Abstract
This study aimed to evaluate the carcinogenic potential of the herbicide Diuron in a two-stage rat medium-term mammary carcinogenesis model initiated by 7,12-dimethylbenz(a)anthracene (DMBA). Female seven-week-old Sprague-Dawley (SD) rats were allocated to six groups: groups G1 to G4 received intragastrically (i.g.) a single 50 mg/kg dose of DMBA; groups G5 and G6 received single administration of canola oil (vehicle of DMBA). Groups G1 and G5 received a basal diet, and groups G2, G3, G4, and G6 were fed the basal diet with the addition of Diuron at 250, 1250, 2500, and 2500 ppm, respectively. After twenty-five weeks, the animals were euthanized and mammary tumors were histologically confirmed and quantified. Tumor samples were also processed for immunohistochemical evaluation of the expressions of proliferating cell nuclear antigen (PCNA), cleaved caspase-3, estrogen receptor-α (ER-α), p63, bcl-2, and bak. Diuron treatment did not increase the incidence or multiplicity of mammary tumors (groups G2 to G4 versus Group G1). Also, exposure to Diuron did not alter tumor growth (cell proliferation and apoptosis indexes) or immunoreactivity to ER-α, p63 (myoephitelial marker), or bcl-2 and bak (apoptosis regulatory proteins). These findings indicate that Diuron does not have a promoting potential on mammary carcinogenesis in female SD rats initiated with DMBA.
Introduction
Breast cancer is an invasive and potentially fatal disease whose incidence in postmenopausal women has gradually increased in most Western societies over the past few decades. The incidence has also sharply increased in younger women in more recent years, mainly in industrialized countries (Bouchardy et al. 2007; Parkin et al. 2005).
Besides genetic/familial factors including the major susceptibility genes (BRCA1 and BRCA2), other influences on breast cancer risk appear to be certain reproductive factors (i.e., older age, later age at first full-term pregnancy, no full-term pregnancies), body size/obesity, alcohol, exogenous hormones (oral contraceptives, hormone replacement therapy), menopause, and possibly, some dietary habits; Parkin et al. 2005). In the early 1990s, it was suggested that exposure to some environmental chemicals such as pesticides could play a causal role in the etiology of breast cancer through estrogen-related pathways or endocrine disruption effects. Substantial evidence from experimental rodent studies indicates that organochlorine pesticides are potential mammary carcinogens. In contrast, direct correlations between human tissue levels of pesticides and the development of breast cancer have not been consistent, with positive and negative associations (Calle et al. 2002; Fenton 2006; Rudel et al. 2007; Salehi et al. 2008).
Diuron (3-[3,4-dichlorophenyl]-1-1-dimethylurea) is a substituted phenyl urea herbicide used throughout the world to control a wide variety of annual and perennial broadleaf and grassy weeds on both crop (i.e., citrus fruit, cotton, asparagus, sugar cane, alfalfa, wheat, and grapes) and noncrop sites, such as roads, garden paths, and railways (Field et al. 2003; Giacomazzi and Cochet 2004; Iyer 2002). In Brazil, it is widely used on many agricultural crops such as sugar cane and soy (Nascimento et al. 2006). Diuron per se has low systemic toxicity to mammals and birds and moderate toxicity to aquatic invertebrates. Its principal biodegradation product, 3-4-dichloroaniline (3-DCA), is highly toxic and relatively persistent in the environment (Iyer 2002; Giacomazzi and Cochet 2004; Valentovic et al. 1997). Thus, environmental contamination by this herbicide may represent an important public health problem (Abass et al. 2007; Sorensen et al. 2008).
In a recent epidemiological study in areas of high agricultural pesticide use, no association between Diuron exposure and breast cancer was observed in Californian women (Reynolds et al. 2005). However, for more than a decade, Diuron has been categorized as a “known/likely” human carcinogen by the U.S. Environmental Protection Agency (US EPA), mostly based on long-term bioassays that indicated increased incidences of urothelial bladder and renal pelvis tumors after continuous dietary high-concentration (2500 ppm) exposure in both sexes of Wistar rats, and a trend toward increased incidence of mammary adenocarcinomas in female NMRI mice (Iyer 2002; US EPA 2003; US EPA 2004). In these long-term studies, female NMRI mice showed increased, although relatively low, incidences of adenocarcinomas: two of fifty, one of forty-seven, one of forty-nine, and six of fifty after exposure through diet to 0, 25, 250, and 2500 ppm of Diuron, respectively (Iyer 2002). The 12% incidence of mammary tumors led the US EPA to assume that a positive oncogenic response was seen in the highest dose group when compared to the control, after discarding the possibility of those tumors being spontaneous (US EPA 2003). Recently, we demonstrated that Diuron does not affect cell proliferation in the non-altered mammary glands of female Swiss mice (de Moura et al. 2010). Although the carcinogenic mode of action of Diuron on the urinary bladder urothelium of the rodent has been studied (Nascimento et al. 2006; de Moura et al. 2010; da Rocha et al. 2010), the mammary carcinogenesis process referred to Diuron exposure has not been adequately explored in a strain of rat susceptible to mammary carcinogenesis.
The most widely used medium-term bioassay to assess the carcinogenic potential of chemicals on mammary carcinogenesis is based on 7-12-dimethylbenz(a)anthracene (DMBA)–induced tumors in the highly susceptible female Sprague-Dawley rat, whose tumors closely mimic human breast cancer (Costa et al. 2002; Russo and Russo 1996). This in vivo bioassay is useful for dissecting the multistep process of carcinogenesis and for detection of potential mammary carcinogens, especially those that act through endocrine disruption (Costa et al. 2002; Fenton 2006; Rudel et al. 2007; Russo and Russo 1996). Thus, the identification of the potential mode of action as well as possible species-specific responses of laboratory rodents may assist in the selection and further development of appropriate models for evaluation of mammary carcinogenicity of Diuron.
As breast cancer is one of the most frequent neoplasms in women and hazard detection and risk assessments of environmental chemicals for breast cancer are high public health priorities, the present study was conducted to assess the modifying effects of the herbicide Diuron on the promotion/progression stages of mammary carcinogenesis induced by DMBA in virgin female Sprague-Dawley (SD) rats. In addition, effects of Diuron on cell proliferation and apoptosis biomarkers were also examined.
Material and Methods
Chemicals
7,12-Dimethylbenz(a)antracene (CAS 57-97-6, ~ 95%) and Diuron (CAS 30-54-1, analytical standard grade, ≥ 98%) were purchased from Sigma-Aldrich Co., USA.
