Abstract
A half-day American College of Toxicology continuing education course presented key issues often confronted by translational neuroscientists when predicting human risk from animal-derived toxicologic pathology data. Two talks correlated discrete structures with major functions in brains of rodents and nonrodents. The third lecture provided practical advice to obtain highly homologous rodent brain sections for quantitative morphometry in developmental neurotoxicity testing. The last presentation discussed demographic influences (eg, species, strain, sex, age), physiological attributes (eg, body composition, brain vascularity, pharmacokinetic/pharmacodynamic patterns, etc), and husbandry parameters (eg, group housing) recognized to impact the actions of neuroactive chemicals. Speakers described common cases of real-world challenges to animal data interpretation encountered when designing studies or extrapolating biological responses across species. The efficiency of translational neuroscience efforts will likely be enhanced as new methods (eg, high-resolution non-invasive imaging) improve our capability to cross-connect subtle anatomic and/or biochemical lesions with functional changes over time.
Keywords
Discovery and development of new molecules for agricultural, environmental, industrial, and medical therapeutic applications require extrapolation of animal-derived data to assess the potential for adverse outcomes in humans. Indeed, animal experimentation is the only viable approach for in vivo investigation of novel chemical entities (NCEs) in which human exposure is unintended (eg, pesticides, solvents) or of new molecular entities (NMEs) in which human exposure is intended (eg, drugs, biologics, medical devices). Thus, translational biology is an essential discipline for evaluating xenobiotic efficacy and toxicity in whole organisms as well as their systems, including the central nervous system (CNS) and peripheral nervous system (PNS).
People may be exposed to possible neurotoxic agents in many venues and by many routes. Global efforts to define neurotoxic properties have accelerated with the growing recognition that neurological damage in adults is a common consequence associated with environmental or occupational exposure to chemicals or chemical mixtures 1,2 or purposeful administration of medicines. 3 –5 Neurotoxicity is also a key hazard to developing embryos, fetuses, infants, and even juveniles. 6 The prospect that new xenobiotics may induce serious injury to individuals along with substantial collateral costs in energy, money, and time to their families and communities assures that the characterization and assessment of any neurotoxic liabilities will remain a key global health issue throughout the 21st century.
Fundamental Principles of Translational Neuroscience
Collective experience with identifying and characterizing neurotoxic hazards and assessing human risk has shown that no one animal species or experimental design is acceptable for efficacy and safety testing in all cases. Effective neurotoxicity testing requires that toxicologists and pathologists choose a suitable animal species (eg, mouse, rat, rabbit, dog, pig, nonhuman primate, etc) and population (eg, adult vs developing) as well as an appropriate study duration (eg, acute, subchronic, chronic) and dosing (eg, single vs repeated dose). An important neglected factor in the experimental design of regulatory toxicology studies is the issue of timing regarding evaluation of neurotoxicity end points. When to evaluate is especially critical in neuropathology assessments. At present, correct mixing and matching among these biological and experimental design factors affords the best means for examining the neurotoxic risks that new chemical entities might pose to heterogeneous human populations.
Regardless of the chosen species, several basic biological factors underpin the use of neural testing in animals as predictors of possible neurotoxicity in humans. Rodents are favored species for screening studies due to the extensive parallels of key brain structures and neurotransmitters in rats and humans, 7 reasonable correlation of neuroanatomic damage with functional insufficiencies in both species, and relatively fewer resources needed to complete long-term studies, reproductive/fertility studies, and carcinogenicity studies. These neuroanatomic and neurophysiological similarities are even closer for larger nonrodent mammals, which have expanded cerebral cortices and white matter tracts to support more complex associational and sensorimotor activities. 7,8 Communal experience has shown that studies in nonrodent species (eg, dog, nonhuman primate) usually forecast potential human outcomes more effectively than rodent bioassays, 9 while agents that produce neurotoxicity in both rodents and nonrodents are better able to predict human responsiveness. 4 Nonetheless, divergence among animals and humans in fundamental neural attributes such as behavior, regional cytoarchitecture and connectivity, and neurochemistry often confounds efforts to extrapolate data among animal species 10 and from animals to humans. 11 Many biological parameters and experimental design variables may affect translation of nonclinical data for human risk assessment, thus necessitating that the nervous system be evaluated using multiple modalities, most frequently including behavioral, biochemical/molecular, electrophysiological, pathologic, pharmacokinetic, and/or pharmacologic methods.
