Abstract
Poloxamer 407 (P407) is a non-ionic detergent that is used widely in pharmaceutical formulations and personal care products. In animals, P407 causes hyperlipidaemia. P407 is taken up by the liver and causes loss of fenestrations in liver sinusoidal endothelial cells (LSEC), which contributes to the pathogenesis of hyperlipidaemia. Here the short-term (1–15 days) effects of P407 on all liver cells were investigated in mice using electron and light microscopy. As expected, P407 was associated with hyperlipidaemia. Kupffer cells became massively engorged with vacuoles and took on a marked honeycomb morphology. LSECs also became engorged with vacuoles and endocytosis was activated. The diameter of lipoproteins in the space of Disse was less than those in the lumen, consistent with a filtering effect of fenestrations. Defenestration of the LSEC was noted. Hepatocyte endocytosis of lipoproteins and P407 particles was also noted; however, hepatocyte steatosis was not evident. Hepatic stellate cells did not appear to be abnormal. In conclusion, P407 is taken up by the liver mostly through endocytosis by LSECs and Kupffer cells.
Keywords
Poloxamer 407 (P407) is a ubiquitous manmade surfactant and non-ionic detergent. Structurally, P407 is a copolymer of ethylene oxide and propylene oxide with a molecular weight of 12,600. P407 has unusual thermoreversible properties. It is a liquid at room temperature, while at body temperature it self-assembles into micelles that then aggregate into a gel. These temperature-dependent micellization and gelation properties have led to the widespread use of P407 in personal care products such as mouthwashes, deodorants, and skin care products and also as an excipient in a variety of pharmaceutical preparations (Dumortier et al. 2006).
P407 has a major adverse effect, which is hyperlipidaemia observed in experimental animals after parenteral administration (Johnston 2004; Johnston and Palmer 1993; Palmer, Emerson, and Johnston 1998). Typically, cholesterol and triglycerides increase by an order of magnitude or more within 1 to 2 days of a single intraperitoneal injection of 0.5 to 1 mg/kg of P407. This is considered to be secondary to direct inhibition of lipoprotein lipase, which is an endothelial enzyme that is responsible for converting chylomicrons to chylomicron remnants (Johnston 2004; Johnston and Palmer 1993). Repeated administration of P407 is associated with the development of atherosclerotic lesions in rodents, and this model has been developed by Johnston and colleagues for the study of the pathogenesis and treatment of atherosclerosis (Palmer, Emerson, and Johnston 1998).
Following parenteral administration, P407 is most avidly taken up by the liver (Li, Palmer, and Johnston 1996). Previously we reported that P407 has a marked effect on the ultrastructure of the liver sinusoidal endothelial cells (LSECs). LSECs are very thin cells that are perforated with pores called fenestrations that are thought to facilitate the transfer of substrates such as lipoproteins from the blood to the hepatocyte. P407 was associated with a loss of fenestrations and with impaired transfer of lipoproteins through fenestrations from the sinusoidal blood to the hepatocytes. It was proposed that one mechanism for P407-induced hyperlipidaemia is related to this reduction in the transfer of lipoproteins from the blood to the hepatocytes (Cogger et al. 2006).
In this study, we examined the effect of P407 and associated hyperlipidaemia on the entire hepatic sinusoid. As P407 forms micelle particles, we hypothesized that endocytosis by LSECs and the liver macrophages, Kupffer cells, would be activated. We also hypothesized that the diameter of lipoproteins on the luminal side of the LSEC would be greater than those in the extracellular space of Disse as a result of filtering by the fenestrations. Finally, we hypothesized that the morphology of the stellate cells and hepatocytes themselves might be influenced by any P407 and/or lipoproteins that are able to traverse the LSEC and enter the space of Disse.
Materials and Methods
Inbred mice were obtained from the Centenary Institute, Sydney, Australia (B10.BR). The study was approved by the Sydney Southwest Area Health Service Animal Ethics Committee. P407 was dissolved in saline (0.5g/mL, 4°C) and administered by intraperitoneal injection at a dose of 0.5g/kg body weight. Injections were given at days 1, 3, and 5; and livers were harvested at days 2, 5, 8, 11, and 15 (n = 2–4 per time point).