Experimental Design
The University Committee for Ethics in Animal Research approved the present study (Protocol number 523). Female five-week-old SD rats were obtained from the Multidisciplinary Center for Biological Investigation (CEMIB/UNICAMP, Campinas-SP, Brazil). They were kept in polypropylene cages (three to five animals/cage) with metallic grid covers and maintained in a room at 22°C ±2°C, 55% ±10% humidity and a twelve-hour light/dark cycle. They were fed commercial Purina chow (Paulínia, SP, Brazil) and water ad libitum during a two-week acclimatization period.
After acclimatization, the animals were randomized based in age (fifty-one days old) and body weight values, allocated to six groups consisting of eighteen rats in groups G1 to G4, ten rats in group G5, and fifteen rats in group G6. Groups G1 to G4 were initiated for mammary carcinogenesis by a single i.g. dose of 50 mg/kg body weight of DMBA; groups G5 and G6 received a single i.g. dose of canola oil (DMBA vehicle, 1 mL/kg). After one week, groups G1 and G5 were kept in the commercial basal diet, and groups G2, G3, G4, and G6 received the same diet with the addition of 250, 1250, 2500, or 2500 ppm of Diuron, respectively, for twenty-five weeks. During the experimental period, the animals were carefully checked once a week for the presence of gross mammary tumors; data on the number and localization of each palpable mass in the six mammary gland complexes were recorded. Sacrificed moribund rats and deceased animals were autopsied; those surviving for twenty-three or more weeks of Diuron treatment were included in the effective number of rats for histopathology and immunohistochemistry analysis. All animals were euthanized by exsanguination under sodium pentobarbital anesthesia (45 mg/kg b.w.). Individual body weights and food consumption were recorded weekly during the experimental period. The food consumption measured was registered as an average of the whole cage consumption.
Hormone Analysis and Tissue Processing for Histology
Immediately before sacrifice, samples of peripheral blood were collected for estrogen and progesterone plasma determinations. Estrogen and progesterone levels were determined automatically (VITROS ECi-Johnson and Johnson Ultra-Sensitive Chemiluminescence analysis, USA) using specific reagents supplied by Johnson and Johnson Orthoclinical (São Paulo-SP, Brazil). At necropsy, the whole skin with mammary glands and tumors, liver, kidneys, spleen, ovaries, uterus, and vagina were removed and fixed for twenty-four hours in 10% phosphate-buffered formalin. Before fixation, mammary tumors, liver, kidneys, spleen, ovaries, and uterus were weighed. Samples of organs/tissues were processed to provide 5-µm–thick paraffin sections for histological (hematoxylin-eosin [HE] straining) and immunohistochemical analysis.
Proliferative or neoplastic lesions in mammary glands and in the reproductive system (i.e., oviduct, ovary, uterus, cervix, and vagina) were classified according to published criteria by The Society of Toxicologic Pathology (SSNDC 2006).
Proliferating Cell Nuclear Antigen, Caspase-3, p63, RE, bcl-2, and bak Immunostaining
Histological sections were put on poly-l-lysine–coated slides, deparaffinized, and rehydrated with graded alcohol. Sections were subjected to microwave antigen retrieval in citric acid buffer at pH 6.0 for three times five minutes (PCNA, cleaved caspase-3, p63, bcl-2, and bak primary antibodies) or Pascal pressure chamber retrieval in citrate acid buffer at pH 6.0 at 120°C for three minutes (ER-α antibody). Endogenous peroxidase was blocked with 3% H2O2 in phosphate-buffered saline (PBS) for ten minutes in the dark. After washing with PBS, slides were incubated with nonfat milk in PBS for sixty minutes. Sections were then incubated with primary antibodies mouse monoclonal anti-PCNA/PC10, 1:200 dilution (DakoCytomation Denmark A/S, Glostrup, Denmark); rabbit polyclonal cleaved anti-caspase-3/Asp 175 rabbit, 1:100 dilution (Cell Signalizing Technology, Inc., Danvers, MA, USA); mouse monoclonal anti-ER-α/6F11, 1:50 dilution (BioCare Medical, Concord, CA, USA); monoclonal mouse anti-p63/4A4, 1:100 dilution (Dako Cytomation); and anti-polyclonal rabbit anti-bcl2/N-19, 1:200 dilution, and polyclonal rabbit anti-bak/G-23, 1:200 dilution (Santa Cruz Biotechnology) overnight. This step was followed by biotinylated secondary antibodies horse anti-mouse or goat anti-rabbit (Vector Laboratories Inc., Burlingame, CA, USA) for sixty minutes and incubated with performed streptoavidin-biotin complex/horseradish peroxidase (Vector Laboratories), both at room temperature. Chromogen color development was accomplished with 3,3′-diaminobenzidine tetrahydrochloride (DAB, Sigma-Aldrich Co.) as the substrate to show the sites of peroxidase binding. The slides were counterstained with Harris's hematoxylin. A negative control was performed in all cases by omitting incubation with the primary antibodies for PCNA, cleaved caspase-3, ER-α, p63, bcl-2, and bak, which in all instances resulted in negative immunoreactivity.
The PCNA S-phase and the cleaved caspase-3 labeling and apoptosis indexes (HE staining) in mammary tumor sections were calculated as the number of positively marked cells divided by the total number of cells scored × 100 (ten representative microscopic fields without necrosis with about 10,000 tumor cells in each tumor section). The immunore activities for p63, ER-α, bcl-2, and bak were evaluated using a score system. The score was based on estimating both the percentage of positively stained cells on a slide (proportion score) and the general strength of the staining (intensity score). The proportion score was ranked as 0 for negative, 1 for <10%, 2 for >10% and <50%, and 3 for >50% of cells stained. The intensity score was ranked as 1 for weak, 2 for moderate, and 3 for strong expression. The combined score was represented by the product of the proportion score and the intensity score (Ip et al. 2000).
Statistical Analysis
Data for body weight and body-weight gain, food consumption, relative liver kidney and spleen weights, tumor weights, and estrogen and progesterone plasma levels were analyzed by analysis of variance when the results showed normal distribution or the Kruskal-Wallis test when they did not. Analyses of the semiquantitative combined scores were performed using the Kruskal-Wallis test. Contrast among groups was analyzed by the Tukey or Student-Newman-Keuls methods. Incidences of mammary tumor were examined using the chi-square or the Fischer test. Significant differences were assumed when p < .05. The statistical analyses were performed using Jandel Sigma Stat software for Windows Version 3.5, 2006 (Jandel Corporation, San Rafael, CA, USA).
