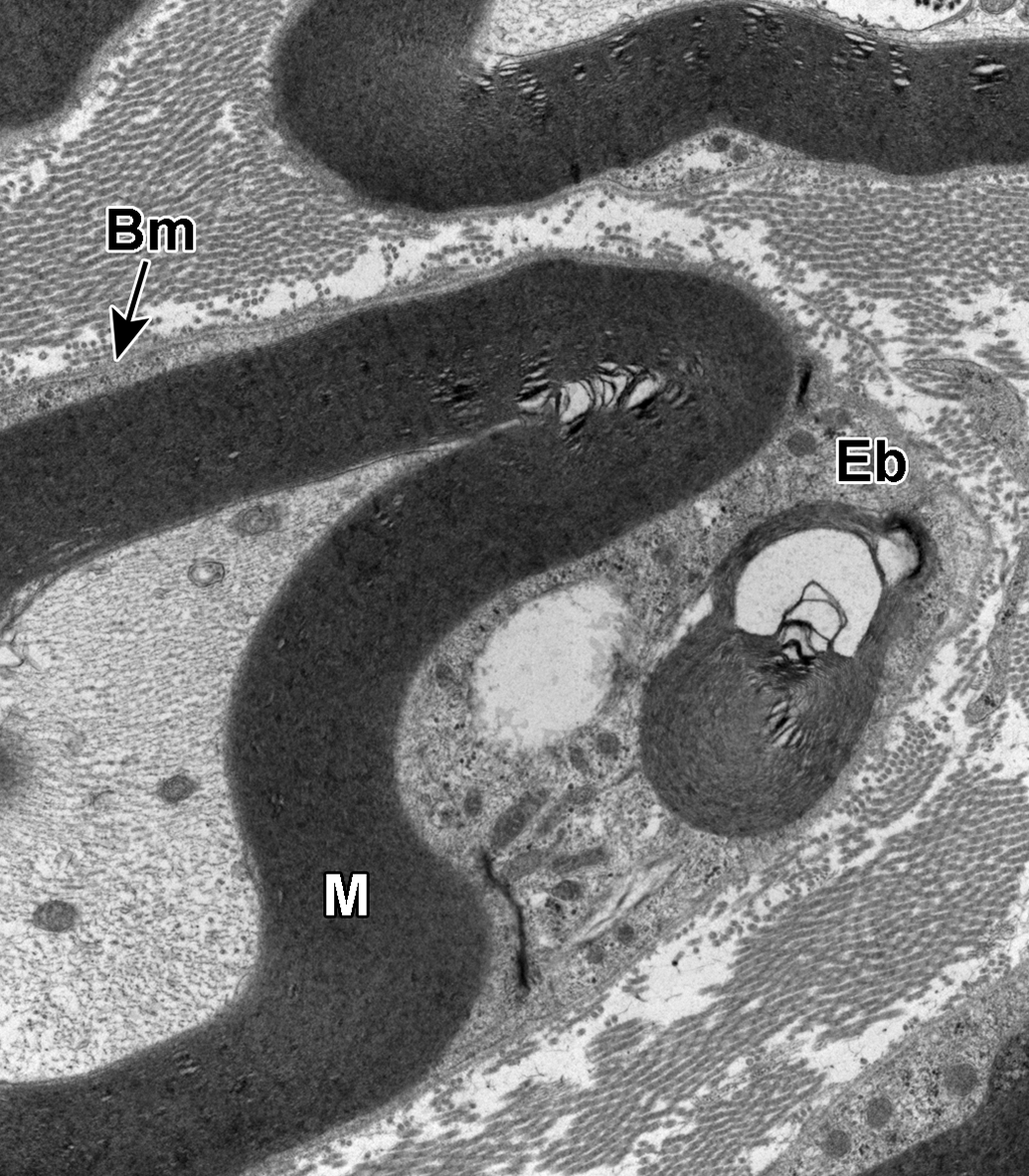
Myelinated sural nerve axon and associated Schwann cell. The Schwann cell contains an intracytoplasmic electron-dense body composed of concentric layers of osmiophilic material with the same periodicity as the adjacent myelin sheath (M), consistent with an Elzholz body (Eb). The distinct basement membrane (Bm) of the Schwann cell is noted separating the cell from the adjacent endoneurium containing transverse sections of collagen fibrils.
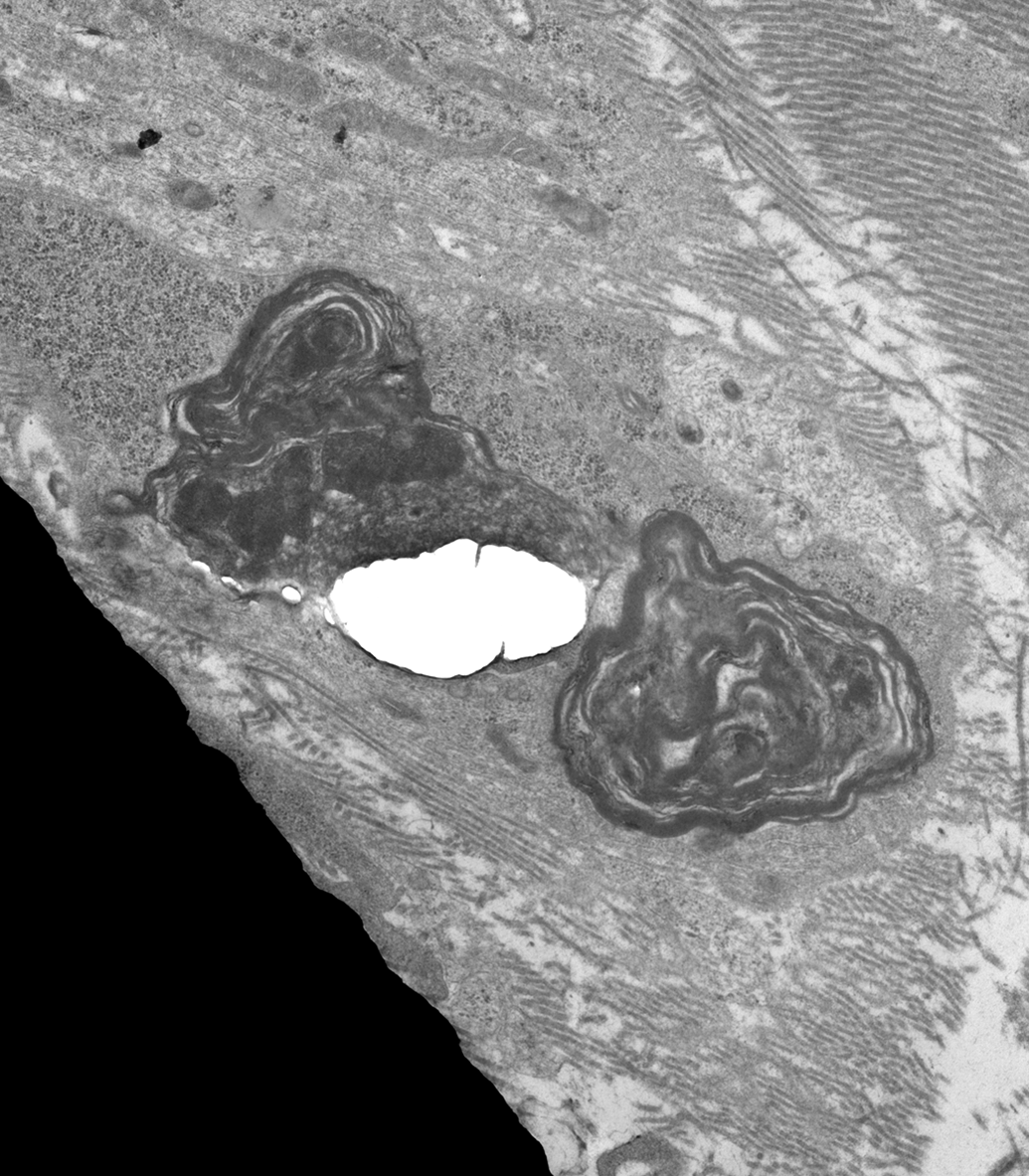
Schwann cell containing phagocytosed debris. The Schwann cell cytoplasm contains ovoid, membrane-bound bodies of variable electron density composed of myelin debris that lacks a regular periodicity. The membrane-bound bodies are consistent with phagolysosomes. Longitudinal sections of collagen fibrils are present in the adjacent endoneurium. The electron-dense region in the lower left is a section of grid bar.
In transverse or longitudinal sections of myelinated axons from aged control rats examined at low magnification (see Figure 5), many of the fibers exhibited irregular infoldings or projections of myelin sheath into the axoplasm. These myelin infoldings were observed in both large- and small-caliber myelinated axons. A few axons with regions of split myelin and/or contracted axoplasm were considered to be consistent with a degenerative change. Normal-appearing nodes of Ranvier (ie, the narrow spaces between 2 adjacent myelinating Schwann cells) were occasionally evident with the nodal gaps covered by projections or microvilli of adjacent Schwann cells (see Figure 6).
