Abstract
A half-day Society of Toxicologic Pathology continuing education course on “Fundamentals of Translational Neuroscience in Toxicologic Pathology” presented some current major issues faced when extrapolating animal data regarding potential neurological consequences to assess potential human outcomes. Two talks reviewed functional–structural correlates in rodent and nonrodent mammalian brains needed to predict behavioral consequences of morphologic changes in discrete neural cell populations. The third lecture described practical steps for ensuring that specimens from rodent developmental neurotoxicity tests will be processed correctly to produce highly homologous sections. The fourth talk detailed demographic factors (e.g., species, strain, sex, and age); physiological traits (body composition, brain circulation, pharmacokinetic/pharmacodynamic patterns, etc.); and husbandry influences (e.g., group housing) known to alter the effects of neuroactive agents. The last presentation discussed the appearance, unknown functional effects, and potential relevance to humans of polyethylene glycol (PEG)–associated vacuoles within the choroid plexus epithelium of animals. Speakers provided real-world examples of challenges with data extrapolation among species or with study design considerations that may impact the interpretability of results. Translational neuroscience will be bolstered in the future as less invasive and/or more quantitative techniques are devised for linking overt functional deficits to subtle anatomic and chemical lesions.
Keywords
Potential neurotoxicants in the environment are likely to be numerous (Roe, Pease, and Silbergeld 1997). Indeed, the recognition that chemically induced neurological deficits are a major occupational health hazard (Connelly and Malkin 2007; Gobba 2003) and a primary cause of neurodevelopmental damage (Bearer 2001) has resulted in a growing international effort to screen for neurotoxic liabilities in currently marketed as well as new chemicals and pharmaceuticals. Estimates based on literature reports suggest that approximately 22% to 29% of chemicals and drugs may be neurotoxic in adult and/or developing animals and humans (Olson et al. 2000; Anger 1984). The potential for widespread injury and the associated familial and societal costs have assured neurotoxicity a prominent place among the most critical global health issues in the 21st century.
Extrapolation of animal data to predict possible human outcomes is the basic problem in product discovery and development. The rationale for using neural findings in animals (especially rodents) as potential predictors of human neurotoxic responsiveness is the similarity of major neuroanatomical areas in the central nervous system (CNS) among these species (Bolon 2000) and the correspondence of structural lesions to functional deficits in both rats and humans. This equivalency is even closer for nonrodent species of mammals (Bolon 2000). However, despite such broad similarities, minor to fairly substantial differences in anatomic, biochemical, and/or functional attributes for various species as well as differing exposure scenarios may complicate extrapolation of data among animal species (Fu et al. 2010; Van der Zee and Keijser 2011; Bonthuis et al. 2010), let alone from test animals to humans (Pryce 2008; Casals et al. 2011). The large number of biological factors and experimental design considerations that may impact study outcomes are the basis for compiling data sets for risk assessment via a multidisciplinary strategy in which neural integrity is surveyed routinely via many modalities: behavior, neurochemistry, neuropathology, neuropharmacology, and pharmacokinetics, to name the most common fields.
Despite this comprehensive approach, data translation from animals to humans often remains challenging because of differences in the available data sets assembled for risk assessment. No single species, animal model, or study design is ideal in all instances for efficacy evaluations or safety assessments. Toxicologists and pathologists must select from a diverse mixture of animal species (e.g., mouse, rat, rabbit, dog, pig, nonhuman primate) as well as many study types/durations (e.g., acute, subchronic, chronic, developmental neurotoxicity (DNT), adult neurotoxicity) in order to best model the potential risks and benefits posed by exposing a diverse human population to new therapeutic and medical device candidates. In addition, possible approaches to evaluating the nervous system and communicating the data can differ among institutions, ranging from a standard screening approach (Rao et al. 2011; Pardo et al. 2012; Bolon et al. 2013) to a much more extensive evaluation (Switzer, Lowry–Franssen, and Benkovic 2011). This overall complexity is also reflected in the diverse regulatory guidelines that exist within and among countries (Bolon, Bradley, Butt, et al. 2011; Salvo and Butt 2011).