Similarly, the design of neurotoxicity experiments will fundamentally impact efforts to utilize animal data for human risk assessment. Basic considerations include the study purpose (eg, general screen vs dedicated neurotoxicity analysis), processing conditions (eg, tissue fixation by immersion for general toxicity studies vs perfusion for dedicated neurotoxicity studies), embedding medium (eg, paraffin vs plastic), and nervous system sampling strategy. 12 –17 In some instances, these details reflect the preferences of the sponsoring institution and/or the scientific team. In other cases, study parameters are dictated by regulatory guidelines. 18,19 For a given country, the nature of the guidance varies between agencies (or even within agency centers/divisions) depending on the nature of the chemical entities being regulated (eg, compare neurotoxicity testing rules for the US Environmental Protection Agency with those of the various US Food and Drug Administration centers). For a given product class, guidance also diverges to some degree in different regions around the world.
This review summarizes the current challenges in translating animal data to assess human neurotoxic risk as presented during a half-day continuing education course on “Fundamentals of translational neuroscience in toxicologic pathology: optimizing the value of animal data for human risk assessment,” which was delivered at the 2015 annual meeting of the American College of Toxicology in Summerlin, Nevada. The course was constituted to provide toxicologists with the most up-to-date information regarding key neurobiology and neuropathology concepts and methods needed for efficient and effective identification and characterization of an agent’s neurotoxic risk to human populations. The material was presented in 4 coordinated lectures.
Structure/Function Correlations: The Foundation of Translational Neuroscience