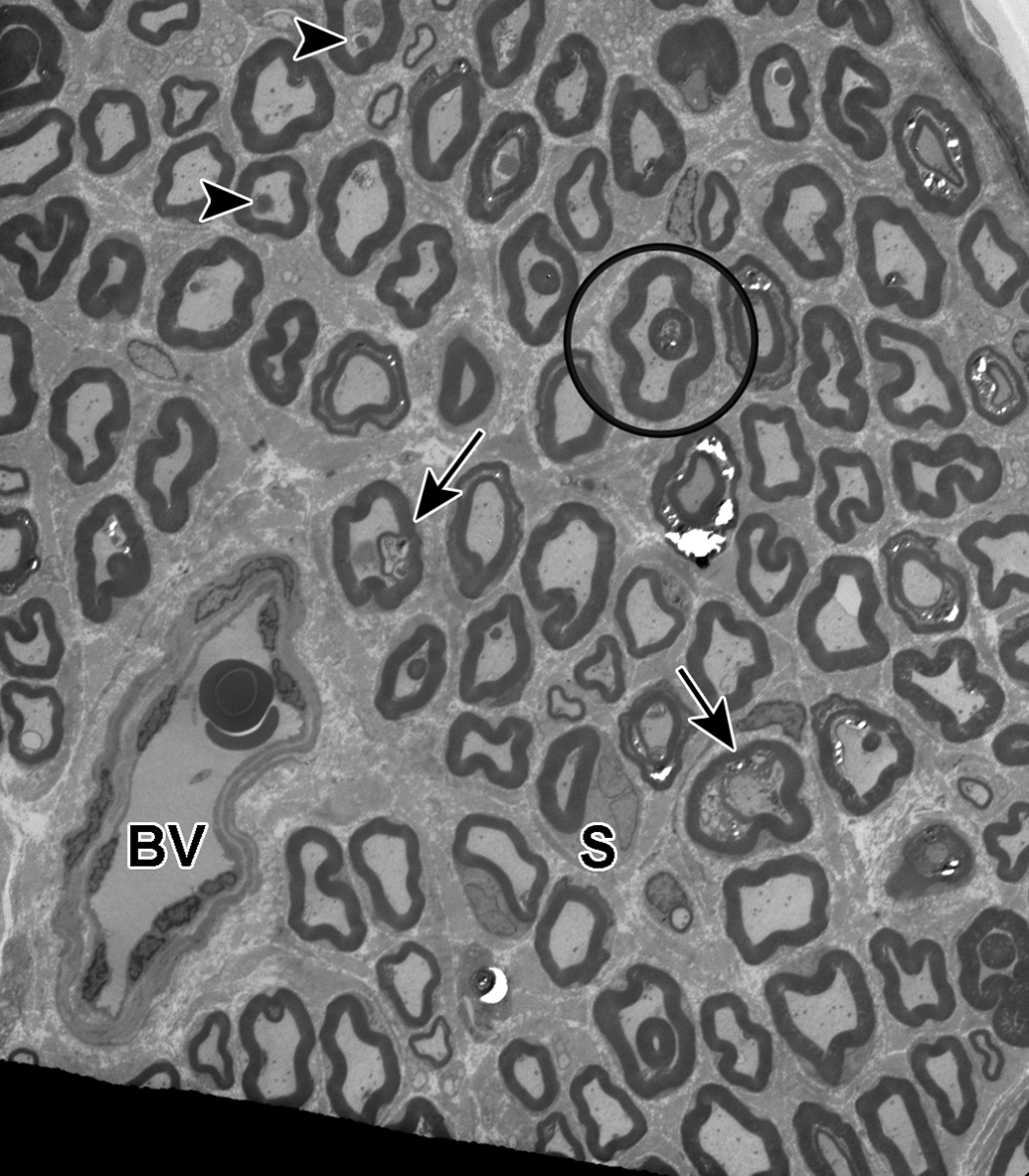
Transverse sections of sural nerve showing irregular infoldings or loops of myelin into the axoplasm (circle). A few axons exhibit splitting or separation of myelin and adaxonal Schwann cell compartments containing degenerative debris (arrows; see also Figure 11). The axon of one myelinated fiber is contracted to one side of the section (arrow, lower right). A few axons contain intra-axonal bodies of less electron density compared to the myelin sheath (arrowhead). These bodies correspond to glycogenosomes and/or polyglucosan bodies (see also Figures 7 and 8). A few myelinating Schwann cells are seen encompassing their associated axons (S). A blood vessel containing a few red blood cells and conspicuous endothelial cells is present along the left lower margin (BV).
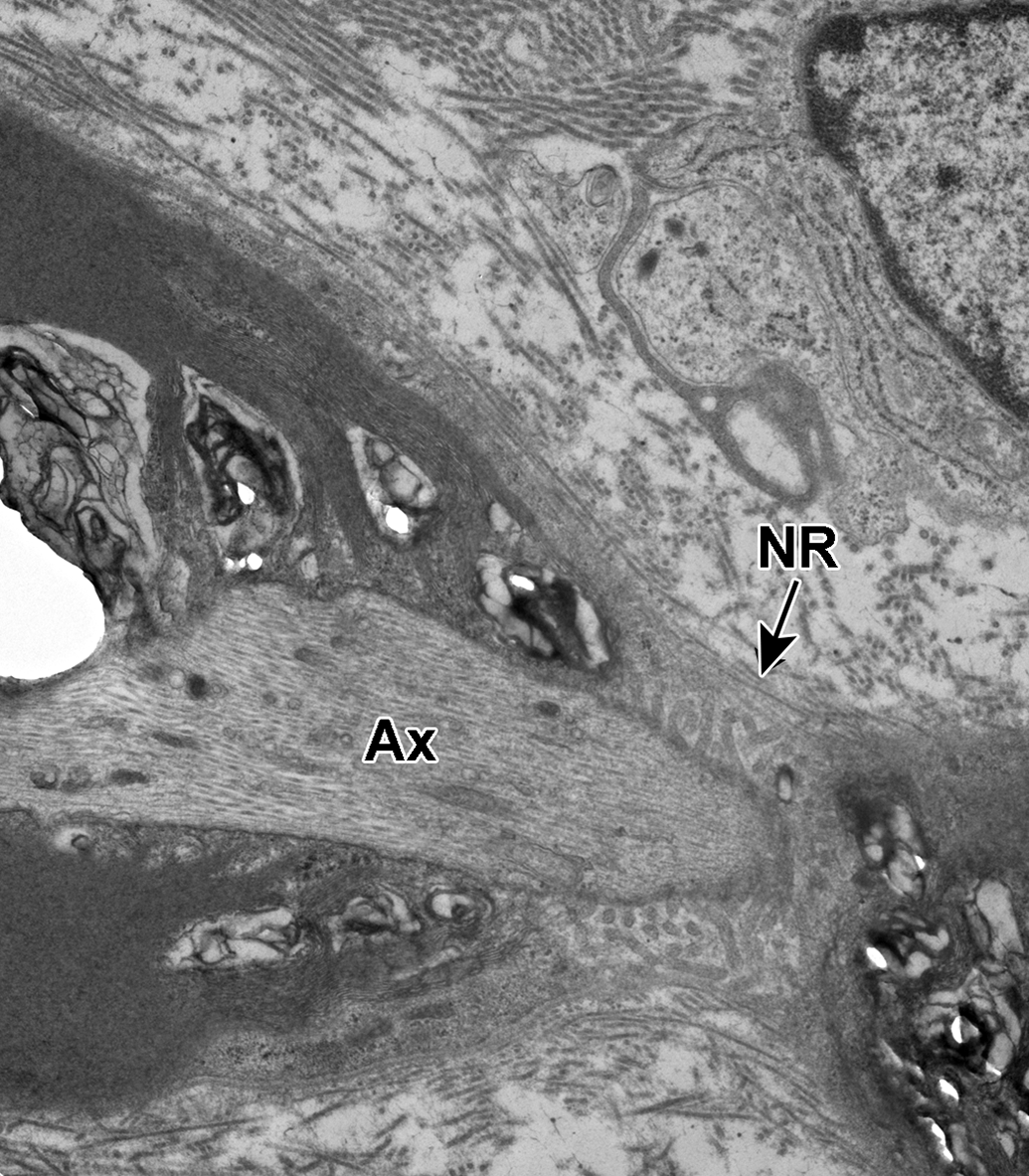
Node of Ranvier. A longitudinal section through an axon (Ax) at the Node of Ranvier is shown (NR). The nodal gap is seen covered by cytoplasmic projections (microvilli) from the associated Schwann cell.
Both transverse and longitudinal sections of myelinated axons frequently contained glycogenosomes that appeared as single to multiple circular structures within the axoplasm composed of large quantities of glycogen circumscribed by a single limiting membrane (see Figure 7). Other axons were disrupted by polyglucosan bodies, apparent as nonmembrane-bound accumulations of filaments that formed circular bodies (see Figure 8). Polyglucosan bodies occurred as both singular structures and in combination with glycogenosomes.
