Abstract
This case study session of the hepatobiliary system was held during the 42nd Annual Society of Toxicologic Pathology Symposium in Summerlin, Nevada. The case studies highlighed potential hepatic and biliary toxicity liabilities. This article comprises several of the case studies that were presented during the session which included copper-associated hepatitis in a dog, sinusoidal obstruction syndrome in non-human primates, hepatic cytoplasmic alteration in mice and rats, and Kupffer cell hyperplasia/granulomatous inflammation in rats. Presenters, when applicable, provided case signalment, anatomic/clinical pathology data, and diagnoses and discussed potential pathogeneses.
Keywords
Dr. Shakirat Adetunji (anatomic pathology resident, College of Veterinary Medicine, Kansas State University, Manhattan, KS, USA) presented a case titled “Copper-associated chronic hepatitis: The tale of a 2-year-old Boston Terrier.” The case consisted of a 2-year-old Boston Terrier that uneventfully whelped five healthy puppies. The dog subsequently developed a distended abdomen, and up to 710 mL of clear, yellow-tinged fluid was intermittently drained from the abdominal cavity. Exploratory surgery revealed a shrunken and nodular liver. The dog deteriorated, was euthanized, and an autopsy was performed at the Kansas State Veterinary Diagnostic Laboratory. On autopsy, the liver was diffusely tan and severely shrunken (Figure 1) with variably sized nodules separated by fibrosis. Microscopically, there was distortion of the normal hepatic cord architecture and organization of the parenchyma into multiple micronodules that were separated by fibrous connective tissue that extended into periportal regions and bridged other portal regions (Figure 2). Hepatocytes within the micronodules were rounded and moderately enlarged and contained pale indistinct intracytoplasmic microvesicles and occasional brown to red granular pigment (Figure 3A) that was strongly positive for Rhodamine stain (Figure 3B). The copper level in the liver was 642 ppm on a wet weight basis and 2247 ppm on a dry weight basis (threshold for established copper-associated hepatitis is 1000 ppm on a dry weight basis). The clinical history, gross and microscopic lesions, and the presence of a toxic level of copper in the liver of the dog were diagnostic for chronic copper toxicosis. Because developmental disorders like ductal plate malformation can also present with bile duct proliferation and extensive hepatic fibrosis, these conditions were ruled out based on the absence of renal cysts and gallbladder lesions in the dog.
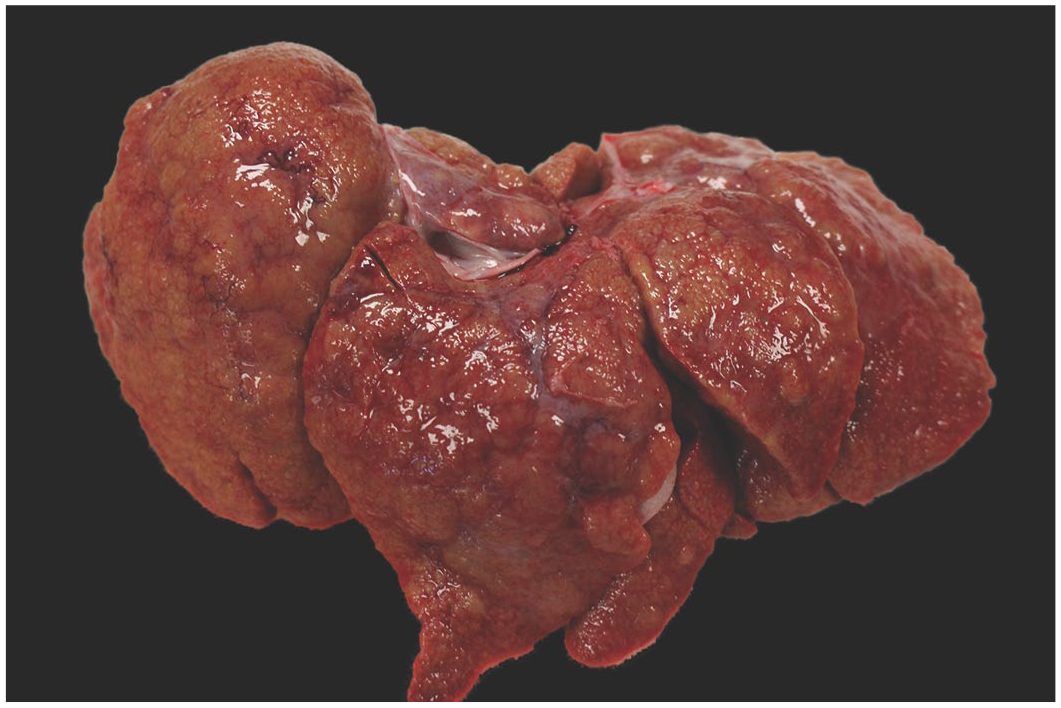
Gross necropsy examination of the liver of a Boston Terrier with copper-associated chronic hepatitis. Diffusely tan and severely shrunken liver with poorly defined, variably sized nodules separated by fibrosis, dorsal surface.