Blood samples were obtained by tail bleeding and analyzed for total cholesterol, total triglycerides, and alanine transaminase (Royal Prince Alfred Hospital Biochemistry Department, MODULAR Analytics system, Roche Diagnostics, Castle Hill, Australia).
For collection of liver samples, mice were sacrificed with carbon dioxide, and livers were immediately perfusion-fixed using a 23 gauge needle inserted into the portal vein. Liver tissue was fixed with 1% glutaraldehyde, 4% paraformaldehyde in PBS (0.1M sucrose), processed for electron microscopy, and paraffin-embedded for light microscopy as described previously (Warren et al. 2007, 2006). For transmission electron microscopy, 2 blocks per liver were studied, and several photographs were obtained for each sample using a Phillips CM10 transmission electron microscope.
For scanning electron microscopy, samples of liver underwent secondary fixation with 2% osmium in 0.1M cacodylate buffer (pH 7.4, 2% sucrose, 2mM CaCl2) were washed and dehydrated with ethanol, then placed in hexamethyldisilazane for 10 min. Specimens were sputter-coated with gold, and observed with a Jeol JSM 6380 scanning electron microscope (JEOL, Tokyo, Japan) at 15-kV acceleration voltage.
Light microscopy was performed after staining with hematoxylin and eosin. Immunohistochemistry was performed to identify Kupffer cells using mouse-specific F4/80 antibody (a gift from R. Ganss, Australia). Sections were pretreated with proteinase K after de-paraffinization. To prevent endogenous peroxidase activity and biotin binding, sections were pretreated with 0.3% H2O2 in PBS and avidin-biotin blocking solutions. Sections were then incubated with the F4/80 (1:10 dilution) for 1 hour. Secondary biotinylated