Effective Nervous System Sampling in Adult Rodents and Nonrodents
The first 2 lectures in combination offered a synopsis of correlative neuroanatomy (ie, links between CNS structures and functions) in rodent and nonrodent mammalian species used in neurotoxicity testing. Despite readily evident differences in structure and function among species, the same neuroanatomic subsites should be selected routinely for pathology examination in all test animals.
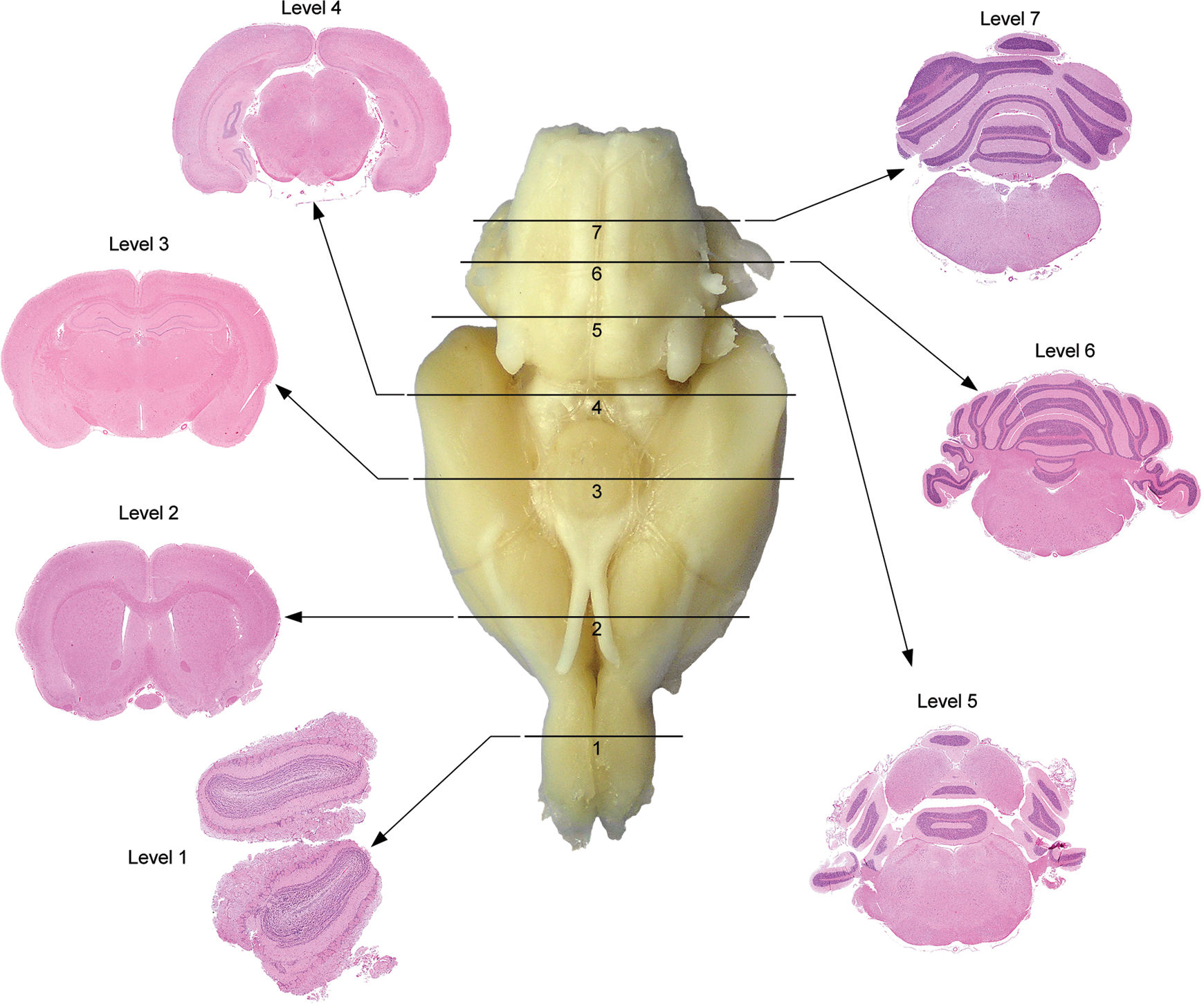
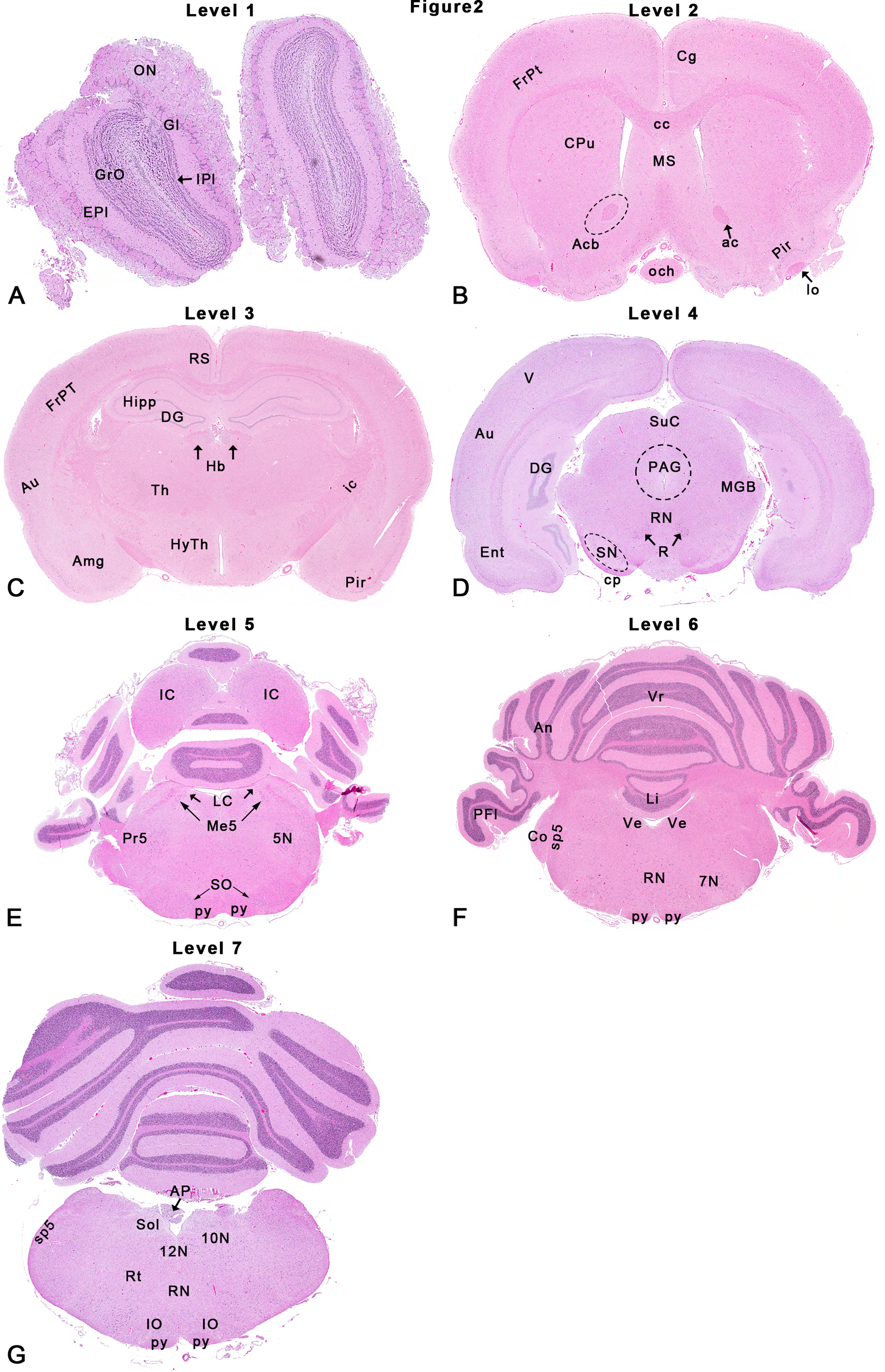
The first talk, given by Dr Deepa Rao, described a modified approach first adopted by the U.S. National Toxicology Program (NTP). The NTP 7-section sampling procedure targets consistent rodent coronal brain sections suitable for neuropathology analysis in general toxicity studies 20 (Figure 1). Compared to traditional rodent brain sampling, 21 which utilizes only 3 of these coronal sections (usually levels 2, 3, and 6 in Figure 1), the NTP 7-section approach includes key neuroanatomical areas based on vulnerability to toxicity (eg, caudal [inferior] colliculus), areas associated with neurodegenerative disorders (eg, substantia nigra for Parkinson disease), and areas associated with neurobehavioral, neurochemical, and neurophysiological end points. Taken together, the 7 sections include approximately 50 specific neuroanatomical areas that represent cognitive, learning and memory, motor, sensory, addiction, depression, and homeostatic neuronal circuitry in the rodent brain 13 (Figure 2). The basis for routine use of NTP 7-section sampling in general toxicity screening is that the expanded sampling will reduce the likelihood that subtle changes 14 or even major lesions 22 will be overlooked, especially in sites not captured by conventional trimming protocols (eg, caudal [inferior] colliculus 23 ).