Results
Mortality, Body and Organ Weights, Food Consumption, and Hormonal Analysis
At the end of the experimental period, survival rates were 72.2% in DMBA-initiated group (G1, n = 13); and 77.8%, 88.9%, and 83.3% in the DMBA-initiated and Diuron-treated groups that received 250 (G2, n = 14), 1250 (G3, n = 16), and 2500 ppm (G4, n = 15), respectively, and survival rates were 100% in both non-initiated groups (G5, n = 10, and G6, n = 15, respectively; Figure 1A and Table 1 ). Despite a loss of almost 30% of the rats in the DMBA-initiated group (G1) by the end of the study, the incidence of palpable mammary tumors was not diferent among DMBA-initiated groups during the experimental period (Figure 1B).
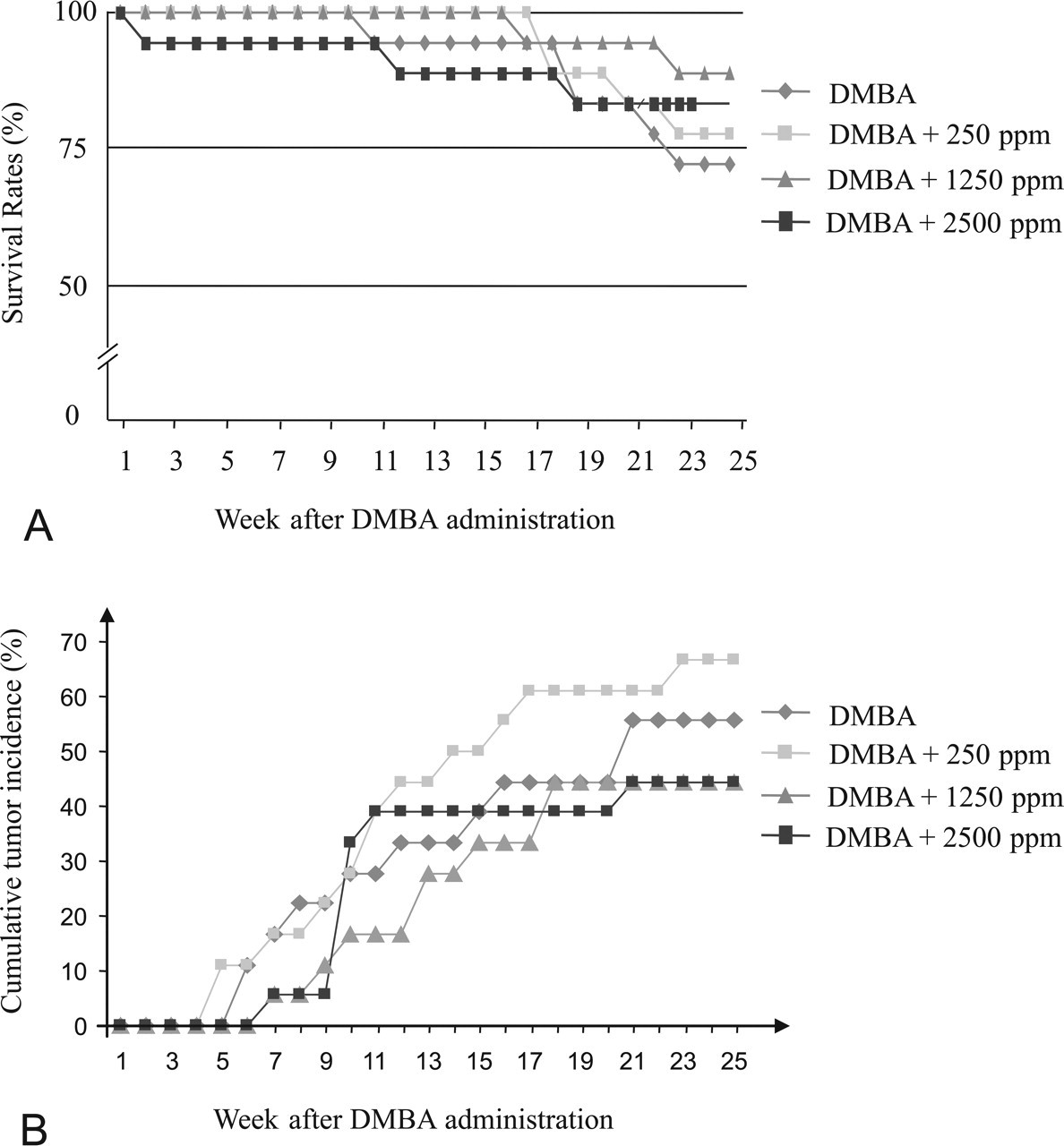
(A) Survival rate (%) of female Sprague-Dawley rats initiated by 7,12-dimethylbenz(a)anthracene. (B) Cumulative tumor incidence (%) calculated from tumors in all mammary gland complexes. Only neoplasms that were subsequently histologically verified as mammary tumors are shown.
