Abstract
Radiological protection standards generally assume that all members of the population are equally sensitive to the adverse health effects associated with radiation exposure, recognising the age- and sex-related differences in sensitivity to radiation-induced cancer. It has become very clear over recent years that genetic and lifestyle factors can play important roles in the susceptibility of individuals to a range of diseases; as such, the same may apply to radiation-associated diseases. Evidence is accumulating from studies at many levels of biological organisation – cells, experimental organisms, and humans – that a range of radiosensitivity exists between individuals in the population. Consideration of improvements in radiological protection practices to take account of such differences will require the availability of robust and accurate ways to assess the sensitivity of an individual or population subgroup. In addition, there will need to be careful consideration of the ethical aspects relating to use of individual sensitivity information. These ethical considerations are very likely to be exposure context dependent, and require careful risk–benefit balance consideration before practical application.
1. INTRODUCTION
Radiological protection aims to ‘manage and control exposures to ionising radiation so that deterministic effects are prevented and the risks of stochastic effects are reduced to the extent reasonably achievable’ (ICRP, 2007). The risks of stochastic effects (cancers and hereditary effects) are controlled through the principle of justification, followed by dose limitation and optimisation below constraints and reference levels using the concept of effective dose. While effective dose is a risk-adjusted dosimetric quantity, it does not take account of recognised age- and sex-related differences in rates of radiation-induced cancer. No account is taken of other factors that may lead to variation in sensitivity between individuals. If there is substantial and widespread variation in risk per unit dose between individuals, a more ‘tailored’ system of protection may need to be considered, or a system that serves to protect the most vulnerable rather than all members of the population based on average sensitivity (Hansson, 2009).
Increasingly sophisticated methods of genetic and epigenetic analysis have been a significant driver of the identification of genes, variants, or epigenetic states that impact on an individual’s risk of cancer in general. Methods to analyse chronic inflammation have also identified potential non-genetic risk modifiers. The genetics of breast cancer risk are an example, in which breast cancer susceptibility gene 1 (BRCA1) and 2 (BRCA2) variants enhance risk. In the UK, close to 50,000 women are diagnosed with breast cancer each year, and one in eight women develop breast cancer in their lifetime (12.5% lifetime risk). Carrying the BRCA1 or BRCA2 variant can raise the lifetime risk to 45–65% or one in 2.2–1.5 (see http://www.cancerresearchuk.org/about-cancer/type/breast-cancer/about/risks/definite-breast-cancer-risks). There is also a growing appreciation of the environmental risk factors for differing cancers, and, in a few cases, knowledge of interaction between environmental risk factors, notably the interaction between radon gas exposure and tobacco smoke exposure in lung cancer risk (AGIR, 2009).
People are different in terms of genetic constitution and exposure history, often referred to as ‘genome’ and ‘exposome’. The extent to which genetic and other environmental factors interact with ionising radiation to impact individual cancer risk is not well understood. This paper aims to provide an overview of the evidence available on variation in radiation sensitivity, and to consider some of the potential implications for radiological protection. The Advisory Group on Ionising Radiation 2013 report, ‘Human radiosensitivity’ (AGIR, 2013) covers similar ground but in much greater depth.
2. DEFINITIONS
The term ‘radiation sensitivity’ can relate to deterministic and stochastic endpoints, and some studies may be relevant to both main types of radiation health effect. It is important to be clear and consistent in definitions and usage. The main categories can be summarised as follows.
Radiosensitivity relating to deterministic effects – as measured in mean lethal dose assays for example, and determining normal tissue radiosensitivity following radiotherapy and associated variations in observed tissue reactions. This category of radiosensitivity is likely to be relevant to protection of the lens of the eye among medical radiographers, and in other exposure situations. Radiosensitivity relating to stochastic effects, principally susceptibility to radiation-induced cancer – variation between individuals or population subgroups observed in epidemiological or experimental studies, with established differences between tissues in their radiosensitivity. Cellular radiosensitivity – defined in specific in-vitro or ex-vivo (e.g. lymphocyte) assays that may relate to either deterministic or stochastic endpoints or both.
When considering radiation sensitivity in a radiological protection context, different categories are relevant for different situations. Normal tissue radiosensitivity, if severe, can limit the use of cancer radiotherapy in individuals, and may also be relevant to circulatory disease risk and cataract risk. Occupational and public protection is generally concerned with susceptibility to radiation cancer, except in accident situations where short-term tissue damage may be of concern. Cellular radiosensitivity assays can be useful for mechanistic investigations and may provide convenient predictive tests. However, the relationships between measures of radiation sensitivity at different levels of organisation are far from clear. Ideally when radiation sensitivity is discussed, the organisational level and specific test employed should be made clear.
