Abstract
Drug development in neuro-oncology remains a challenge for neoplasms of the central nervous system (CNS). Drugs can be administered peripherally (i.e., oral or intravenous) or locally (into the tumor or the adjacent neuropil). Each of these routes has advantages and disadvantages. Like the treatment for non-CNS cancers, peripheral side effects are encountered (i.e., diarrhea, myelosuppression, rash); however, there also may be neural-specific side effects for patients that may be acute or delayed (i.e., seizures, somnolence, hearing loss). The nervous system is also a privileged site protected by the blood-brain barrier, so many agents developed for peripheral administration will not penetrate into the CNS due to issues of size, charge, or lack of lipid solubility. In addition, the abnormal vasculature, increased interstitial pressure, and inherent mechanisms of tumor resistance (methyl-guanine-methyl transferase [MGMT], P-glycoprotein, etc.) within brain neoplasms reduce the efficacy of many agents designed for neuro-oncologic indications. Each of these issues alone, and all of them in aggregate, are reasons for the limited success of therapeutic agents directed against CNS tumors despite promising data acquired using cell lines and animal models.
Keywords
Introduction
There are many different types of brain tumors, arising as either primary neoplasms from the intracranial contents or as secondary metastases originating from another location. Certain primary brain tumors preferentially occur in patients of a specific age range (CBTRUS 2010). For example, medulloblastomas and intracranial germinomas are much more common in the pediatric population, whereas gliomas and primary central nervous system (CNS) lymphomas are more common in adults. Similarly, the biological behavior of CNS neoplasms varies with the cell of origin and, to a lesser degree, the location. Meningioma, the single most frequent primary brain neoplasm, evolves from the dura mater and develops as a localized mass that compresses (rarely invading) the underlying neural tissue and can be readily removed in most cases. In contrast, the most common class of CNS neoplasms is that of glial tumors, which arise mainly from astrocytes and invade along white matter tracts so that complete resection is nearly impossible.
The most common glial tumor is the highly malignant glioblastoma multiforme (GBM), which accounts for approximately 50% of all gliomas in adults. Other astrocytic gliomas account for another 10% to 15% of gliomas (CBTRUS 2010). Gliomas develop in one of two ways. Primary GBMs develop de novo and frequently have either PTEN (phosphatase and tensin homolog) deletions or EGFR (epidermal growth factor receptor) amplification or mutations and are commonly seen in older individuals. Secondary GBMs arise from a preexisting low-grade astrocytoma or anaplastic astrocytomas, typically occur in younger patients, and more commonly have P53 mutations.
In adults, almost all astrocytomas are of grades 2 to 4 (where grade 2 is slow-growing and grade 4 is extremely aggressive). The differences between these grades are related to the cumulative pattern of histopathologic factors like cellularity, pleomorphism, mitotic activity, vascular proliferation, and intraneoplastic necrosis. Low-grade gliomas have only increased cellularity, while GBMs are highly cellular, anaplastic, rapidly proliferating, and prone to necrosis (Louis et al. 2007). Survival for patients with astrocytomas ranges from about 5 to 10 years for low-grade tumors and 12 to 18 months for GBM.
Treatment Options for Gliomas
Historically, the preferred treatment for brain tumors has been attempting to maximize the degree of surgical resection. Retrospective data indicate that patients who have undergone total gross resection of their tumors have better outcomes than those patients who have received subtotal resections (Keles, Anderson, and Berger 1999; Lacroix et al. 2001). This approach has its limitations with respect to gliomas due to the invasiveness of these tumors and their tendency to reside in or near important brain centers.
Traditional postsurgical therapy for gliomas involved standalone radiation therapy. Radiation has evolved to employ a three-dimensional treatment plan with an approximately 2-cm-diameter margin around the tumor, thereby avoiding whole brain radiation (which does not improve the outcome but does increase neurotoxicity; Walker et al. 1980; Nieder, Mehta, and Jalali 2009). Since 2005, the standard postsurgical regimen for GBMs has involved radiation and chemotherapy (with temozolomide). For anaplastic astrocytomas, no standard treatment protocol exists; radiation alone or chemotherapy alone or both modalities in tandem (i.e., adoption of the GBM-based protocol) are deemed acceptable (Stupp et al. 2005; Wick et al. 2009). Prior to 2005, the utility of chemotherapy had been debated as a treatment for GBM due to an apparent modest increase in survival time over radiation-alone regimens as determined by a meta-analysis of published clinical studies (Fine et al. 1993; Stewart and Brush 2002; Stupp et al. 2005).
