Abstract
In preclinical studies of pharmaceutical agents, the beagle dog is a commonly used model for the detection of cardiotoxicity. Incidental findings, postmortem changes, and artifacts must be distinguished histopathologically from test item–related findings in the heart. In this retrospective analysis, cardiac sections from 88 control beagles (41 male, 47 female; ages 5–18 months) in preclinical studies were examined histopathologically. The most common finding was thickening of the tunica media of intramural coronary arteries, most likely a postmortem change. The second most common finding was the presence of vacuoles within Purkinje fibers. Dilated lymphatic and blood vessels at the insertion of chordae tendineae were noted more commonly in males than in females and were considered a normal anatomic feature. Mesothelial-lined papillary fronds along the epicardial surface of the atria were present in several dogs, as were small infiltrates of inflammatory cells usually within the myocardium. In summary, control beagles’ hearts frequently have incidental findings that must be differentiated from test item–related pathologic changes. Historical control data can be useful for the interpretation of incidental and test item–related findings in the beagle heart.
Introduction
The beagle dog is used extensively as a non-rodent model in preclinical toxicity studies. When histopathologic examination, electrocardiography, and other techniques are combined, dogs are a sensitive species for the detection of cardiotoxicity of pharmaceutical agents intended for humans (Greaves 2007). Preclinical toxicity studies using beagle dogs typically use a small number of animals per treatment group. Incidental findings and artifacts in histological sections of beagles’ hearts may be difficult to distinguish from genuine test item–related changes. For instance, distinguishing coronary arterial lesions induced by vasodilating agents from lesions of spontaneous arteritis can be problematic (Clemo et al. 2003; Isaacs, Joseph, and Betton 1989; Joseph 2000). The presence of these incidental findings and artifacts in dogs dosed with the test compound may confound a toxicologic pathologist’s interpretation of toxicity study results, particularly if these findings are absent in the control animals in the study. The pathologist must recognize the more common incidental findings to interpret study results accurately. The aim of this article is to describe the features and incidence of some of these background findings in untreated dogs.
Materials and Methods
In this retrospective analysis, representative sections of hearts from 88 control beagles (41 male and 47 female) included in 17 toxicity studies from 2001 to 2008 were reviewed. The dogs’ ages at necropsy ranged from 5 to 18 months, with a median age of 9 months. The study durations ranged from 10 days to 3 months. Thirty-six of the males and 36 of the females were obtained from Marshall BioResources (formerly Marshall Farms USA, Inc., North Rose, NY), and the other 5 males and 11 females were obtained from Covance Laboratories, Inc. (Kalamazoo, MI). All procedures were in compliance with the Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals. Dogs were housed individually in animal rooms that were set to maintain a temperature of 18° to 25°C, with a 12-hr light/12-hr dark cycle. Dogs were fed rations of Harlan 2025C Teklad Global 25% Protein Dog Diet (Harlan Teklad, Madison, WI) and were allowed free access to water. The dogs’ vaccination status from both breeders included an intranasal Bordetella-Parainfluenzavirus-Adenovirus vaccine, repeated Canine Parvovirus modified live vaccine, and a Bordetella vaccine injection as a puppy. With age, dogs were vaccinated against rabies with repeated vaccinations against distemper-adenovirus-parainfluencavirus-parvovirus-leptospirosis. Dogs from Marshall BioResources additionally received a recombinant canine papillomavirus vaccine. The dogs’ anthelmintic status from both breeders included regular treatments with pyrantel pamoate anthelmintics at the breeder’s facility. Dogs from Marshall BioResources temporally received amprolium, sulfadimethoxine, decoquinate, and ivermectin.
At the completion of the in-life portion of the studies, the dogs were anesthetized, exsanguinated, and had necropsies performed. The dogs’ hearts were placed in 10% neutral buffered formalin for a minimum of 24 hr. Representative sections of each heart were then embedded in paraffin, sectioned at approximately 5 µm, placed onto glass slides, and stained with hematoxylin and eosin (H&E). Glass slides were digitally scanned using a ScanScope XT (Aperio Technologies, Vista, CA). In most instances, the left atrium, right atrium, left ventricle (including apex and papillary muscle), right ventricle, interventricular septum, and interatrial septum were represented. All sections were examined retrospectively by a pathologist (J.D.) using ImageScope slide viewer (Aperio Technologies). Another pathologist (K.B.) performed a peer review of selected slides. Additionally, representative serial sections of the left ventricular papillary muscle of a male dog were stained with Masson Goldner (Boeck 1989), Congo red (Puchtler, Sweat, and Levine 1962), and an antibody against Ki-67 (Monoclonal Mouse Anti-Human Ki-67 Antigen, Dako Denmark A/S, Glostrup, Denmark). Representative serial sections of the right ventricle of another male dog were stained with a Periodic Acid-Schiff (PAS) stain, and were stained with antibodies against α-actin (Monoclonal Mouse Anti-sarcomeric Actin, DakoCytomation Denmark A/S, Glostrup, Denmark), desmin (Desmin [DE-R-11], Santa Cruz Biotechnology Inc, Santa Cruz, CA), full-length dystrophin (Dystrophin [H-300], Santa Cruz Biotechnology Inc, Santa Cruz, CA), protein gene product 9.5 (PGP 9.5; Anti-Protein Gene product 9.5, Millipore, Temecula, CA), lysosome-associated membrane protein 2 (LAMP-2; Rabbit Anti-Lamp-2, Invitrogen Corporation, Frederick, MD), and adipophillin (Mab to Adipophillin/ADRP, Progen Biotechnik GmbH, Heidelberg, Germany). The antibodies were applied using an immunohistochemical technique according to standard laboratory procedures including representative positive control tissue. The antigen–antibody complex was visualized by applying Biotin and alkaline phosphatase streptavidin complexes followed by fast red substrate (Biogenex, San Ramon, CA), and nuclear staining with Mayers hematoxylin (Merck KGaA, Darmstadt, Germany). Glass slides with special stains were digitally scanned using a ScanScope XT (Aperio Technologies, Vista, CA).
Results
The incidence of microscopic findings in male and female dogs by anatomic site is listed in Tables 1 and 2, respectively. Since there was no significant difference in the incidence of findings between the dogs from the two different breeders, no further differentiation was performed. In both males and females, the most common finding was thickening of the tunica media of arteries (Figure 1). This finding was noted in 21 of 41 males (51%) and 22 of 47 females (47%). The most frequent site for this finding was the left ventricular wall, particularly in the papillary muscle. Smooth myocytes in affected vessels were plump and often had vesicular nuclei with prominent nucleoli. The lumina of affected vessels were often narrowed. The media of affected vessels was negative for Masson Goldner stain (only adventitial tissue was positively stained for connective tissue as expected; see Figure 2) and was also negative for Congo red (Figure not shown). The nuclei of the smooth myocytes in affected vessels were negative for Ki-67 (see Figure 3).
