Abstract
The International Commission on Radiological Protection (ICRP) mandated a task group (Task Group 64) to review recently published epidemiological studies related to cancer risk and incorporated alpha emitters, and to evaluate whether the results might consolidate or challenge assumptions underlying the current radiation protection system. Three major alpha emitters were considered: radon and its decay products, plutonium, and uranium. Results came mainly from cohorts of workers, while for radon, major studies of the general population contributed to a better understanding of the risk of lung cancer at low and chronic exposure. Selection criteria for the review were: assessment of individual exposure of the target organ, long duration of the health survey, availability of attained age at end of follow-up, and adjustment for major co-factors. Task Group 64 is composed of members from ICRP Committees 1 and 2 (because epidemiological and dosimetric expertise were needed) and external experts. A first report (ICRP Publication 115) considered the risk of lung cancer related to inhalation of radon and its decay products. As the estimated excess risk per unit of exposure was higher by a factor of 2 compared with a previous ICRP estimate in 1993, Task Group 64 suggested a reconsideration of the reference levels for the workplace and for the general population. A second report, using the same standardised methodology (lung cancer baselines, population, life expectancy), will include estimation of the cancer risk of nuclear workers exposed to plutonium, focusing on the risk of lung cancer. A comparison of these risks with those of populations exposed to external gamma radiation alone will be made in the near future. For uranium, the results related to the organ-specific dose were too sparse to draw reliable conclusions, despite a recent publication. More research is needed on this topic.
1. PRESENT WORK OF TASK GROUP 64
1.1. Introduction
Committee 1 of the International Commission on Radiological Protection (ICRP) deals with health effects after exposure to ionising radiation, and their relevance for radiation protection. Major efforts include consideration of both biological and epidemiological studies, the aim being a synthesis of recently published results and explaining mechanisms of induction of tissue/organ damage. Task Group 64 was launched in order to study and compare results from populations internally exposed to different alpha emitters. The health effects to be considered are specific cancer sites (mortality or morbidity) observed in large populations with different levels of exposure. Effects other than cancer are also discussed if enough data are available. Precise individual exposure data on an annual basis, long duration of follow-up, and validation of the dosimetric models used for individual dose assessment were some of the criteria necessary for inclusion in the analysis. Three major alpha emitters have been considered to date: radon and its progeny, uranium, and plutonium.
Each report will include a detailed executive summary, a glossary, and a description of the studies considered before formulating conclusions and possible recommendations. Some of the selected radionuclides and their related health risk have also been considered by other international expert groups, such as the World Health Organization (International Agency for Research on Cancer) and the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR). In each case, a summary of the conclusions of these committees is presented. However, the efforts of Task Group 64 are mainly based on more recent studies, not yet published when the above-mentioned international reviews were conducted, as they may have some influence on the general conclusions.
1.2. Composition of Task Group 64
Task Group 64 is a collaborative group including experts from ICRP Committees 1 and 2 as well as external experts. The current composition of Task Group 64 is as follows: Margot Tirmarche, France (Chair); Iulian Apostoaei, USA; Eric Blanchardon, France (Committee 2); Betsy Ellis, USA; Dominique Laurier, France (Main Commission); James Marsh, UK (Committee 2); Mikhail Sokolnikov, Russia (Committee 1); and Richard Wakeford, UK (Committee 1).
Previous members were: François Paquet, France (Committee 2); Dan Stram, USA (Committee 1); Ethel Gilbert, USA; and John Harrison, UK (Committee 2).
Several corresponding members contributed to the work of Task Group 64: Sergey Zhivin, France; Bernd Grosche, Germany; Jay Lubin, USA; Colin Muirhead, UK; and Irina Guseva-Canu, Switzerland.
1.3. Radon and its progeny
The conclusion of Task Group 64 on the risk of lung cancer and radon was published in 2010 (ICRP, 2010). After having described the cancer risk observed, mainly lung cancer, in a large number of independent studies and the corresponding risk coefficients calculated from studies of underground hard-rock miners, and also from numerous studies of the general population exposed in dwellings, the results were compared with those published in a previous ICRP publication based on results observed before 1993 (ICRP, 1993).