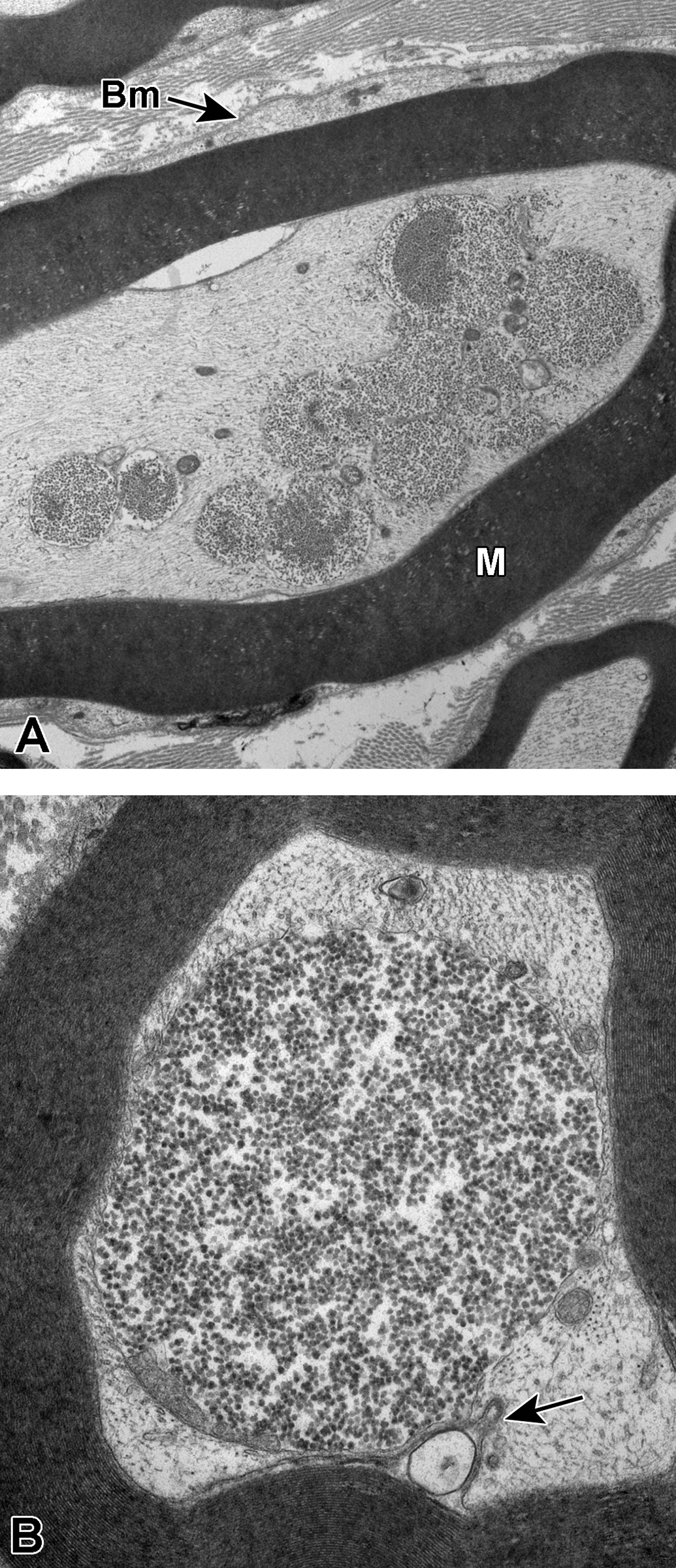
A and B, Sural nerve myelinated axons. In A, several glycogenosomes are seen disrupting the axoplasm. The Schwann cell cytoplasm and its prominent basement membrane (Bm) is seen circumscribing the concentric layers of compact myelin of the axonal sheath (M). In B, a single very large glycogenosome encompasses the central region of axoplasm. In the lower margin of the image, a small infolding of mesaxon (arrow) contains a membrane-bound structure likely originating within the associated Schwann cell cytoplasm.
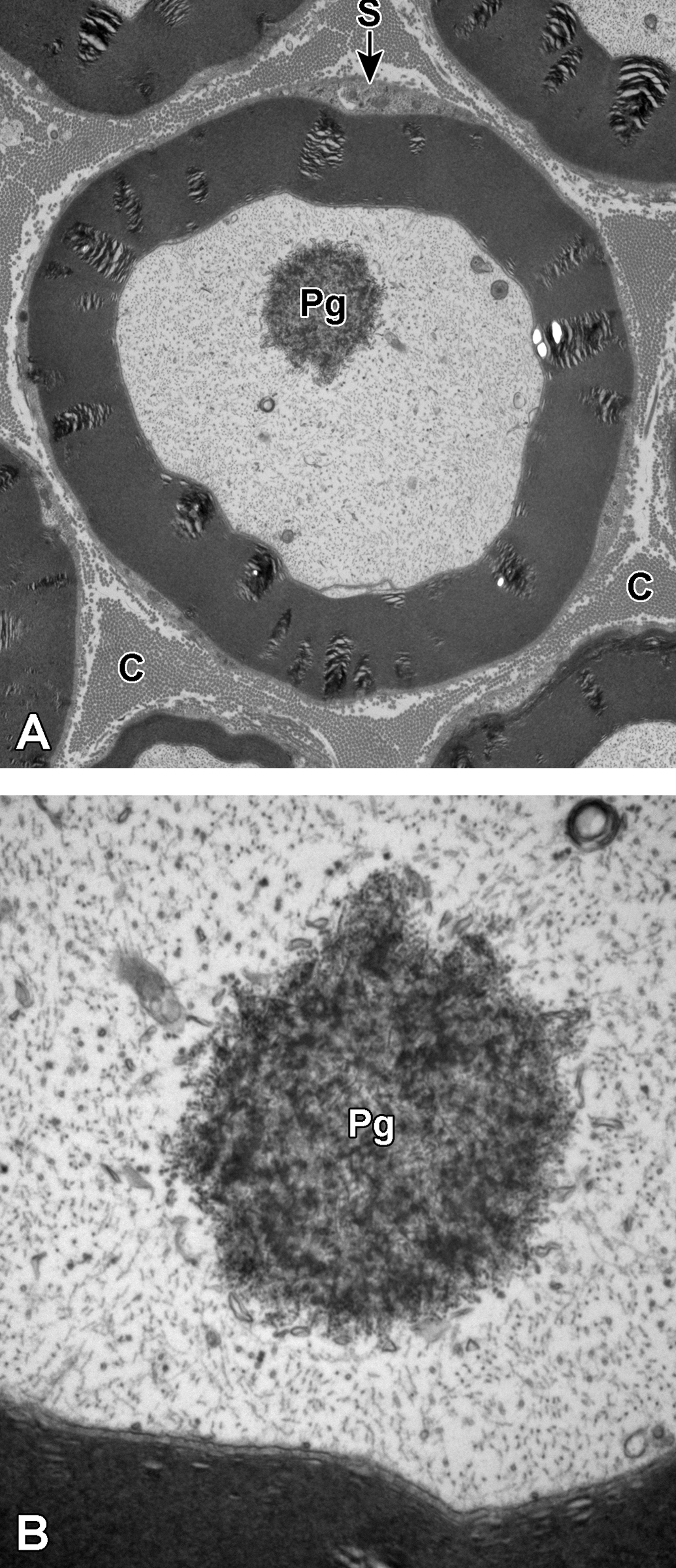
A and B, Sural nerve myelinated axons. In A, a single nonmembrane-bound filamentous body consistent with a polyglucosan body is present within the axoplasm (Pg). The myelin sheath is surrounded by small quantities of Schwann cell cytoplasm (S). The collagen fibrils within the endoneurium are prominent (C). In B, a higher magnification of the polyglucosan body shows its filamentous structure and the lack of a limiting membrane (Pg). In both A and B, neurofilaments are easily recognized throughout the axoplasm.
Occasional myelinated axons containing glycogenosomes and/or polyglucosan bodies were observed in association with degenerative changes such as swollen mitochondria and/or double membrane-bound lysosomes consistent with autolysosomes (see Figure 9). Additional changes, which were interpreted to be consistent with potential axonal degeneration, included splitting of myelin (some of which can be artifactual) and contraction/compression of the axoplasm by myelin debris either within the adaxonal compartment (see Figure 10) or along the inner region of the sheath (see Figure 11).
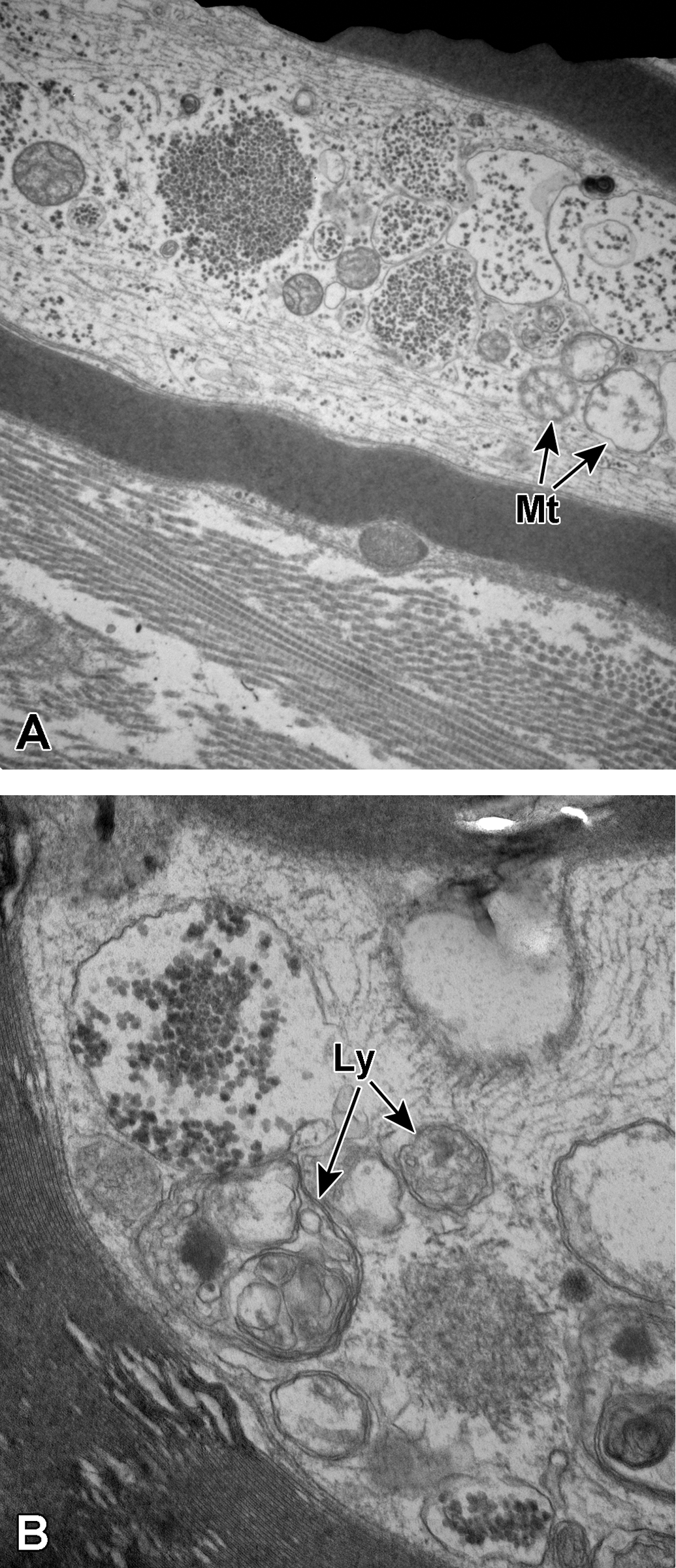
A and B, Sural nerve myelinated axons. In A, a longitudinal section through a myelinated axon shows disruption of the axoplasm by numerous glycogenosomes. There are several swollen mitochondria (Mt) in the surrounding axoplasm. In B, the axoplasm contains a glycogenosome and a polyglucosan body. There are also several double-membrane-bound structures consistent with autolysosomes (Ly).