In the past 15 years, three drugs have been approved for used in GBM or anaplastic gliomas (Table 1). The initial agent, Gliadel®, is a wafer of biodegradable polymer impregnated with carmustine. The second, Temodar® (temozolomide), is an alkylating agent that incorporates at the 06 position of guanine to exert its cytotoxic effects. This drug is now a standard part of the conventional chemoradiation protocol used in GBM; concentrations in the brain and CSF reach 20% to 30% of those in the plasma (Ostermann et al. 2004; Portnow et al. 2009). The last molecule, Avastin® (bevacizumab), is a mononclonal antibody that inhibits vascular endothelial growth factor (VEGF). It is used in GBM upon the first relapse based on data from two studies (Friedman et al. 2009; Kreisl et al. 2009). Some other agents used in the treatment of GBMs are listed in Table 1.
Approved a chemotherapeutic agents for treating gliomas.
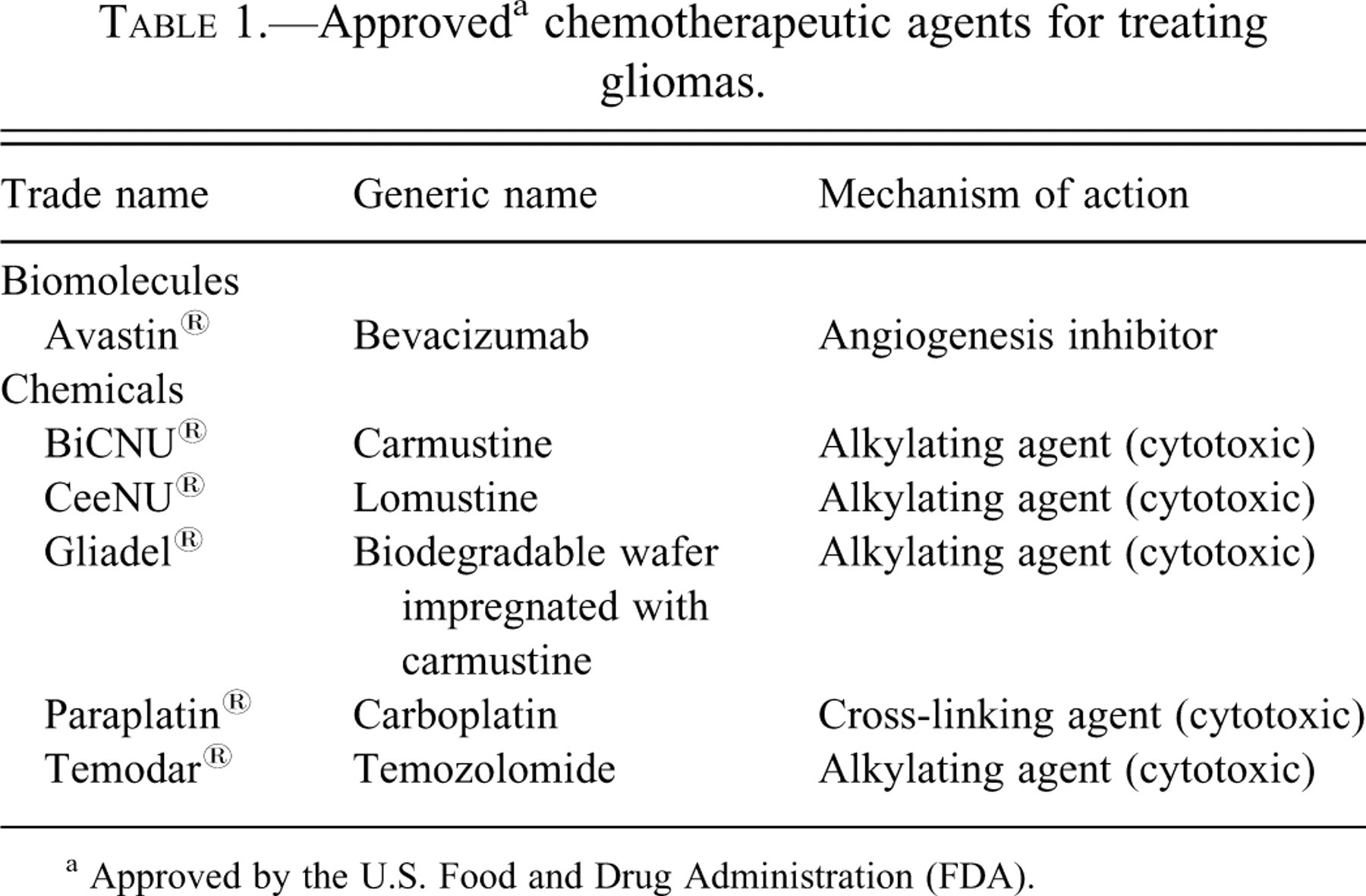
a Approved by the U.S. Food and Drug Administration (FDA).