The challenges in extrapolating animal data to assess the potential risk to humans of novel neurotoxic hazards led to a decision by the Society of Toxicologic Pathology (STP) to present a continuing education (CE) course on “Fundamentals of Translational Neuroscience in Toxicologic Pathology” at its 2014 annual meeting in Washington, DC. This half-day CE session was designed to give STP members and regulatory scientists from the U.S. Food and Drug Administration a stronger background in the subject, including the most up-to-date information regarding basic neurobiological principles and practical techniques needed to address neurotoxic hazard identification and risk assessment. The symposium was split into 5 lectures to fulfill this purpose.
The first 2 talks provided a coordinated overview that examined the functional relevance of neuroanatomic structures in the CNS of animal species utilized in toxicity testing. Dr. Deepa Rao delivered the initial lecture, which focused on structure–function correlations for brain regions that are commonly evaluated by toxicologic pathologists from conventional brain preparations in rodent general toxicology studies. Specifically, the review of neuroanatomic areas was based on the 7-section approach adopted recently by the National Toxicology Program (2011; Rao et al. 2011), which later was adapted in large part as the recommended STP strategy for sampling rodent brains in general toxicity testing (Bolon et al. 2013). Principal regions selected for evaluation include olfactory bulbs, frontal pole (frontal cortex and basal nuclei), anterior commissure, parietal cortex (for motor and sensory regions), hippocampus, rostral and caudal thalamus/hypothalamus, occipital lobe, cerebellum, and brain stem (pons and medulla oblongata). Expansion of brain sampling to include additional sectioning is warranted because the traditional 3-section screen (Morawietz et al. 2004) will miss subtle (Switzer, Lowry–Franssen, and Benkovic 2011) or even major lesions (Tiedeken, Muha, and Ramsdell 2013) that would be detected when using a 7-section sampling scheme. Within these seven brain sections, approximately 50 to 60 distinct neuroanatomic structures serving the most key cognitive, homeostatic, motor, and sensory functions (Rao, Little, and Sills 2014) may be identified for evaluation during routine screening of toxicological studies; this expanded sampling approach permits examination of the main brain structures known to be quite vulnerable to neurotoxicant exposure (Switzer, Lowry–Franssen, and Benkovic 2011), and thus may increase the possibility of detecting previously unknown neurotoxicants. Such analyses will require that pathologists become familiar with annotated neuroanatomy atlases of multiple species (e.g., Palazzi 2011; Paxinos, Huang, and Toga 2000; Paxinos et al. 2008; Paxinos and Watson 2009; Franklin and Paxinos 2008). Examples of site-specific brain lesions were given, including carbonyl sulfide in the caudal (inferior) colliculus (Morgan et al. 2004) and trimethyltin in white matter tracts (Johnson et al. 2014). Of particular importance was Dr. Rao’s assertion that future studies may perform morphologic analyses by combining neuropathology with novel neuroimaging end points such as magnetic resonance microscopy (MRM; Badea and Johnson 2013; Johnson et al. 2014), positron emission tomography (PET; Xi, Tian, and Zhang 2011), and ultrasound biomicroscopy (UBM; Powell and Wilson 2012). These strategies will allow high-resolution anatomic evaluation post mortem, commonly using contrast agents to enhance the tissue detail (MRM) or repeated examination of the same anatomic structure at multiple times during longer toxicological evaluations (PET and UBM), which could increase the possibility of determining which areas might be targets of potential neurotoxicity prior to brain sampling. Such special studies may necessitate the use of satellite groups since the routine fixation and processing methods for neuropathology specimens may not mesh well with the technical aspects for these novel methods. While time constraints limited the discussion to the brain, corresponding function–structure correlations also can be drawn for spinal cord, nerves (sciatic and trigeminal), and autonomic (intestinal) ganglia (Rao, Little, and Sills 2014).