Rat: hematoxylin and eosin (H&E). The U.S. National Toxicology Program (NTP) 7-section approach depicting neuroanatomical subsites at each level as shown in Figure 1. Each coronal brain slice was embedded with the rostral surface facing down. Abbreviations below correspond to abbreviations used in the Paxinos rat brain atlas. 28 Dotted outlines are close approximations, and use of an atlas is recommended for accuracy. 5N indicates motor trigeminal nucleus; 7N, facial nucleus; 10N, vagal nucleus or dorsal motor nucleus of vagus; 12N, hypoglossal nucleus; ac, rostral (anterior) commissure; Acb, accumbens nucleus (area indicated by dotted line); Amg, amygdaloid nuclei; An, ansiform lobe; AP, area postrema; Au, auditory (temporal) cortex; cc, corpus callosum; Cg, cingulate cortex; Co, cochlear nucleus; cp, cerebral peduncle; CPu, caudate–putamen; DG, dentate gyrus; Ent, entorhinal cortex; EPl, external plexiform layer; FrPt, frontoparietal cortex; Gl, glomerular layer; GrO, granule cell layer; Hb, habenular nucleus; Hipp, hippocampus; HyTh, hypothalamus; IC, caudal (inferior) colliculus; ic, internal capsule; IO, inferior olivary nucleus (now formally designated in animals as the olivary nucleus [Nomina Anatomica Veterinaria] 78 ); IPl, internal plexiform layer; LC, locus coeruleus; Li, lingual (first cerebellar lobule or 1Cb); Me5, mesencephalic trigeminal nucleus; MGB, medial geniculate body; och, optic chiasm; ON, olfactory nerve layer; lo, lateral olfactory tract; PAG, periaqueductal gray; PFl, paraflocculus; Pir, piriform cortex; Pr5, principal sensory nucleus of CN V; py, pyramidal tract; R, red nucleus; Rt, reticular formation; RN, raphe nuclei; RS, retrosplenial cortex; SN, substantia nigra; SO, superior olivary nucleus (now formally designated in animals as the dorsal nucleus of the trapezoid body [Nomina Anatomica Veterinaria]); SpN, septal nuclei; Sol, solitary tract nucleus; sp5, spinal trigeminal tract; SuC, rostral (superior) colliculus; Th, thalamus; V, visual (occipital) cortex; Ve, vestibular nucleus; Vr, vermis. Figure is reproduced from Rao et al. 13
Rat: hematoxylin and eosin (H&E). The U.S. National Toxicology Program (NTP) 7-section approach. Seven coronal sections corresponding to levels based on anatomic target landmarks are taken by embedding with the rostral surface facing down. Level 1 is at the midlevel olfactory bulb (OB). Level 2 is approximately 1 to 2 mm rostral to the optic chiasm (OC). Level 3 is taken at the infundibulum (IF) and/or
The companion talk by Dr Ingrid Pardo described structural/functional correlations that dictate routine brain sampling practices in nonrodent mammals (dog, nonhuman primate). Routine microscopic evaluation of brain from these species typically evaluates 6 samples in coronal orientation from only 1 hemisphere (ie, hemisections). The rationale for unilateral assessment is that structures that are present bilaterally usually exhibit equal sensitivity to potential neurotoxicants; obviously, where warranted both sides of the brain should be examined. Areas to be evaluated should include all major structures/regions that are assessed in rodents 15,18 except for the olfactory bulbs, which are relatively small in nonrodents relative to those of rodents. In general, the neuropathology examination in rodent toxicity studies should routinely incorporate olfactory bulbs since the importance of olfaction in these species is supported by 7% of brain parenchyma by weight. 16 Nonetheless, the decision regarding whether to assess olfactory bulbs should be left to the discretion of the sponsoring institution.
