Abstract
The ALLIANCE working group on effects of ionising radiation on wildlife brings together European researchers to work on the topics of radiosensitivity and transgenerational effects in non-human biota. Differences in radiation sensitivity across species and phyla are poorly understood, but have important implications for understanding the overall effects of radiation and for radiation protection; for example, sensitive species may require special attention in monitoring and radiation protection, and differences in sensitivity between species also lead to overall effects at higher levels (community, ecosystem), since interactions between species can be altered. Hence, understanding the mechanisms of interspecies radiation sensitivity differences may help to clarify mechanisms underpinning intraspecies variation. Differences in sensitivity may only be revealed when organisms are exposed to ionising radiation over several generations. This issue of potential long-term or hereditary effects for both humans and wildlife exposed to low doses of ionising radiation is a major concern. Animal and plant studies suggest that gamma irradiation can lead to observable effects in the F1 generation that are not attributable to inheritance of a rare stable DNA mutation. Several studies have provided evidence of an increase in genomic instability detected in germ or somatic cells of F1 organisms from exposed F0 organisms. This can lead to induced radiosensitivity, and can result in phenotypic effects or lead to reproductive effects and teratogenesis. In particular, studies have been conducted to understand the possible role of epigenetic modifications, such as DNA methylation, histone modifications, or expression of non-coding RNAs in radiosensitivity, as well as in adaptation effects. As such, research using biological models in which the relative contribution of genetic and epigenetic processes can be elucidated is highly valuable.
1. Introduction
Species radiosensitivity has been studied for decades. While an acute dose of 10 Gy is lethal to most vertebrates, including humans, other organisms are much less sensitive to ionising radiation (Harrison and Anderson, 1996). The most radioresistant organism known is the extremophile bacterium
Invertebrates and plants are generally more radioresistant than vertebrates (Garnier-Laplace et al., 2006, 2010), either in acute or chronic exposure conditions. However, species radiosensitivity can be highly variable. For example, for chronic exposure and sublethal effects, species radiosensitivity in laboratory-controlled conditions was shown to vary over six orders of magnitude (Garnier-Laplace et al., 2010).
The multiple factors conferring radioresistance to organisms have not been fully identified. However, the data from various studies indicate that radiation tolerance of the deinococcus bacteria might result from a combination of different molecular mechanisms and physiological determinants, including efficient DNA repair, protection of proteins against oxidation, and a highly condensed nucleoid structure (Cox and Battista, 2005; Slade and Radman, 2011; Daly, 2012). However, a complete understanding of these factors is necessary for the development of robust predictive methods that aim to extrapolate radiosensitivity between species, in order to protect all sensitive species, as well as ecosystem functioning, efficiently.
This paper will present the main possible features that may determine species radiosensitivity or radioresistance. As radiosensitivity or radioresistance can also evolve under long-term exposure, recent data obtained on transgenerational effects will also be presented, considering genetic and epigenetic factors.
2. Cellular and physiological features determining radiosensitivity
Radiosensitivity of organisms is influenced by a number of different factors, and is known to vary for different endpoints (e.g. reproduction vs mortality), life stage (embryo, larva, and juvenile stages are the most sensitive), and individuals and species.
Some general parameters known to determine the sensitivity of an organism to radiation are: the DNA content (i.e. mean chromosome volume) of the cell; the efficiency and types of DNA repair pathways; the cell repopulation capacity; and the ability of tissue and organs to regenerate (reviewed in Harrison and Anderson, 1996). It is also recognised that prior or combined exposure to other stressors can influence an organism’s radiosensitivity (Pereira et al., 2014). For vertebrates, it is believed that this priming response is induced through the immune system; however, it remains to be seen how the immune response is triggered by radiation as such. Nevertheless, immune system priming is not the only factor likely to contribute to radioresistance (for review, see Hilker et al., 2016).