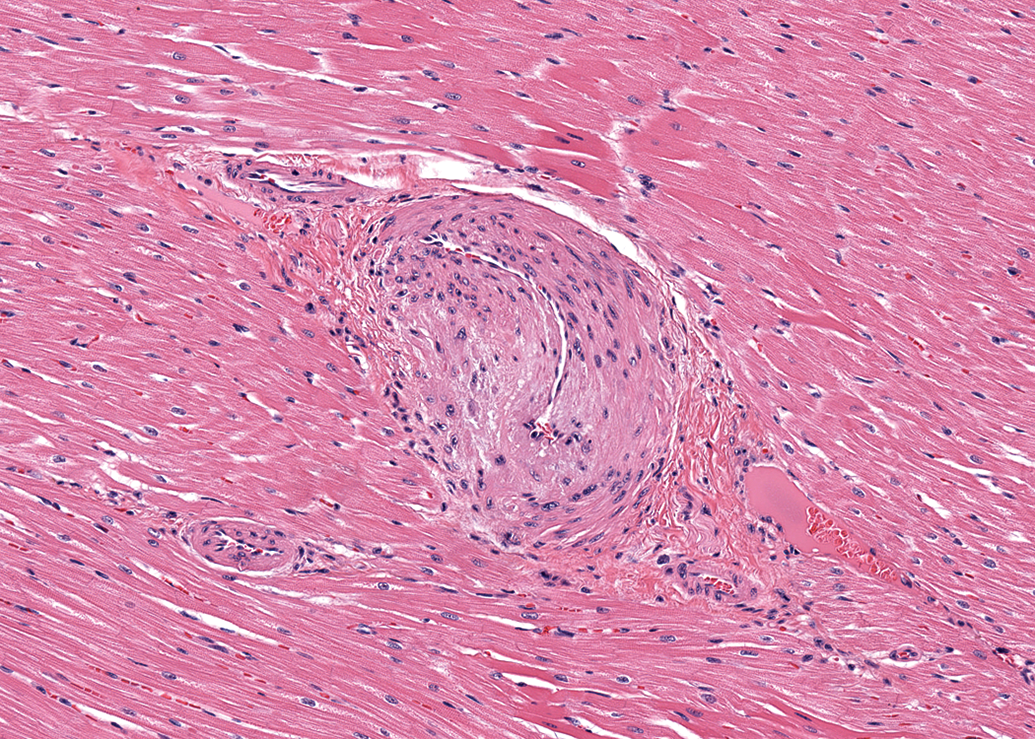
Left ventricle; female dog. Thickened wall and narrow lumen in an intramural coronary artery. Hematoxylin and eosin (H&E). 180×.
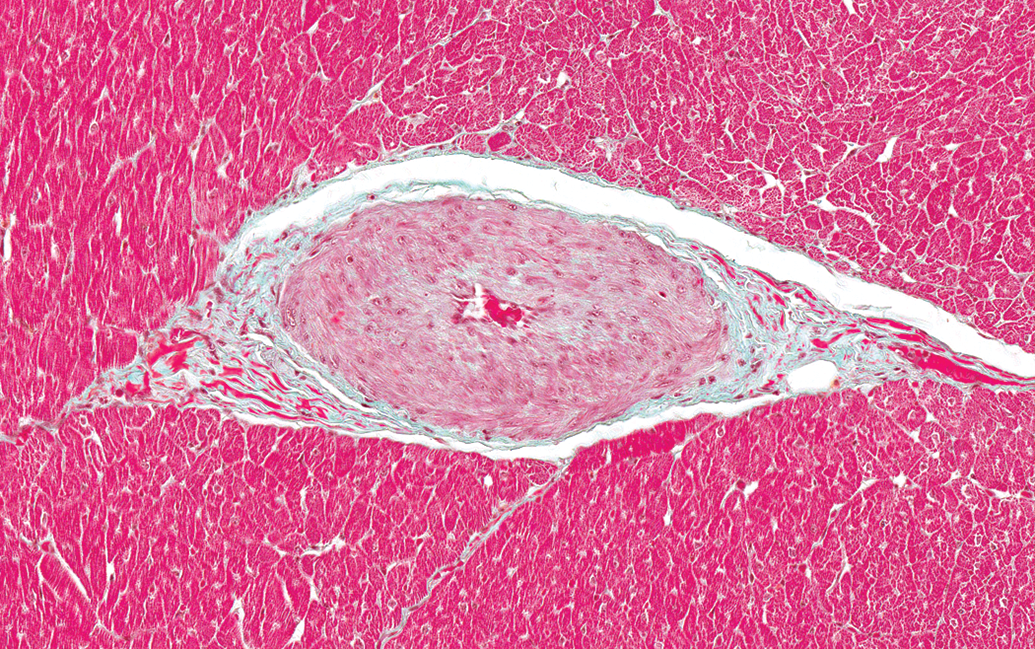
Left ventricle; papillary muscle; male dog. Thickened wall and narrow lumen in an intramural coronary artery. Masson Goldner. 200×.
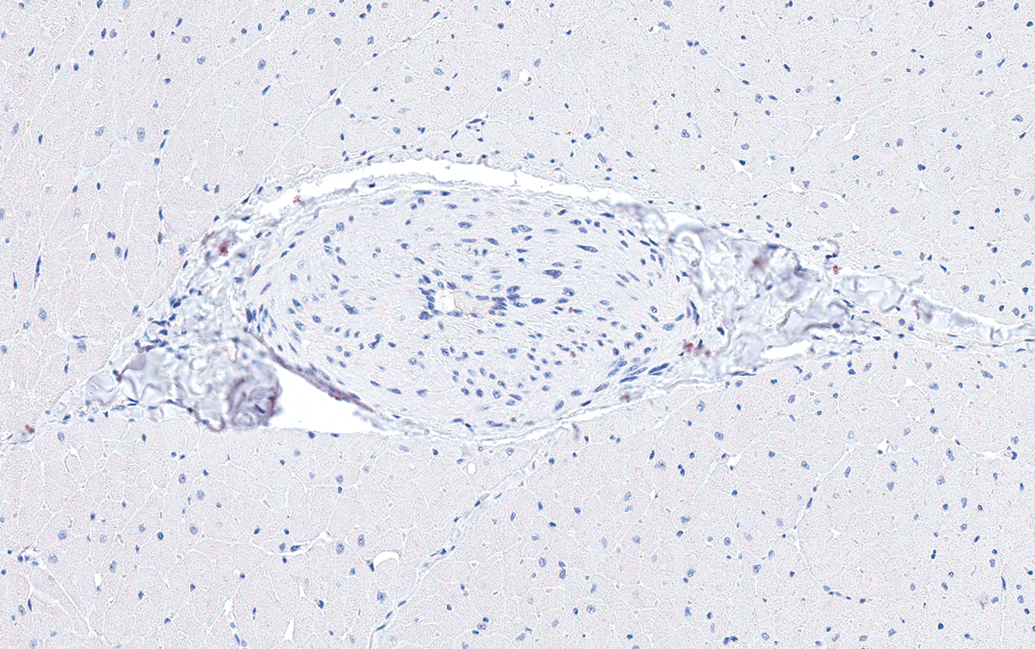
Left ventricle; papillary muscle; male dog. Thickened wall and narrow lumen in an intramural coronary artery. Ki-67 Immunohistochemical stain. 200×.