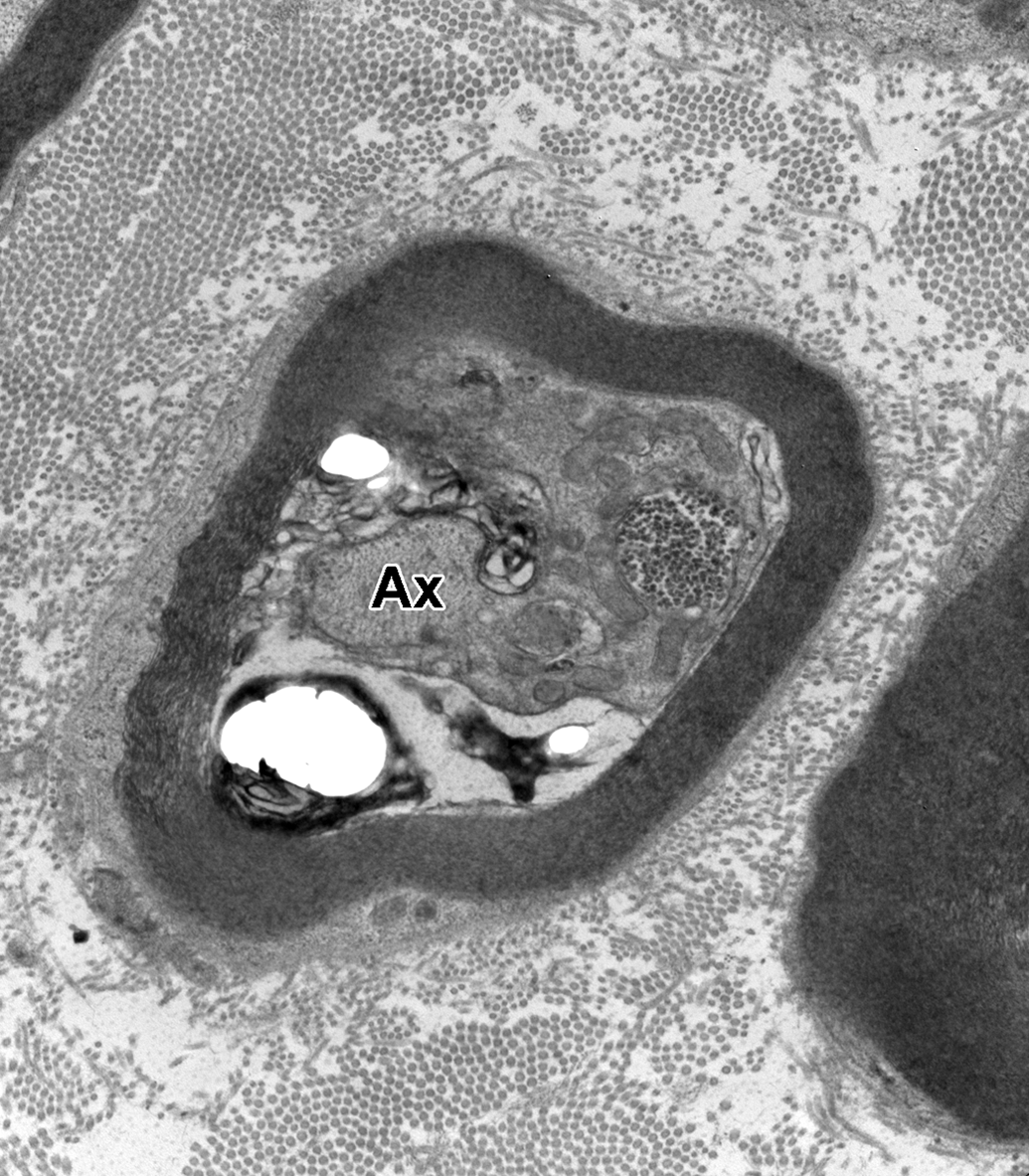
Sural nerve myelinated axon with potential degenerative changes. The axoplasm (Ax) is focally separated from a large portion of the sheath and appears contracted or atrophied, and the neurofilaments appear condensed. There are numerous mitochondria and a few small glycogenosomes within what appears to be Schwann cell cytoplasm extending into the axon. There is electron-dense material, likely vesicular myelin debris, within what could be an adaxonal Schwann cell compartment.
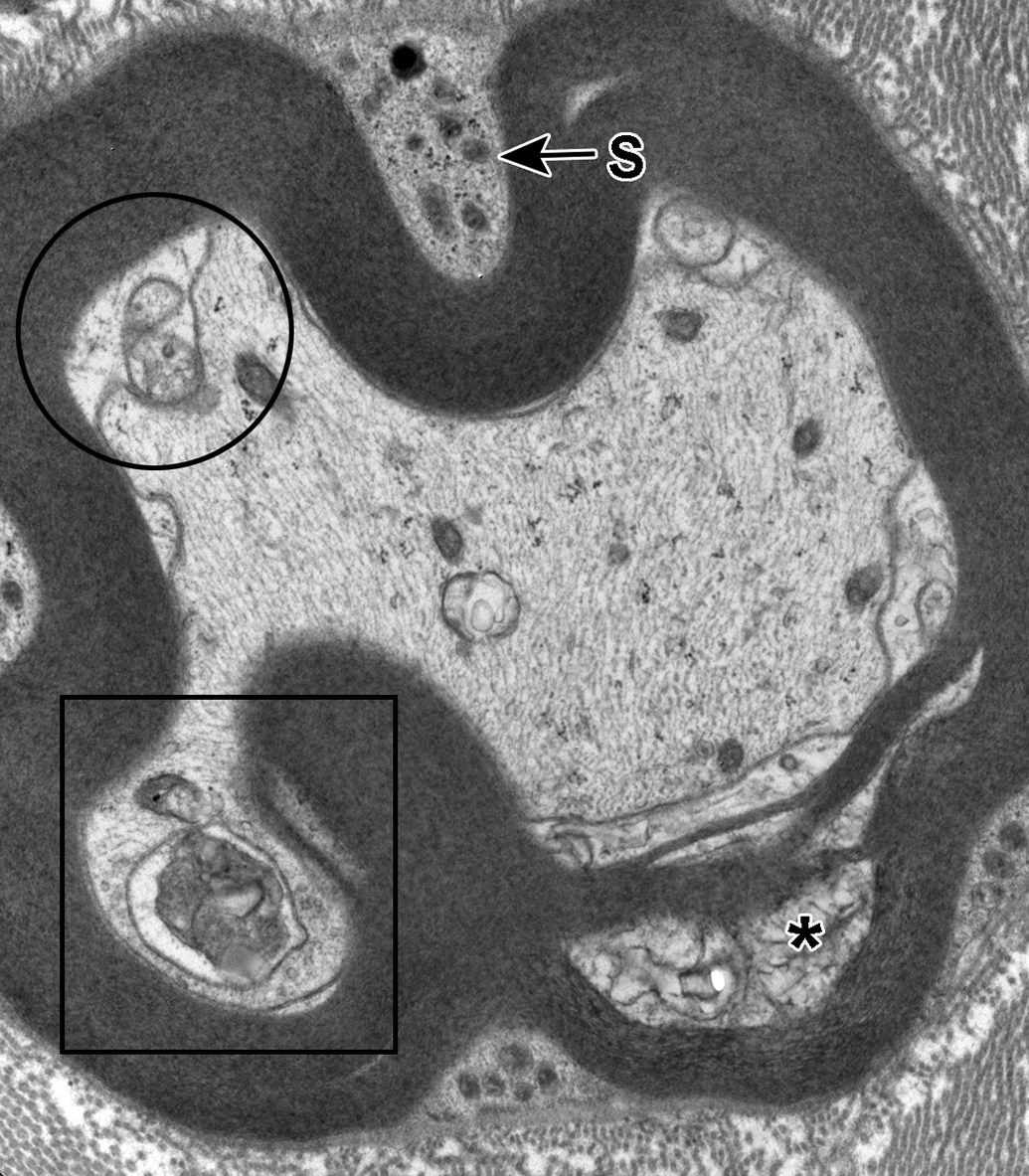
Sural nerve myelinated axon with degenerative changes. A focal region of split myelin contains vesicular debris (*). There is separation of the mesaxon along the inner margin of the myelin sheath and the resulting space may represent an adaxonal Schwann cell compartment (circle). One double-membrane-bound vacuole contains electron-dense debris and is consistent with an autolysosome (square). A small section of Schwann cell cytoplasm (S) is evident above and below the image.