Although time constraints in both talks limited the discussion to considerations for brain sampling, analogous structure/function correlations have also been established for other neural organs. Both Dr Rao and Dr Pardo acknowledged that a comprehensive analysis of neurotoxic liabilities would necessitate sampling of the spinal cord (cervical, thoracic, and lumbar segments; Figure 3), dorsal root ganglia (especially those associated with major nerve trunks), and somatic nerve (at minimum to include the sciatic trunk). 12,13,15,16 A preferred practice is to sample additional somatic nerves as well, which may serve different purposes depending on the species (eg, sural nerve carries only sensory fibers in nonhuman primates and humans but contains both motor and sensory fibers in rodents). Although nonsystematic in nature, autonomic ganglia and nerves often may be evaluated within other organs and tissues.
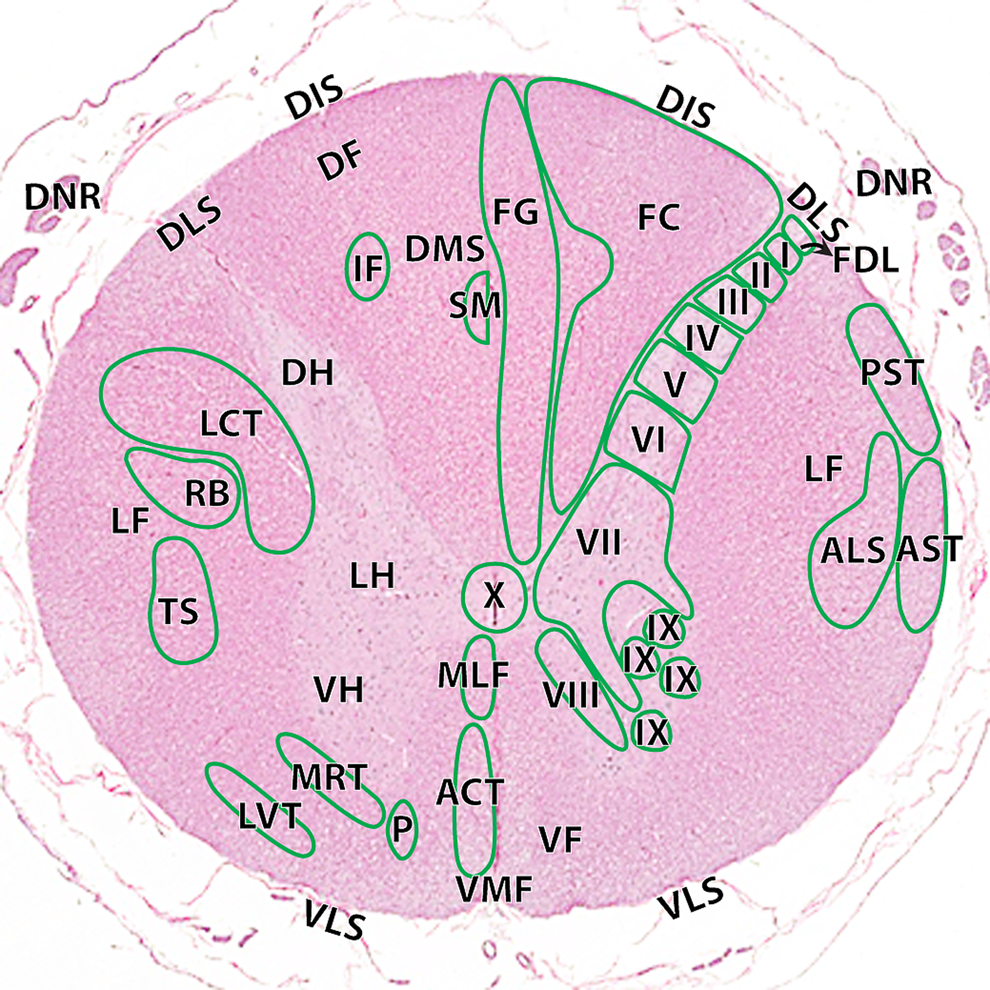
Hematoxylin and eosin cross-section of the cynomolgus monkey spinal cord at the level of C1–C2. ACT indicates anterior corticospinal tract; ALS, anterolateral system; AST, anterior spinocerebellar tract; FC, fasciculus cuneatus; FDL, fasciculus dorsolateralis; FG, fasciculus gracilis; I, lamina 1; II, lamina II; III, lamina III; IV, lamina IV; V, lamina V; VI, lamina VI; VII, lamina VII; VIII, lamina VIII; IX, lamina IX; X, lamina X. Ascending tracts (right): MLF indicates medial longitudinal fasciculus; PST, posterior spinocerebellar tract. Descending tracts (left): DF indicates dorsal funiculi; DIS, dorsal intermediate septa; DH, dorsal horn; DLS, dorsolateral sulcus; DMS, dorsal median septum; DNR, dorsal nerve roots; IF, interfascicularis; LCT, lateral corticospinal tract; LH, lateral horn; LVT, lateral vestibulospinal tract; MRT, medullary (lateral) reticulospinal tract; P, pontine (medial) reticulospinal tract; RB, rubrospinal tract; SM, septomarginalis; VF, ventral funiculi; VH, ventral horn; VLS, ventrolateral sulcus; VMF, ventral median fissure. Figure is reproduced from Pardo et al. 15
Many veterinary neuroanatomy and veterinary neurology texts provide descriptions and visual representations correlating vital functions with particular brain regions. 24 In like manner, annotated neuroanatomy atlases are available for multiple animal species, 25 –29 which can be used to localize the brain regions that should be evaluated when performing the neuropathology portion of nonclinical toxicity studies. Principal neuroanatomic regions that should be evaluated in the brain slide sets advocated above (ie, 7 coronal sections for rodents, 6 coronal hemisections for nonrodents) have been defined in recent papers. 13,15,16 Structural/functional correlations have also been demarcated for spinal cord. 15 Pathologists who frequently participate in neuropathology analysis for nonclinical studies should also be conversant with the many other resources that are available for data translation across species. 18,30,31