Incidence of microscopic findings in control males by anatomic site.
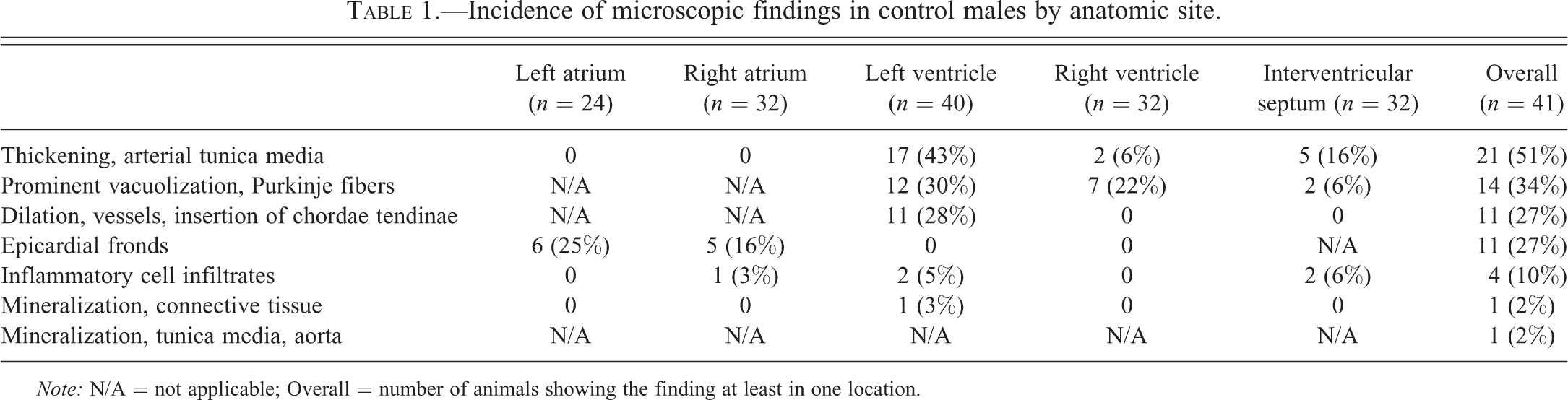
Note: N/A = not applicable; Overall = number of animals showing the finding at least in one location.
Incidence of microscopic findings in control females by anatomic site.
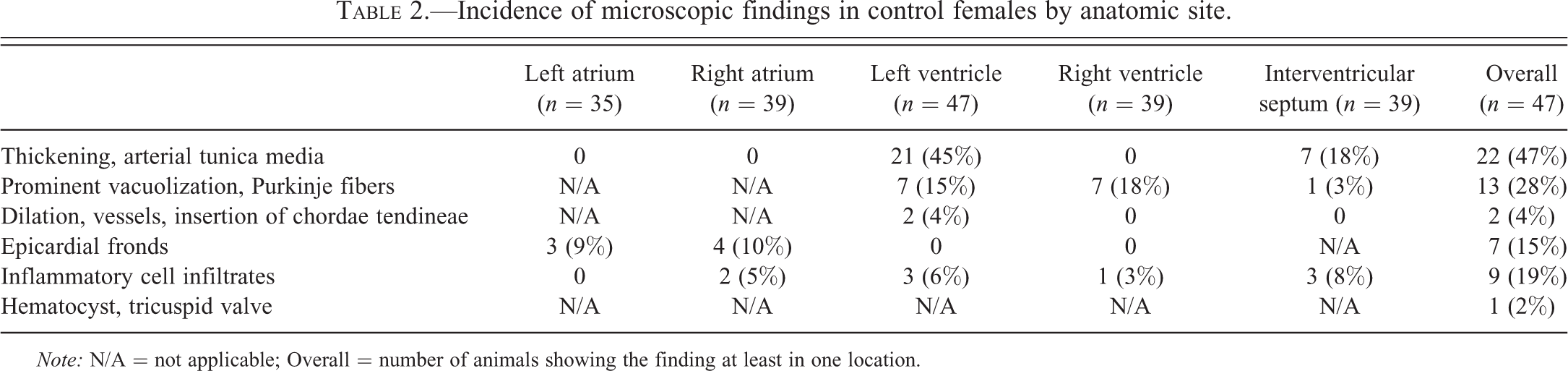
Note: N/A = not applicable; Overall = number of animals showing the finding at least in one location.
The second most common finding in both males and females was the presence of prominent vacuoles within Purkinje fibers (Figure 4). This finding was present in 14 of 41 males (34%) and in 13 of 47 females (28%). These cytoplasmic vacuoles were generally large (often >50% of the total cross-sectional diameter of the cell), round, clear, and centrally located. The Purkinje fibers exhibited a strong cytoplasmic reactivity for α-actin and desmin and PGP 9.5 (Figures not shown), as well as strong cytoplasmic and marginal reactivity for dystrophin (see Figure 5). The Purkinje fibers had abundant granular PAS-positive material in the cytoplasm and also the vacuoles contained PAS-positive granular material (see Figure 6). The vacuoles were negative for LAMP-2 and adipophilin (Figures not shown).