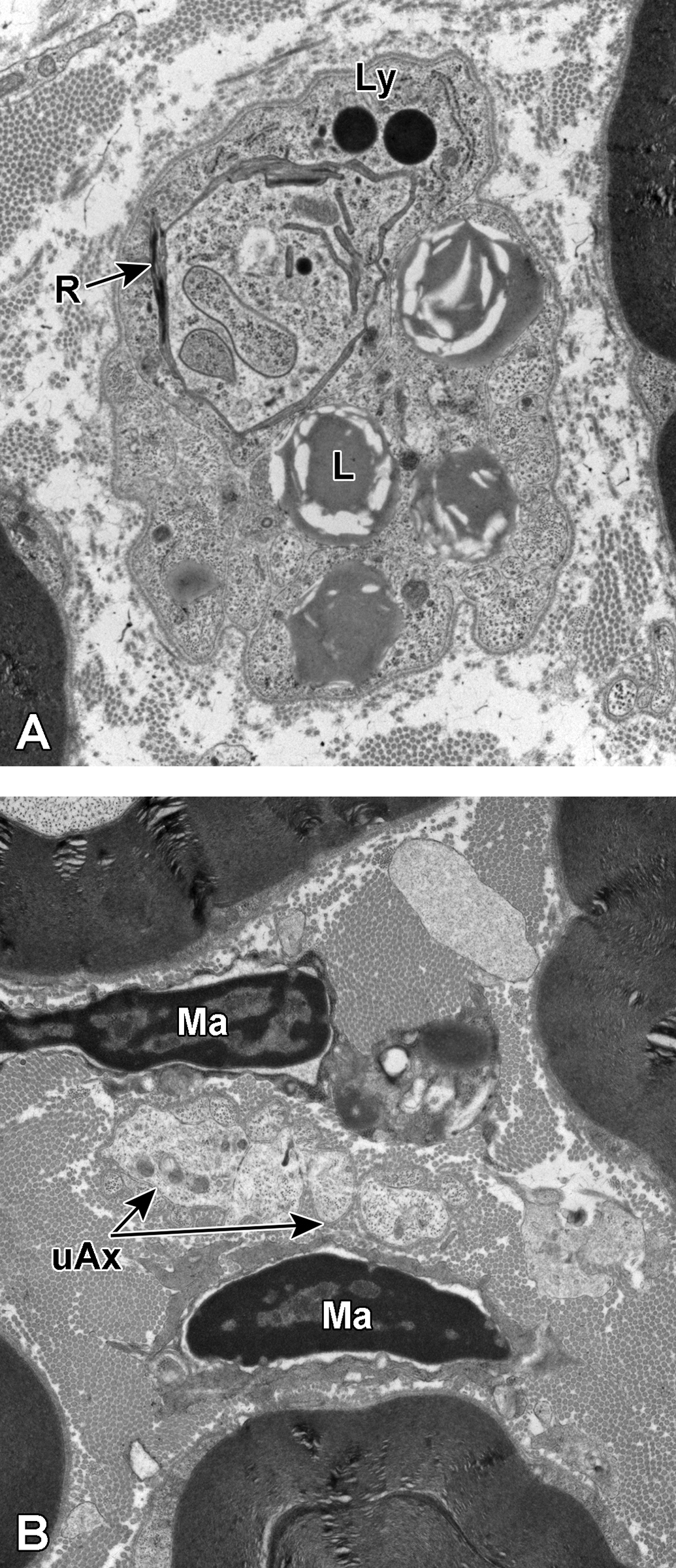
Further evidence of ongoing degeneration/regeneration was the presence of macrophages containing phagocytosed debris within the endoneurium, between myelinated and unmyelinated fibers. These macrophages were occasionally present in the vicinity of degenerative axons and contained a mixture of lipidic material, paracrystalline inclusions (consistent with ingested Reich granules), and lysosomes containing variably electron-dense material comprised of degenerate mitochondria and/or accumulations of myelin debris (see Figure 12).
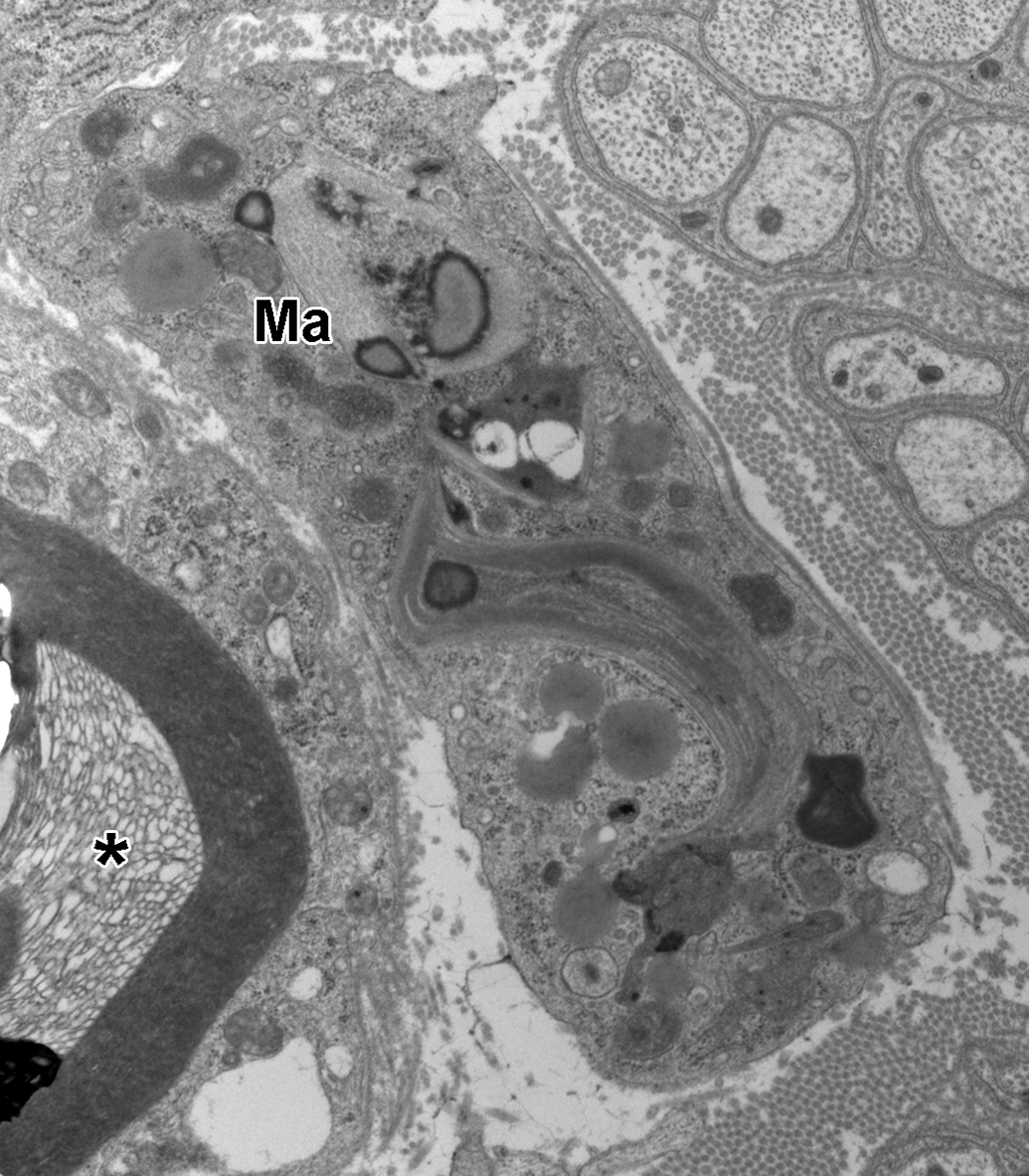
Macrophage within the endoneurium with phagocytosed debris. The macrophage (Ma) cytoplasm shows numerous dense bodies consistent with lysosomes containing lipidic material, several Reich (Pi) granules, and a large, curved, paracrystalline inclusion that also contains lipidic densities. A myelinated axon with split myelin and the formation of vesicular myelin (*) is present in the lower left. The associated Schwann cell contains several lysosomes, dilated endoplasmic reticulum, and mitochondria with vacuolated cristae (degenerative change). Unmyelinated axons are present in the upper right of the image.
Unmyelinated Sural Nerve Fibers: TEM
Similar to myelinated axons, normal unmyelinated axons are also wrapped by Schwann cell processes (see Figure 13) and are evident throughout the endoneurium as clusters or subunits between the myelinated fibers. Compared to those described in myelinated axons, very few ultrastructural changes were noted in unmyelinated axons from aged control rats. A few unmyelinated axons exhibited the presence of electron-dense bodies, filamentous bodies, and/or glycogenosomes, similar to those noted in myelinated axons. Pockets of collagen invested by Schwann cell membrane were infrequently noted (see Figure 13). Rarely, degenerative changes, such as accumulations of lipidic material, filamentous bodies, dense bodies, and/or paracrystalline inclusions, were noted within the cytoplasm of Schwann cells surrounding unmyelinated axons (see Figure 14).