Revision of life-long risk calculation performed in the past was a major task; as the estimate was to be compared with that obtained in ICRP (1993), the same methodology had to be applied, taking into account time since exposure, age at exposure, and the updated background rates of lung cancer mortality given in ICRP (2007). The scenario of exposure (for a typical occupational exposure) was also the same as in Publication 65 (ICRP, 1993): 2 working level months (WLM) year−1 from 18 to 64 years of age.
The associated life-long excess absolute risk (LEAR) of lung cancer was close to 5 per 10,000 per WLM, a value fairly comparable to that obtained with the BEIR VI model (Time Since Exposure-age-concentration) of 1999 or the model developed by Tomášek et al. (2008) based on the joint Czech and French uranium miner studies. As the LEAR estimate published by ICRP (1993) was 2.8 per 10,000 per WLM, Task Group 64 recommended dividing the radon reference levels proposed in 1993 by a factor of 2.
Calculations depending on the choice of the individual studies, and on the reference population, using, for example, the rates of a male and female Euro-American population instead of the reference rates of a composite Euro-American-Asian population of Publication 103 (ICRP, 2007), showed that the estimated LEAR of lung cancer can vary between 3 and 7 per 10,000 per WLM.
These recommendations were discussed in detail with members of Committee 2 in charge of calculating the values of effective dose related to inhalation of radon progeny derived when using the Human Respiratory Tract Model; the calculated estimate of Publication 115 (ICRP, 2010) was between 10 and 20 mSv per WLM depending on the exposure conditions. More detailed information on the dose conversion factor for radon was published by ICRP in December 2017 in Publication 137 (ICRP, 2017).
During recent years, recommendations from Committee 4 and from other organisations have provided a framework of risk management linked to radon progeny in both dwellings and workplaces. One of the complications when using effective dose linked to radon is that this approach has to be understood by decision makers who are not working in the field of nuclear industry. Decision makers have to learn that specific working conditions can change the value of calculated effective dose despite the same measured radon concentration in the environment.
The message should be relatively clear and simple for most working conditions, but several measurements of radon concentration taken over a relatively short period should reflect, at least, the average annual value. Indeed, epidemiological studies have demonstrated an excess risk of lung cancer for a measured radon concentration in the air if inhaled over more than 20 years. The cumulated exposure at organ level is the best indicator of the risk of lung cancer.
As radon progeny represent the major component of natural background radiation exposure of the general public, the corresponding value of effective dose will demonstrate the relatively large contribution of radon progeny to the total effective dose of a national or regional population. This requires further explanation, as this value is often larger than the so-called ‘population limit’ of 1 mSv linked to industrial releases.
Communication is presently a major issue, particularly as Europe is currently in the process of applying the new basic safety standards. Members of Task Group 64 continue to participate in various meetings, often in close collaboration with dosimetrists and regulators (Marsh et al., 2017), to make clear that even if smoking is a major risk factor for lung cancer in the general population, the contribution of radon progeny in cancer development has to be explained and managed carefully.
In fact, interaction between radon and tobacco smoke has been studied in the past in animals (rodents and dogs) as well as in epidemiological studies (Tirmarche et al., 2010). Several European and North American case–control studies have shown, both in the general population and in miners, that adjustment for smoking habits is necessary to obtain a clear view of the role of both lung carcinogens. Note that a clear dose–response relationship related to radon exposure has been evidenced for non-smokers in the joint European study (Darby et al., 2006), and an interaction of a submultiplicative or more than additive effect has been observed between radon and cigarette smoking, mainly in the uranium miner studies (Leuraud et al., 2007, 2011). For uranium and plutonium, the same discussion of the role of tobacco is relevant because both of these radionuclides are also inhaled in most cases.
More studies should be performed to clarify the risk at low dose rates, and how the cumulative doses to the target organ may finally contribute to the development of lung cancer.