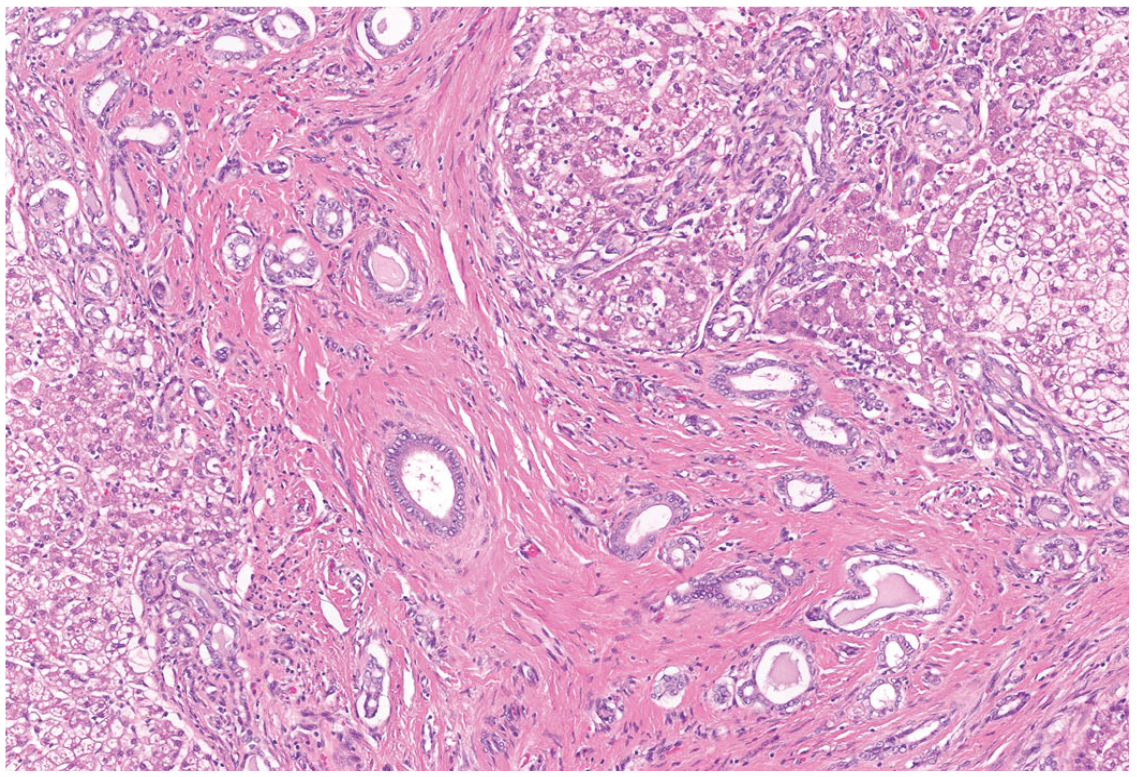
Microscopic evaluation of the liver of a Boston Terrier with copper-associated chronic hepatitis. Hepatic cord architecture is distorted, and there is organization of the parenchyma into multiple micronodules that are separated by bands of collagen fibers and fibrous connective tissue.