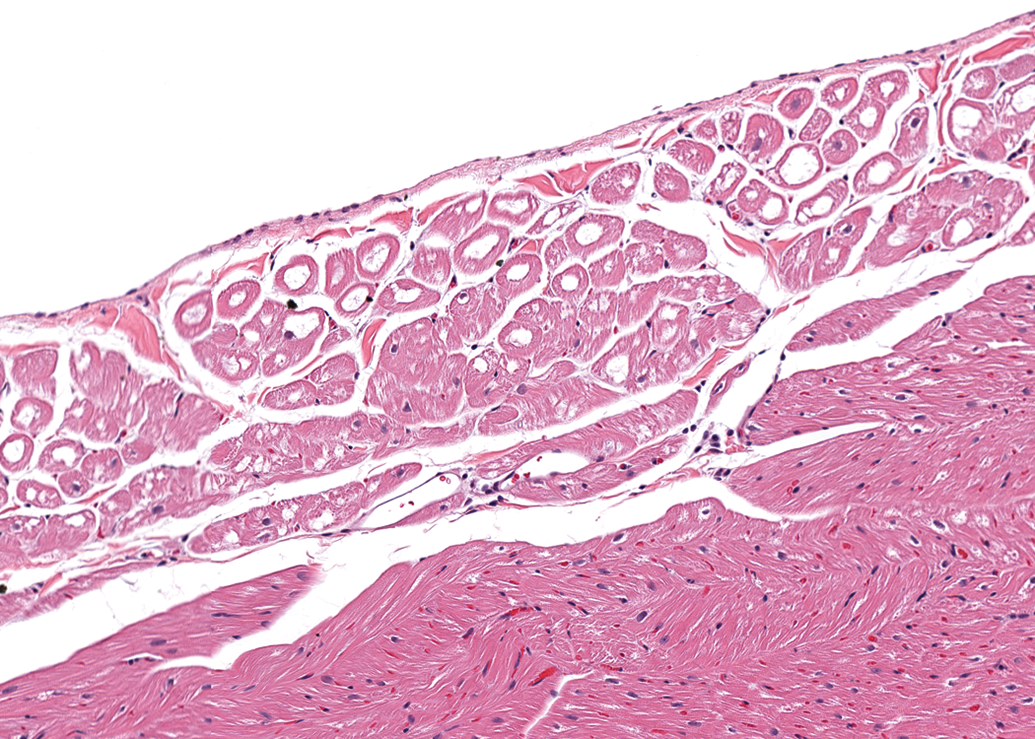
Right ventricle, male dog. Prominent distinct round vacuoles in Purkinje fibers subjacent to the endocardium. H&E. 164×.
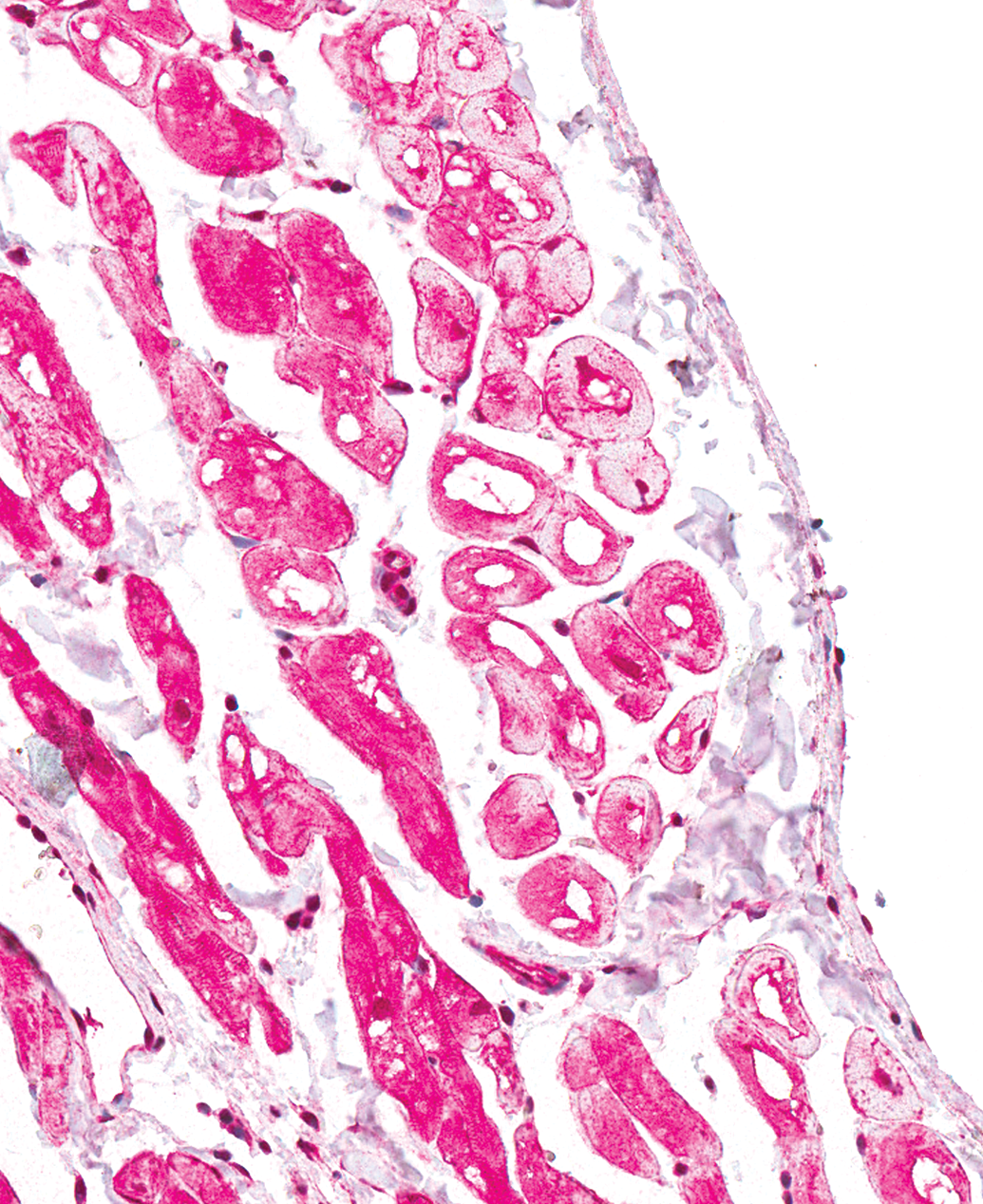
Right ventricle, male dog. Prominent distinct round vacuoles in Purkinje fibers subjacent to the endocardium. Dystrophin Immunohistochemical stain. 200×.