anti-rat antibody (1:200, Sigma, St. Louis, MO) was applied for 45 min. Sections were incubated with peroxidase-conjugated streptavidin (30 min, 1:50, Sigma), and peroxidase activity was revealed using 3,30-diaminobenzidine. Staining was also performed using Green Dye to identify P407 in liver tissue (code 3408-1, Bradley Products Inc., Bloomington, MN), as has been described elsewhere for visualizing exogenous surfactants in lung tissue (Krause et al. 2000).
Results are presented as mean ± standard error and comparisons performed using the Student’s T-test and Kruskal Wallis analysis of variance on ranks with a post hoc Dunn’s test when indicated using SigmaPlot (version 11.0, Systat Software Inc., Chicago, IL). P < .05 was considered significant.
Results
As expected, there was an increase in blood levels of cholesterol and triglycerides following treatment with P407, which normalized following cessation of treatment. Alanine transaminase was also elevated slightly (Figure 1 ).
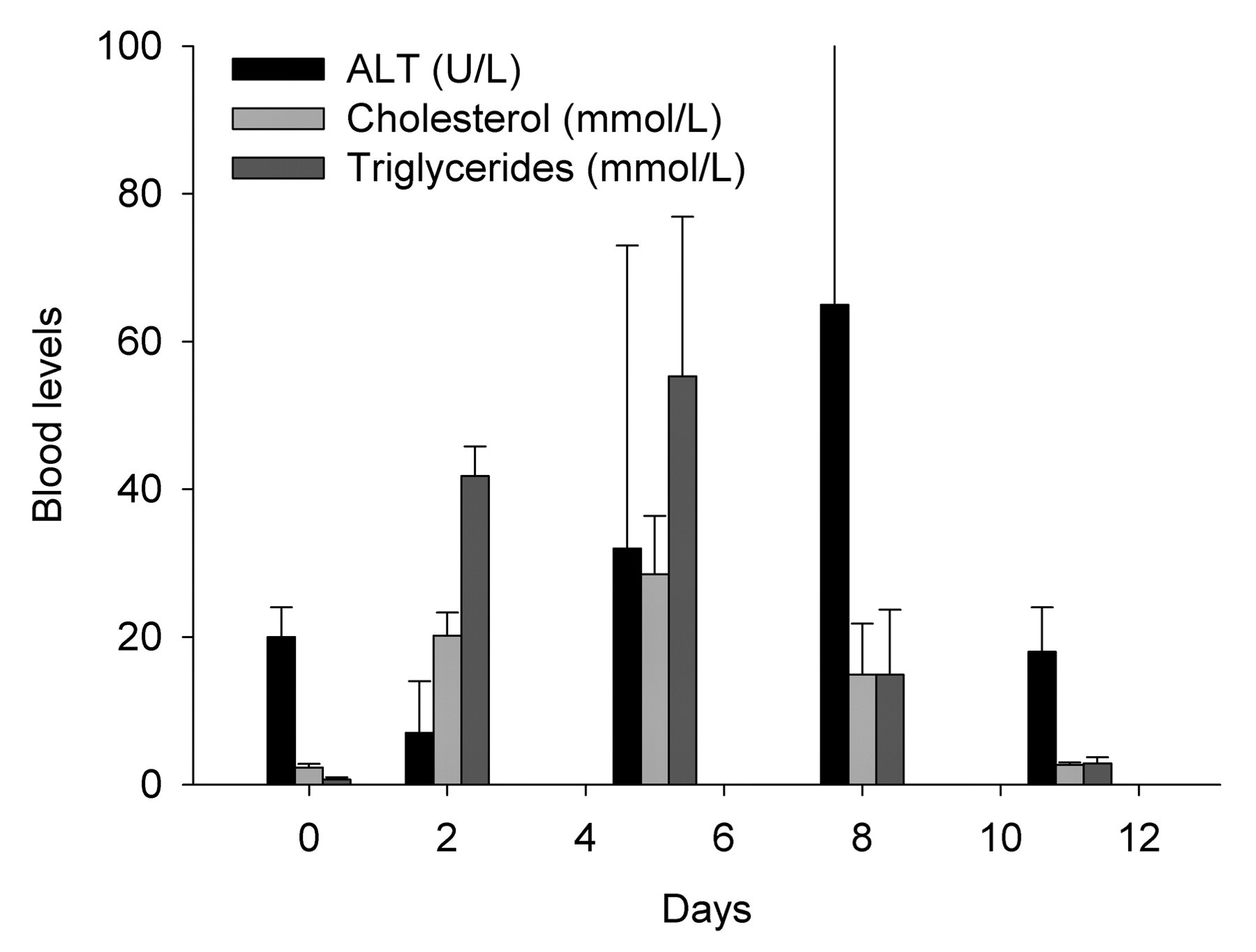
The effect of P407 on cholesterol, triglycerides, and ALT. There were significant increases in triglycerides at day 2 and day 5, and cholesterol at day 5 (p < .001, Kruskal-Wallis test) but no statistically significant change in ALT.
Light microscopy revealed the presence of large foamy Kupffer cells and the absence of hepatosteatosis (Figure 2 ). To determine the distribution of P407, staining with Green Dye was performed. This showed that P407 was distributed along the hepatic sinusoids with uptake in the Kupffer cells. There was no apparent staining of hepatic stellate cells or hepatocytes. Immunohistochemistry for Kupffer cells (F4/80) confirmed that the foamy cells were Kupffer cells.