In the future, lesion identification by routine histopathologic evaluation is likely to be supplemented by noninvasive or minimally invasive methods. The chief modalities that are likely to be deployed during nonclinical studies include noninvasive imaging platforms such as magnetic resonance microscopy (MRM 32 –34 ), positron emission tomography (PET 35 ), or ultrasound biomicroscopy (UBM 36 ). Addition of imaging to nonclinical neurotoxicity assessment will increase the likelihood that subtle lesions in vulnerable cell populations can be found prior to brain trimming (MRM) and/or followed progressively in vivo over the course of longer studies (PET, UBM). Moreover, digitized image files offer the possibility for automated quantitative acquisition of linear, areal, or volumetric data, 37,38 although the utility of such information for estimating human risk will need careful validation prior to widespread adoption of imaging technology for translational neuroscience. Over time, development of minimally invasive techniques to harvest small fluid or tissue samples from the CNS should prove important in detecting novel nucleic acid 39 –41 or protein 42,43 biomarkers of neurotoxicity. At present, predictive translational molecular biomarkers are unavailable for routine use with neural specimens.
Effective Nervous System Sampling in Developing Rodents
The next lecture, by Dr Robert Garman, provided practical tips for performing the pathology portion of a rodent developmental neurotoxicity (DNT) study. Two unique considerations apply to the neuropathology assessment of developing animals. First, in adult animals, toxic agents usually lead to qualitative evidence of cell degeneration and death, which in chronic lesions may be accompanied by glial cell (astrocytic, microglial) activation and/or proliferation. In contrast, toxic lesions in the developing brain usually present as anomalies in cell numbers (abnormal regional size), location (ectopia), and/or differentiation (eg, defective myelination or synaptogenesis; Figure 4). Degenerative lesions that transpire in the CNS prior to maturity may be repaired (partially or fully), often without invoking either an inflammatory response or a glial reaction. Accordingly, rodent DNT studies are conducted using a relatively limited battery of histological stains, usually including hematoxylin and eosin and a stain to highlight myelin integrity (eg, Luxol fast blue; Rohm and Hass Chemicals, LLC, Philadelphia, PA, USA), although silver stains for neuronal processes are often also used in order to look for alterations in neuron polarity. Other stains commonly used to evaluate neurotoxic effects in adult animals, such as Fluoro-Jade (Histo-Chem, Inc, Jefferson, AR, USA) for neuron necrosis or cell type-specific markers to reveal glial reactions (eg, glial fibrillary acidic protein [GFAP] for astrocytes, ionized calcium-binding adaptor molecule 1 for microglia), are used in DNT studies on a case-by-case basis.