Effects of Anti-Neoplastic Agents to Treat Gliomas
The CNS can be subdivided into the parenchyma (brain and spinal cord), meninges (which include the leptomeninges [arachnoid and the pia] as well as the dura), special sense organs (ears and eyes), cranial nerves, spinal nerves, and the ventricular system with its contents (choroid plexus and cerebrospinal fluid [CSF]). Multiple boundaries serve to isolate these compartments, including a blood-brain barrier (BBB) and a blood-CSF barrier under normal conditions as well as a blood-tumor barrier that exists within CNS neoplasms. The location of a neural tumor—intra-axial (i.e., developing within the brain parenchyma) or extra-axial (i.e., arising from the dura or within the subarachnoid space)—impacts whether systemically delivered therapies are able to reach it.
Several limitations exist in finding highly effective treatments for GBM and other gliomas (Table 2 ). Preclinical development is carried out in vitro in cell lines or in vivo using a variety of animal models. Each of these approaches has limitations. Cell lines often consist of relatively homogeneous populations relative to the heterogeneous constitution of actual neural neoplasms, and they lack the local microenvironmental conditions that exist in and around tumors as well as the systemic influences that affect drug metabolism and distribution. Most animal models are based on human tumor xenografts where tumor cells are inserted into an animal’s flank (thereby avoiding the several barriers) or brain (equivalent to a “metastasis”); both these situations are distinctly different from human neoplasms that have arisen de novo. These inherent disparities between genuine CNS tumors and model systems frequently result in failure of a promising agent in the clinical setting despite preclinical efficacy. A major advance within the past 10 years has been the generation of genetically engineered mouse models (GEMM) that spontaneously develop glial tumors, mostly of high grade, after a variable latent period (Huse and Holland 2009). Such GEMM are the best current models we have for approximating the biology of CNS neoplasms in humans.
Issues of drug development for gliomas.

Drug delivery in the treatment of malignant gliomas is rife with potential problems that are not relevant in most other malignancies. One major issue is the effectiveness of the BBB. When intact, the BBB prohibits many agents from penetrating the CNS. Agents that are small, uncharged, and lipophilic enter the brain and CSF more readily. However, for patients with malignant tumors of the brain, neoplastic regions that exhibit contrast enhancement on imaging studies have dysfunctional blood-brain and blood-tumor barriers that permit many anti-neoplastic drugs to penetrate easily. On the other hand, tumors that lack contrast enhancement have intact barriers and thus may allow access of systemic agents. Methods to facilitate drug penetration into areas with intact blood-brain barriers represent a strategy to increase our ability to treat these tumors.
Several other considerations render CNS neoplasms resistant to standard therapies. Increased intracranial pressure and cerebral edema, as well as higher intratumoral pressure, may limit drug penetration, as drugs typically must move down rather than against a pressure gradient. Abnormal vasculature within tumors may prevent distribution of molecules into some tumor domains. Systemic (oral or intravenous) administration of anti-neoplastic agents may induce systemic toxicities (myelosuppression, pneumonitis, etc.) or even neurotoxicity (e.g., encephalopathy, seizures) even if the neoplasm is not impacted. Finally, limitations due to biological pathways within tumor cells that promote drug resistance (methyl-guanine-methyl transferase [MGMT], P-glycoprotein, etc.) may reduce the efficacy of agents even if they reach the tumor parenchyma.