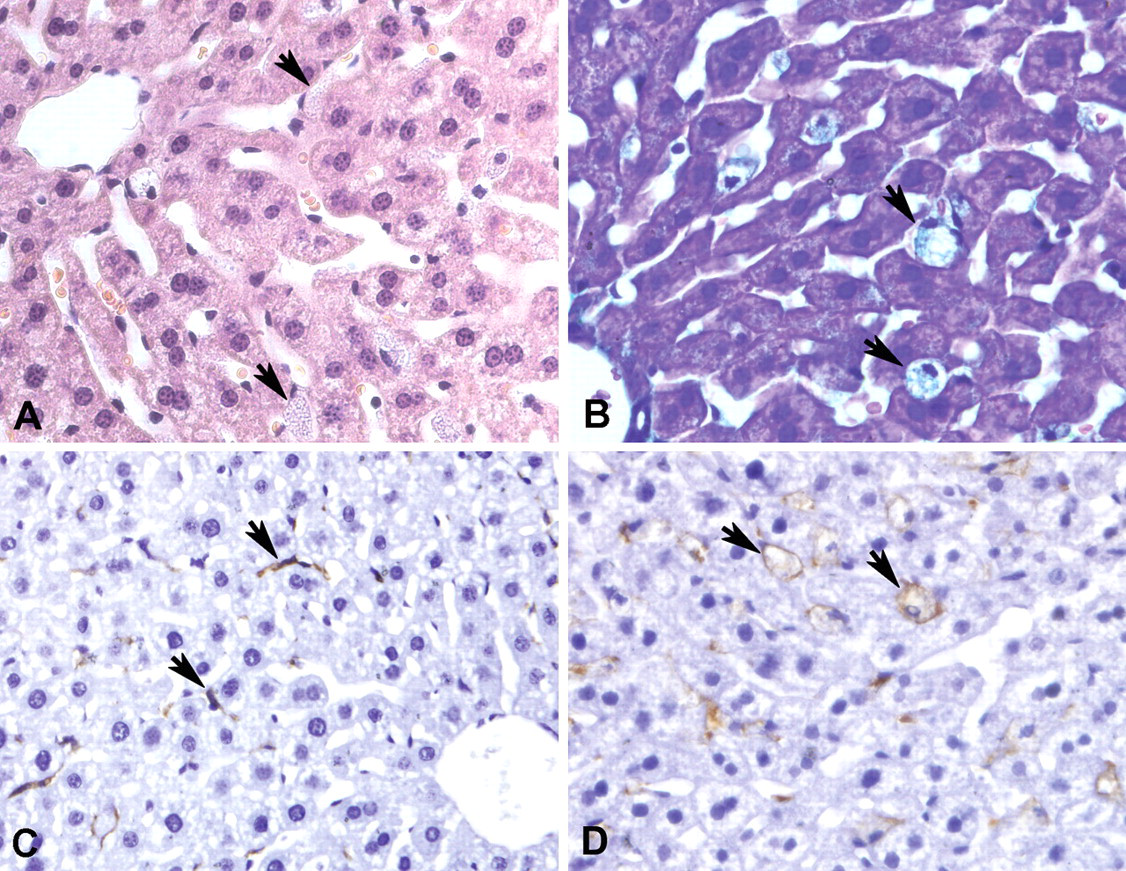
Light microscopy of livers following administration of P407 (original magnifications × 400). [A] Histological section stained with H&E demonstrating foamy Kupffer cells (arrows, day 5). [B] Kupffer cells stained with Green Dye showing uptake of P407. The insert shows an hepatic stellate cell with no staining (day 11). [C–D] Immunohistochemistry with F4/80 to demonstrate Kupffer cells. In the control liver (C), the Kupffer cells are elongated and do not occlude sinusoids, while in the treated animal (D), they are large and foamy (day 5).
The effect of P407 on the morphology of Kupffer cells is shown in Figure 3 . Figure 3A shows a normal Kupffer cell within the sinusoidal lumen. The cell membrane is ruffled with lamellopodia and filopodia, and there are only a few vacuoles in the cytoplasm. Figures 3B, 3C, and 3E show transmission electron micrographs of Kupffer cells in livers after administration of P407. There is less ruffling of the cell membranes, and the cytoplasm is engorged with large vacuoles. The vacuoles contain circular inclusions that appear to be a mixture of P407 micelles and lipoproteins. Figure 3F shows the electron microscopic appearance of P407 studied at 4°C and at concentration of 0.025 g/ml. The in vitro diameter was 17.4 nm (range 7–66 nm), which was consistent with the size of P407 particles seen in vacuoles and space of Disse and the size and appearance of the P407 particles in vitro was similar to that reported by Rizwan et al. (2007). Moreover, the appearance of P407 in vitro is identical to that seen in some of the Kupffer cell vacuoles particularly at the earlier time points (Figure 3E). Figure 3D shows a scanning electron micrograph of a Kupffer cells from a liver pretreated with P407. The Kupffer cell has a remarkable honeycomb appearance. In many cases, Kupffer cells were so swollen that they appeared to occlude the sinusoidal lumen. It is possible that the slight increase in ALT that we observed after treatment with P407 reflects ischemia caused by this process of Kupffer cell enlargement.