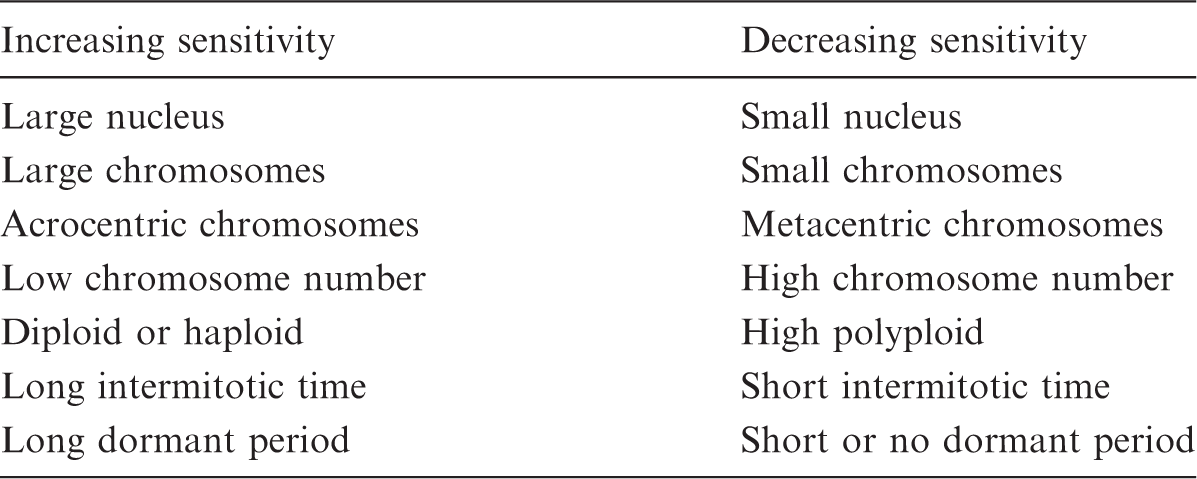
Genetic and reproductive factors influencing the sensitivity of plants to radiation (based on Sparrow and Miksche, 1961).
2.1. Initial infliction of DNA damage
Double-strand breaks (DSBs) formed in

Number of DNA DSBs per haploid genome that an organism can survive.
In different cell types, the number of radiation-induced DNA damages might vary to some extent depending on physical protection afforded by the different degree of packaging of DNA (e.g. histones vs protamines etc.), as well as the presence of a physical barrier around the cell (e.g. a cell wall, trichomes, or a cuticula) (Arena et al., 2014). For example, the high radioresistance of mature spermatozoa (≥25 Gy is required to produce significant levels of DNA damage in spermatozoa) has been attributed to the extremely condensed sperm chromatin (e.g. Haines et al., 2002). In a similar fashion, a highly condensed nucleoid, observed in radiation-resistant
2.2. Checkpoint control mechanisms and DNA repair
The consequence of the induced DNA damage after irradiation depends on whether the lesions are repaired, and if the mode of repair is error free or error prone (see review, Arena et al., 2014). First, the damage has to be recognised, leading to a block in the cell cycle before the cells enter S phase (G1 block) or mitosis (G2 block). These checkpoints apparently give the cells additional time for DNA repair. The various types of DNA damage are repaired by different repair pathways, which differ with respect to speed and fidelity. It is clear that inherent radiosensitivity relates to the efficiency of checkpoint control mechanisms and the repair capability of the cells (reviewed by UNSCEAR, 2000). DNA repair pathways govern the efficiency and fidelity of DNA repair, and, as such, are fundamental in determining cell and organism sensitivity to ionising radiation. This is because when both strands of the DNA are damaged in the same location, no template strand is available for repair. Homologous recombination repair and non-homologous endjoining are the two pathways for DSB repair, of which the former is more complex and considered to be less error prone than the latter (reviewed in UNSCEAR, 2000). The extreme radioresistance of
2.3. Reactive oxygen species scavenging
In addition to direct effects, ionising radiation can induce the production of reactive oxygen species (ROS) when it interacts with different biomolecules or with water. The cellular ROS concentrations are kept under control by a series of mechanisms which consist of both protein and metabolite scavenger components (Noctor and Foyer, 1998). The reason for this strict control is two fold. On the one hand, the balance between ROS formation and ROS removal in the cell has to be controlled to avoid an increase in oxidative damage to the cellular components, such as lipids, proteins, and DNA. On the other hand, correct signalling relies on modulation and control of ROS concentrations to obtain a transient signal (Mittler, 2002; Mittler et al., 2004; Miller et al., 2010). It has been widely shown that activities of antioxidative enzymes are generally increased in various organisms exposed to enhanced levels of ionising radiation (Kovacs and Keresztes, 2002; Zaka et al., 2002; Kim et al., 2005; Vandenhove et al., 2010; Vanhoudt et al., 2010, 2014; Van Hoeck et al., 2015, 2017; van de Walle et al., 2016). As an example, transcriptomic studies in