Final body weights, body-weight gain, food and Diuron consumption, and feed efficiency of different experimental groups at the end of the twenty-five–week experiment.1
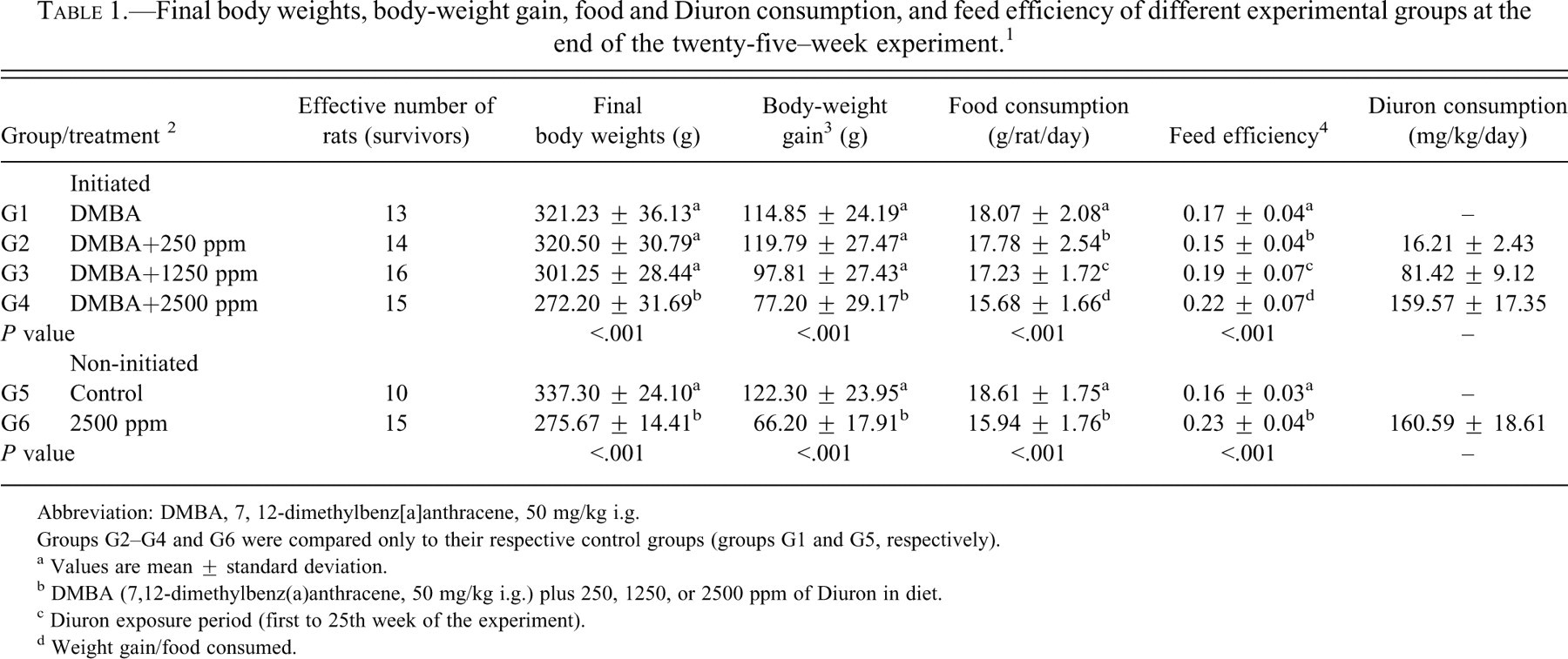
Abbreviation: DMBA, 7, 12-dimethylbenz[a]anthracene, 50 mg/kg i.g.
Groups G2–G4 and G6 were compared only to their respective control groups (groups G1 and G5, respectively).
a Values are mean ± standard deviation.
b DMBA (7,12-dimethylbenz(a)anthracene, 50 mg/kg i.g.) plus 250, 1250, or 2500 ppm of Diuron in diet.
c Diuron exposure period (first to 25th week of the experiment).
d Weight gain/food consumed.
During Diuron treatment, food consumption and body-weight gain (Figure 2 ) were significantly reduced (p < .001) in the DMBA-initiated/Diuron-treated 1250 and 2500 ppm and non-initiated/Diuron-treated 2500 ppm groups (G3, G4 and G6, respectively) when compared to the respective control groups (Table 1). At week 25, DMBA-initiated/Diuron-treated 2500 ppm and non-initiated/Diuron-treated 2500 ppm groups (G4 and G6, respectively) had lower final body weights (p < .001) than their respective control groups (Table 1). Increased relative liver and spleen weights (p < .001) were observed in the DMBA-initiated/Diuron-treated 1250 and 2500 ppm groups (G3 and G4) and in the non-initiated/Diuron-treated 2500 ppm group (G6; Table 2 ). Increased relative right and left kidney weights (p = .026; p = .006, respectively) and decreased relative left ovary weight (p = 0.004) were observed in non-initiated/Diuron-treated 2500 ppm group (G6) when compared to the non-initiated group (G5; Table 2). Diuron treatment did not cause any significant alterations inestrogen (pg/mL) or progesterone (ng/mL) plasma levels when compared to the respective control groups (Table 3 ).
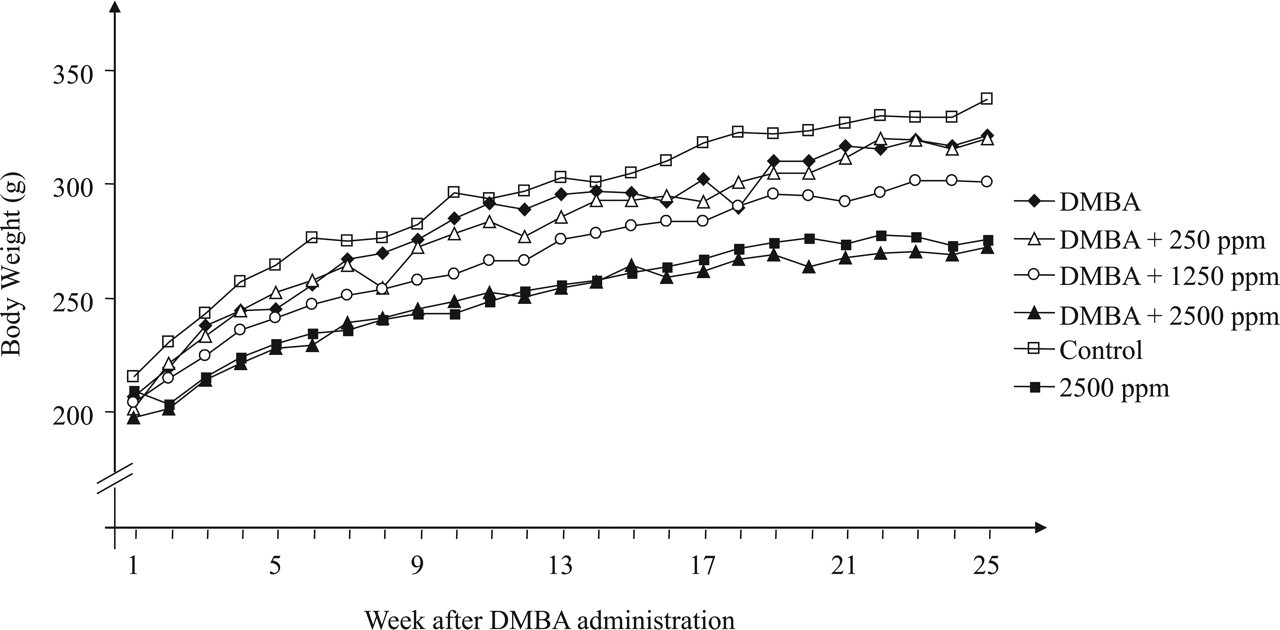
Evolution of body weight in different groups during the experimental period.