Risks other than lung cancer due to radon inhalation have been suggested by some studies, but more information is needed before drawing a final conclusion. This holds, for example, for the potential link between radon and leukaemia because an association was observed in some miner studies. The contribution of other radiological components, such as external gamma radiation exposure, must be considered carefully. This is one of the reasons why the follow-up of cancer risks from uranium miner studies has to be continued at international level.
1.4. Plutonium exposure
The review of studies of workers exposed to plutonium has been a major task of Task Group 64 over the last 4 years. It is mainly based on studies of the workers at the Mayak Production Association (in the Southern Urals of Russia), who had experienced relatively high exposures in the past, particularly during the period 1948–1958. In addition, the Task Group 64 review will consider other studies published over the last 15 years if dose values for specific organs are available; this is the case for plutonium workers at the Sellafield plant of British Nuclear Fuels, and for several US nuclear worker groups.
Organ dose calculations in these studies depend on the quality of past bioassays, as well as on working conditions. Aerosols of plutonium have different absorption characteristics and chemical properties, with the major distinction being between plutonium nitrates and plutonium oxides. The calculations also depend on the dosimetric models used and on the time between exposure and measurement. In the TG64 report, experts in dosimetry will summarise all aspects relevant for calculation of organ doses linked to plutonium exposure, including the historical changes in the Mayak dosimetry, in order to consider the most important modifying factors as well as dose uncertainty.
A recent international effort compared the health effects observed in cohorts of Sellafield (UK) and Mayak workers. A paper presenting the findings on lung cancer in the joint Mayak–Sellafield workers cohort was published very recently by Gillies et al. (2017). A European case–control study (from the European Commission Framework Programme 6) by Grellier et al. (2017) included deaths from lung cancer among nuclear workers from the UK, France, and Belgium exposed to either uranium or plutonium. This study allowed comparison of the results with those observed in cohort studies.