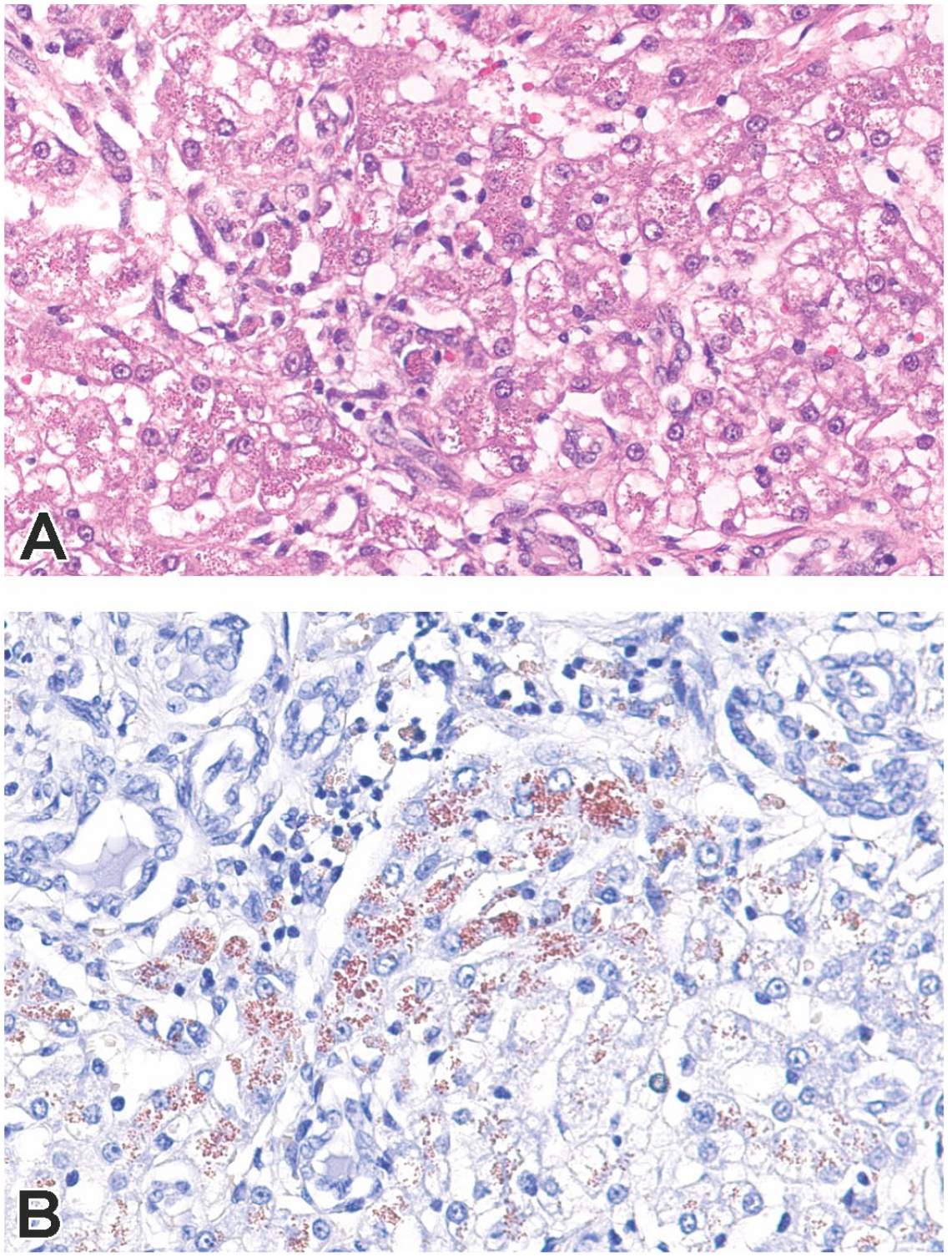
Special stain evaluation of the liver of a Boston Terrier with copper-associated chronic hepatitis. Hepatocytes contain intracytoplasmic brown to red granular pigment (A) that stained strongly positive for Rhodamine stain that identifies copper (B). H&E stain and Rhodanine stain.
Physiologic stressors including immunosuppression, chronic infection, and inflammation have been implicated in the exacerbation of copper-associated hepatitis in animals. Hepatocellular injury and turnover can release the stored copper from hepatocytes leading to oxidative damage and cell death.1,2,6 Due to the temporal association with parturition, it is tempting to associate pregnancy and lactation stress as a predisposing health risk factor that contributed to the clinical progression of this case. To date, there are no clinically reliable diagnostic biomarkers for evaluation of copper status in dogs, especially in early stages of hepatocellular injury. Therefore, histologic examination and copper quantification in liver tissue evaluated together remain the gold standard for diagnosis of copper-associated hepatitis. 7 In summary, the clinical history, gross and microscopic lesions, positive rhodamine staining of liver sections, and the presence of a toxic level of copper in the liver of this dog were diagnostic for chronic copper-associated hepatitis.