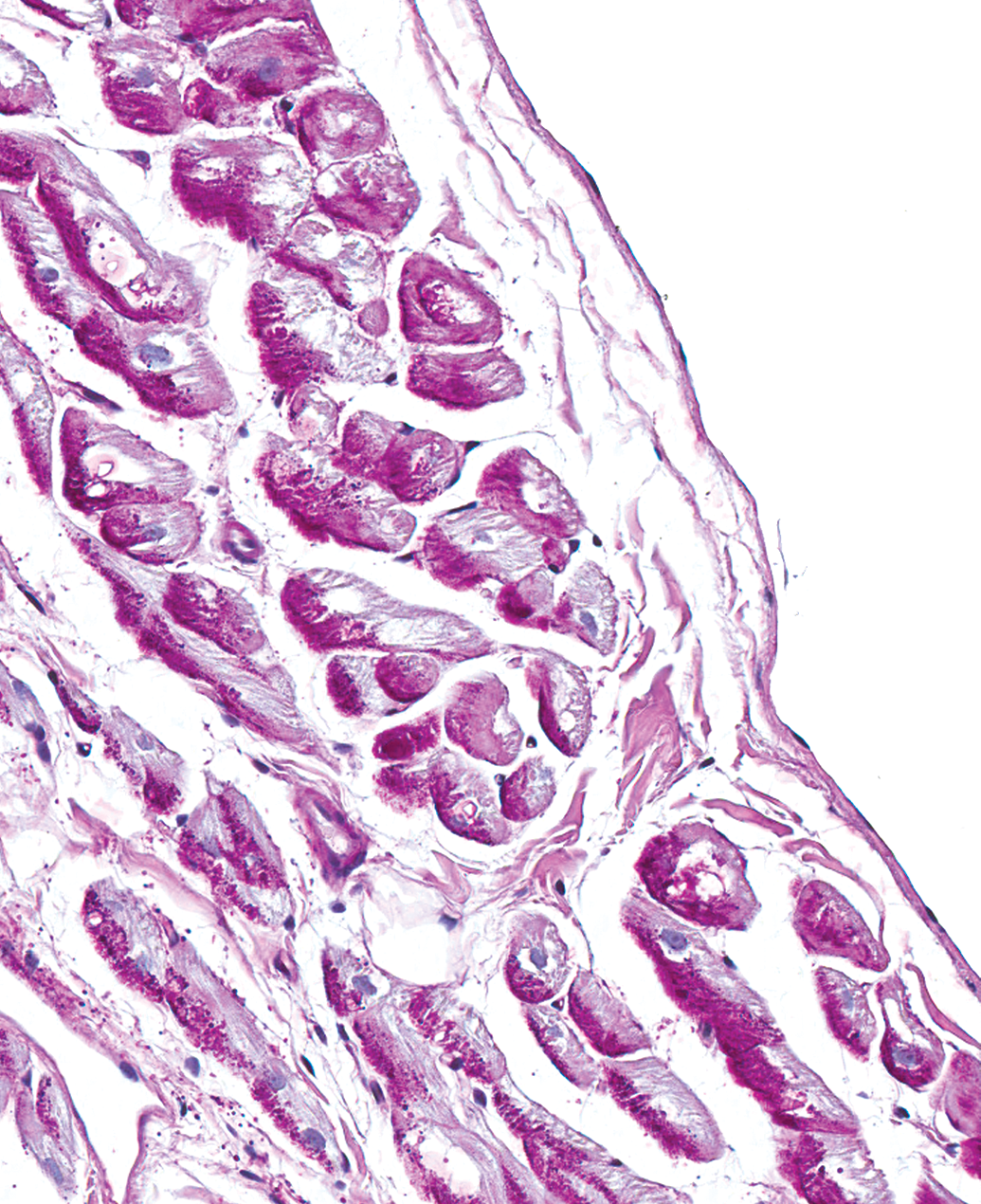
Right ventricle, male dog. Prominent distinct round vacuoles in Purkinje fibers subjacent to the endocardium. Periodic Acid-Schiff (PAS). 200×.
Epicardial fronds (Figure 7) on the surface of either atrium were another common finding, with a slight predilection for males (11 of 32 [34%] males in which at least one atrium was examined) over females (7 of 39 [18%]). These fronds were composed of a poorly vascular, loose collagenous stroma lined by a single layer of low cuboidal mesothelium.
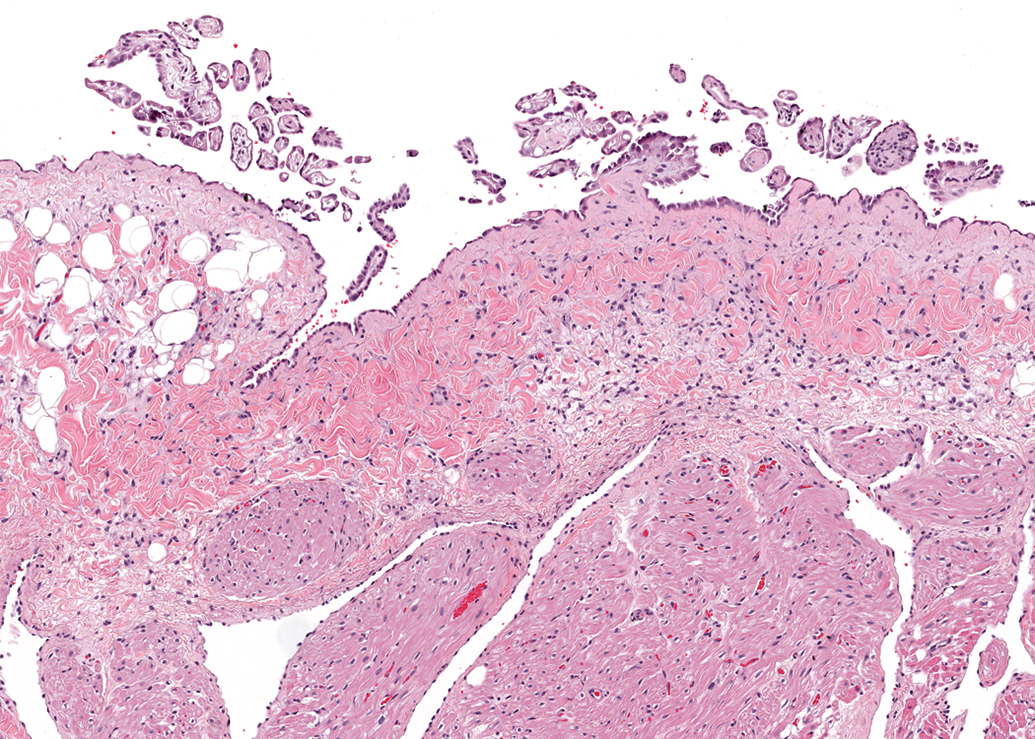
Right atrium, male dog. Papillary fronds lined by a layer of mesothelial cells along the epicardial surface. H&E. 98×.
Dilated lymphatic vessels and blood vessels at the insertion of the mitral valve’s chordae tendineae into the left papillary muscle (Figure 8) were also observed more frequently in males than in females. This finding was noted in 11 of the 40 males in which the left ventricle was examined (28%) and in 2 of the 47 females (4%). Occasional vessels contained scant numbers of erythrocytes, while the remainder was empty. These vessels were lined by an inconspicuous layer of endothelium.