Dr. Ingrid D. Pardo gave a follow-up overview of “Structural-Functional Correlations in Neuropathology Evaluations in Non-rodents” that complemented the first rodent-based talk. Studies conducted in nonrodent species (e.g., dog, nonhuman primate) typically predict human toxicity better than studies conducted in rodents, although instances in which concordant toxicity is observed in both rodent and nonrodent studies have the highest likelihood of successfully anticipating human outcomes (Olson et al. 2000). The recommendations of the STP for routine sampling of brain, spinal cord, and peripheral nerve in nonrodents during regulatory-type general toxicity studies (Bolon et al. 2013) were reviewed. For this purpose, the STP advocates routine evaluation of 6 brain sections (including all major structures evaluated in rodents except for olfactory bulbs); 3 segments of spinal cord (cervical, thoracic, and lumbar intumescence); and cross- and longitudinal sections of at least 1 mixed peripheral nerve (Pardo et al. 2012; Bolon et al. 2013). Dr. Pardo then presented 3 examples from nonhuman primate studies in which the animal data effectively predicted the pattern of human toxicity, with emphasis on the neuroanatomic and neurofunctional similarities between nonhuman primates and humans. These examples were (1) emesis caused by a nicotinic receptor partial agonist, (2) spinal cord injury with functional (motor) end points resulting in decreased progression of Parkinson disease and improved responsiveness to treatment (Courtine et al. 2007), and (3) drug-induced sensory neuropathy (where the sural nerve is exclusively sensory in nonhuman primates and humans but has mixed sensory and motor functions in rodents). Other case examples described morphologic brain lesions caused by glucokinase activators (Pettersen et al. 2014) or systemic administration of a monoclonal antibody leading to immune complex deposition in the choroid plexus (Figure 1). Strategies and assays that can be used early in the drug development process to predict, understand, and mitigate nervous system toxicity include a comprehensive literature review of the known targets and mechanisms of intended action; early target characterization (molecular evaluation correlated with appropriate in vitro and in vivo studies); review of any genetically modified rodent models relevant to the intended mechanism of action; evaluation of physiochemical properties of the test article and related molecules (Hughes et al. 2008); receptor binding assays to detect off-target pharmacology; and in vivo discovery studies to permit sampling of major brain areas to measure tissue drug concentrations. Pathologists who regularly engage in neuropathology assessments of nonrodent species should become familiar with the wide array of resources available for cross-species extrapolation (Bolon, Bradley, Garman, et al. 2011; Bolon and Butt 2011).
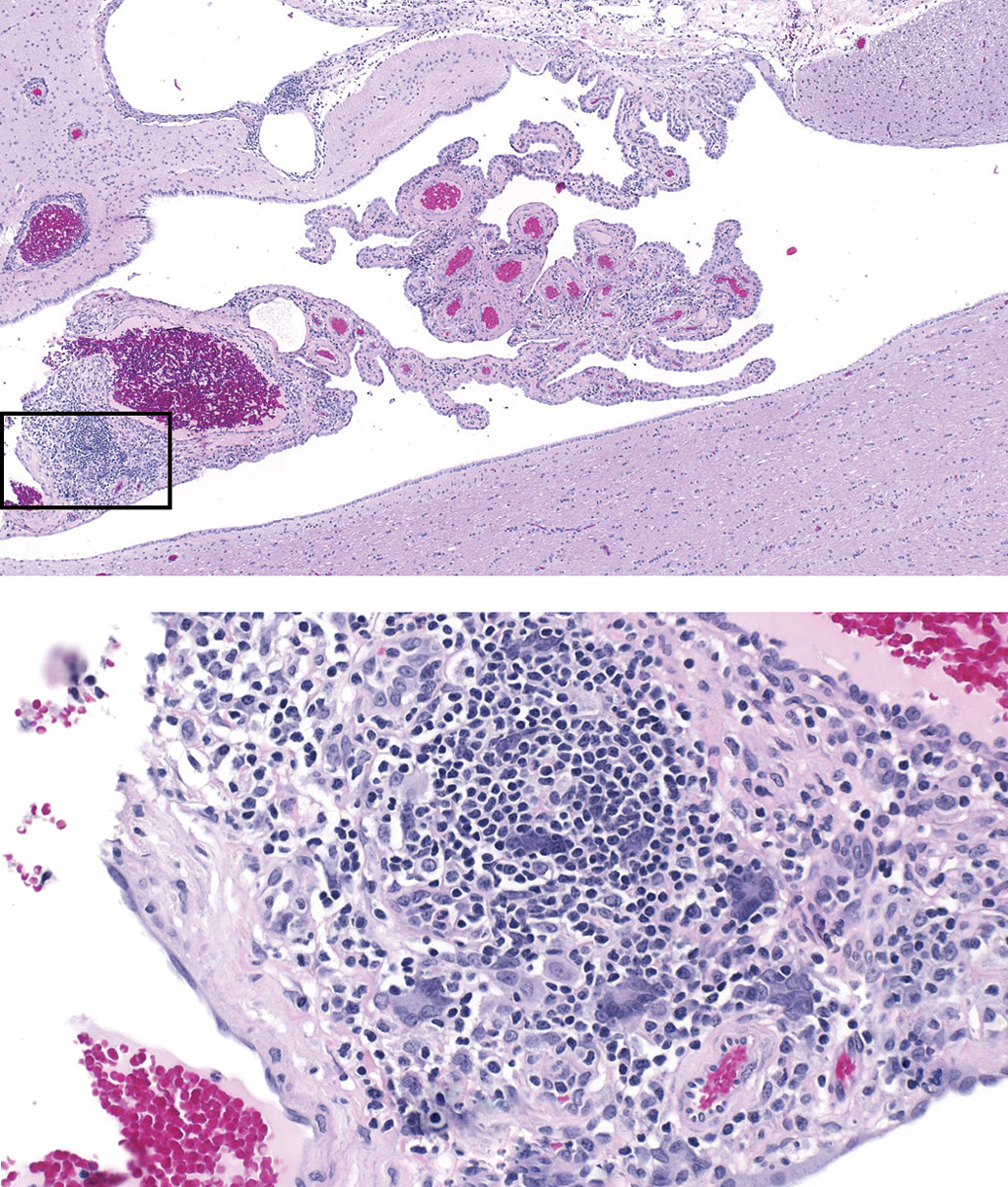
Monoclonal antibody–induced mononuclear cell infiltrate in the choroid plexus of a cynomolgus monkey, thought to be caused by immune complex deposition. (The box in the upper image denotes the field shown in the lower image.).