Organ relative weights of the different experimental groups at the end of experiment. 1
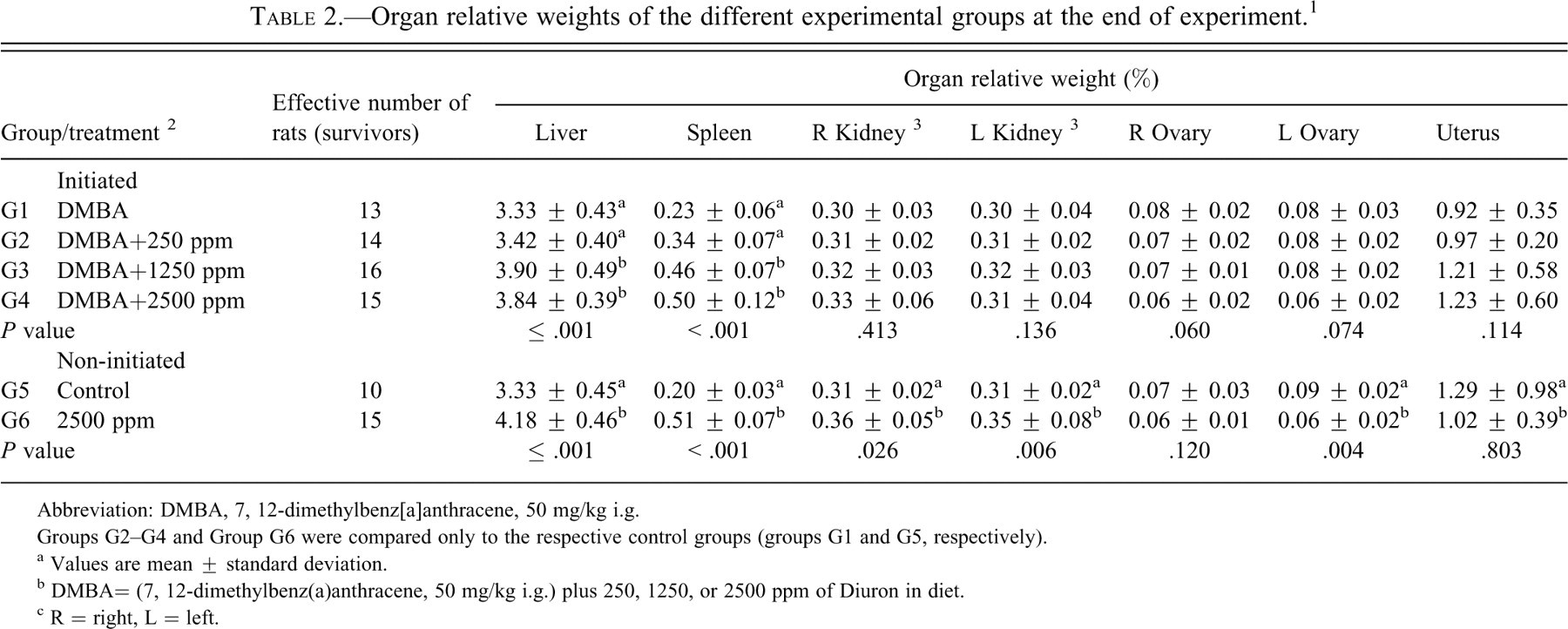
Abbreviation: DMBA, 7, 12-dimethylbenz[a]anthracene, 50 mg/kg i.g.
Groups G2–G4 and Group G6 were compared only to the respective control groups (groups G1 and G5, respectively).
a Values are mean ± standard deviation.
b DMBA= (7, 12-dimethylbenz(a)anthracene, 50 mg/kg i.g.) plus 250, 1250, or 2500 ppm of Diuron in diet.
c R = right, L = left.
Hormonal plasma levels and mammary tumor data of DMBA-initiated groups.
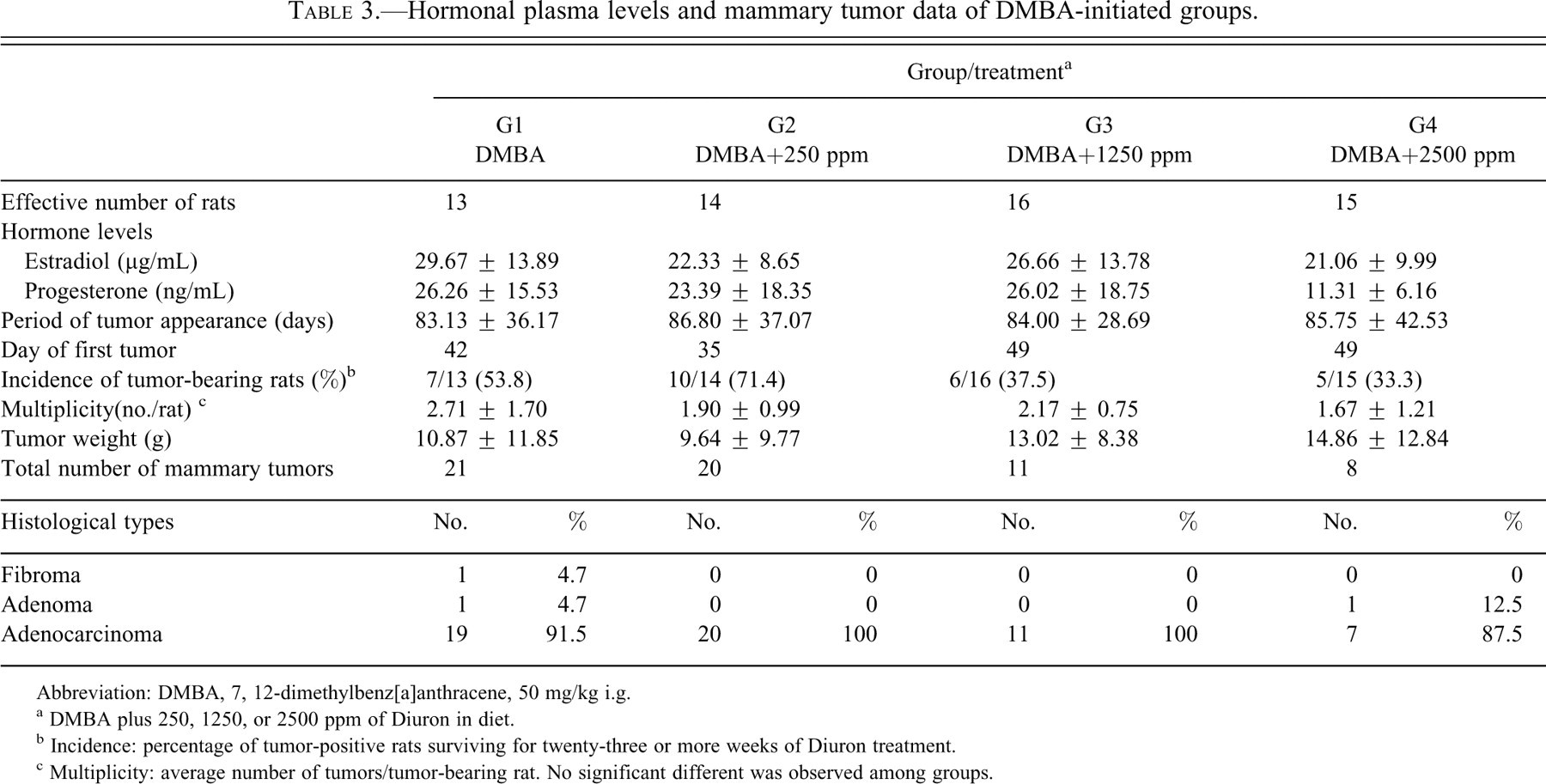
Abbreviation: DMBA, 7, 12-dimethylbenz[a]anthracene, 50 mg/kg i.g.