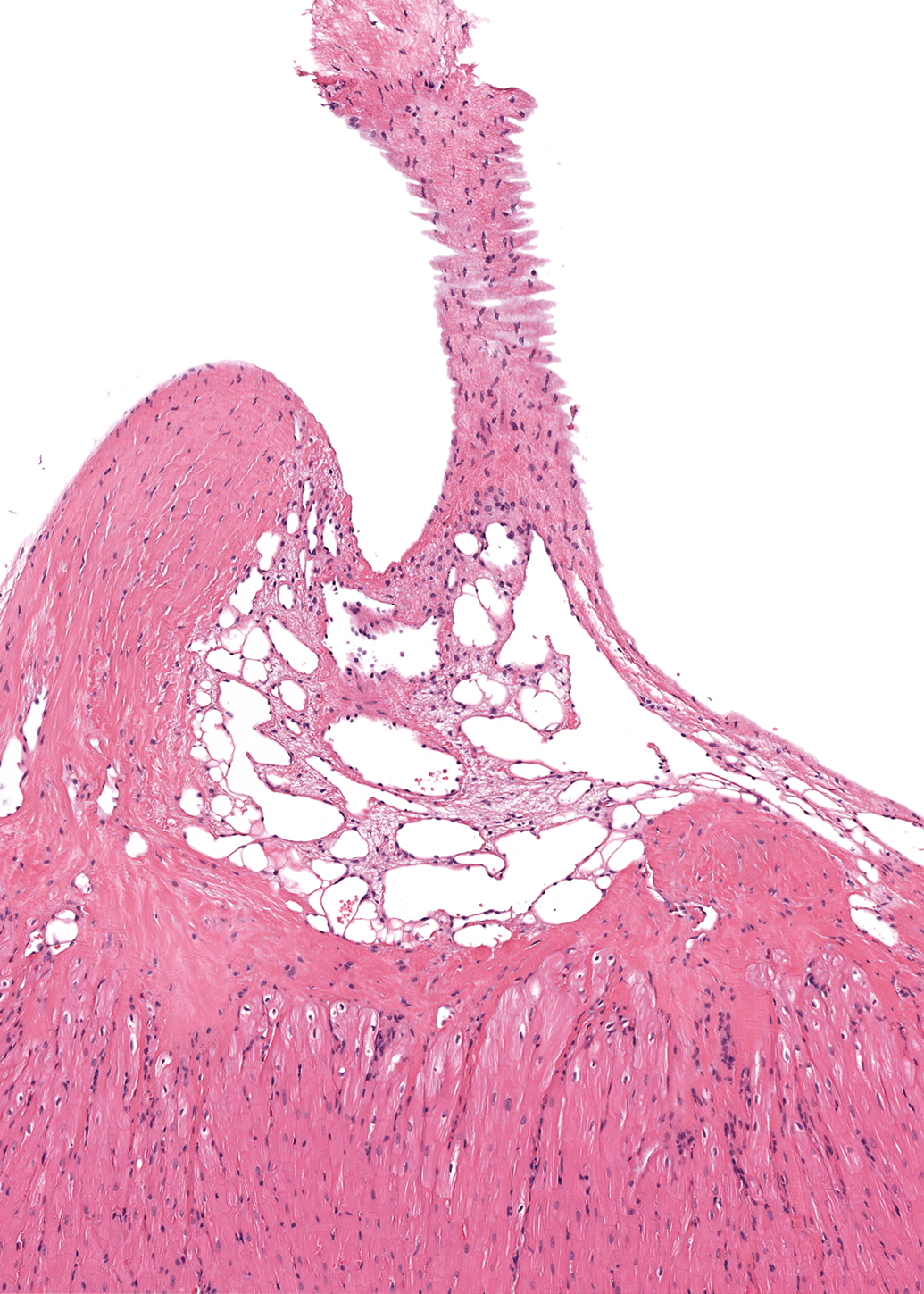
Chorda tendinea and left papillary muscle, male dog. Dilated lymphatic vessels and blood vessels are present at the point of insertion of a chorda tendinea into the papillary muscle. H&E. 74×.
Small foci of inflammatory cell infiltrates, usually solitary, were occasionally present in the myocardium at all sites except the left atrium (Figure 9). These foci consisted mostly of macrophages and lymphocytes. Four of the 41 males (10%) and 8 of the 47 females (17%) were affected. Similar mononuclear cell infiltrates were also present in the epicardial fat and in the subendocardium in 2 females and in 1 female, respectively. An additional female had a mild focal infiltrate of eosinophils, macrophages, lymphocytes, and plasma cells within a mitral valve leaflet (Figure 10).
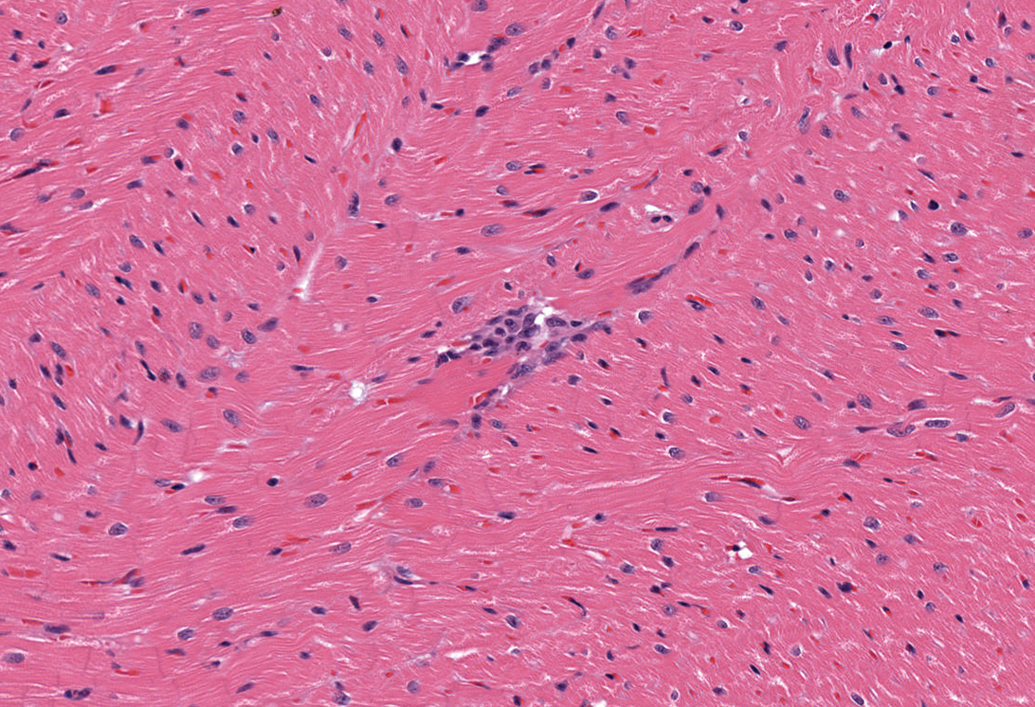
Left ventricle, female dog. A small focus of mononuclear inflammatory cells (primarily macrophages and lymphocytes). H&E. 200×.