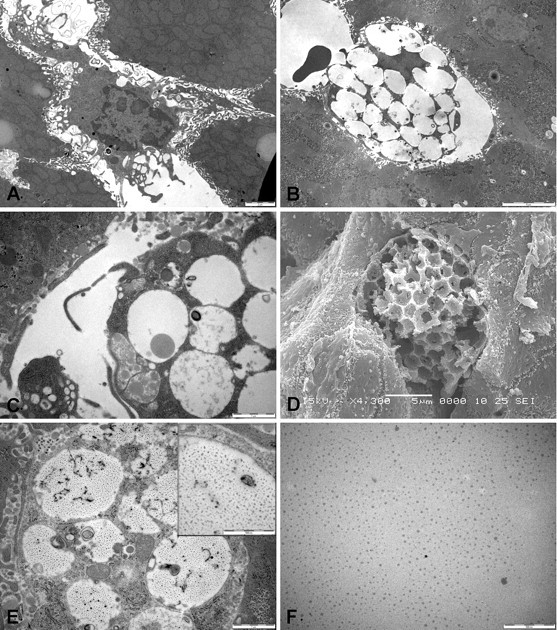
Electron microscopy of Kupffer cells. [A] Kupffer cell in control liver with the characteristic lamellopodia and filopodia (bar = 2 µm). [B, D] Transmission and scanning electron micrographs of Kupffer cells following P407. Kupffer cells became completely engorged with large vacuoles (bar = 5 µm, day 11, day 8). [C] Details of Kupffer cell vacuoles containing lipoproteins (arrows) (bar = 1µm, day 5). [E] Some Kupffer cells contained vacuoles filled with particles (see also insert, bar = 0.5 µm) similar to P407 micelles (day 5). [F] Transmission electron micrograph of P407 (0.025g/ml) dissolved in distilled water (bar = 0.5 µm).
The morphology of the LSECs and hepatic stellate cells is shown in Figure 4 . Figure 4A shows a normal LSEC with its nucleus protruding into the sinusoidal lumen and very thin cytoplasmic extension lining the sinusoidal lumen. Fenestrations within these cytoplasmic extensions are visible. Figures 4C and 4D show LSECs that have numerous large vacuoles swelling the cytoplasm and loss of fenestrations. Hepatic stellate cells are visible in Figures 4B and 4D. The appearance of the hepatic stellate cells, containing only a few large fat-engorged vacuoles, is normal. Figures 4B and 4C also provide additional examples of Kupffer cells that are filled with large vacuoles.
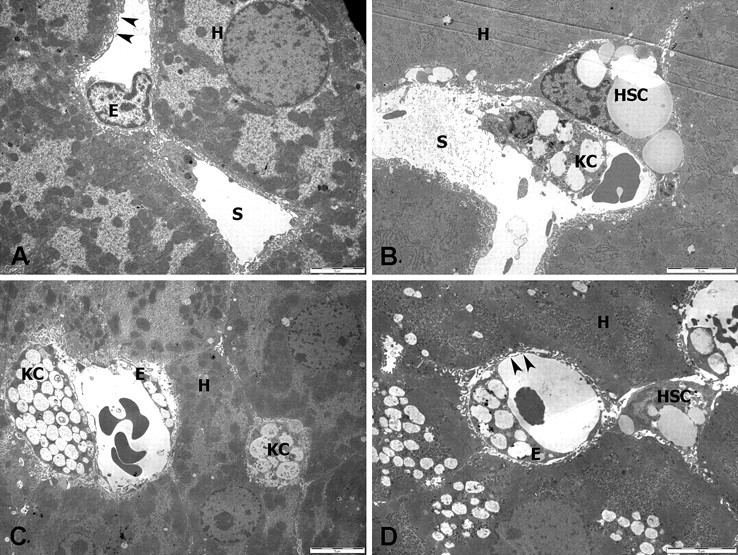
Electron microscopy of LSECs and hepatic stellate cells (low magnification, bar = 5 µm). [A] Hepatic sinusoid (S) in control liver. The normal LSEC (E) has thin cytoplasmic extensions perforated by fenestrations (arrows). [B–D] Livers from treated mice (day 2, day 5, day 11). LSECs have swollen cytoplasm rich in vacuoles. Kupffer cells are also filled with vacuoles. Hepatic stellate cells are morphologically normal. Hepatocytes contain lipid droplets. (E, LSEC; KC, Kupffer cell; HSC, hepatic stellate cell; L, lipid filled vacuole; S, sinusoidal lumen.)