Once drugs have been designed to treat CNS lesions, they must be tested extensively in model systems and then in human patients. Anti-neoplastic agents are usually either cytotoxic or cytostatic. Therefore, the degree of efficacy is assessed by reductions in tumor volume (or cell numbers in vitro) and/or increased survival. Human clinical trials for a single agent or combination of agents determine the maximal tolerated dose and potential spectrum of toxic effects (Phase I) and the efficacy against a given tumor type (Phase II). Positive data is then vetted in a Phase III trial where the experimental agent, either alone or as part of a treatment combination, is compared to a “standard” treatment to determine whether there is benefit to be gained (e.g., improved survival, decreased toxicity). Positive Phase III results are may be followed by U.S. Food and Drug Administration (FDA) approval. Drugs that receive accelerated approval—a common scenario in the development of anti-neoplastic agents—often require follow-up (Phase IV) studies to confirm that the efficacy warrants continued availability of the new drug.
Complexities encountered in developing the three approved anti-glioma drugs used to treat malignant gliomas exemplify the trade-offs encountered when designing new agents to attack CNS neoplasms (Table 2). Gliadel® was initially approved for recurrent malignant gliomas, and subsequently for newly diagnosed gliomas, based on a marginal improvement (8 to 12 weeks) in survival (Brem et al. 1995; Westphal et al. 2003). This carmustine-impregnated wafer degrades over the course of approximately 3 weeks. Neurotoxic effects include an increase in seizures the first 5 days after wafer implantation, as well as increased contrast enhancement of the tumor (in 10% to 15% of patients), cyst formation (10%), and wound infections (Sabel and Giese 2008). Temodar® is approved for treating recurrent anaplastic astrocytomas and newly diagnosed GBM. Systemic toxicities associated with its use are modest, thereby allowing the drug to be used for protracted periods. Protracted schedules may improve the therapeutic response by the depletion of MGMT while increasing the overall dose of drug that penetrates the tumor over a given period of time. Pseudo-progression (i.e., an increase in tumor size related to treatment and not to growth of tumor) typically occurs anywhere within the first 6 months after the initial radiation/temozolomide regimen (most cases within the first 3 months). Pseudo-progression is characterized by increased contrast enhancement and cerebral edema related to augmented treatment-related permeability around the tumor; it appears to be related to a patient’s MGMT status, with a higher frequency of necrosis as well as improved survival in methylated patients (Chamberlain et al. 2007; Brandes et al. 2009). In true cases of pseudo-progression, the contrast enhancement and cerebral edema will spontaneously improve over time or with the use of corticosteroids. Nonetheless, the occurrence of this false-positive entity is important, as misunderstanding its implications may lead to unnecessary surgical procedures in 15% to 30% of GBM patients who develop this effect following chemoradiation (Chamberlain et al. 2007; Brandes et al. 2009).
Methotrexate (MTX) is another cytotoxic drug that causes various neurotoxic effects (used in primary CNS lymphoms). This agent can be administered by the intra-arterial, intravenous, intrathecal, and oral routes. When given intravenously or orally at conventional high doses, the main side effect of MTX is leukoencephalopathy, which can often be worsened if radiation therapy is given prior to its use. The effect can be acute and transient or more chronic and persistent. When given intra-arterially, hemorrhagic cerebral infarctions can occur. When given intrathecally or sometimes intravenously, acute toxicities include aseptic meningitis, paraplegia, seizures, as well as stroke-like disturbances. Other chronic toxicities may include brain atrophy and calcification (DeAngelis and Posner 2008).