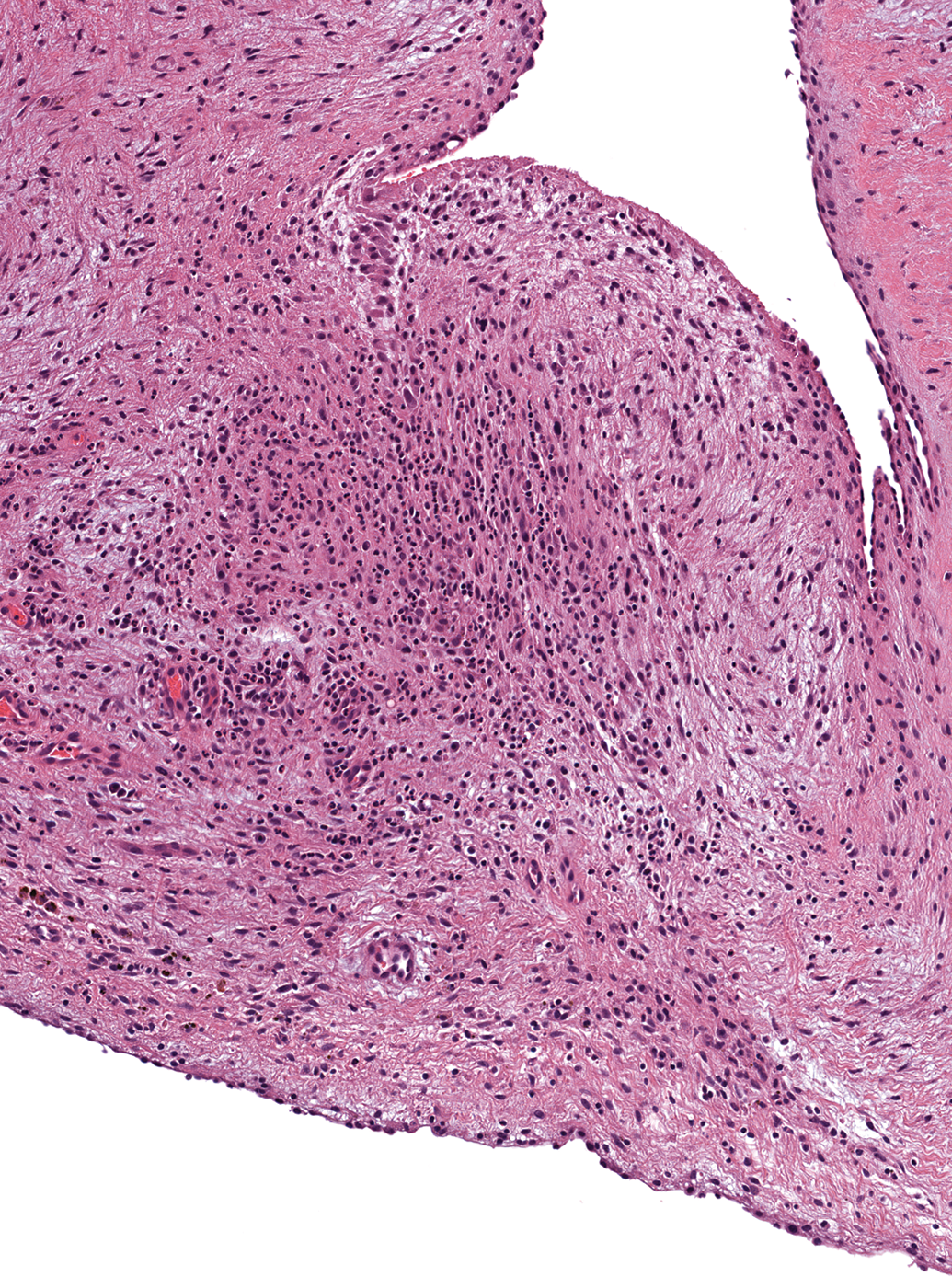
Mitral valve, female dog. Infiltration by a mixed inflammatory cell population composed of eosinophils, macrophages, lymphocytes, and plasma cells. H&E. 96×.
The following findings were observed in 1 animal each: focal mineralization of the aortic tunica media, focal mineralization of dense connective tissue within the papillary muscle, and a hematocyst on a tricuspid valve leaflet (Figure 11).
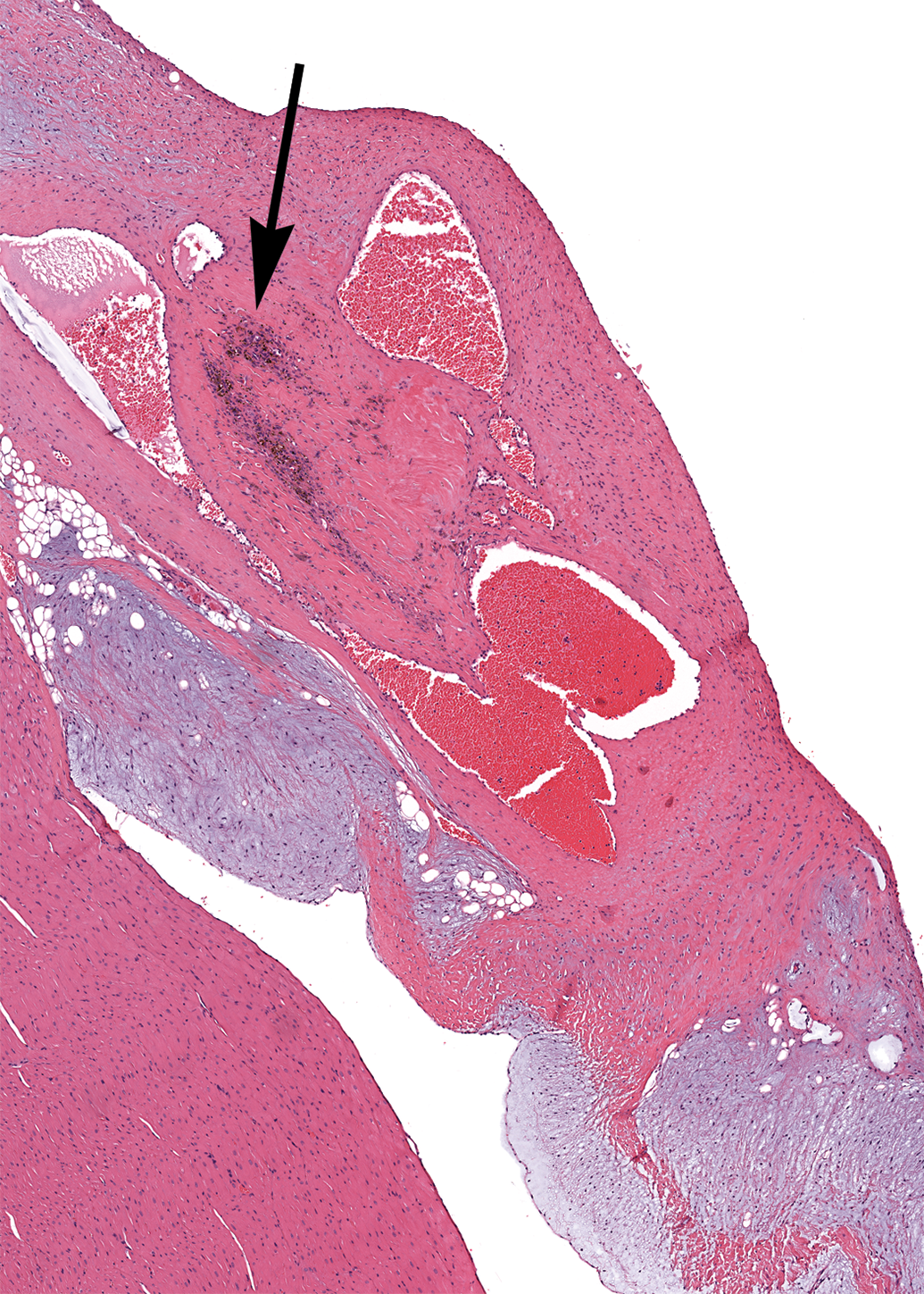
Tricuspid valve, female dog. A hematocyst with dilated vascular spaces filled with blood. A collection of hemosiderin-laden macrophages is present (arrow). H&E. 26×.