a DMBA plus 250, 1250, or 2500 ppm of Diuron in diet.
b Incidence: percentage of tumor-positive rats surviving for twenty-three or more weeks of Diuron treatment.
c Multiplicity: average number of tumors/tumor-bearing rat. No significant different was observed among groups.
Histopathologic and Immunohistochemical Analysis
Sixty mammary tumors were histologically confirmed in the DMBA-initiated groups. Most of them were classified as adenocarcinomas with either tubular, papillary, or comedo-cribriform patterns or mixed structures (fifty-seven of sixty); the others were adenomas (two of sixty) and fibroma (one of sixty; Table 3). The prevailing histological tumor pattern was considered for classification. In general, adenocarcinomas induced by DMBA presented an expansive pattern with local invasive areas. In non-initiated/Diuron treated 2500 ppm animals (G6), no hyperplastic lesion (lobular or atypical) or benign or malignant neoplasms were observed in the mammary gland complexes.
Treatment with Diuron did not alter the latency period (i.e., time to first palpable mammary tumor), incidence, multiplicity, tumor weights, or histological patterns in female SD rats initiated by DMBA (Table 3). Also, malignant mammary neoplasms were further characterized by a number of immunohistochemical markers such as PCNA, cleaved caspase-3, p63 (myoepithelial marker), bcl-2 and bak (apoptosis regulatory proteins), and ER-α (Figure 3A–F ). The values of cleaved caspase-3–positive apoptotic cells correlated well with the corresponding values of HE-stained apoptotic cells (Figure 3B1). Thus, apoptotic indexes (AI%) were determined through both positive staining for cleaved caspase-3 expression and morphological aspects (Eckle et al. 2004).
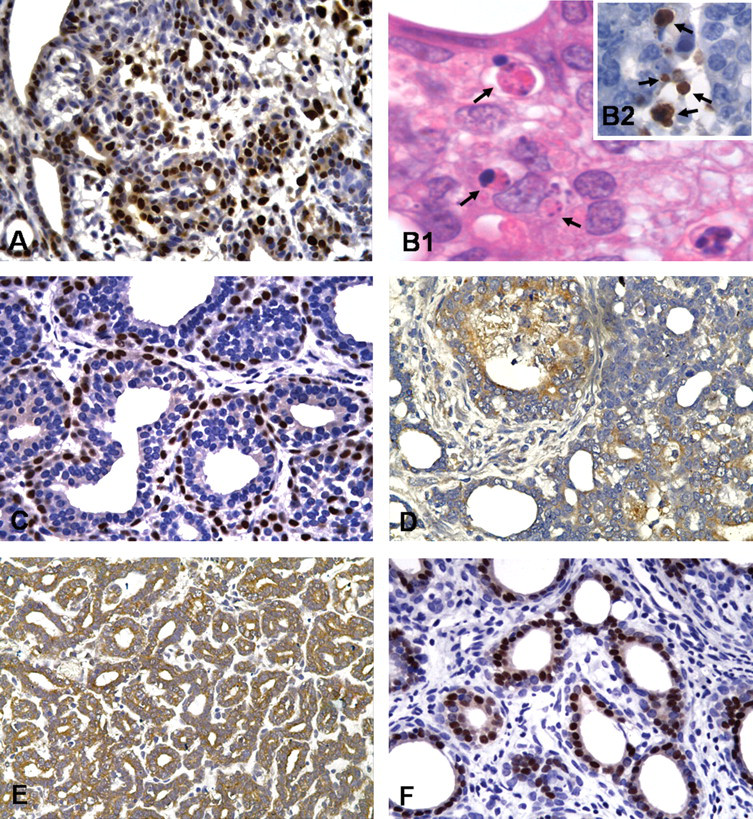
Immunohistochemically or HE-stained sections of mammary tumors from DMBA-initiated rats. (A) Proliferating cell nuclear antigen–positive neoplastic cells (brown nuclei, 40× objective). (B1and 2) Apoptosis cells identified in HE-stained section (black arrows, 100× objective) or by expression of caspase-3–cleaved (black arrows, 60× objective), respectively. (C) p63-Positive myoepithelial cells (brownnucleus; 40× objective). (D, E) Immunostaining for bak and bcl-2 mitochondrial proteins (brown-stained cytoplasm, 40× objective). (F) Immunostaining for ER-α (brown nuclei; 40× objective).
The different levels of Diuron in the diet did not change the indexes of cell proliferation (PCNA LI%) and apoptosis (AI%) in mammary tumors. Immunoreactivity for p63 nuclear protein and for apoptosis regulatory proteins bcl-2, bak, and ER-α were variably expressed in different regions of the tumor (central, periphery, or in proliferating or necrotic areas), irrespective of their histological patterns. The semiquantitative combined score system herein adopted indicated that treatment with Diuron also did not change the patterns of immunoexpression of p63, bcl-2, bak, or ER-α in the DMBA-induced neoplasms (Table 4 ).