Considerations in the Development of Novel Anti-Neoplastic Agents
Several approaches have been developed to bypass barriers and reduce systemic toxicities while greatly increasing intra-neoplastic drug accumulation (Table 1). One obvious example is local implantation of toxic drugs directly into a neoplasm (e.g., impregnated wafers [Gliadel®]). Direct delivery by intra-arterial catheterization of the carotid or vertebral arteries that feed the tumor bypasses the systemic circulation and leads to significantly higher concentrations of drug in the brain and many fewer systemic side effects. This approach is not without toxicities, however, including a potentially fatal necrotizing encephalopathy and blindness (Miller et al. 1985; Rosenblum et al. 1989). The encephalopathy is characterized by disseminated miliary necrotic foci with mineralizing axonopathy involving the white matter, which becomes diffusely and severely edematous; a common clinical presentation is progressive hemiparesis and decline in mental status, generalized seizures, and, in some instances, aphasia. The leukoencephalopathy is indistinguishable from pure radiation necrosis but is restricted to the distribution of the injected arterial tree. Much of the toxicity is by vascular injury, but the disseminated lesion probably reflects direct damage to the neural parenchyma as well (Rosenblum et al. 1989). The encephalopathy may be delayed by weeks to months and occur only after repeated courses of therapy at high doses (e.g., “intra-carotid carmustine syndrome” requires doses greater than 100 mg/m2). Prior or coincident ocular damage is associated with infraophthalmic injections that expose the retinal vasculature to particularly high drug concentrations (Rosenblum et al. 1989). Targeted immunotherapy using a fusion protein conjugated to a potent biotoxin (Kunwar et al. 2010) or a radioisotope (Akabani et al. 2005; Fiveash et al. 2006) has been used to attack tumor cells that highly overexpress a cell type–specific receptor, but to date they have not proven to be effective treatments for GBM. The use of such local therapies often leads to treatment-related non-neoplastic changes on magnetic resonance imaging (MRI) that resemble tumor progression (Parney et al. 2005). Finally, vaccines directed against a specific protein (e.g., CDX110, a vaccine targeting the EGFR VIII; Heimberger et al. 2003) may be used to promote an immune response against neoplastic tissue. This vaccine had good activity in preclinical animal models and in early trials with GBM (Lai et al. 2010). The efficacy of this approach was comparable to, if not better than, that produced by the standard therapy. However, the patients eligible for this study were a very select population with an unknown natural history, so the exact relevance of these data remains to be defined.
As our understanding of CNS tumor biology increases, we have begun to define the molecular pathways that drive tumorigenesis. This new understanding has led to an increased usage of biologic agents and less traditional chemotherapies to attack neoplasms within the CNS. The number of potential targets continues to grow, as does the number of drug candidates in development directed against specific target pathways. Targeted molecular therapies are most effective when they work directly against the target to which they are directed. Examples include imatinib (Gleevec®), which targets c-kit and BCR-ABL in gastrointestinal stromal tumors and chronic myelogenous leukemia, respectively; cetuximab (Erbitux®) and erlotinib (Tarceva®), which inhibit EGFR in primary lung neoplasms; and trastuzumab (Herceptin®), which binds the HER-2 receptor in many breast tumors. Some of these targets are expressed in gliomas; for instance, EGFR is amplified in approximately 50% of glioblastomas and is also overexpressed in many high-grade malignant gliomas. However, although logic suggests that this should be a good target for inhibiting malignant gliomas, results to date of several studies have shown very limited activity (Raizer et al. 2010a, 2010b).
So how do we account for the limited activity of such agents against CNS neoplasms? Several possible explanations for a lack of efficacy have been identified. The considerable redundancy among intracellular pathways means that blocking one pathway is insufficient to fully impede the neoplastic cells. Insufficient quantities of drug may gain entry to the tumor tissue (Kuhn et al. 2007; Lassman et al. 2005; Raizer et al. 2010a), or the molecule that enters may be inactive. We have also learned, particularly with EGFR inhibitors, that certain patient populations respond better to treatment (Haas-Kogan et al. 2005; Mellinghoff et al. 2005), although the enhanced efficacy is not always a consistent occurrence (van den Bent et al. 2009)—thereby highlighting the complexity of the issue. Furthermore, the specific EGFR mutations that arise in lung cancer have not been found in glioblastoma, suggesting that the glioma-based genetic changes will not produce sensitivity to drugs directed against the proteins expressed in lung tumors. Interestingly, novel missense mutations in the ectodomain of EGFR have been identified in approximately 14% of GBM that promote both tumorigenicity and sensitivity to EGFR-inhibiting small molecules (Lee et al. 2006). However, at least two patterns of EGFR resistance have been proposed (Mellinghoff, Cloughesy, and Mischel 2007). In addition, it has been shown that activation of multiple tyrosine kinase receptors occurs at one time, so blocking one has little effect as other receptors remain active to drive tumor growth (Stommel et al. 2007). These findings confirm that a very complex network of genetic interactions directs the extremely aggressive behaviors of glioblastomas (Bredel et al. 2009).