Interestingly, it has been shown that
Other ROS scavenging systems exist for other organisms. Many fungi, especially melanised ones, have LD10 values approaching or exceeding 1 kGy, and have been found in highly contaminated areas of the Chernobyl Exclusion Zone (Dadachova and Casadevall, 2008). Other pigments, such as carotenoids, play a major role in ROS scavenging, and have been shown to be linked with radiotolerance in many bacteria (e.g.
2.4. Induction of cell death
Exposure to ionising radiation can lead to different types of cell death (i.e. necrosis, apoptosis, and mitotic death). Apoptosis (programmed cell death) can be viewed as a complementary way of getting rid of cells with DNA damage (UNSCEAR, 2000). At low doses, this is probably the most efficient response as it removes damaged cells from the population and reduces the probability of misrepair.
Cells that are seriously damaged by ionising radiation (e.g. containing asymmetric exchange type chromosome aberrations) will undergo mitotic death, leading to loss of proliferation capacity. It is well known that rapidly dividing cells are more radiosensitive than non-dividing cells, and that sensitivity varies within the cell cycle (UNSCEAR, 1996). When it comes to mitotic death, the highest and lowest sensitivity is apparent during mitosis (and late G2 phase) and S phase, respectively, whereas the opposite pattern applies to apoptosis. Furthermore, mitotic death shows a substantial dose-rate effect, whereas the induction of apoptosis for a given dose appears to be independent of dose rate and dose fractionation (UNSCEAR, 2000; Hall and Giaccia, 2006). In response to radiation, the relative importance of the two mechanisms varies with dose and dose rate, as well as with cell type and developmental stage (UNSCEAR, 2000).
2.5. Tissue regeneration
For higher, multi-cellular organisms, the ability and capacity of cells to repopulate themselves, to replace cells damaged or killed by radiation, and to orchestrate tissue and organ regeneration can influence both organ and species radiosensitivity. Many primitive organisms tend to show more efficient tissue and organ regeneration, several of which appear to be relatively radioresistant as adults (Harrison and Anderson, 1996).
2.6. Cell cycle sensitivity
Cells are generally found to be most sensitive to radiation at the beginning of mitosis and least sensitive during S phase. The varying sensitivity of cells in different phases of the cell cycle is not fully understood, but several correlations have been found. The varying amount (duplication during S phase) or form (relaxed vs condensed) of DNA during a cell cycle might influence the sensitivity.
There is also a correlation between radiosensitivity and the levels of naturally occurring sulfhydryl compounds in the cell through the cell cycle. They act as free radical scavengers, and can facilitate direct chemical repair at sites of DNA damage by hydrogen atom donation (Hall and Giacca, 2006).