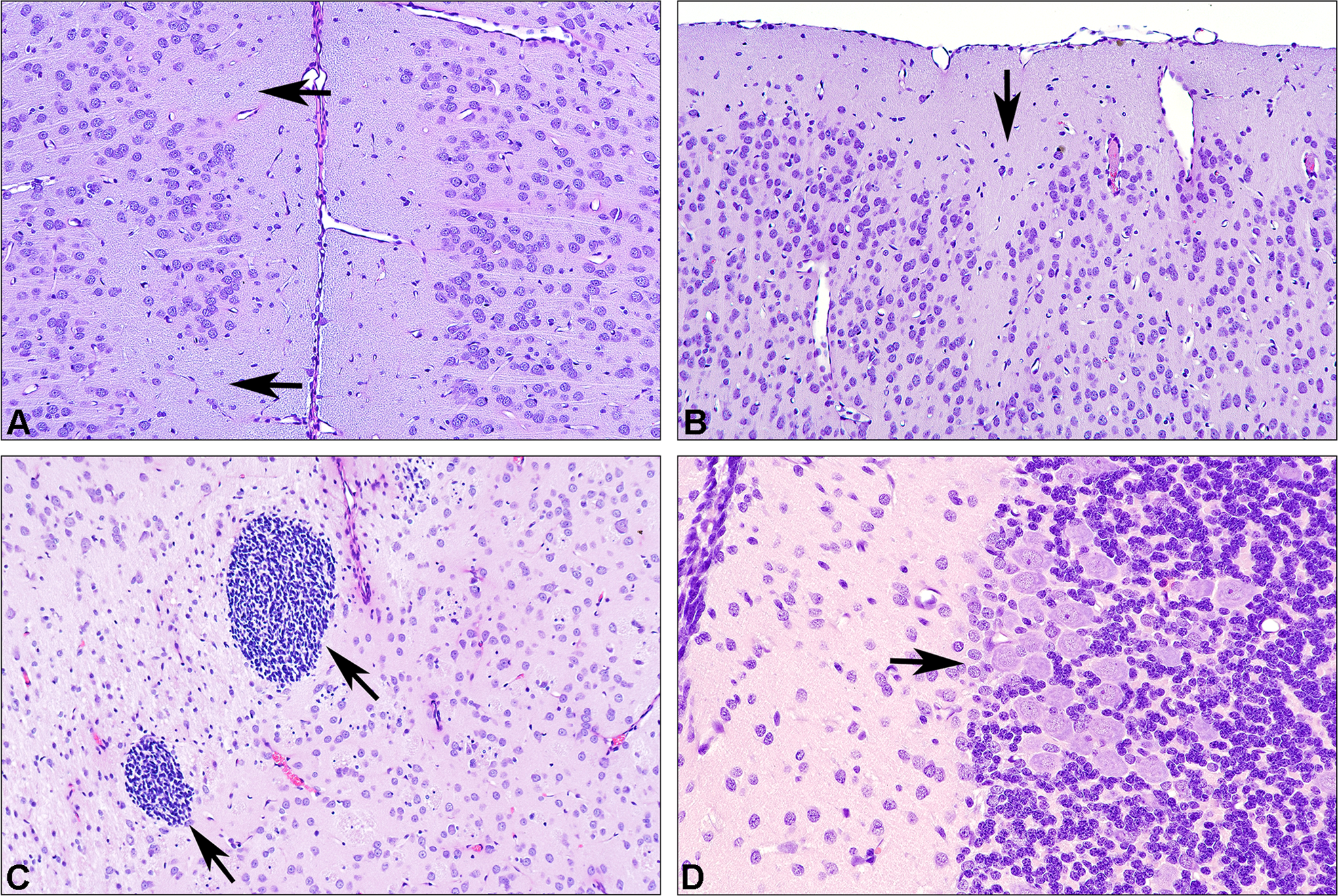
Rat: hematoxylin and eosin (H&E). A, Micrograph of the cingulate gyrus in a young adult (day 70) rat with normal cortical architecture on the right side but with an irregular contour to layer II on the left. Arrows point to defects in the superficial portions of layer II suggestive of incomplete neuronal migration. Based on the H&E-stained section alone, this would be classified as “neuronal dysplasia.” B, Another example of neuronal dysplasia in a young adult (day 70) rat involving layers II and III of the occipital cortex. C, Foci of small round cells within the caudate–putamen of a juvenile (day 22) rat. Morphologically, the constituent cells resemble blastic cells rather than lymphocytes, although they are of uncertain lineage. These nests are only encountered as normal (albeit uncommon) findings in juvenile rats and are not seen in adults. These nests are interpreted as representing evidence of delayed or incomplete migration. A diagnosis of cellular ectopia would not have been considered for a juvenile brain such as this since the constituent cells either degenerate or complete their migration by the time of adulthood. D, An unusually dense cluster of Purkinje neurons found focally within the cerebellar cortex (arrow) of a juvenile (day 22) rat. This was judged to not represent a tangential cut of the Purkinje neuron layer. This alteration was interpreted as representing a neuronal migratory defect leading to neuronal dysplasia.
Second, in the absence of easily recognized qualitative findings, quantitative changes to simple morphometric measurements (eg, linear or areal dimensions) may indicate the presence of neurotoxicity. 12,44 The evidence is more trustworthy if many end points are affected at the same time. 44 The essential prerequisite for effective morphometric assessment is that sections from all animals must be highly homologous, which requires considerable technical expertise in identifying key neuroanatomic structures in embedded tissues or on unstained floating sections. 44 The choice of measurements to procure will depend on such factors as regulatory guidance, biological relevance of the propounded dimensions, and the reliability of measurement capture, as well as pathologist and institutional preferences. Although proposed as a potentially more sensitive index of DNT than simple morphometric measurements, 45 stereological enumeration of selected neural cell populations is less practical for conventional DNT screening studies because they require much more time to collect 36 and will not detect alterations in such common noncellular indices of DNT as deficient white matter myelination, reduced synaptic numbers, and/or diminished dendritic branching, 44 unless specifically designed to do so.