Additional morphological changes in LSECs are shown in Figure 5 . Figure 5A shows a normal LSEC. Figures 5B, D, E, and F show increased endocytic activity of the LSEC. Figure 5D shows all phases of endocytosis, while Figure 5E shows a vesicle packed with P407 micelles and lipoproteins. Lipoproteins and P407 particles are also apparent in the space of Disse (Figures 5D–F). All of these changes in the cells of the hepatic sinusoid, particularly the numbers of vacuoles in Kupffer cells, LSECs, and hepatocytes increased after each injection and had not resolved by 10 days after the final injection of P407.
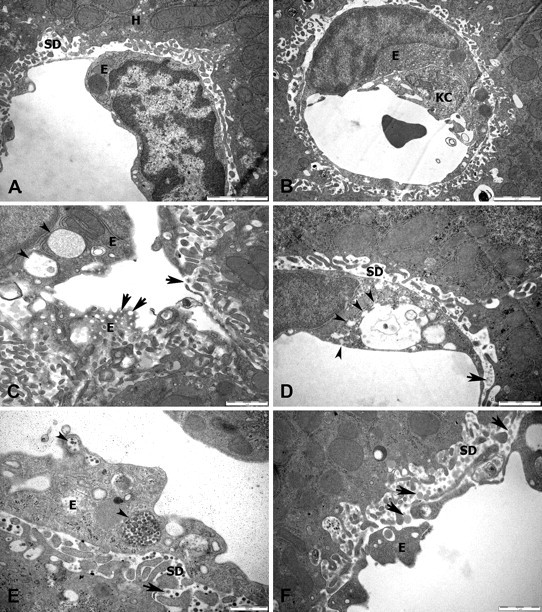
Electron microscopy of LSECs. [A] LSEC from a control mouse demonstrating limited endocytotic activity and thin cytoplasm (bar = 2 µm). [B] LSEC following P407 showing increased small vesicles in the cytoplasm (bar = 2 µm, day 2). [C] LSECs containing enlarged vacuoles engorged with P407 particles (small arrows) and fenestrations (large arrows) (bar = 1 µm, day 5). [D] Different stages of endocytosis with small vesicles (small arrows) fusing with a large central vesicle. Small lipoproteins and P407 particles are present in the space of Disse (bar = 1 µm, day 15). [E] Lipoprotein and P407 particles in LSEC vesicles (small arrows) and space of Disse (large arrow) (bar = 0.5 µm, day 2). [F] Lipoproteins (arrow) in the space of Disse (bar = 1 µm, day 5). (E, LSEC; SD, space of Disse; KC, Kupffer cell; H, hepatocyte.)
The effect of P407 on the ultrastructure of the hepatocytes is shown in Figure 6 . There are numerous lipoproteins in the space of Disse, with some endocytosis of these particles by hepatocytes. Increased autophagy was also noted, with electron dense inclusions and lamellar myelin figures within the lysosomes (Figure 6D).