2.7. Life stage
Reproduction is known to be one of the most radiosensitive biological functions, and it might be impaired at doses corresponding to less than 10% of the dose causing mortality. This can be explained, in part, by the fact that actively dividing cells are most sensitive, and consequently the highest radiosensitivity is likely to be found in cell systems undergoing rapid cell division for either reproduction (e.g. spermatogonia) or growth (e.g. the developing embryo) (UNSCEAR, 1996). It is well known that irradiation leads to apoptosis of cells in the early stages of spermatogenesis, thereby reducing the production of spermatozoa. Furthermore, the successful transfer of the genome to the offspring may be disturbed by gene mutations induced in the germ cells. Induction of recessive and dominant lethal mutations in male and female germ cells may lead to early death of the developing embryo. Other more ‘subtle’ mutations may lead to the development of abnormalities influencing the fitness and fertility of the offspring in the next and subsequent generations. Understanding the underlying mechanisms for differences in sensitivity of germ cells (reproductive organs) and embryonic tissues between different species would therefore be of high relevance since these factors would also be of importance for reproductive strategy (e.g. sexual vs non-sexual reproduction, parthenogenesis).
2.8. Metabolic rate
Other global methods have been used to try to understand species radiosensitivity. Empirical relationships based on allometry (Higley and Bytwerk, 2007), describing but not explaining interspecies variations in radiosensitivity, were used to predict radiosensitivity in untested species or endpoints (Vives i Batlle, 2012; Alonzo et al., 2016). However, the cause for the observed variability among species was not elucidated. This understanding is necessary for the development of robust predictive methods that aim to extrapolate radiosensitivity from laboratory species to natural biota.
Understanding differences in sensitivity to toxicity among species is a challenge for environmental protection against ionising radiation, as well as chemical contaminants. In this context, two mechanistic approaches, namely the GUTS (General Unified Threshold Model of Survival) and DEBtox (Dynamic Energy Budget in Toxicology) models, addressing lethal and sublethal effects, respectively (Jager and Zimmer, 2012), have given an insight into the role of metabolism on intra- and interspecies variability in sensitivity to chemical pollutants (Jager, 2013; Gergs et al., 2015). A study has recently tested the link between no-effect concentrations (NEC), which measure species sensitivity in the long term, and parameters of the dynamic energy budget (DEB) in organisms exposed to four pesticides (Baas and Kooijman, 2015). As a result, NEC values for mortality were particularly well correlated with ‘volume-specific maintenance rate’. This parameter is defined as the amount of energy that is needed to allow survival for one unit of volume. In the DEB theory, it governs the cost of survival and the maximum body size in organisms (Kooijman, 2013). One could hypothesise that, like sensitivity to pesticides, changes in radiosensitivity among species could be explained by differences in volume-specific maintenance rate. If this correlation was confirmed, metabolic rate values could potentially be used to predict radiosensitivity in non-tested species (in more than 400 species that have been described using the DEB theory) (Vrije University Theoretical Biology, 2014).
3. RADIOSENSITIVITY AND TRANSGENERATIONAL EFFECTS
By comparing two sets of chronic radiotoxicity data in terrestrial non-human species, one acquired under controlled conditions in the laboratory, and another acquired from field studies in the Chernobyl Exclusion Zone, field species were shown to have eight-fold higher radiosensitivity (Garnier-Laplace et al., 2013). This suggests that populations in their natural environment were more sensitive than organisms in laboratory-controlled conditions (Fig. 1). One cause for this discrepancy might link to the fact that responses to toxicity might not be comparable between the organism and the population levels (Stark et al., 2004), and this is also true for radiotoxicity (Alonzo et al., 2008a, 2016; Lance et al., 2012). This occurs, among other reasons, because demographic endpoints often change as a response to several effects induced at the same exposure level. Exposure effects relationships in the field may be modified due to the combination of radiotoxicity effects on growth rate/reproduction and geographic gene diversity, competition, predation, and abiotic factors including pollutants other than radionuclides. Moreover, species sensitivity is often the result of evolutionary adaptation or increased sensitivity in different environmental extremes, and these processes may be genetic or epigenetic.