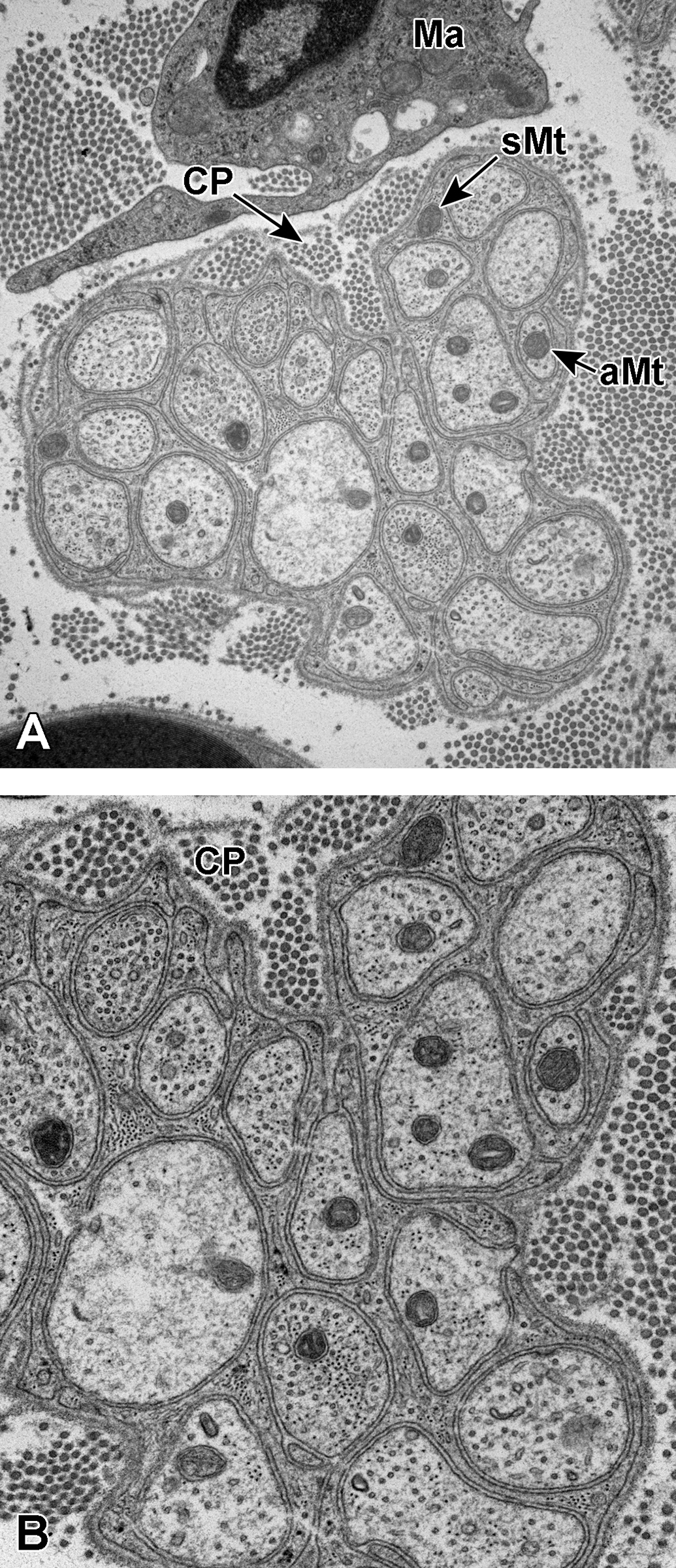
A and B, Normal unmyelinated sural nerve axons. Unmyelinated axons are completely invested by nonmyelinating Schwann cell processes. The distinct basement membrane of the Schwann cell can be seen wrapping around the axons, and the Schwann cell cytoplasm can be seen between basement membrane projections. In A, mitochondria can be seen within the axoplasm (aMt) as well as within the Schwann cell processes (sMt). A macrophage (Ma) can also be seen along the top margin of the image. In A and B, collagen pockets (CP) are present along the top margin of the cluster of axons. These structures are defined by the presence of Schwann cell membranes investing collagen fibrils.
A and B, Degenerative unmyelinated sural nerve axons. In A, a Schwann cell subunit is shown. The nonmyelinating Schwann cell basement membrane is seen investing what appear to be several small, irregularly shaped, unmyelinated axons. These are best visualized in this image along the right and bottom margins of the Schwann cell subunit. The cytoplasm of the nonmyelinating Schwann cell contains several large lipidic vacuoles (L), a few electron-dense lysosomes (Ly), and several paracrystalline inclusions (R). This debris likely represents phagocytosed material from damaged nerve fibers taken up by the Schwann cell. In B, several irregularly shaped, variably sized, unmyelinated axons (uAx) are present between 2 macrophages (Ma).
Dorsal Root Ganglion: TEM
Neuronal cell bodies of varying sizes are typically present in normal DRG of rats, and the subcellular constituents are similar to those observed in other neurons found throughout the nervous system (see Figure 15). Dorsal root ganglia neurons typically contain lysosomes with either electron-dense material or electron-dense material in conjunction with lipid (ie, lipofuscin) and variably distributed bundles of Nissl substance (ie, granular/rough endoplasmic reticulum). Neurofilaments and microtubules are generally present in regions of lower electron density (see Figure 16). Ganglion cells are completely invested by satellite cell cytoplasmic processes. Ultrastructural changes observed in neurons of the DRG of aged control rats that were considered possibly degenerative in nature included dilation or swelling of mitochondria (see Figure 17), mitochondria with vesicles containing dense bodies (possibly consistent with glycogen), or flocculent densities within the mitochondrial matrix (see Figure 18).
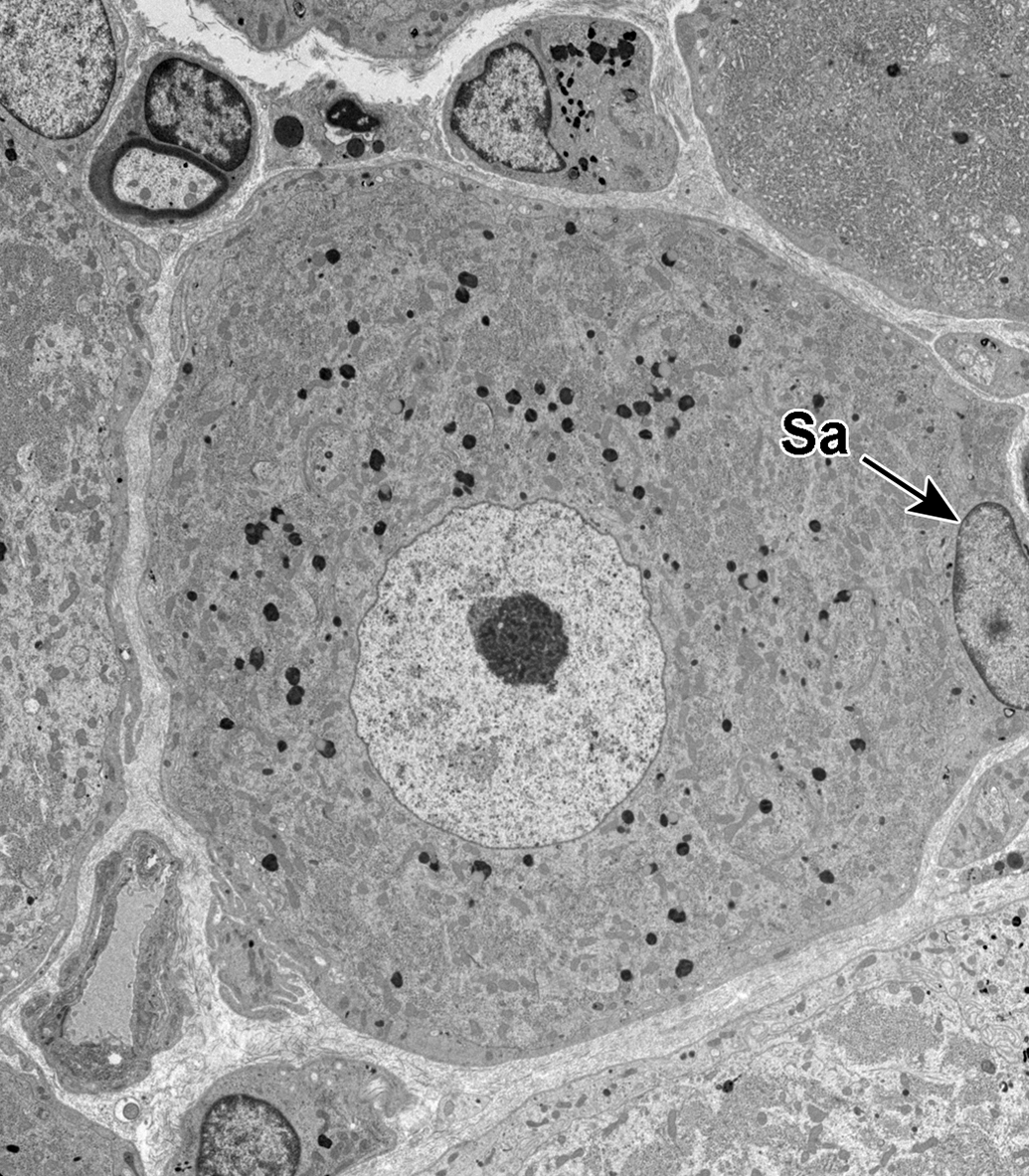
Dorsal root ganglion cell. A small dorsal root ganglion cell is shown with numerous small electron-dense bodies (ie, lysosomes) scattered throughout the cytoplasm. The nucleus contains a prominent nucleolus. The nucleus of a satellite cell (Sa) is present along the right margin of the image.