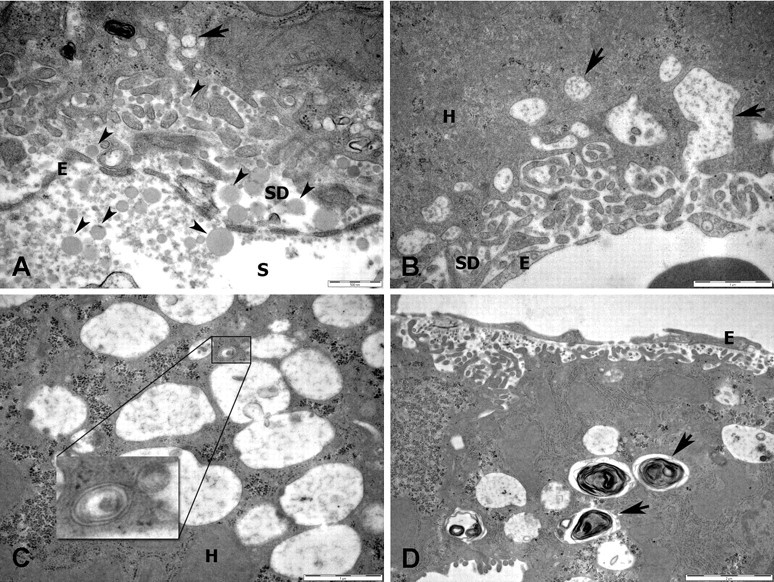
Electron microscopy of hepatocytes from mice treated with P407. [A] Numerous lipoproteins (small arrows) are present in the sinusoidal lumen and space of Disse and are being endocytosed by the hepatocytes (large arrows) (bar = 0.5 µm, day 5). [B] Large endocytotic vesicles are present in the hepatocytes (arrows) (bar = 1 µm, day 2). [C] Numerous lysosomes are present in the hepatocyte (bar = 1 µm, day 11). [D] Dense lamellar figures are present within the lysosomes indicating autophagy (arrows) (bar = 2 µm, day 11). (E, LSEC; SD, space of Disse; KC, Kupffer cell; H, hepatocyte).
To determine whether the diameter of particles in the space of Disse and sinusoidal lumen was consistent with a sieving action by fenestrations in the LSEC, the diameter of lipoproteins was measured. Lipoproteins in the sinusoidal lumen had a diameter of 121 ± 47 nm, while those in the space of Disse where significantly smaller at 90 ± 27 nm (p = .02).
Discussion
After intraperitoneal injection, P407 is found predominantly in the liver. About 5% of a single dose of P407 was reported to be found in the liver after 24 hours, which was 5 times more than found in the kidney (Li, Palmer, and Johnston 1996). In this study, we show that intraperitoneal injection of P407 causes marked ultrastructural changes in hepatic sinusoids. P407 undergoes extensive endocytosis by Kupffer cells and LSECs, while hepatocytes and hepatic stellate cells remain relatively unaffected. LSECs are defenestrated, with fewer fenestrations of smaller diameter, and P407 lipoproteins in the extracellular space of Disse are smaller than those in the sinusoidal lumen. These findings suggest that the LSECs are selectively permeable to P407 and control the entry of these lipoproteins into the space of Disse.
The most obvious morphological change was in the Kupffer cells. Kupffer cells are resident hepatic macrophages that are involved in hepatic inflammatory responses and the uptake of large circulating substrates (McCuskey 2008; Smedsrod et al. 1994). Kupffer cells, particularly when they are activated, have a large irregular surface caused by numerous microvilli, filopodia, and lamellopodia extending from the cellular surface. They ingest circulating particulate substrates by endocytosis and phagocytosis (McCuskey 2008). Previously Johnston et al. (2002) reported that P407 increased the number of foamy Kupffer cells in mice. In their study, C57BL/6 mice were treated with P407 for 4 months, and light microscopy was used to study the liver. Likewise, we found on light microscopy that there were increased numbers of Kupffer cells that had a foamy appearance. Scanning electron microscopy showed that the Kupffer cells had a remarkable honeycomb appearance. Transmission electron microscopy revealed that the Kupffer cells were engorged with numerous vacuoles that contained particles similar in appearance to P407 as well as other particles that more typical of lipoproteins. It is of note that Johnston et al. found that the Kupffer cells stained positive for Oil Red O, indicating that there was at least some uptake of fat. In addition, we found that Green Dye, a stain for surfactants, was also present in Kupffer cells, adding evidence that Kupffer cells actively take up P407.
LSECs were found to be thickened and defenestrated, confirming our previous finding following acute administration of P407 (Cogger et al. 2006). In this earlier study, fenestrations were reduced by about 80% in vivo following P407, and fenestrations were also reduced following in vitro exposure of isolated LSECs to P407 (Cogger et al. 2006). Here we also found that endocytosis was markedly increased in the LSECs, which eventually developed steatosis with large vacuoles. Similarly, Vilaro and Llobera (1988) observed increased endocytosis and steatosis in LSECs following intravenous injection of Intralipid, a fat emulsion that forms micelles approximately 200 to 1,000 nm in diameter.