Extrapolation approach proposed for modelling interspecies radiosensitivity variation. (a) species sensitivity distribution (SSD) fitted to minimum values of EDR10 (dose rate giving 10% change in observed effect) for terrestrial species exposed under controlled conditions to external gamma irradiation; (b) SSD fitted on EDR10 data set acquired in real field conditions from the Chernobyl Exclusion Zone (taken from Garnier-Laplace et al., 2013).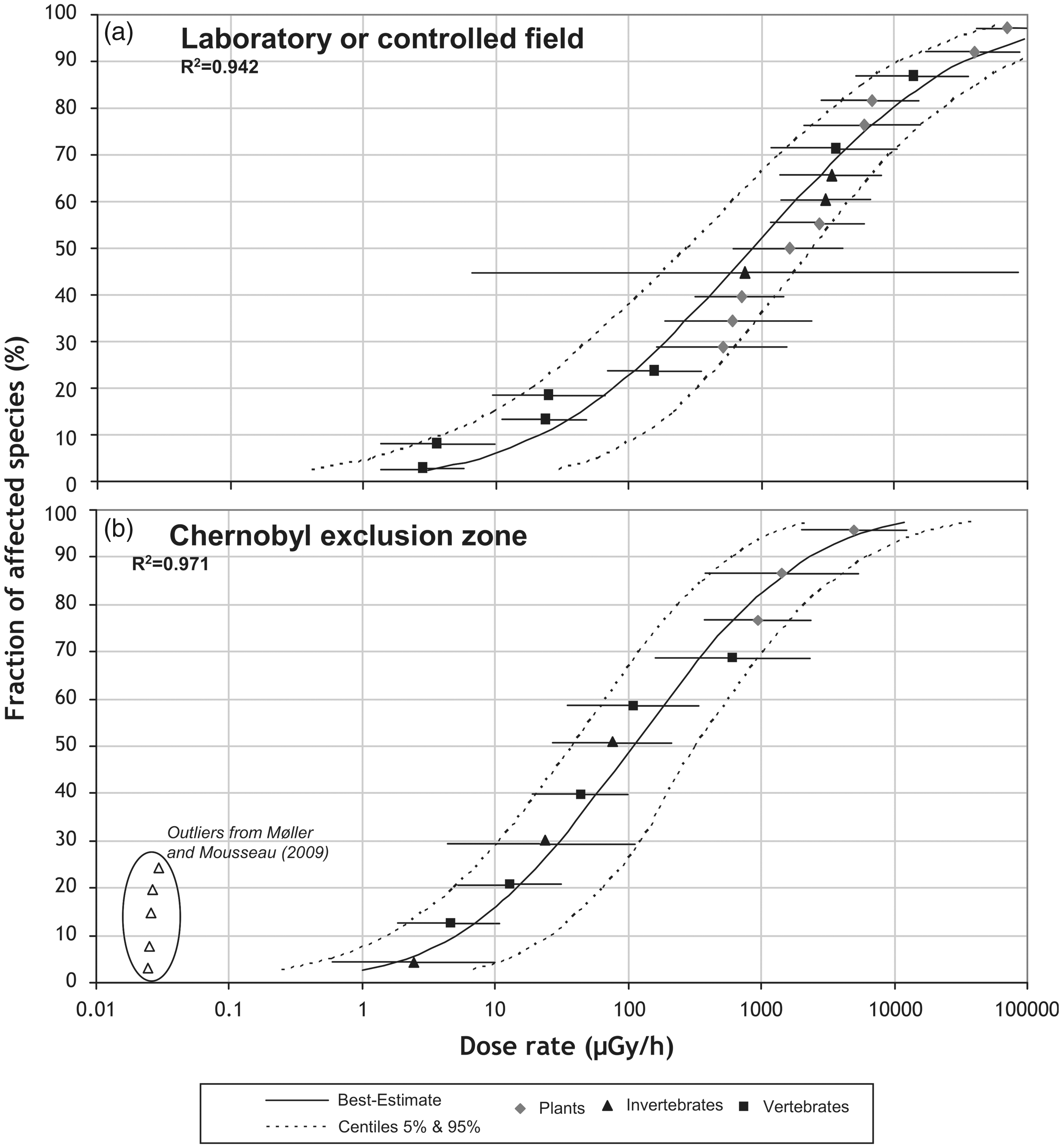
Out of concern that radiation might induce long-term hereditary effects, a number of studies have focused on studying generational sensitivity for continuously exposed populations (multi-generational studies) or even unexposed progeny (transgenerational studies). For example, an increased sensitivity towards gamma radiation, Am, or U was found for the second and third generations of daphnids compared with the parental generation (Pane et al., 2004; Alonzo et al., 2008b; Massarin et al., 2010; Parisot et al., 2015). Studying reproduction in nematodes exposed to gamma radiation either continuously or only the F0 generation showed transgenerational effects from parental exposure in the second generation, indicating that the second non-exposed generation was indeed more affected than the second exposed generation (Buisset-Goussen et al., 2014). Increased sensitivity towards acute doses of gamma radiation was also found in pea plants (
Also, in a transgenerational set up where only the first generation was exposed, differential responses have been shown in the offspring or even in further generations. For example, in a recovery experiment, the vegetative progeny of
4. EPIGENETIC CONTROL OF RADIOSENSITIVITY
Radioresistance of cancer cells is a topic of primary importance in cancer therapy. Recent studies have shown that DNA methylation can regulate gene expression, and modulate cancer cell biology and chemotherapy resistance. For example, cisplatin resistance has been shown to have an epigenetic DNA methylation component in ovarian cancer. Other evidence suggesting that DNA methylation plays a role in radiation resistance has been found in studies showing that treatment with 5-azacitidine, to reduce global methylation, has a radiosensitising effect (discussed in Antwih et al., 2013).