3. EVIDENCE
There is substantial evidence for variation in radiation sensitivity at many levels of organisation, from the whole organisation down to the individual cell level.
3.1. Normal tissue radiosensitivity following radiotherapy
There are many manifestations of clinical morbidity, some early (occurring within 90 days of radiotherapy exposure) and others late (presenting more than 6 months after radiotherapy), and partly dependent upon the site therapeutically irradiated (West and Barnett, 2011). A typical range of clinical radiosensitivity is shown in Fig. 1, which represents the results of a study of >1000 patients with breast cancer treated with intensity-modulated radiotherapy (Barnett et al., 2012). In reviewing the factors that affect normal tissue radiosensitivity (AGIR, 2013), it was concluded that (i) increasing age in adults, (ii) smoking, (iii) diabetes, and (iv) collagen vascular disease tend to increase clinical radiosensitivity. However, insufficient data are available to conclude if sex, ethnicity, body mass index, or alcohol consumption have any influence. There is also evidence that genetic factors influence clinical radiosensitivity, with heritability estimates generally in the range of 60–80%. Studies that aim to identify specific genetic risk factors have tended to be inconclusive, as reported associations have not been replicated in independent studies (Barnett et al., 2012). One more recent study that included second and third stages of replication in the original report suggested that a region on Chromosome 2, 2q24.1 including the tetratricopeptide repeat, ankyrin repeat, and coiled-coil containing 1 Example of the range of human clinical radiosensitivity in a sample of 1010 patients with breast cancer treated with intensity-modulated radiotherapy. The residual score is a measure of clinical radiosensitivity (toxicity) that standardises several clinical indicators into a single score having accounted for patient- and treatment-related factors (Barnett et al., 2012). A score of 0 indicates the expected level of sensitivity, positive scores indicate higher-than-expected radiosensitivity, and negative scores indicate relative radioresistance. Redrawn from Barnett et al. (2012).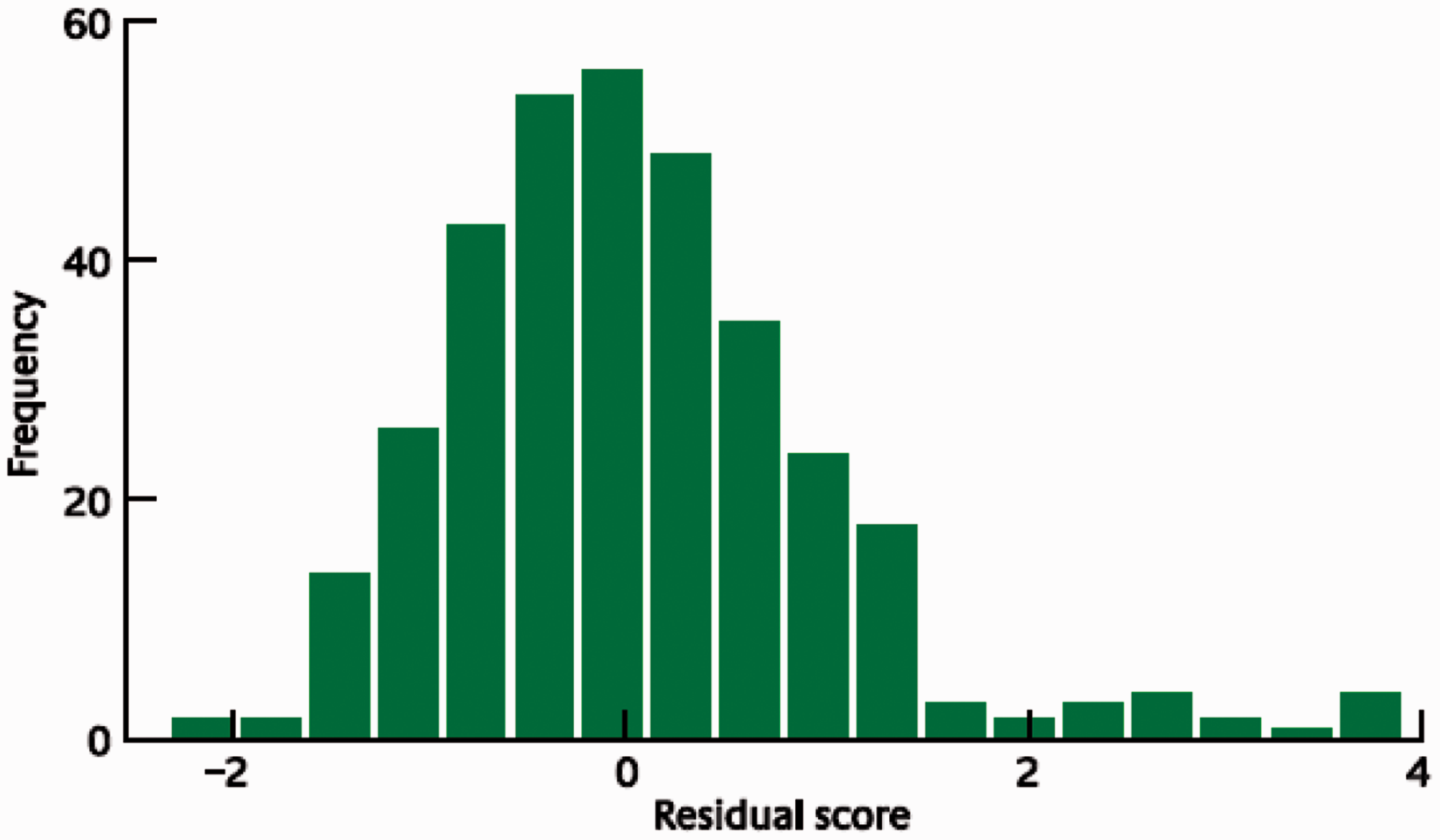
3.2. Radiosensitivity syndromes
A number of rare recessive genetic disorders that have increased cellular radiosensitivity and, in some cases, normal tissue radiosensitivity following radiotherapy have been described. These include the following well-established syndromes: ataxia telangiectasia, Fanconi anaemia, and Nijmegen breakage syndrome. If these conditions are undetected and the individuals receive radiotherapy, very severe effects may occur. Some of these disorders are genetically complex, with multiple genes and mutations leading to a clinically similar phenotype. More recently, further radiosensitivity syndromes have been described, such as Cornelia de Lange syndrome, Warsaw breakage syndrome, and Cernunnos [see AGIR (2013) for further examples and their genetic basis]. These disorders have been important in helping identify mechanisms of radiosensitivity. To date, four major groupings have been identified: (i) those that affect the non-homologous end-joining pathway of DNA double-strand break repair (e.g. some severe combined immunodeficiencies and Cernunnos); (ii) those that affect homologous recombination repair of DNA (e.g. Fanconi anaemia and radiation 51 repair
While these disorders are genetically recessive, at least in some cases, there is evidence for radiosensitivity in heterozygous carriers of variants of these genes. One example is that of modestly increased chromosomal radiosensitivity in ataxia telangiectasia and Nijmegen breakage syndrome carriers (Neubauer et al., 2002).
3.3. Predisposition to radiation-associated cancer
A number of paediatric subpopulations have been found to have increased frequencies of cancers arising in the radiation fields and surrounding ‘penumbra’ of collaterally exposed non-target tissue used for treatment of a primary cancer (Kleinerman, 2009). These include (i) retinoblastoma cases, where soft tissue sarcomas are observed within radiation fields; (ii) neurofibromatosis type 1, where second cancers are associated with primary glioma radiotherapy; (iii) Li-Fraumeni syndrome, where high frequencies of secondary and tertiary cancers are related to radiotherapy; and (iv) Gorlin syndrome, where multiple basal cell skin cancers can occur within radiation fields. There can be similar issues arising due to frequent diagnostic radiography during treatment. It should also be noted that radiation may, in some cases, be one amongst multiple environmental contributory risk factors for cancer, so interactions between these risk factors and genetic risk variants are possible.
The genetics of radiosensitivity for cancer in adults have been best studied in breast cancer, where a number of genes encoding proteins involved in aspects of DNA double-strand break repair and the signalling of repair have been implicated. While the incidence of the clinically obvious radiosensitivity syndromes, which frequently present with many obvious features, is low [e.g. one in 300,000 live births for ataxia telangiectasia in the UK (Woods et al., 1990)], the frequency of heterozygous carriers of risk variants can be substantial (0.5–1% in this case). It has been established that ataxia telangiectasia mutated (ATM) heterozygous carriers are at approximately two-fold higher risk of breast cancer (Renwick et al., 2006; Goldgar et al., 2011). More recently, it has been found that rare mis-sense variants of ATM and radiation exposure from breast cancer therapy together confer a greater risk of contralateral second breast cancer than the sum of the individual effects (Bernstein et al., 2010). Evidence for other breast cancer susceptibility genes, such as BRCA1, BRCA2, and checkpoint kinase 2, have been found but are generally weaker (AGIR, 2013).