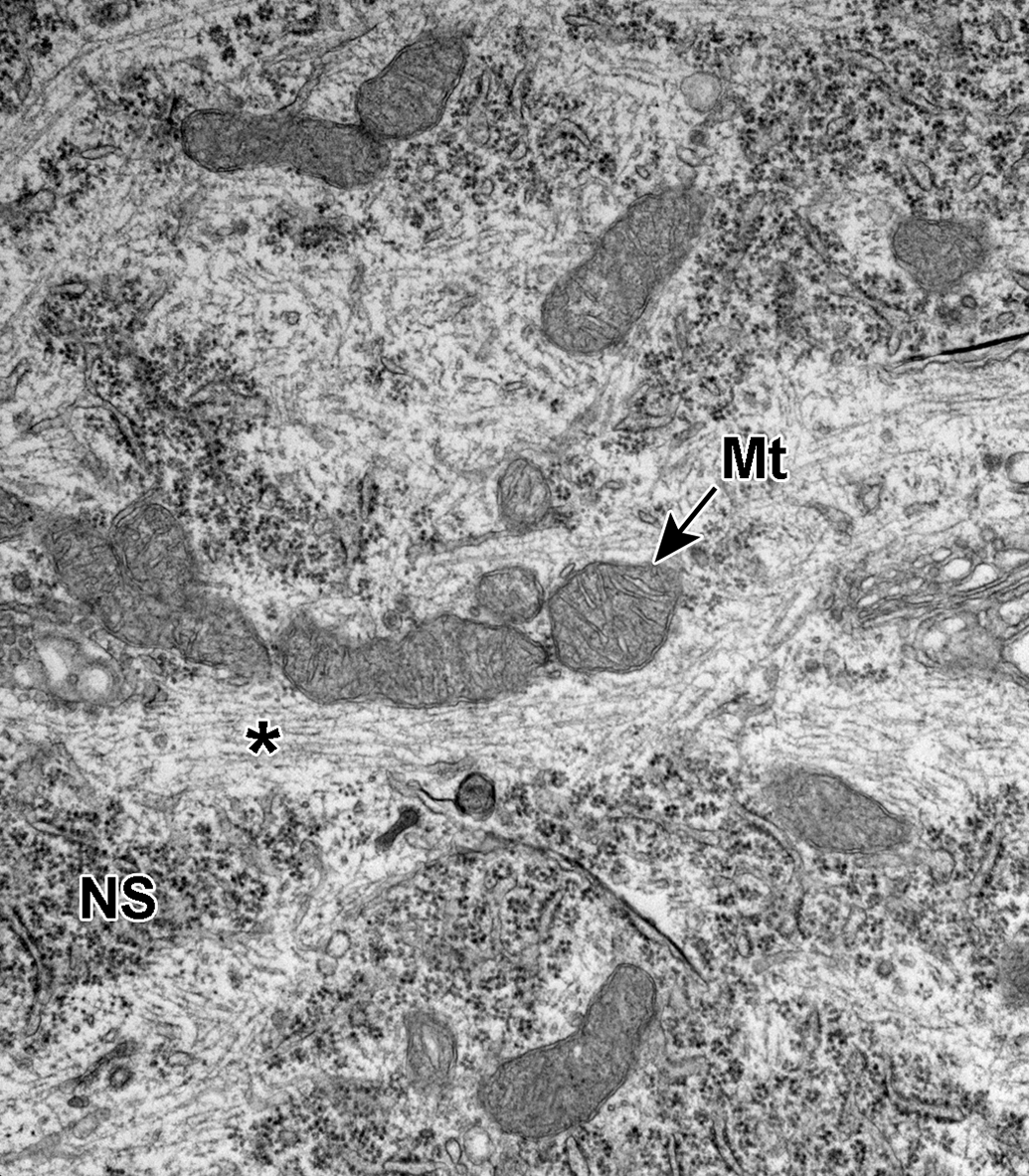
Dorsal root ganglion cell. Neurofilaments and microtubules are present coursing through the cytoplasm (*) between the Nissl substance (NS). Numerous mitochondria are present (Mt). Neurofilaments have a smaller diameter than the microtubules.
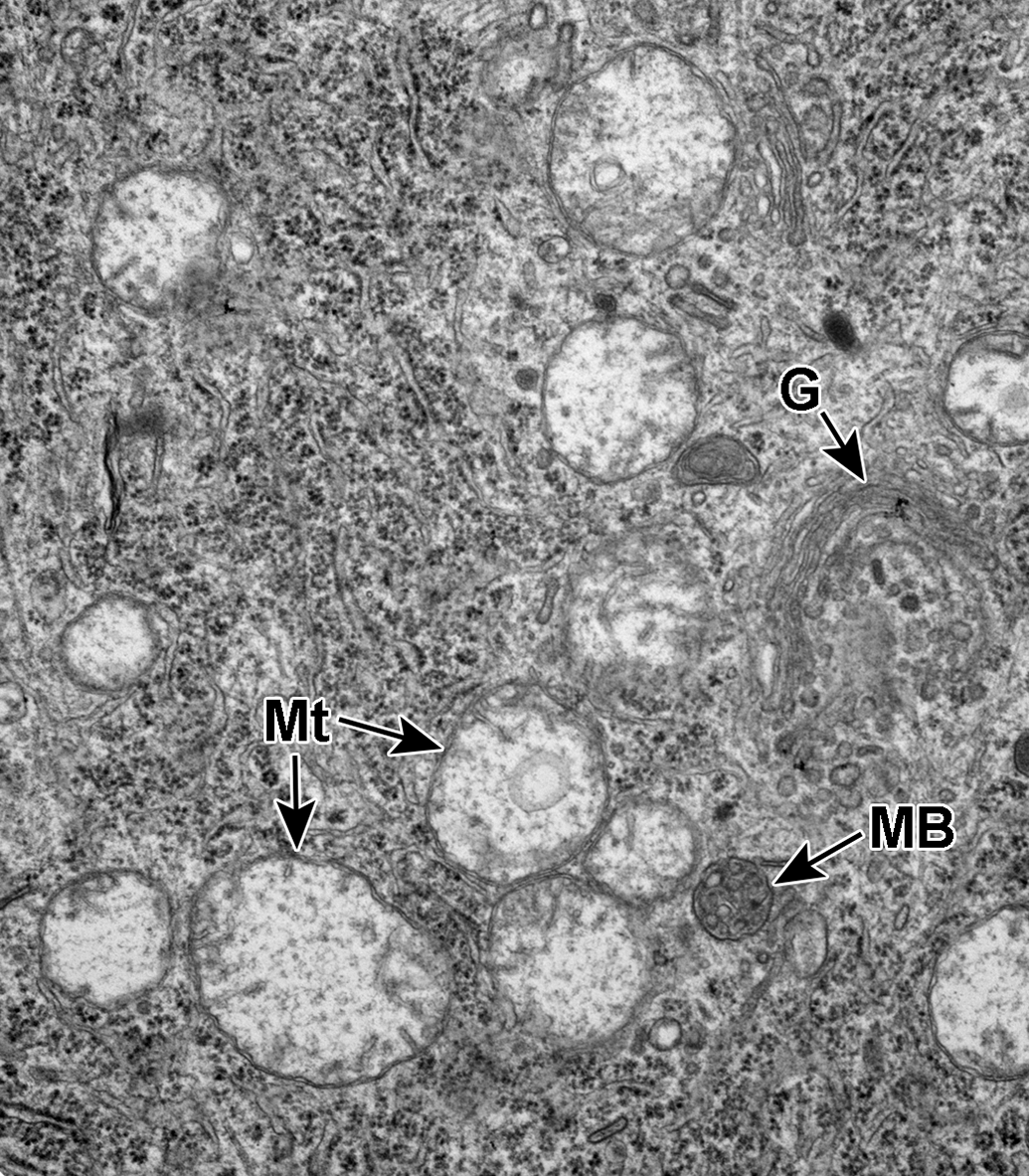
Dorsal root ganglion cell. Mitochondria exhibit swelling and expansion of the cristae and matrix (Mt). A Golgi complex (G) and a single multivesicular body (MB) are also present.
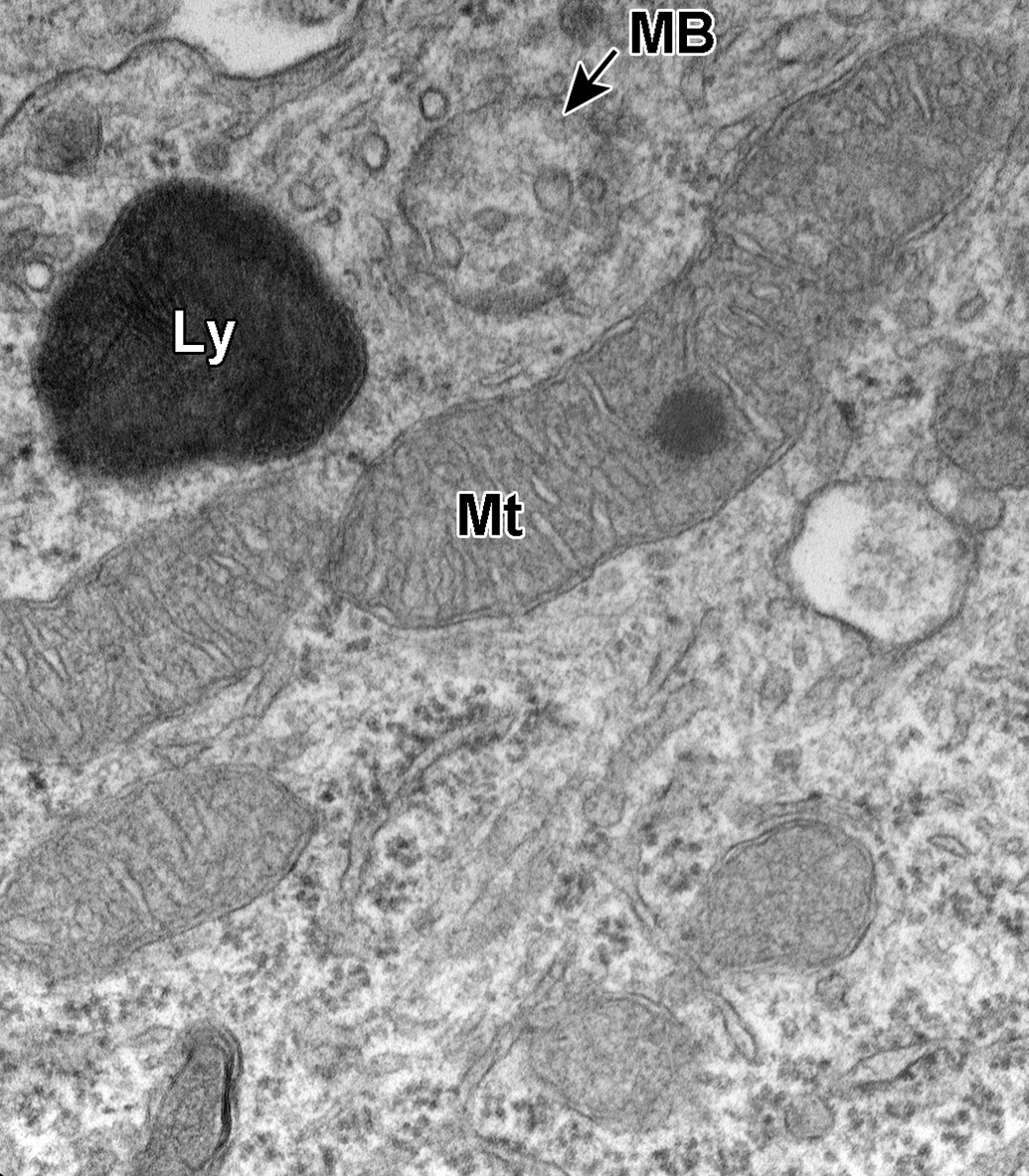
Dorsal root ganglion cell. A mitochondrion (Mt) contains a small density within the matrix, and a lysosome (Ly) and a multivesicular body (MB) are also evident.