The third presentation on “Pathology Considerations in Developmental Neurotoxicity (DNT) Testing” was delivered by Dr. Robert H. Garman. Whereas the mission of pathologists involved in the majority of neurotoxicity studies in adults is to detect the presence of degenerative processes (or reactions to the same), toxicant-induced damage in the developing brain often is cleared prior to the postnatal time points at which the CNS typically is evaluated during DNT studies, and such developmental effects seldom invoke a lasting telltale response by either astrocytic or microglial cells. Thus, the typical morphologic lesions in DNT studies are treatment-related morphometric differences in subregions of the brain. Although, volumetric measures and cell counting methods (such as those utilized in optical or physical dissector stereology) or computer-based areal measures frequently may be used as targeted investigational tools, simple linear measures are employed by most laboratories for obtaining morphometric data in DNT studies on rats (Bolon et al. 2006). The ability to take such linear measures accurately requires highly homologous brain sections, which in turn depends on adequate technician training and the accurate identification of specific neuroanatomic regions within unstained sections floating on a water bath. Once homologous sections are obtained and stained, consideration must be given to which measurements should be taken—not only which brain regions to measure, but also the number and orientation of the lines of measurement. In determining which measurements to take, considerations should include whether or not the measurements have biologic relevance (from both the standpoints of the test article and basic principles of developmental neurobiology), whether or not the measures can be taken consistently or are excessively affected by slight differences in section level, and possibly issues relating to historical data already present within the laboratory undertaking the work. Because only a small number of neuroanatomic regions are actually measured in the course of a DNT study, other methods of subjective assessment also are warranted, including comparing sizes of different brain regions and some special staining procedures (e.g., Luxol fast blue to highlight myelinating white matter tracts). Newer methodologies for evaluating brain developmental morphology were also mentioned, concentrating on a discussion of high-resolution magnetic resonance-based microimaging (MRI) techniques that allow for the 3-dimensional digital reconstruction of the brain and facilitate the determination of volumes for a relatively large number of neuroanatomic regions (Bolon, Garman, et al. 2011; Calabrese et al. 2013). Although very few contract laboratories currently perform such high-resolution imaging, MRI techniques likely will become valuable tools for performing DNT studies in the not too distant future. The ability to discriminate multiple nucleic acids (Shiraki et al. 2014; Contet 2012; Youdim 2003) or proteins (Jha and Suk 2013; Li et al. 2012) in small quantities of fluid or tissue offers the promise that relatively noninvasive micro-sampling of brain or cerebrospinal fluid (CSF) eventually might produce biomarkers of use for assessing neurotoxic risk. At present, such premonitory biomarkers are unavailable for routine use with neural specimens.