Grellier et al. (2017) found that the excess odds ratio per Gy was 49 (90% confidence interval 16–106); it was reduced to 37 (90% confidence interval 0.18–121) when workers from the UK Atomic Weapons Establishment (AWE) were excluded from the analysis. The average lung dose from plutonium was low (median dose 1.27 mGy) when compared with the average cumulative lung dose from external gamma radiation (median dose 33.9 mGy).
Mayak plutonium and lung cancer dose–response analyses published since 2000.
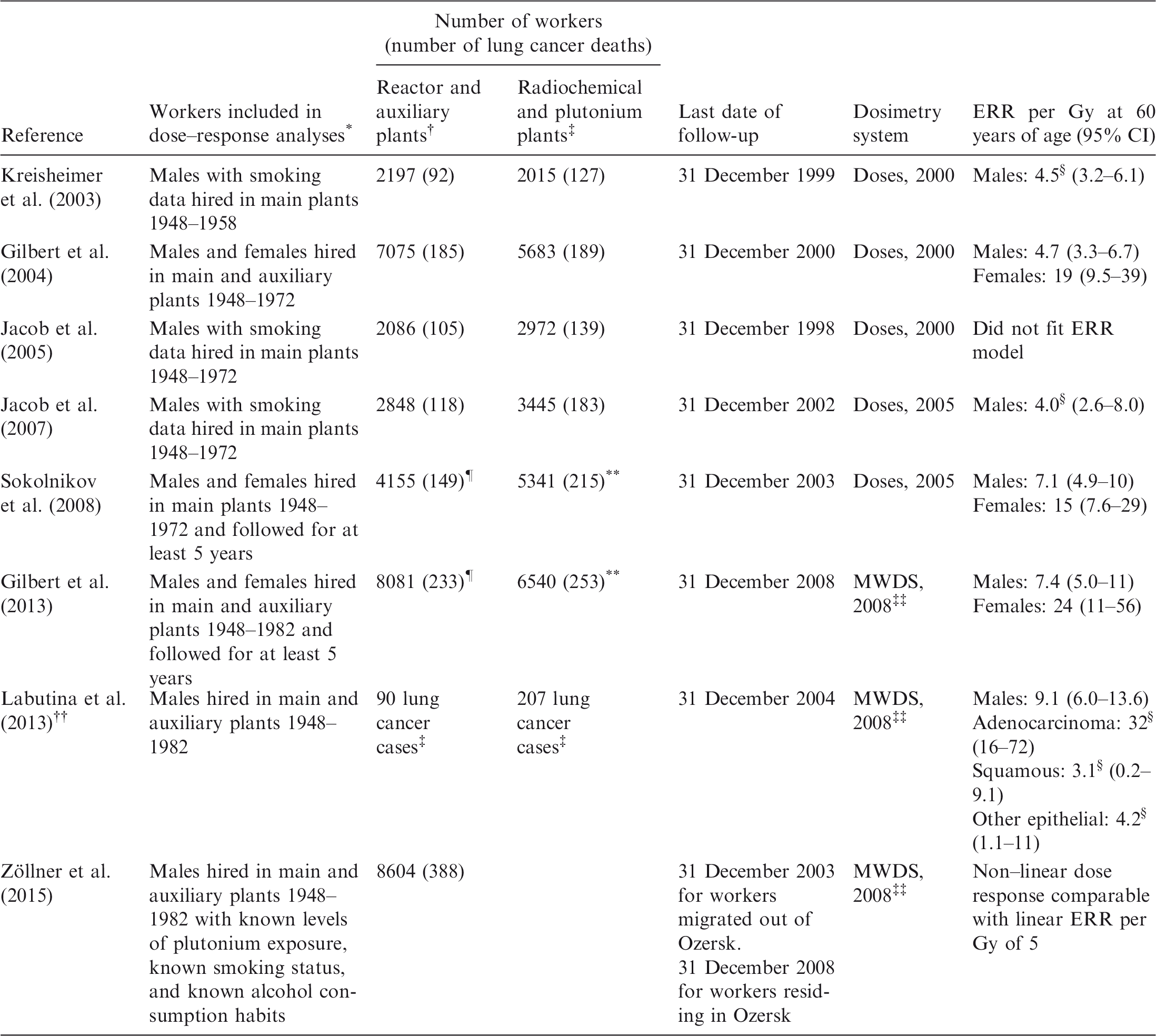
ERR, excess relative risk; CI, confidence interval; MWDS, Mayak Workers Dosimetry System 2008.
Workers were either monitored for plutonium, or worked solely in the auxiliary or reactor plants with little potential for plutonium exposure. With the exception of Kreisheimer et al. (2003), workers with potential for plutonium exposure had to have been monitored for at least 2 years before the end of follow-up.
Little potential for plutonium exposure.
Potential exposure to plutonium.
ERR per Gy is for all ages.
Number with plutonium doses of 0, including a few workers in the radiochemical and plutonium plants.
Number with positive plutonium doses, including a few workers in the reactor and auxiliary plants.
Based on cancer incidence data for workers who were diagnosed with lung cancer in the city of Ozersk.
The number of workers contributing to these analyses is not given in the paper.
1.5. Uranium
Task Group 64 also reviewed the cancer risk linked to uranium. This review is limited to occupational exposure to uranium resulting from the processing of uranium ore through milling and refining, chemical conversion, enrichment, fuel fabrication, and reprocessing.
In 2012, the International Agency for Research on Cancer concluded that there was sufficient evidence for uranium carcinogenicity in experimental animals, and that evidence was limited in humans exposed to mixtures of natural, enriched, and depleted uranium isotopes (IARC, 2012). Recently, UNSCEAR (2017) published an extensive report focusing on the biological effects of uranium in studies of animals, nuclear workers, and the general population.
Uranium exhibits both chemical and radiological effects. The chemical effects are assumed not to occur below a certain concentration. Uranium compounds vary greatly in solubility, which can lead to differences in the bioavailability of the compound after inhalation or ingestion.