Confounding Influences That Impact Translation of Animal-Derived Neuroscience Data
The last presentation, by Dr Brad Bolon, reviewed fundamental biological and study design factors that, if neglected, may impair efforts to extrapolate animal data for human risk assessment.
46,47
Known neuroanatomic attributes that drive mammalian variations in neurotoxic responsiveness include species, strain, sex, and age (or developmental stage). For instance, the lissencephalic (smooth) rodent cerebral cortex supports 5-fold fewer neocortical interneurons and receives 4-fold less blood than the gyrencephalic (folded) cortex of nonhuman primates.
48
–50
Similarly, the mass of the corpus callosum and the hippocampus in rodents depends on the strain and substrain (eg, BALB/cJ, BTBR T/+
Unfortunately, many other physiological parameters tend to be ignored during the experimental design phase, if they are known at all. For example, divergent neural cell populations exhibit differing metabolic capabilities across species, 61,62 strains, 63 and under shifting husbandry conditions. 64 Similarly, the categories and/or receptor sensitivities of neurotransmitters for discrete brain regions often vary among species 65,66 and strains. 67,68 The tissue distribution and retention of lipophilic molecules, which can readily enter the CNS, fluctuate among individuals depending on individual differences in body composition such as quantities of adipose tissue 69 versus lean muscle. 70 The lung serves as a depot for lipid-soluble xenobiotics (eg, anesthetics, opioids), acting as a slow-release reservoir to bind approximately 25% of such agents following intravenous administration. 71,72 Neurotransmitter classes and/or sensitivities exist in homologous brain regions of different species 65,66 and strains. 67,68 Recent publications have even reported that dissimilarities in intestinal microflora may substantially influence the presentation of many neurological disorders, including autism spectrum disorders 73,74 and neurodegenerative diseases. 75,76
The influence of neglected factors on translational neuroscience may be limited in 2 major fashions. First, scientists will need to continuously recall that the neurological response elicited in a single study can be considered characteristic only for the strain (or stock or breed) utilized in the assay; assessment of a species’ representative reaction in most cases will entail conduct of equivalent experiments in 2 or more strains of the species. This approach is analogous to the traditional product development strategy in which the toxicity of new test articles is examined in multiple species, typically in both sexes and sometimes at 2 or more ages. Second, data extrapolation will be facilitated by ensuring that study reports and future publications will completely and exactly relate key demographic traits (eg, genetic background, sex, age, and husbandry conditions) as well as full methodological particulars. 77 Attention to such details will allow all stakeholders (nonclinical and clinical scientists, corporate managers, and regulators) to better appreciate the full weight of evidence, including animal-derived data, regarding the neurotoxic potential of NCEs and NMEs.
Conclusions
In summary, this half-day continuing education session highlighted several important fashions in which toxicologic pathology facilitates modern translational neuroscience, recognizing that the pathology analysis is only a single means by which experimental animal tissues are evaluated in producing a comprehensive data set for neurotoxic risk assessment. Current best practice recommendations for nervous system sampling and microscopic analysis in nonclinical general toxicity studies involve the analysis of approximately 50 or more key brain regions, which support critical physiological functions, in multiple brain sections (6 or 7, depending on species). 13,16 In the future, subtle neurotoxic lesions may be identified more easily by expanding the conventional testing strategy to include such technical innovations as noninvasive neuroimaging and neural tissue-specific biomarkers. Confidence when extrapolating animal-derived data to predict human risk will also be improved by thorough communication of experimental design and procedural details as well as regular remembrance of structural/functional differences among species and individuals.
Footnotes
Acknowledgments
The authors thank the members and staff of the American College of Toxicology (ACT) for selecting this Society of Toxicologic Pathology (STP) continuing education course for re-presentation to ACT members. The authors also appreciate the support originally provided to formulate this course by the STP Annual Symposium Committee and STP Continuing Education Subcommittee. Dr Rao acknowledges the contribution and support from the National Toxicology Program where the work on modified rodent neuropathology evaluation was conducted.
Authors’ Note
This article reflects the views of Deepa B. Rao on research conducted during her tenure at the National Toxicology Program (NTP) and should not be construed as representing views or policies of her current employer—the US Food and Drug Administration. This works represents a substantially recrafted version of a work first published by Morrison et al, “Fundamentals of translational neuroscience in toxicologic pathology: optimizing the value of animal data for human risk assessment.”
Author Contributions
All authors contributed to the conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agree to be accountable for all aspects of the work. Alok K. Sharma and James P. Morrison have equally contributed to the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