Dr. Rebecca Kohnken (senior principal pathologist at AbbVie) described a case of sinusoidal obstruction syndrome (SOS) in monkeys resulting from administration of a cytotoxin-bearing antibody-drug conjugate (ADC). 3 The title of her presentation was, “ADC-Associated Liver Sinusoidal Injury in Monkeys.” Histologic features of this toxicity in monkeys include multifocal sinusoidal dilation, midzonal hepatocellular atrophy (Figure 4A), and occasional nodular regenerative hyperplasia (Figure 4B). The proposed pathogenesis of SOS involves initial injury to liver sinusoidal endothelial cells (LSECs), and in this case, it was due to non-target-mediated uptake of the ADC by endocytosis. Following LSEC injury and loss, platelets sequester in dilated sinusoids (leading to decreased circulating platelets), and hepatocellular plates are inadequately perfused (leading to often small increases in aspartate aminotransferase (AST) and alanine transaminase (ALT)). This can be further demonstrated by loss of VEGFR2 positivity in areas of sinusoidal dilation (Figure 5B; Figure 5A is the matching H&E stained section). Ultimately, LSEC injury promotes recruitment of CD34+ cells from the bone marrow (Figure 5D; Figure 5C is the matching H&E-stained section) and may completely resolve or remain as hepatocellular atrophy. In humans with SOS, histologic features are very similar to those seen in monkeys (sinusoidal dilation, hepatocellular atrophy, nodular regeneration); however, due to the frequent late-stage diagnosis, the patient is often in liver failure. Due to the clear drug-relationship and translation from nonclinical toxicology studies in monkeys to the clinic, it is critical for toxicologic pathologists to be familiar with and recognize SOS. This can pose a significant diagnostic challenge due to the temporal variability of the LSEC injury and manifestations of SOS and the frequent lack of prominent histologic findings which may necessitate special stains and/or immunohistochemistry (IHC) to aid in diagnosis. Knowledge of the mechanism of action of the therapeutic being evaluated (such as ADCs), and careful study design to capture transient decreases in platelets and increases in liver enzyme activities, should help prompt the pathologist to carefully evaluate for histologic evidence of LSEC injury.