On the other hand, there were few if any changes in the hepatic stellate cells. These cells are present in the space of Disse and store vitamin A and lipids in large vacuoles. In response to inflammation, hepatic stellate cells become activated, losing their fat droplets and producing substances such as collagen involved in hepatic fibrosis. Here we found that hepatic stellate cells did not take up any P407 and were morphologically unchanged following administration of P407.
There were also only few changes in the hepatocytes following P407. There was no hepatosteatosis despite the hyperlipidaemia. This is consistent with the finding that hepatic cholesterol and triglycerides do not increase following P407 (Millar et al. 2005; Palmer, Emerson, and Johnston 1998). However, endocytosis of lipoproteins and P407 particles by hepatocytes were observed with evidence of autophagy, including lamellar myelin figures. It is of interest that similar findings were seen following administration of Triton X. Like P407, Triton X is a non-ionic detergent that causes hyperlipidaemia. Electron microscopic studies of the rat liver following Triton X administration found that it had been taken up by hepatocyte lysosomes and underwent autophagy (Trout and Viles 1979a, 1979b). Overall, our findings suggest that hepatocytes are involved primarily with endocytosis of P407, while Kupffer cells and LSECs are involved with the endocytosis of both P407 and larger lipoproteins. By contrast, hepatic stellate cells were relatively unaffected by P407. The main pathway for the clearance of P407 by the liver is endocytosis, mostly by Kupffer cells and LSECs.
Are there any functional implications of these ultrastructural changes? We noted that the Kupffer cells were engorged but not activated. Moghimi (1999) showed that an injection of P407 prevents subsequent hepatic uptake of microspheres, presumably by blocking macrophage uptake. Here we found that after P407, Kupffer cells appeared to be less activated, as evidenced by the observation that the number of membranes ruffles was substantially reduced. This is also consistent with the observation that P407 reduces the nitric oxide response of peritoneal macrophages to lipopolysaccharide (Johnston 2004). Overall, it appears that P407 depresses the inflammatory response of Kupffer cells
The data also provide some support for the concept that fenestrations act as a filter. Fenestrations are patent pores approximately 100 nm in diameter that facilitate the access of sinusoidal lipoproteins, dissolved substrates, and lymphocyte projections to the space of Disse and hepatocytes (Cogger and Le Couteur 2009; Fraser, Dobbs, and Rogers 1995). To determine whether there was any evidence of selective filtration on the basis of diameter, we measured the average diameter of lipoproteins in the space of Disse and the sinusoidal lumen. The lipoproteins in the space of Disse were about 25% smaller than those in the sinusoidal lumen, which is consistent with permselectivity.
We also noted that there was no hepatosteatosis associated with the marked hyperlipidaemia, confirming other reports. The absence of any change in hepatocyte lipids is somewhat puzzling. Previously, we have hypothesized that P407 contributes to hyperlipidaemia by decreasing fenestrations in the LSEC. We found that P407 was associated with reduced uptake of lipoproteins, such as chylomicrons and chylomicron remnants, by the liver (Cogger et al. 2006). Thus, defenestration of the LSEC could potentially protect hepatocytes from hyperlipidaemia, by reducing the passage of lipoproteins from the sinusoidal lumen to the space of Disse. The fact that Kupffer cells and LSECs become engorged with fat while hepatic stellate cells and hepatocytes did not is consistent with this speculation.
In conclusion, P407 was associated with marked changes in the Kupffer cells and LSECs. The findings included increased endocytosis, vacuolization, and steatosis, with defenestration of the LSEC. The main mechanism for the hepatic clearance of P407 appears to be endocytosis by Kupffer cells and LSECs. By contrast, the hepatic stellate cells and hepatocytes were relatively unaffected, which supports the concept that defenestration of the LSEC reduces access of lipoproteins to the extracellular space.
Footnotes
Abbreviations
The work was funded by the Australian National Health and Medical Research Council and the Ageing and Alzheimers Research Foundation (a division of the Medical Foundation of the University of Sydney).