Discussion
In humans, nerve biopsy is considered the gold standard in investigations of peripheral neuropathies. Sensory-predominant nerves are known to differ by species. 1 Long distal nerves that are composed primarily of sensory fibers are most frequently chosen, as they are located away from regions susceptible to localized trauma. The sural nerve represents a good candidate to screen for morphologic changes associated with the neuropathy being investigated. 24,25 For these reasons, examination of distal peripheral nerves, such as the sural nerve, should be considered for new therapeutic candidates that have a known potential to induce peripheral neuropathies. An understanding of the possible ultrastructural changes associated with normal aging in control rats is therefore imperative in order to distinguish any potential test article–related changes that might occur.
Given this heightened awareness of the potential for peripheral nerve damage resulting from the administration of xenobiotics, there has been a renewed effort to establish standardized preclinical methodologies to help identify the relative risks for drug development candidates. Although the occurrence of age-associated peripheral neuropathy is a well-recognized condition in humans, 26,27 its definitive cause in aging experimental animals is less well understood. 28,29 Few studies with experimental animals have definitively separated age-associated distal nerve changes from those induced by pressure neuropathy (ie, localized trauma). Since the sural nerve is anatomically isolated from the tibial/plantar nerve complex, a site known to be associated with compression neuropathy and likely “dying back” type lesions in more proximal segments of nerve, any ultrastructural abnormalities identified in the sural nerve of control rats should represent age-associated lesions.
In the current study, the majority of myelinated sural nerve fibers from aged control rats had no ultrastructural abnormalities. Aged control rat Schwann cells exhibited occasional paracrystalline inclusions such as Reich (Pi) granules, Elzholz bodies/Mu granules, and lysosomes that contained phagocytosed myelin debris and/or lipidic inclusions.
Reich (Pi) granules have been shown to stain with acid phosphatase, thus identifying them as elongated or polygonally shaped lysosomes 23,30 containing sulfate and/or phosphate moieties. 31 Bilbao and Schmidt reported that Pi granule-like inclusions have been noted in macrophages associated with active nerve degeneration and that these inclusions likely originated from degenerative Schwann cells. 23 Interestingly, several examples of macrophages (within the endoneurium) and degenerative Schwann cells with Pi-like granules were noted in the present study of aged control rats. It is generally agreed that Reich (Pi) granules increase with age 28,30,32,33 and thus are likely to be seen in aged control rats.
Elzholz bodies or Mu granules were noted in myelinating Schwann cells, but likely have no significance, as they occasionally are present in the cytoplasm of normal nerves, possibly occurring secondary to myelin remodeling or glancing sections through loops of myelin just out of the plane of section. They are reported to have the same periodicity as normal myelin. 23 However, Schwann cells with lysosomes containing engulfed myelin debris should not be confused with Elzholz bodies, as they are known to participate in the early clearance of damaged nerves prior to the appearance of macrophages. 34,35 As in this study, these types of Schwann cells present with lysosomal accumulations of degenerate myelin, often coincident with other lipidic material or degenerate cellular organelles, and likely represent on-going degenerative processes being cleared by resident Schwann cells.
The other frequently observed ultrastructural findings noted in both myelinated and unmyelinated sural nerve axons in control rats of this study included intra-axonal round bodies enclosed within a single-limiting membrane containing electron-dense particles of glycogen (ie, glycogenosomes) and nonmembrane-bound electron-dense bodies composed of filamentous material (ie, polyglucosan bodies). Both of these findings were noted at greater incidence in myelinated axons compared to unmyelinated axons. The presence of intra-axonal glycogenosomes and polyglucosan bodies has been regarded as evidence of a primary axonal abnormality 28 ; whereas other investigators considered glycogenosomes to arise from perturbations of mitochondria as evidenced by several toxic or metabolic states. 29,36 Further evidence that glycogenosomes may occur secondary to some altered metabolic state is supported by the observations of Powell et al 37 and Sharma et al, 38 which both concluded that while axonal glycogenosomes increased with age in nondiabetic control animals, they occurred with greater frequency in both diabetic rats and diabetic mutant mice, respectively. In the study by Grover-Johnson and Spencer, 29 they further postulated that glycogen accumulation in axonal injury may be a source of energy for maintenance of function. Others have suggested that glycogen deposition in axons may reflect Ca+2 mediated interference of glycogen synthase activity and evidence of compromised axonal transport. 39 In the diabetic rats of the Powell et al study, 37 polyglucosan bodies (ie, Lafora body-like inclusions) were not observed in age-matched controls, whereas, in the diabetic mutant mice of the Sharma study, 38 polyglucosan bodies were present in controls and increased with age. Polyglucosan bodies are composed of abnormally branched glycogen, protein, phosphate, and sulfate. 40,41 They have been identified in myelinated and unmyelinated axons, Schwann cells, perineurial cells, macrophages, and endothelial cells. 40
Other changes in sural nerve axons of aged control rats in the current study included swollen mitochondria, the presence of autolysosomes containing effete cytoplasmic structures, splitting of myelin, the presence of adaxonal compartments composed of Schwann cell processes with/without debris, and axonal atrophy or collapse.
Splitting of myelin is generally considered an early change that often is noted near the nodes of Ranvier. 42 Some degree of myelin separation, as well as frequent infoldings or loops of myelin noted in normal axons of this study, may represent artifact and not a degenerative change; however, these minor changes have been described with increased incidence in neuropathy 23,43,44 and with increasing age, 45 so noting their presence may be important.
Adaxonal Schwann cell compartments (ie, Schwann cell–axon networks) were noted in a few myelinated axons in the aged control rats in this study. Adaxonal Schwann cell compartments may represent evidence of axonal damage with the extension of Schwann cell cytoplasmic processes into the axoplasm of a nerve for the removal of damaged organelles and/or debris 34,46 but also may be important in maintaining normal axon function in the absence of a pathologic process.
In the current study, myelinated axons and a few dorsal root ganglion cells occasionally contained mitochondria that exhibited dilated regions of cristae containing small electron-dense particles suggestive of glycogen. It may be that glycogen accumulation in axonal mitochondria and glycogenosomes in damaged axons represent a manifestation of the same process, namely, providing energy for maintenance of function or, in the more extreme examples of glycogenosome formation, overt evidence of compromised axonal transport mechanisms. 39
Fewer ultrastructural changes were noted in unmyelinated axons from aged control rats. However, occasional unmyelinated axons exhibited the presence of electron-dense bodies, filamentous bodies, and/or glycogenosomes, similar to those noted in myelinated axons as well as a few collagen pockets. Although pockets of collagen invested by Schwann cell membrane can be seen in normal unmyelinated axons, they have been reported to increase with age and neuropathy. 26 Rarely, degenerative changes, such as accumulations of lipidic material, filamentous bodies, dense bodies, and/or paracrystalline inclusions, were noted within the cytoplasm of Schwann cells surrounding unmyelinated axons (see Figure 14).
In the PNS, nonmyelinating Schwann cells are much more abundant than myelinating Schwann cells, 43,47 and unmyelinated axons are more numerous than myelinated axons in the sural nerve. 25 This suggests that the examination of unmyelinated nerves is as informative as examination of myelinated nerves, maybe even more so. In the current study, fewer ultrastructural changes were noted in unmyelinated axons of aged control rats compared to myelinated axons, but some of the same changes were noted.
The ultrastructural changes noted in neurons of the lumbar DRG of aged control rats were sporadic, often with 1 affected ganglion cell surrounded by ganglion cells lacking ultrastructural changes. It may be that individual ganglion cells with ultrastructural changes, such as swelling of mitochondria, intramitochondrial glycogen, or other mitochondrial inclusions, might represent the cell bodies of distal nerve axons that also exhibit ultrastructural changes. If so, then this would highlight the importance of examining the ultrastructure of neurons of the DRG in any nonclinical toxicity study, where peripheral neuropathy is an expected or potential outcome.
In conclusion, age-associated sural nerve changes, although rare, should be expected when examining rats of advanced age. This article is intended to help establish a baseline understanding of incidental age-associated sural nerve and DRG ultrastructural changes. Similar changes in aged rats treated with any new drug candidate known, or suspected to induce peripheral neuropathy, should be represented by an increased incidence or severity of the types of changes described in this study.
Footnotes
Acknowledgments
The authors would like to thank Ashley Reise, Kathleen Komro, and Joseph Heintz for their assistance in electron microscopy sample processing as well as Steve Van Adestine for assistance in figure formatting.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