The fourth talk, by Dr. Brad Bolon, provided an overview of “Neglected Factors That May Confound Translational Neuroscience Research.” Conventional neurotoxicity testing is conducted successfully using a blend of functional testing (Moser 2000, 2011) and structural evaluation (Bolon et al. 2013; Fix and Garman 2000; Hale et al. 2011), although in some cases highly localized lesions may be missed by one or both of these approaches (Switzer, Lowry–Franssen, and Benkovic 2011). While the occurrence of artifacts can represent a confounding influence to neuropathology assessment (Garman 1990), many other factors may, either individually or in combination, lead to confusion when trying to utilize animal data for human risk assessment (Calabrese 1991; Claassen 1994). Commonly considered sources of divergence include species, strain, sex, and developmental stage (age). For example, the gyrencephalic brain of nonhuman primates has 5-fold more neostriatal interneurons and receives 4-fold more blood from the heart relative to the lissencephalic rodent brain (Forsyth et al. 1968; Graveland and Difiglia 1985; Stott, Dryzga, and Ramsey 1983). In rodents, the sizes of the corpus callosum and hippocampus differ among strains, and these anatomic discrepancies may impact behavior (Jamot, Bertholet, and Crusio 1994; Fairless et al. 2008; Wahlsten, Ozaki, and Livy 1992). Male animals, including primates, tend to have more neurons, while female animals seem to have more connections among neurons; this relationship may explain the lower incidence of learning disabilities in female humans (de Courten-Myers 1999; Pakkenberg and Gundersen 1997; Pakkenberg et al. 2003). Age-related atrophy due to neuronal depletion is a feature of dog (González-Soriano et al. 2001) and primate (Henderson, Tomlinson, and Gibson 1980; Anderson et al. 1983) brains, while rodent brains generally are spared this process (Jucker and Ingram 1997). In general, researchers do a reasonable job of accounting for these obvious factors in designing their experiments.
In contrast, other sources of divergence are often forgotten or ignored in the course of experimental design. For example, approximately 25% of typical lipophilic chemicals (which includes most neuroactive agents) are retained by the lung tissue for later slow release following intravenous exposure (Benowitz et al. 1974). Variations in body composition among strains with relatively more adipose (Decad, Birnbaum, and Matthews 1981) or lean muscle (Lionikas et al. 2013) mass also may impact the distribution and retention of lipid-soluble molecules. The neurotransmitter complement and/or sensitivity often differs for a given site among species (Berger, Gaspar, and Verney 1991; DeFelipe, Alonso–Nanclares, and Arellano 2002; Fitzgerald 2009) and strains (Finn, Kovács, and Pearce 2010; Anderzhanova et al. 2013), while the metabolic capacities of various neural cell populations vary among species (Colton et al. 1996; Van der Zee and Keijser 2011), strains (Sato et al. 1996), and husbandry conditions (Lokiec, Cohen, and Jacquot 1981). Recent reports even indicate that the gut microbiome may profoundly impact brain functions in various disease states, including autism spectrum disorders (Douglas-Escobar, Elliott, and Neu 2013; Hsiao et al. 2013) and neurodegenerative diseases (Hill et al. 2014; He and Balling 2013).