Growth kinetic and expression immunohistochemical of biomarkers in mammary neoplasms from DMBA-initiated groups. a
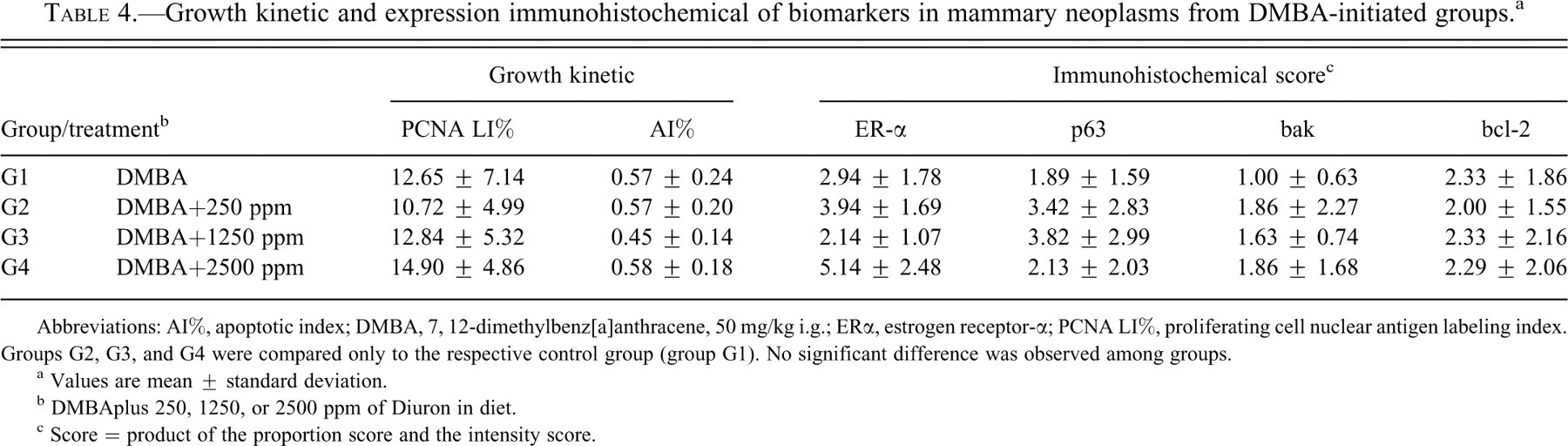
Abbreviations: AI%, apoptotic index; DMBA, 7, 12-dimethylbenz[a]anthracene, 50 mg/kg i.g.; ERα, estrogen receptor-α; PCNA LI%, proliferating cell nuclear antigen labeling index. Groups G2, G3, and G4 were compared only to the respective control group (group G1). No significant difference was observed among groups.
a Values are mean ± standard deviation.
b DMBAplus 250, 1250, or 2500 ppm of Diuron in diet.
c Score = product of the proportion score and the intensity score.
Some female SD rats exposed to Diuron 2500 ppm (groups G4 and G6) developed liver centrilobular hypertrophy, splenomegaly, and urothelial hyperplasia in the urinary bladder and renal pelvis. These changes were related to the influence of high concentrations of Diuron since they have been already reported in our previous studies (Nascimento et al. 2006; da Rocha et al.; 2010; de Moura et al. 2010).
Discussion
The results of the present study indicate that a twenty-five–week exposure to high dietary concentrations of Diuron does not modify the mammary carcinogenesis process initiated by DMBA in the highly susceptible female SD rat. Also, different Diuron exposure levels did not alter the histological patterns, the indexes of cell proliferation and apoptosis, and the immunoreactivity of ER-α, p63, bcl-2, and bak biomarkers in developing tumors. These findings were obtained using a dietary concentration of Diuron reported to be carcinogenic to the urinary bladder of male and female Wistar rats and to the mammary gland of female NMRI mice in long-term feeding studies (Iyer 2002; US EPA 2003; US EPA 2004).
Oral exposure to Diuron at 2500 ppm for twenty-five weeks also did not initiate the mammary carcinogenesis process, since no preneoplastic or neoplastic lesions were histologically detected in the mammary gland complexes of non-initiated female SD rats. It is not surprising that Diuron treatment alone did not initiate the carcinogenesis process in this organ. Although some reports in the literature suggest that Diuron is genotoxic (Agrawal et al. 1996; Agrawal and Mehrota 1997; Bouilly et al. 2007), studies performed in our laboratory indicated that the herbicide does not damage DNA in vivo and does not induce crosslinks in vitro (Nascimento et al. 2006; da Rocha et al. 2010) when tested by the comet (single-cell gel eletrophoresis) assay. Besides, Diuron did not exert initiating or promoting potentials in the liver of male Wistar rats or the skin of male Swiss mice (Ferrucio 2010; Grassi et al. 2007). These observations are in line with others reported in the literature stating that Diuron is a nongenotoxic agent (Iyer 2002; Liu 2002; US EPA 2003).
In the present study, oral exposure to Diuron did not exert any promoting effect on the mammary carcinogenesis process initiated by DMBA when latency, incidence, multiplicity and histological patterns were used as parameters. In fact, fewer, although not significantly fewer, tumors (tumor burden) were observed in DMBA-initiated groups fed higher dietary levels of Diuron (1250 and 2500 ppm). This finding could be directly associated to the reduction of body-weight gain (−15 % and −33%, respectively) and food consumption (−4% and −13%, respectively) observed in these two groups at the end of the experiment (Table 1). In fact, chemical treatments that decrease body-weight gain associated with toxicity, nonpalatability, or indirect food restriction are likely to inhibit the development of rodent mammary tumors in medium- and long-term bioassays (Rudel et al. 2007).
Cell proliferation and apoptosis play an important role during the progression of rat mammary gland tumorigenesis (Al-Dhaheri et al. 2008; Strange et al. 2001). Proliferating cell nuclear antigen, a co-factor for DNA-polymerase-delta leading to DNA replication and DNA damage repair, has been considered a feasible marker for cell proliferation in mammary carcinogenesis (Al-Dhaheri et al. 2008). Bcl-2 family members are pivotal in the regulatory processes that either repress (e.g., bcl-2 and bcl-x1) or induce (e.g., bak, bax, and bad) apoptosis (Brunelle and Letai 2009; Kuwana and Newmeyer 2003). In premalignant lesions and during mammary tumor progression in the female rat, increases in cell proliferation and imbalances of the anti-apoptotic bcl-2 and bcl-x and pro-apoptotic bax or bak proteins have been described (Al-Dhaheri et al. 2008; Shilkaitis et al. 2000; Strange et al. 2001; Xie et al. 1999). However, in the present study, Diuron did not significantly alter cell proliferation or apoptosis indexes or the expression of bcl-2 and bak proteins in DMBA-initiated mammary tumors.
p63 is a p53-related DNA-binding protein that has been described as a nuclear transcriptional factor involved with differentiation, maintenance, and proliferation of epithelial progenitor cells, or stem cells (Barbieri and Pietenpol 2006). This myoepithelial cell marker has been observed in normal and neoplastic tumors of both human and rat mammary tissues (Chan et al. 2005; Rakha et al. 2006; Ribeiro-Silva et al. 2005). In human breast cancer, p63 has been shown to be expressed in in situ ductal carcinoma and in benign neoplasms, but it is frequently absent in invasive lesions (Rakha et al. 2006; Ribeiro-Silva et al. 2005). In this study, p63 immunoreactivity was observed in basal cells of most neoplastic duct-like structures or in focal areas not directly lining epithelial mammary neoplastic cells. Thus, mammary tumors induced by DMBA in female SD rats can be constituted by a mixed cellular lineage (i.e., epithelial and myoepithelial cells) as previously described for mammary neoplasms induced by N-methyl-N-nitrosourea (Chan et al. 2005), and this structural pattern was not altered by exposure to Diuron.