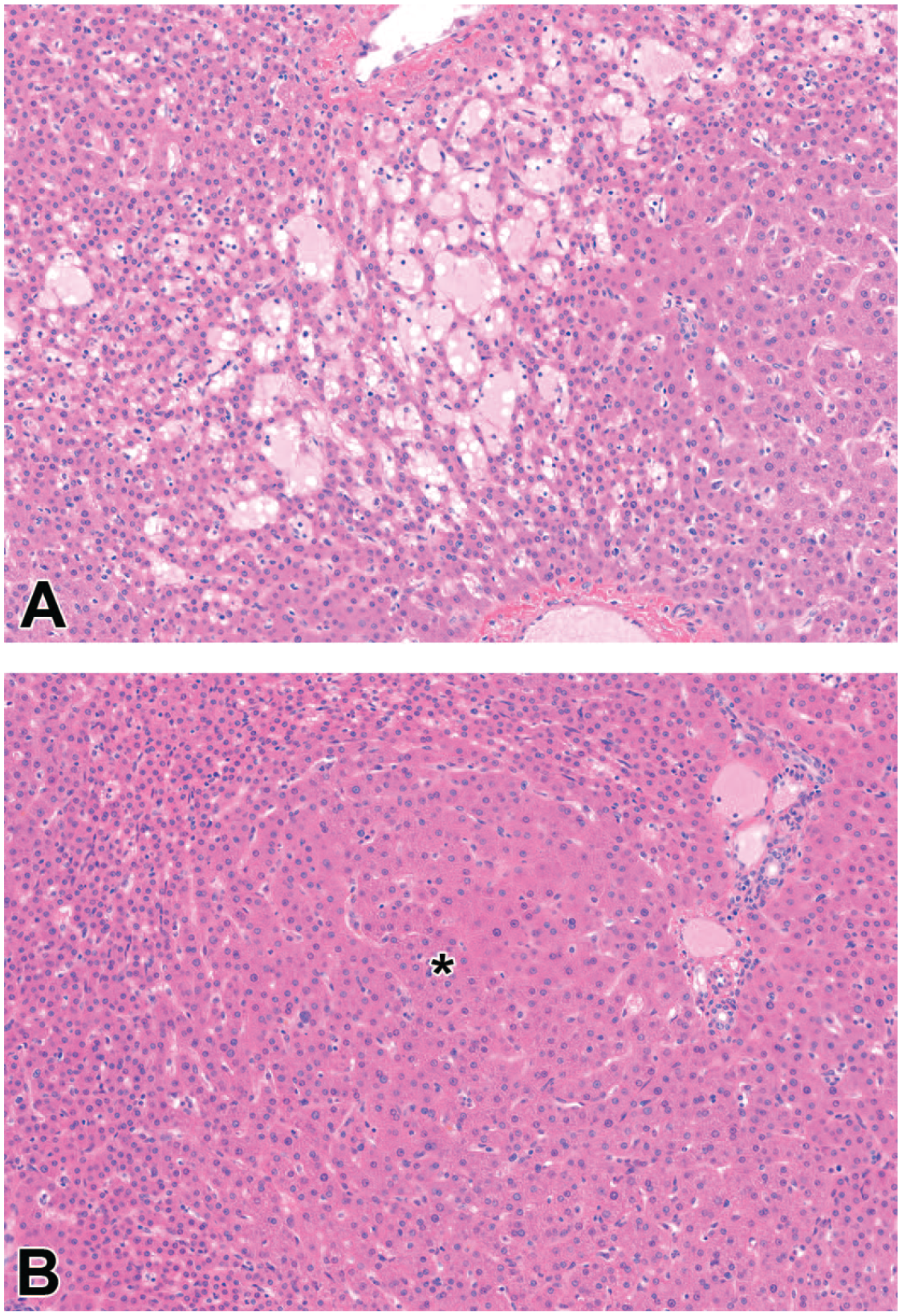
Characteristic light microscopic findings of sinusoidal obstruction syndrome (SOS), liver, cynomolgus monkey, recovery phase. Hematoxylin and eosin (H&E) stain. (A) Area of sinusoidal dilation. The affected sinusoids are variably dilated, filled with homogeneous eosinophilic material (plasma) or less commonly red blood cells (congestion—not shown), and separated by thin cords of atrophic hepatocytes. (B) Nodular regeneration. Nodule of regenerative double cell-thick hepatocellular plates (*) limited at the periphery by atrophic plates. Reprinted with permission from Toxicologic Pathology. 3
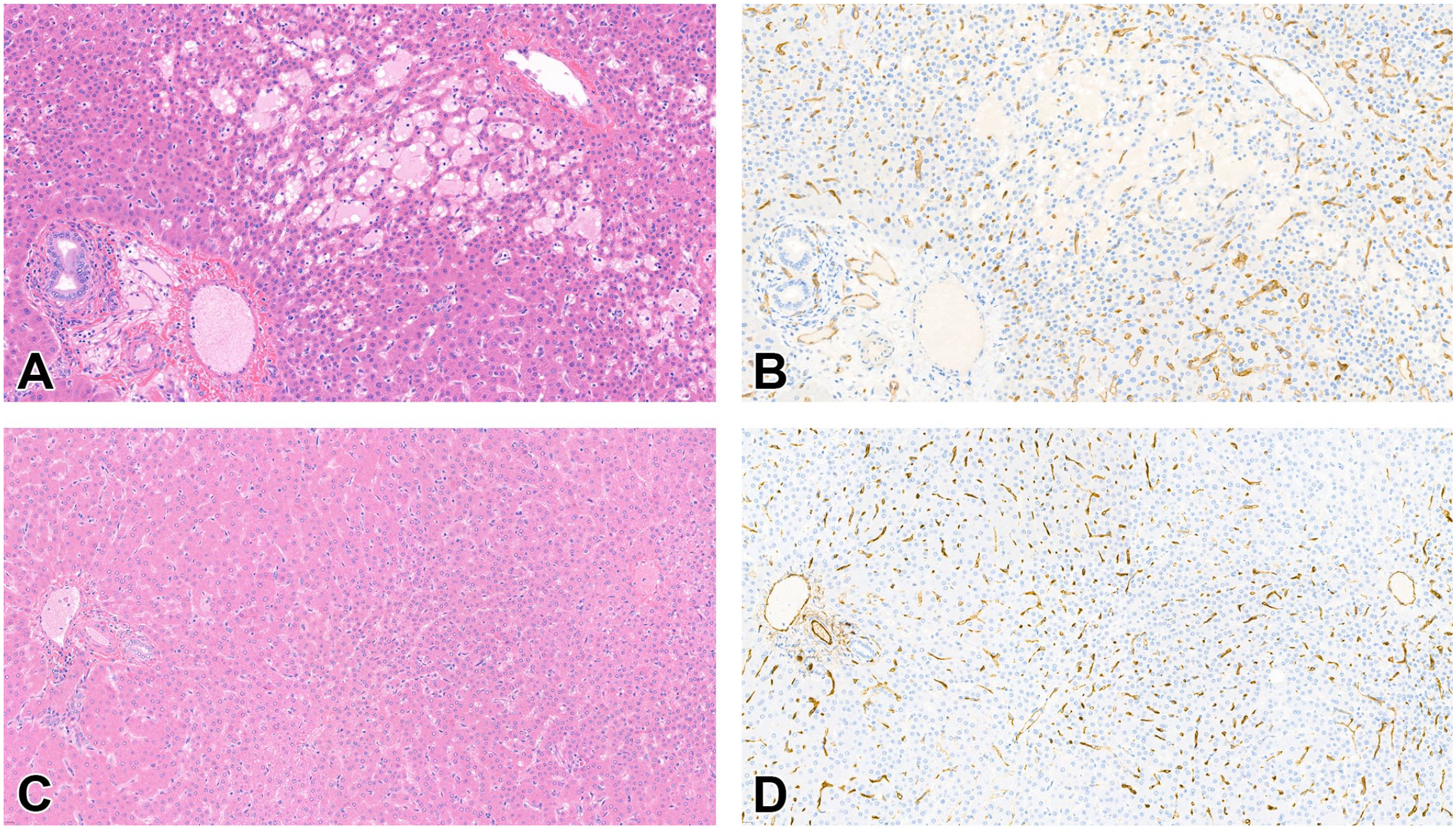
VEGFR2 and CD34 immunohistochemistry (IHC) demonstrate alterations of sinusoidal endothelial cells (SECs) in sinusoidal obstruction syndrome (SOS), liver, cynomolgus monkey, recovery phase. (B) VEGFR2 IHC. (D) CD34 IHC. (A, C) Hematoxylin and eosin (H&E) stain. (B) VEGFR2 IHC shows staining disruption and loss of VEGFR2 immunoreactivity in an area of sinusoidal dilation in ADC-008-dosed monkey, which is indicative of loss of endothelial cells. (A) The area of sinusoidal dilation is confirmed at evaluation of the matching H&E-stained section. (D) CD34 IHC shows moderate to marked CD34 immunostaining of SECs throughout the liver lobules in areas of parenchymal alterations in ADC-008-dosed monkey, which is indicative of sinusoidal capillarization. (C) The matching H&E-stained section demonstrates alterations in hepatocellular plate thickness. Reprinted with permission from Toxicologic Pathology. 3
Dr. Allison C. Boone (employed by Experimental Pathology Laboratories, Inc., Morrisville, NC, USA, as a Division of Translational Toxicologic Pathologist for the National Toxicology Program (NTP) in the Comparative and Molecular Pathogenesis Branch, Molecular Pathology group at the National Institute of Environmental Health Sciences in Research Triangle