At lower levels of exposure in medical diagnostics, it is perhaps unsurprising that evidence of breast cancer genes affecting radiation cancer susceptibility is less clear. Despite some indications of increased breast cancer risk in BRCA1/2 carriers subjected to medical diagnostics (Andrieu et al., 2006), overall evidence is mixed and, at most, potentially indicates that mutation carriers may be at increased risk following diagnostic exposure (Goldfrank et al., 2006; Narod et al., 2006).
3.4. Cellular radiosensitivity
Numerous studies have investigated the range of human cellular radiosensitivity using several different endpoints; for example, cell survival, chromosome aberration induction, induction of apoptosis, and cell cycle checkpoint induction. In some cases, cellular radiosensitivity appears to be associated with increased cancer risk (AGIR, 2013). Studies combining pedigree analysis or twin study designs with cellular radiosensitivity assays suggest that on the order of 70% of the observed variation may be due to genetic factors. One assay deserves particular note: the G2 chromosomal radiosensitivity assay. This ex-vivo lymphocyte test assesses chromatid aberration arising in the G2 phase of the cell cycle following radiation exposure. High G2 aberration scores have been observed in several known cancer predisposition syndromes and a high proportion of women with breast cancer [see AGIR (2013) for further discussion].
3.5. Summary of evidence
There are many lines of evidence for the variation in human radiosensitivity in terms of tissue reactions (e.g. to radiotherapy) and cancer, certainly following radiotherapy exposure. In terms of risk to normal tissues during radiotherapy, other pre-existing conditions or exposures, notably smoking, can affect radiation risk. This is paralleled by the strong interaction between residential and occupational radon exposure and smoking (AGIR, 2009). In addition, there is substantial evidence for genetic variation contributing to radiosensitivity, although identifying all the genetic influences is challenging.
4. IMPLICATIONS
The growing evidence on variation in human radiosensitivity has potential implications for many areas of radiological protection. Those highlighted below are drawn from a more detailed consideration presented by AGIR (2013).
4.1. Medical sector
In radiotherapy, normal tissue damage can limit the ability to control tumour growth, and regimes are developed to avoid tissue injury in the majority of cases. There are sometimes options to modify the treatment plan in cases of adverse normal tissue reaction occurring during therapy. Robust and reliable assays to predict individual sensitivity would be beneficial, and could help inform selection of the most appropriate therapeutic strategy. On the basis of current evidence, patients can be provided with advice on potentially modifiable risk factors, and this could help optimise the benefits of therapy.
4.2. Diagnostics
The increasing use of medical diagnostics involving ionising radiation, especially computed tomography scans, could present problems for highly radiosensitive individuals in the population, although these are few in number. In some cases, alternative methods can be available, such as in the case of magnetic resonance imaging as an alternative to x-ray mammography for breast cancer screening. Again, having assays to accurately determine or predict sensitivity would be beneficial in this respect. There remains, of course, the need to maintain justification of diagnostic use on the basis of clinical benefit outweighing risk.
4.3. Occupational
Epidemiological studies provide good evidence of females and younger people being at greater risk of radiation-associated cancer [evidence not discussed in this paper, but see AGIR (2013)]. As prospective radiological protection aims to protect the population, it is reasonable to use age- and sex-averaging in defining effective dose for control of exposures. However, when retrospective assessment of risk in individuals is being undertaken, these factors should be taken into account.
More specifically, concerns in the occupational sector centre around the ability to identify those at greater (or even lesser) risk of radiation health effects. The evidence that ATM carriers can be at increased radiation cancer risk suggests that genetic testing may become feasible. It is, of course, a highly contentious issue whether genetic testing is used appropriately in employment selection. Indeed, the International Labour Organisation strongly discourages the practice, and it is legislated against in many countries. This would not preclude individuals seeking genetic testing on an individual, private basis to inform their individual employment choices.
4.4. Emergencies
The availability of rapid and reliable tests of radiation exposure is valuable in emergency situations. The availability of tests that are predictive of tissue injury or cancer risk on an individual basis could enhance the robustness of immediate and longer-term clinical decision making in emergencies.