The influence of these neglected factors on neurotoxicity testing and neurotoxic risk assessment may be diminished in 2 fashions. First, researchers will need to constantly remind themselves that the response in a single animal type (e.g., strain, stock, or breed) is not representative of the whole species, which may require the completion of parallel studies in 2 or more strains to approximate the responsiveness for the entire species. Indeed, this necessity is acknowledged in current product development paradigms by the need to test new materials in multiple species, and often both sexes and at 2 or more ages. The second means of minimizing translational difficulties is to ensure that study documentation includes exact descriptions of important demographic traits, such as the genetic background, sex, and age (as defined both by chronological means and by body weight) as well as meticulous recordings of husbandry conditions that might impact neurobiological functions and processes. Such details will permit nonclinical scientists, clinicians, and regulators to make better extrapolations of possible or likely human responses based on animal-derived data.
In the last talk, Dr. Wolfgang Kaufmann considered the implications of “Polyethylene Glycol (PEG)-associated Vacuolation in Epithelial Cells of the Choroid Plexus and Its Impact on Development of PEG-conjugated Therapies.” PEG is an inert, essentially nontoxic, and nonimmunogenic polymer of variable length and conformation (branching, linear, etc.). It has amphiphilic (both hydrophilic and hydrophobic) properties and thus shows good solubility in both aqueous and organic milieus. PEG is conjugated to macromolecules as a successful strategy to overcome some of their intrinsic properties that limit their broad therapeutic use: instability, degradation by proteolytic enzymes, rapid excretion, and potential immunogenicity; the addition of the PEG moiety minimizes the exposure of antigenic determinants and the generation of neutralizing antibodies. PEGs used for conjugation to biomolecules typically have a molecular weight (MW) ≥ 5 kDa and are therefore unlikely to be metabolized to any significant degree. Clearance instead occurs largely through passive glomerular filtration and excretion in the urine and/or by uptake into phagocytes or certain secretory cells, with the choice of route depending chiefly on the shape and size of the combined PEG-biomolecule construct and perhaps also on other currently unknown or incompletely understood factors. Hepatobiliary removal represents a minor route of PEG excretion and is dependent on MW with hepatic clearance reaching a minimum at about 50 kDa molecular mass relative to the increased hepatobiliary clearance of lower and higher MW PEGs (Webster et al. 2007). The increased size of PEGylated proteins causes a clear slowing of renal ultrafiltration, which leads to a prolonged plasma circulating time and half-life. Therefore, the PEGylation strategy effectively alters the in vivo efficacy of large molecules by shifting the balance between their pharmacodynamic and pharmacokinetic properties, which results not only in improved efficacy but also yields reduced dosing frequency and increased patient compliance (Fishburn 2008).
Nonclinical toxicology studies with PEGylated proteins have revealed no PEG-specific toxic (adverse) findings in animals (Webster et al. 2007). However, such studies have demonstrated a vacuologenic capacity of PEG-conjugated biomolecules, the extent of which depends on the size and shape of the PEG moiety (Bendele et al. 1998; Johanson et al. 2011; Rudmann et al. 2013). Chronic use of high MW PEGs (≥10 kDa MW) is associated with accumulation of foamy (vacuolated) macrophages in regional lymph nodes, spleen, and thymus as well as adrenal glands, uterus, and cervix, and/or cytoplasmic vacuolation of renal epithelium and/or choroid plexus epithelial cells (CP; Figure 2). The degree of vacuolation (as indicated by a histological severity score) and reversibility is dose and time dependent. Over time, vacuoles remain but the proteins to which they were conjugated do not, suggesting that the PEG and/or degradation by-products are retained long after the biomolecule is degraded. The pathology evaluation generally is considered to show that cytoplasmic vacuolation is an adaptive response to an increased demand for PEG clearance rather than a toxicologic response due to effects of PEG on cellular physiology. The conundrum is that the functional consequences (if any) of sudden PEG release from many cells at once are unknown.