Park, NC, USA) presented a study on “Reevaluating the Diagnoses of Hepatic Cytoplasmic Alteration.” The term “cytoplasmic alteration” is a common descriptive diagnosis used in toxicologic pathology to describe various forms of hepatocellular cytoplasmic changes including glycogen accumulation, ground glass change, granular change/degeneration, and hyaline degeneration. 5 Hence, cytoplasmic alteration is used as a catch-all phrase that reflects diverse pathophysiological processes. It is possible that one or more types of cytoplasmic alteration may be related to a specific toxicological process, in the context of a single test article/study. Therefore, a retrospective review of several NTP rodent bioassays that reported significant incidences of hepatocellular cytoplasmic alteration in rats and/or mice at 90 days was conducted. The aim of the review was to examine if the use of additional terms (qualifiers) in the diagnosis could provide more information and clarity, which may assist with more accurate tabulation and comparison of group effects, forego the need/shorten explanation in the narrative of the report, and preclude any clarifying questions. 4 The review revealed various histological descriptions used for the diagnosis of cytoplasmic alteration (Table 1). Thus, the study showed that a variety of histological lesions representing different pathogeneses are given the same diagnosis of cytoplasmic alteration. The current International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice (INHAND) criteria recommend a descriptive diagnosis over an interpretative diagnosis. 5 To capture the underlying mechanisms, the use of qualifiers (e.g., glycogen depletion or accumulation, cytoplasmic eosinophilia) should be considered to reflect the major morphological features, provide a better understanding of the pathogenesis of the lesion, and add greater precision to the study. However, these suggestions may be study or lesion specific and not applicable for all situations. Nonetheless, the study does warrant revisiting the diagnosis of cytoplasmic alteration by the hepatobiliary INHAND group.