We know from preclinical data that combination therapy with multiple anti-neoplastic agents can be significantly more effective in inhibiting tumor growth. However, translating this knowledge to the human clinical setting is problematic, as combining agents often leads to intolerable side effects resulting from either unwanted synergistic toxicity against one organ system or unwanted, nonoverlapping toxic effects on more than one system. Future advances that ameliorate or prevent these toxicities may enable us to adapt combination regimens to treat glioblastomas.
One area that has recently received significant attention as a potential target in treating brain neoplasms is angiogenesis, using agents that block this pathway at the ligand or receptors. Avastin® (bevacizumab), the first such agent approved by the FDA, was developed for use in colon cancer but has since been extended to several other tumor types including relapsed glioblastomas. This anti-VEGF antibody elicits a high response rate but can cause numerous unique MRI changes that complicate the interpretation of posttreatment follow-up imaging studies. In some patients (15% or so), disease progression behaves in a manner similar to gliomatosis cerebri rather than as an expanding mass; this pattern often occurs in association with a significant clinical decline. Areas of diffusion restriction within the tumor and adjacent neural parenchyma have been noted; these have been correlated with hypercellular tumor domains or regions of ischemic necrosis as opposed to the more diffuse pattern of radiation-induced necrosis (Gerstner, Frosch, and Batchelor 2010; Videnovic et al. 2005; Futterer et al. 2009).
Finally, we have learned over time the numerous side effects that impact both the CNS and peripheral organ systems following exposure to agents that are routinely used in neuro-oncology. Many of these adverse effects are well described (DeAngelis and Posner 2008). Some side effects were not initially recognized and were only discovered following widespread use of these agents either alone or in combination. Examples include ifosfamide and methotrexate, which can cause acute encephalopathy and delirium, as well as cytarabine and carmustine, which can incite chronic encephalopathy. Visual loss may be a side effect of tamoxifen and cisplatin. Drugs, such as retinoic acid, can cause headaches without meningitis (“pseudotumor cerebri”). Seizures are common with busulfan, so anticonvulsants are prescribed for patients who are being treated with this agent. Thiotepa can cause myelopathy, particularly when given intrathecally. Finally, one of the most common neurologic complication following anticancer therapy is peripheral neuropathy, which occurs with such agents as oxaliplatin and vincristine. Much of the toxicity is related to the total administered dose. Many agents (e.g., cisplatin, vincristine) elicit a litany of side effects that can occur acutely, subacutely, or chronically. Even agents initially felt to be devoid of neural-based side effects can produce them. One example is capecitabine, a pro-drug that is broken down into 5-fluorouracil (5-FU). This agent can cause distinct (acute and chronic) variants of encephalopathy. The mechanism that predisposes an individual to the acute versus the chronic form is not clear.
Conclusions
The development of anti-neoplastic agents that target primary CNS neoplasms or extra-neural cancers can have profound effects on the CNS and/or PNS. These effects are often different depending on whether the drug is administered locally (e.g., intra-arterially, or into the tumor tissue or peri-tumor parenchyma) or remotely (intravenously, intrathecally, or orally). Many side effects or treatment-related changes may not be initially apparent but become obvious over time. Certainly many agents have been shown to be effective against cultured or implanted cell lines and in animal models. However, a big gap exists between these promising preclinical data and efforts to translate the therapeutic agents and strategies into humans. Fortunately, this gap is slowly closing. Specific considerations for treating gliomas, the most resistant of primary CNS neoplasms, are the unique perturbations of the brain microenvironment, tumor vasculature, and BBB integrity as well as the intrinsic molecular pathways that increase the aggressiveness and capacity for drug resistance. To overcome these obstacles, new or novel approaches to treatment need to be developed. These include presurgical testing, where patients are treated with an agent prior to surgery and then the resected tumors are evaluated for the extent of target expression and the degree of drug penetration. Another way to improve therapeutic outcomes is to design studies based on certain molecular characteristics of the tumor, thereby increasing the likelihood of attaining antitumor activity and avoiding treatment of patients in whom the drug is likely to have little benefit due to the absence of its target.