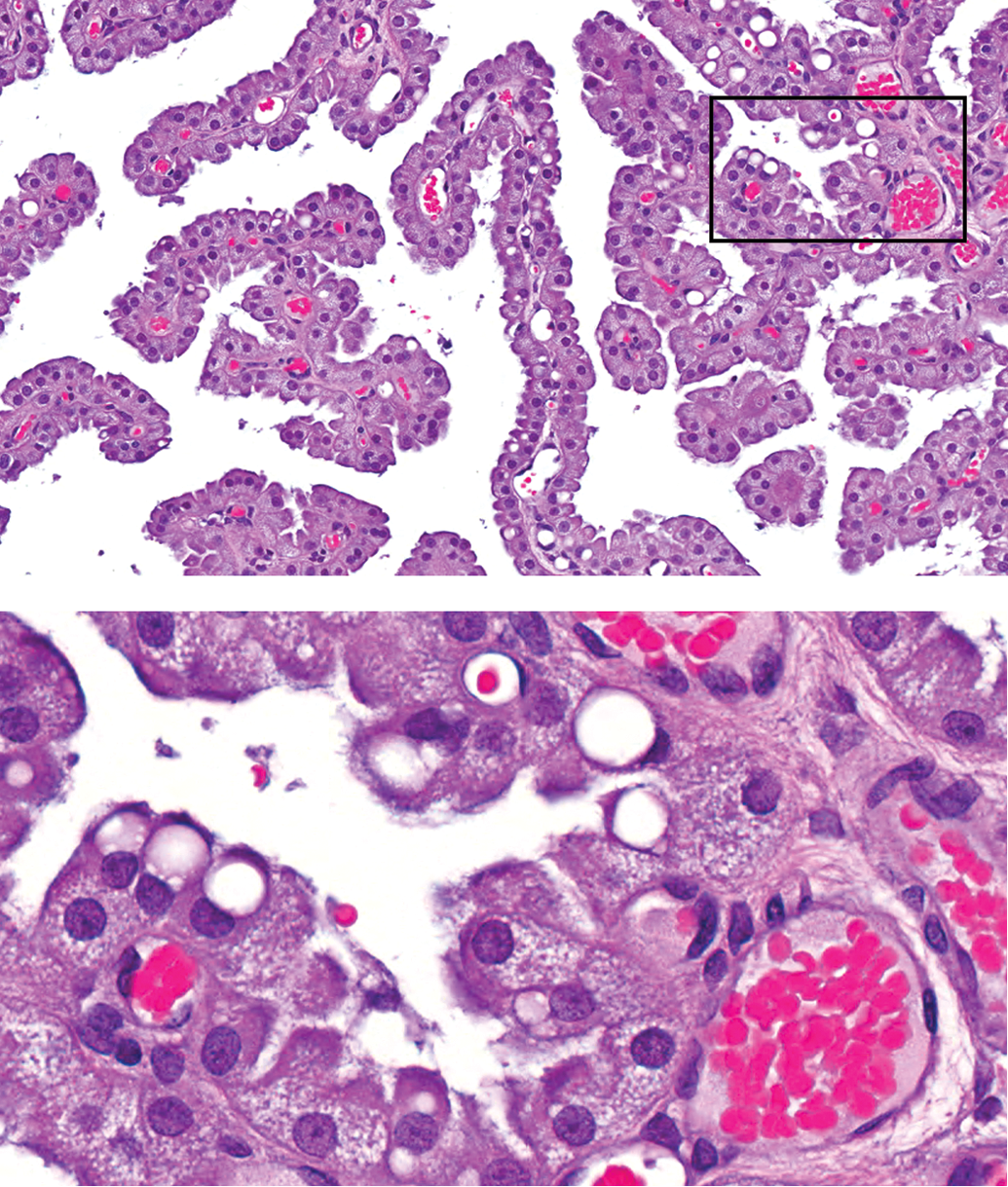
PEG–related vacuole formation in the choroid plexus (CP) epithelium of a rat. At higher magnification (inset showing field outlined by the box in the upper image), the cytoplasm of most epithelial cells contains fine vacuoles leading to pallor.