Apart from cancer domain, a number of studies have shown an active role of the DNA methylome in response to radionuclide exposure. For example, a study with mice exposed for 10 days to 50 mGy day−1 showed hypermethylation of promotors of genes involved in DNA repair processes, cellular cycle, and apoptosis (Wang et al., 2014), while in another study, gravid mice of the Agouti line were exposed to low doses of ionising radiation (0.7–7 cGy). In the latter, epigenetic modifications of the Avy locus were detected in offspring in correlation with exposure dose (Bernal et al., 2013), suggesting that epigenetic modifications could be used as biomarkers of radio-induced alterations in the fetus. Global DNA methylation changes have also been observed in several organisms (plants, worms, fish, frogs) exposed to IR, either in the laboratory or in the field (Saenen et al., 2017).
In the non-exposed F3 and F4 progeny of F0 daphnids irradiated at 40 mGy h−1, DNA methylation profile was also significantly different from offspring coming from non-exposed parents. Moreover, a pattern of differentially methylated genes was transmitted among generations; in particular, three could be linked to responses to radiological stress or diseases identified in human cells or rodents: HEAT shock 70 kDa protein, involved in cellular cycle control and inhibition of radiation-induced cell death; 60S ribosomal protein L28, involved in translation and already dysregulated in the developing murine heart at a dose of 0.1 Gy; and mitochondrial-processing peptidase subunit beta, playing an important role in the biogenesis of most mitochondrial proteins (Trijau et al., in press).
5. CONCLUSION
Radiosensitivity varies greatly between species, and is dependent on the exposure scenario as well as the radiation history of the organisms and their parents. It is apparent that further studies are needed to better characterise the factors that govern species radiosensitivity. For that, it is necessary to rely on the most realistic exposure conditions (lower dose rates, high number of generations, use of complex systems, and in-situ studies in observatories). Additionally, the use of high-throughput analyses should enable the identification of fingerprints of ionising radiation effects and early and sensitive biomarkers, which will potentially be used in risk assessment strategies and will lead to better protection of the environment from the impact of ionising radiation.