Due to variations in the type and size of airborne uranium particles found throughout the nuclear fuel cycle, the solubility and resulting biokinetic distribution of uranium in the human body differ significantly. Soluble uranium compounds are incorporated through the lungs, and transported by the blood to the kidney for excretion. Insoluble uranium, in contrast, is retained in the lungs, deposited in tracheobronchial or other thoracic lymph nodes, or swallowed after mucociliary transport. Consequently, health hazards are likely to vary across the nuclear fuel cycle because of the different forms of uranium present in each stage.
Among the 14 cohort and two case–control studies of uranium workers selected, several activities linked to the uranium nuclear cycle were covered: uranium milling and refining, uranium enrichment via gaseous diffusion, chemical conversion and fuel fabrication, and research and development of nuclear reactors and uranium and plutonium fuel fabrication. One study was able to cover all steps of the nuclear fuel cycle (Samson et al., 2016). The solubility of the uranium used in these different activities varied from predominantly soluble uranium in uranium enrichment to insoluble uranium in uranium processing.
Unfortunately, very few studies provided information on uranium-specific health risks, due to the fact that uranium-specific exposure estimates were missing or very sparse because of incomplete historical recording of individual information. Therefore, in the Task Group 64 report, it is proposed to comment only on those studies that reported uranium-specific risks for the three most plausible cancer outcomes after chronic uranium exposure: lung cancer (organ of entry following inhalation), kidney cancer (organ of accumulation and elimination), and leukaemia and other lympho-haematopoietic malignancies (outcome of general interest after radiation exposure).
The recent paper of Grellier et al. (2017) provides results from a case–control study of lung cancer mortality among uranium workers from the UK, France, and Belgium. The excess odds ratio per Gy for uranium was found to be 4.2 (90% confidence interval −2.5–17), after adjustment for smoking and socio-economic status, and with a lag time of 10 years. The average lung dose from uranium was low (median dose 2.2 mGy), while the average cumulative external gamma dose to the lung was higher (median dose 33.9 mGy).
The preliminary conclusions of Task Group 64 are as follows.
The majority of selected studies have shown no increase in the risk of lung cancer with exposure to uranium. A French study (Guseva Canu et al., 2011) revealed a significant increase in the risk of lung cancer after exposure to reprocessed uranium but not to natural uranium. Cohorts of uranium enrichment workers in France (Zhivin et al., 2016) and the USA (Yiin et al., 2017) exposed mainly to rapidly soluble uranium compounds are in line with this finding.
The studies of Grellier et al. (2017) and Silver et al. (2013) indicate a positive dose–response relationship, but both with a large confidence interval that cannot exclude the absence of any trend. In the study of Grellier et al. (2017), when testing for the influence of specific worker groups, the UK AWE and British Nuclear Fuels Limited workforces influence the risk coefficient in an opposite direction.
In order to increase the statistical power and take into account the uncertainty linked to estimated individual doses, a large international effort with a common protocol for collection of data, focusing solely on those uranium oxide components that will contribute to the lung dose, may help to produce a better estimate of the risk of lung cancer linked to uranium.
2. ONGOING AND FUTURE WORK OF TASK GROUP 64
2.1. How far are we able to compare the risk of lung cancer from different alpha emitters with other exposures?
Several members of Task Group 64 are involved in life-long calculations of the risk of lung cancer in order to apply the same methodology to plutonium-exposed workers as applied in the past to radon-exposed workers: a scenario of chronic exposure, considering the same ages of exposure (18–65 years), a life expectancy up to 90 years, and the same background data of lung cancer rates. The aims of this work are: (1) to compare the calculated LEAR from each alpha emitter; and (2) to compare these values with those obtained from a scenario considering external gamma exposure alone, and using the risk coefficient of lung cancer per Gy from the atomic bomb survivors, from the Mayak workers exposed to external gamma exposure alone, and from other nuclear workers (e.g. INWORKS study) (Richardson et al., 2018). This work is being undertaken in close collaboration with other task groups of ICRP, mainly Task Group 102 on Detriment Calculation Methodology.
Discussion of the influence of dose uncertainty on these values, as well as the possible interaction with tobacco which may have influenced some of the dosimetric models, will be important. Task Group 64 welcomes ongoing research on these topics.