Histological descriptions representing cytoplasmic alteration from NTP studies.

Dr. Kenji Koyama, from the Laboratory for Drug Discovery and Development, Shionogi & Co., Ltd., Osaka, Japan, concluded the case presentations with a talk titled, “Kupffer cell hyperplasia/granulomatous inflammation associated with drug-induced crystals in the liver in Sprague-Dawley rats.” In vivo toxicity studies have shown that active pharmaceutical ingredients and their metabolites may precipitate and crystallize in various organs and tissues depending on their physicochemical properties and toxicokinetics. A 7-day oral toxicity study of Compound A, which is a small-molecule compound and prodrug with improved bioavailability, was conducted. The study used 6-week-old, female Sprague-Dawley rats. The prodrug is metabolized to a poorly soluble active form (<0.4 μg/mL at pH 6.8), and neither the prodrug nor its active form are cationic amphiphilic drugs (CADs). Clinical findings included mildly increased levels of aspartate aminotransferase, alanine aminotransferase, and γ-glutamyltransferase. Necropsy revealed an enlarged and pale liver. Histopathology showed diffuse to multifocal proliferations of foamy Kupffer cells/macrophages in the sinusoids and scattered granulomas with multinucleated cells. Round to polygonal cytoplasmic vacuoles and needle-like clefts were observed in these foamy Kupffer cells/macrophages and a few hepatocytes. Ultrastructurally, needle-like or polygonal clefts were observed in the Kupffer cells/macrophages and a few hepatocytes, although the number of lamellar bodies was not increased. Evaluation of frozen sections under polarizing light revealed bi-refringent micro-crystals in the Kupffer cells/macrophages. Foamy macrophages were also observed in the lungs and lymphoid organs, and bi-refringent micro-crystals were confirmed in the lung. Given the findings of foamy cells in multiple organs, phospholipidosis was the most likely diagnosis. However, this presumed diagnosis was ruled out based on the electron microscopy data. The physicochemical and pharmacokinetic properties as well as the histopathology data suggest precipitation of the administered compound, or its active metabolite and a subsequent foreign body reaction may be involved in the pathogenesis of this lesion. This study emphasizes the importance of a careful pathology approach in the differential diagnosis of foamy cells, especially those induced by substances other than CADs.
Footnotes
Acknowledgements
The authors acknowledge the following expertise in the respective case study. Dr. Scott Fritz from the Kansas State Veterinary Diagnostic Laboratory and Department of Anatomy and Physiology, College of Veterinary Medicine, Kansas State University, and Dr. Kristen Hill-Thimmesch from the Department Diagnostic Medicine/Pathobiology, College of Veterinary Medicine, Kansas State University and Kansas State Veterinary Diagnostic Laboratory in the case titled “Copper-associated chronic hepatitis: The tale of a 2-year-old Boston Terrier.” Dr. Magali Guffroy from Preclinical Safety, AbbVie Inc., North Chicago, Illinois, in the case titled, “ADC-Associated Liver Sinusoidal Injury in Monkeys.” Dr. Arun Pandiri from the Comparative and Molecular Pathogenesis Branch, Division of NTP, National Institute of Environmental Health Sciences, Research Triangle Park, North Carolina, in the case titled, “Reevaluating the Diagnoses of Hepatic Cytoplasmic Alteration.” Drs. Ryo Obara, Miho Mukai, Minako Tajiri, Yuki Kato, Emi Kashiwagi, Kae Fujisawa, Yoshiji Asaoka, Tamio Fukushima, and Mikinori Torii from the Laboratory for Drug Discovery and Development, Shionogi & Co., Ltd., Osaka, Japan, and Dr. Hirotada Murayama from the Research Area for Drug Candidate Generation I, Shionogi TechnoAdvance Research & Co., Ltd., Osaka, Japan, in the case titled, “Kupffer cell hyperplasia/granulomatous inflammation associated with drug-induced crystals of the liver in Sprague-Dawley rats.”
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported [in part] by the Intramural Research Program of the NIH (ES103376-02).