In rodent studies, some pesticides with potential endocrine disruption properties have been categorized as mammary carcinogens (Rudel et al. 2007; Ueda et al. 2005). These pesticides, including some substituted urea herbicides, are able to block or inactivate steroid hormone receptors and/or affect the levels of pituitary hormones with potential to alter the development and function of the male and female reproductive systems (Bauer et al. 1998; Cook et al. 1993; Kojima et al. 2004; Noguerol et al. 2006; Vinggaard et al. 1999; Vinggaard et al. 2000). In vitro assays indicate that Diuron did not inhibit 5α-reductase activity but that it has the capability to connect to the androgen receptor (Bauer et al. 1998) or to induce direct male reproductive toxicity (Couse and Korach 1999), thus acting as a male endocrine disruptor. Also, Diuron did show ER-mediated response in human MCF-7 breast cancer cells, Chinese hamster ovary cells, and recombinant yeast strains (Kojima et al. 2004; Noguerol et al. 2006; Vinggaard et al. 2000). However, Diuron did not affect CYP19 aromatase activity in the human placental microsome assay, indicating that it does not interfere with the conversion of androgens to estrogens, which could alter the balance between male and female sex hormones (Vinggaard et al. 1999). The present findings showing absence of promoting effects of Diuron on mammary carcinogenesis are in agreement with these negative in vitro assay results.
Estrogens have important physiological effects on the growth and function of hormone-dependent tissues, including the mammary gland, but they are also associated with the development and progression of breast cancer (Couse and Korach 1999; Matthews and Gustafsson 2003; Pettersson and Gustafsson 2001). Estrogens exert their carcinogenic effects by both ER-dependent and ER-independent pathways. In particular, the ER-dependent pathway involves activation of the ER by estrogens, leading to the expression of estrogen-responsive genes, as well as stimulation of cell growth and proliferation (Khan et al. 1998). Estrogen receptor-α is a ligand-dependent transcription factor that regulates a large number of genes in many different target tissues and is important in the development and progression of estrogen-responsive neoplastic cells (Iwase 2003). In the present study, the positive expression rate of ER-α was approximately 80% of a total of sixty tumors. In fact, as most mammary tumors induced by DMBA are ER positive and dependent on hormonal status (Russo and Russo 1996), our results also indicate that the rat mammary gland is not a potential target for the carcinogenic potential of Diuron since serum estradiol levels, tumor burden, and the immunoexpression of ER-α in the tumors were not altered.
Finally, no histological evidence of the Diuron’s toxicity on reproductive organs such as the ovary, uterus, and vagina were observed in this study. This finding differs from findings with the herbicide atrazine, which has been categorized as an endocrine disruptor and a potential mammary carcinogen for female SD rats in chronic feeding studies. In high doses, atrazine has been related to an acceleration of age-related endocrine changes leading to an earlier onset and/or increased incidence of chemically induced mammary tumors in females SD rats. This sex/strain endocrine-mediated response, which appears to be unique to the female SD rat, occurs only at high doses that interfere with normal estrous cycling, promoting prolonged exposure to endogenous estrogen and prolactin (Stevens et al. 1999; Wetzel et al. 1994). Therefore, the present results suggest that Diuron also does not have a female endocrine disruptor potential like the atrazine.
The results of the current study indicate that twenty-five weeks of Diuron treatment did not promote mammary carcinogenesis in ovary-intact SD rats, but this finding may be interpreted under two main points: It has been well established that 85% to 90% of the palpable mammary tumors in rats disappear or diminish significantly in size/volume after the ovaries are surgically removed (Thordarson et al. 2001). In fact, ovariectomy abrogates promoting effects of endogenous estrogens on chemically induced rat mammary carcinogenesis (Thordarson et al. 2001; Ueda et al. 2005), and ovariectomized female rats are useful for assessing the impact of endocrine-disrupting agents, since they are more sensitive than their intact counterparts (Ueda et al. 2005). In a study recently finished, we observed that female SD rat pups exposed to Diuron in early life (i.e., in utero, in lactation, and prepubertal periods) did not present changes in complete vaginal opening, estrous cycle length, or uterus and ovary relative weight and morphology, indicating a possible lack of estrogenic action for this herbicide (unpublished results). Therefore, further studies are necessary to determine if Diuron exerts an estrogenic, tumor promoting effect on the growth of DMBA-induced mammary tumors in ovariectomized female SD rats, since weak enhancing effects may be masked by endogenous hormone in ovary-intact rats. In a two-year feeding study (2500 ppm), female NMRI mice were exposed to an approximate dose of 867 mg/kg/day, resulting in an increased incidence of mammary tumors (Iyer, 2002). In our previous thirteen-week feeding study (2500 ppm), female Swiss mice were exposed to an approximate dose of 150 mg/kg/day, without a clear promoting effect in DMBA-initiated mice (de Moura et al. 2010). In the present study, female SD rats were exposed to an approximate dose of 160 mg/kg/day for twenty-five weeks. Therefore, the discrepancy between present and previous studies could be related the different levels of exposute to Diuron.
Since in the present study no initiating or promoting influences of Diuron on the chemically induced mammary gland carcinogenesis process were registered in female SD rats, it can be assumed that the rat mammary glands are not potential targets for the toxicity of this herbicide. Studies designed to clarify the mode of action of high doses of Diuron on the mammary glands of female NRMI mice, as reported by others (Iyer 2002), may lead to better understanding of the possible species-specific mammary carcinogenic activity of this herbicide and its potential relevance to humans.
Footnotes
Acknowledgments
This study was supported by FAPESP (State of São Paulo Research Foundation) and TOXICAM (Centre of the Evaluation of the Impact of the Environment on Human Health, Department of Pathology — Botucatu Medical School, UNESP, Brazil). T. F. Grassi was the recipient of a fellowship from FAPESP (2006/01330-0). We also acknowledge PROINTER/PROPe/UNESP (168/2010).