Dr. Kaufmann’s talk highlights the difficulty of translational neuroscience in instances when human risk assessment depends chiefly or entirely on animal data. A series of studies in cynomolgus monkey studies showed vacuolation of the CP as the only histopathological finding. With increasing treatment duration (up to 52 weeks), the vacuole score rose from minimal up to moderate degree, and the vacuolation persisted or progressed after 8- or 13-week recovery periods. This pattern was considered to have significant relevance for human risk assessment as the similarity of the CP in humans and cynomolgus monkeys suggests that administration of a PEGylated agent to humans likely would result in CP vacuolation, yielding a comparable distribution to that seen in monkeys. Prolonged administration of a PEG-conjugated molecule might not alter the viability of the CP epithelium, but it cannot be confirmed definitively that the function of the CP epithelial cells will not be impaired by the accumulating PEG. There is no firm evidence (e.g., PEG accretion in CSF, infiltration of macrophages) that PEGylated molecules pass through the CP epithelial cells into the ventricular system. The rates of PEG removal, replacement of CP epithelial cells during life, and the effect of PEG accumulation on protein degradation by epithelial cells are not known in monkeys or in humans. Therefore, in this case, it is extremely difficult to forecast the fate of the vacuolated CP cells and to clearly predict the relevance of a potential long-term liability for the human choroid plexus and brain due to treatment with PEGylated molecules. Additional studies to examine the nature of the material within vacuoles (e.g., anti-PEG immunohistochemistry [IHC], oil red O); the CP cell turnover rate (e.g., IHC for markers of apoptosis [cleaved caspase-3] and/or cell proliferation [Ki67, PCNA]); and the integrity of cell architecture (e.g., transmission electron microscopy) may provide further evidence to indicate that the accrual of PEG in vacuolated CP cells is not associated with a change in neural cell function or structure.
Pediatric development of PEGylated drug products, in which agents that induce CP epithelium vacuolation will need to be administered for many years, should only be initiated in cases where a sufficiently large safety margin can be established in a long-term toxicity study (e.g., 104 weeks) using animals representative for the pediatric population in question. More importantly, product development should continue only if vacuolation can be demonstrated to be reversible if administration of the PEGylated material is stopped. Ex vivo/in vitro data evaluating the effect of PEG vacuolation on the viability and function of CP epithelial cells might contribute to the risk assessment process, but it will serve as a supplement to the in vivo data.
In conclusion, this half-day session highlighted several important aspects of toxicologic neuropathology as applied to modern translational neuroscience, recognizing that pathology is one major element but not the sole platform needed to produce the full set of data needed for a comprehensive neurotoxic risk assessment. Current conventions in brain sampling for neuromorphologic assessment of adult and developing animals are suitable means of screening as long as specific brain regions mediating critical functions are identified and evaluated (Rao, Little, and Sills 2014). New investigational tools, particularly biochemical assays and noninvasive imaging, may promote a more efficient and effective means of assessing neurotoxic risk in the future. While instances are likely to arise regularly in which a morphologic finding is observed for which the consequences to neural function in humans are unknown (e.g., accumulation of PEG-filled vacuoles in the choroid plexus epithelium), our ability to predict the potential for human adverse events due to neurotoxic complications should continue to improve with ongoing technical innovation to develop less invasive and/or more specific techniques for linking overt functional deficits to subtle anatomic and chemical lesions.
Footnotes
Acknowledgments
The authors thank the Society of Toxicologic Pathology’s (STP) Annual Symposium Committee, the Continuing Education Sub-Committee, and the Scientific Program Planning Committee for their support of this program. We would also like to thank the staff of the STP for organizational support in producing the materials and arranging the meeting space and Steve Van Adestine of Covance Laboratories Inc. for assisting with the figures.
Author Contribution
JM, AS, DR, IP, RG, and WK< BB contributed to conception or design; JM, AS, DR, IP, RG, WK, and BB drafted the manuscript; JM, AS, DR, IP, RG, WK, and BB critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Two authors (JM, AS) contributed equally to the article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