4.5. Public
In provision of public advice on radon risk, inclusion of information on the role of smoking is clearly relevant. In the UK, Public Health England routinely provides smoking cessation advice to the public, alongside radon risk and remediation advice. The ability to determine radiosensitivity by simple tests, obtained privately by individuals, could be used by members of the public when considering medical treatment options or in other situations that include exposure to ionising radiation.
4.6. Ethics
Evolving evidence for variation in human radiosensitivity prompts questions about the approach to radiological protection. Currently, protection standards focus on the population rather than the individual. Furthermore, within the population, it is a notional average individual that is used in the definition of effective dose and the control of radiation exposures. Some may argue that the most vulnerable (i.e. most sensitive) in the population should be protected (Hansson, 2009). However, disproportionate allocation of resources and budgets protecting small high-risk groups may be seen to lack justice for the wider population.
As reliable routine tests for radiosensitivity are not available at present, the main focus should be on provision of information about risk and how it may be reduced. This can be seen to be particularly important where individuals are at liberty to change their behaviour or lifestyle to reduce their own risk.
5. PREDICTION OF RADIOSENSITIVITY
The preceding section noted that the ability to reliably test for and assess individual radiosensitivity could prompt consideration of substantial changes to the current approaches taken in some areas of radiological protection. The radiation biology literature has many reports of potential tests for radiosensitivity, but none of them have entered routine practice.
In the case of normal tissue reaction to radiotherapy, for example, an assay of apoptosis in blood lymphocytes has been reported to be predictive only for particular individual biological endpoints (Ozsahin et al., 2005; Azria et al., 2008). In a diagnostic setting, it has been claimed that phosphorylated histone H2AX foci levels in mammary epithelial cells are associated with breast cancer risk (Colin et al., 2011). As noted, none of these or similar tests has yet reached routine clinical practice.
In addition to cellular assays, genetic testing could play a role, as noted earlier. Also, tests based on gene expression hold promise. For example, post-irradiation expression of the cell cycle regulator cyclin-dependent kinase inhibitor 1A was found to increase much more in a small sample of breast cancer patients with a severe skin reaction to radiotherapy compared with those with a normal reaction (Badie et al., 2008), but further validation is needed. More generally useful would be an assay of radiation cancer risk. The study of Kabacik et al. (2011) suggests that there may be prospects for such a test. The study used mouse strains modified to carry between zero and four copies of p53, a key tumour suppressor gene and transcriptional regulator. In these strains, p53 copy number determines lifetime cancer risk. A strong linear relationship was found between post-irradiation response of selected p53 regulated genes and p53 copy number. It was therefore possible to correlate 2-h post-irradiation cancer risk with lifetime cancer risk with a high degree of statistical confidence in these specific mouse strains.
6. FUTURE PROSPECTS
It is likely that scientific understanding of human radiosensitivity will improve in the future, and the long-held hope to develop robust and reliable tests for radiosensitivity will be achievable.
In parallel with the accumulation of scientific evidence, other considerations are necessary before such knowledge is applied in the context of radiological protection. It needs to be demonstrated that applying knowledge on radiation sensitivity and its variation will be beneficial in some or all aspects of radiological protection.
Any changes would require a robust framework in which to translate and implement the knowledge. Such a framework is likely to be exposure-situation dependent, and will need sound justification in scientific evidence and ‘gain’ in terms of radiation health protection. Finally, acceptance of both the principle and approach will be needed.
Several needs can be identified before individual human radiosensitivity can be applied in radiological protection.
Improved information on the range of human radiosensitivity, particularly in relation to radiogenic or radiation-associated diseases of concern and at exposure levels of relevance. Robust and reliable predictive assays that are accurate, rapid, and reproducible. These could be genetic, cellular, or based on gene expression. Careful consideration of the benefits of moving to a more individualised or stratified approach to protection (e.g. considering what could be gained or lost). Development of frameworks in which to implement a stratified or individualised approach to protection. Widespread dialogue with all stakeholder groups to achieve acceptance of any such change to the approach to protection.
There are therefore challenges to translation of knowledge of human radiosensitivity into practical radiological protection. These are both scientific and ethical/societal, and it will be important that these are considered together if translation into effective radiological protection practice is to be achieved. Any move to a more personalised approach to radiological protection is likely to be more challenging than moves to personalised medicine, and is arguably a more distant prospect.