Discussion
Histologic examination of the heart is an essential component of preclinical toxicity studies performed in beagle dogs. Test item–related heart findings in dogs can result in the discontinuation of development of a drug before human trials begin. Distinguishing such test item–related findings from spontaneous findings, agonal changes, and artifacts is critical for accurate risk assessment. The study pathologist must have knowledge of incidental background findings, so they do not incorrectly identify them as test item–related findings.
In this retrospective analysis, hearts from control dogs on toxicity studies were examined. The most common finding was thickening of the tunica media of intramural coronary arteries. This finding may be the manifestation of postmortem hypercontraction rather than a true pathologic process (Keenan and Vidal 2006). Masson Goldner and Congo red stains were both negative, indicating that neither connective tissue nor amyloid deposition were responsible for medial thickening. Immunohistochemistry with Ki-67, a proliferation marker, did not show positive nuclei of smooth muscle cells of the media of affected vessels and the media of affected vessels was therefore not considered hyperplastic (Gerdes et al. 1983). Nevertheless, hyperplasia of the tunica media can be a true pathologic finding resulting from administration of vasoactive drugs like minoxidil (Mesfin et al. 1989). The presence of other correlating findings such as hemorrhage and vessel wall necrosis can help the pathologist determine if the arterial wall thickening is indeed a pathologic change. Vessel wall hemorrhage and necrosis were not observed in this retrospective analysis, and all instances of tunica media thickening were considered to have occurred postmortem.
Another incidental finding that may be difficult to differentiate from a true pathologic condition is Purkinje fiber vacuolization. Purkinje fiber vacuolization in the hearts of control dogs has been previously reported (Keenan and Vidal 2006). Regular round vacuoles are frequently present in normal Purkinje fibers in dogs and humans, and these vacuoles actually correspond with the regions of the sarcoplasm that contain few myofibrils (Urasawa et al. 2008). The cytoplasm of normal Purkinje fibers contains large quantities of glycogen (Samuelson 2007). The PAS stain of the Purkinje fibers showed PAS-positive granular material in the cytoplasm and in some vacuoles consistent with glycogen (McManus 1948). However, Urasawa et al. (2008) also describe a colony of dogs with a form of X-linked muscular dystrophy in which a different pattern of Purkinje fiber vacuolization exists. In affected dogs, Purkinje fiber vacuoles are more frequent and irregularly shaped and they lack full-length dystrophin. The vacuolated Purkinje fibers evaluated here showed strong cytoplasmic and marginal immunohistochemical reactivity for full-length dystrophin, which ruled out the presence of vacuolar degeneration in dystrophin-deficient canine Purkinje fibers (Urasawa et al. 2008). LAMP-2 staining is consistent with accumulation of phospholipid in lysosomes and adipophilin staining is consistent with lipid accumulation in the cytoplasm of cells (Obert et al. 2007). Both LAMP-2 and adipophilin staining were negative. In summary, staining results support the presence of glycogen in the vacuoles of Purkinje fibers.
Mesothelial-lined epicardial fronds are considered an incidental finding in beagle dogs. Their pathogenesis is uncertain, but they may arise in response to friction between the atria and pericardium (Mesfin 1990).
A frequent finding involving valves in this analysis was dilation of lymphatic and blood vessels at the insertion of the chordae tendineae of the mitral valve into the left papillary muscle. The connective tissue at the insertion of the chordae tendineae contains numerous tortuous lymphatic vessels in dogs (Uhley and Leeds 1976). Therefore, this should be considered a normal finding and should not be confused with a pathologic process. Indeed, it is possible that this finding would have been noted in almost all the dogs in this analysis if more extensive sampling of the papillary muscle had been done. Another incidental valvular finding in this analysis was the presence of a hematocyst on a tricuspid valve leaflet in 1 female. These hematocysts are believed to arise from the dilatation of valvular blood vessels and are not known to compromise valvular function except in rare cases (Takeda et al. 1991). Similarly, mineralization of the aortic tunica media is considered incidental and has been shown to increase in incidence with age among various breeds (Schwarz et al. 2002).
One commonly reported finding that was not noted in any of the dogs evaluated for this publication was arteritis affecting the extramural coronary arteries. This condition has been called canine juvenile polyarteritis syndrome, idiopathic canine polyarteritis, and beagle pain syndrome, among other terms (Clemo et al. 2003; Snyder et al. 1995). This spontaneous finding can also confound the interpretation of toxicity studies, particularly if there is a question of whether the test compound is vasoactive. Vascular injury induced by vasoactive drugs is often characterized by hemorrhage and may be localized to the coronary arteries (Joseph 2000), whereas idiopathic canine polyarteritis is less likely to involve hemorrhage and is more likely to affect arteries in other tissues in addition to those in the heart (Clemo et al. 2003; Son 2004).
One limitation of this retrospective evaluation was that tissue sectioning was not consistent across studies. Six sections of heart were available from 70 dogs, but only one heart section (left ventricle) was available from 16 dogs (8 males and 8 females) from some of the older toxicity studies.
In conclusion, the hearts of beagle dogs frequently have incidental findings and postmortem changes that could be confused with pathologic processes. In this retrospective evaluation, there were few true pathologic findings; most of the findings such as Purkinje fiber vacuolization and thickening of arterial walls were in fact normal features or postmortem changes. If available and robust, historical control data can be used to support the pathologist’s interpretation of a histopathologic finding as incidental or test item–related.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Abbreviations
Acknowledgments
We would like to thank Jackie O'Connor for histology services, Cordula Geschwill and Katrin Goetzke for the immunohistochemistry and the special stains, and Amanda Niquette for the operation of our ScanScope system.
