Abstract
Solitary fibrous tumor is relatively rare, and solitary fibrous tumor of the parotid gland is even less common. The clinical features of solitary fibrous tumor are non-specific, making early diagnosis difficult. The diagnosis mainly depends on pathological histology and immunohistochemistry. The solitary fibrous tumor is considered as a borderline tumor. Most tumors are benign, while malignant transformation is less common. Currently, surgical treatment is the main approach, and radiotherapy may be used for malignant patients after surgery. This study presents 2 cases of solitary fibrous tumor arising in the parotid gland. Case 1 is a 49-year-old woman who presented with right cervical mass. Imaging studies demonstrated a well-defined mass in the deep lobe of the parotid gland extending into the right parapharyngeal space. The patient underwent right total parotidectomy and had manifestations of facial paralysis after surgery, but recovered about 5 months later. Case 2 is a 16-year-old female who presented with left cervical mass. Imaging studies showed a round mass in the superficial lobe of the parotid gland with significant enhancement. The patient underwent left superficial parotidectomy and recovered well without any manifest facial nerve palsy. The accurate diagnosis of benign solitary fibrous tumors was obtained by the immunohistochemistry. These 2 patients were regularly followed up after the operation, and no obvious recurrence changes were observed.
Introduction
Solitary fibrous tumor (SFT) is a rare spindle cell mesenchymal tumor that was first reported by Klemperer and Rabin in the visceral pleura in 1931. 1 In recent years, SFT has been reported in many extrapleural sites. SFT of the head and neck accounts for approximately 6% of all SFTs, and the most frequently affected areas in the head and neck are the nasal cavity (30%) and orbit (25%), followed by the oral cavity (15%) and salivary glands (14%). SFTs in salivary glands are rare, and most reported cases involve the parotid gland, followed by the submandibular gland and the sublingual gland.2,3 This article reports the clinical data of 2 cases of parotid SFT encountered by the author, and discusses its clinical characteristics, diagnosis, treatment plans and prognosis in combination with relevant literature.
Clinical Data
Case 1
A 49-year-old woman was admitted to hospital due to a right cervical mass that had been detected for over 1 year and had significantly increased in size for about 3 months. Medical history was non-contributory. Physical examination revealed a smooth, hard mass in the right parotid gland without evidence of facial nerve paralysis. B-ultrasound of parotid gland showed a low-echo mass about 43*26 mm in size, with clear boundaries and irregular shape. Enhanced computed tomography (CT) demonstrated a well-circumscribed mass, 60*36 mm in size, in the right parotid gland, extending into the right parapharyngeal space with non-uniform progressive enhancement (Figure 1A). Fine-needle aspiration cytology was performed, but the results were unspecific.
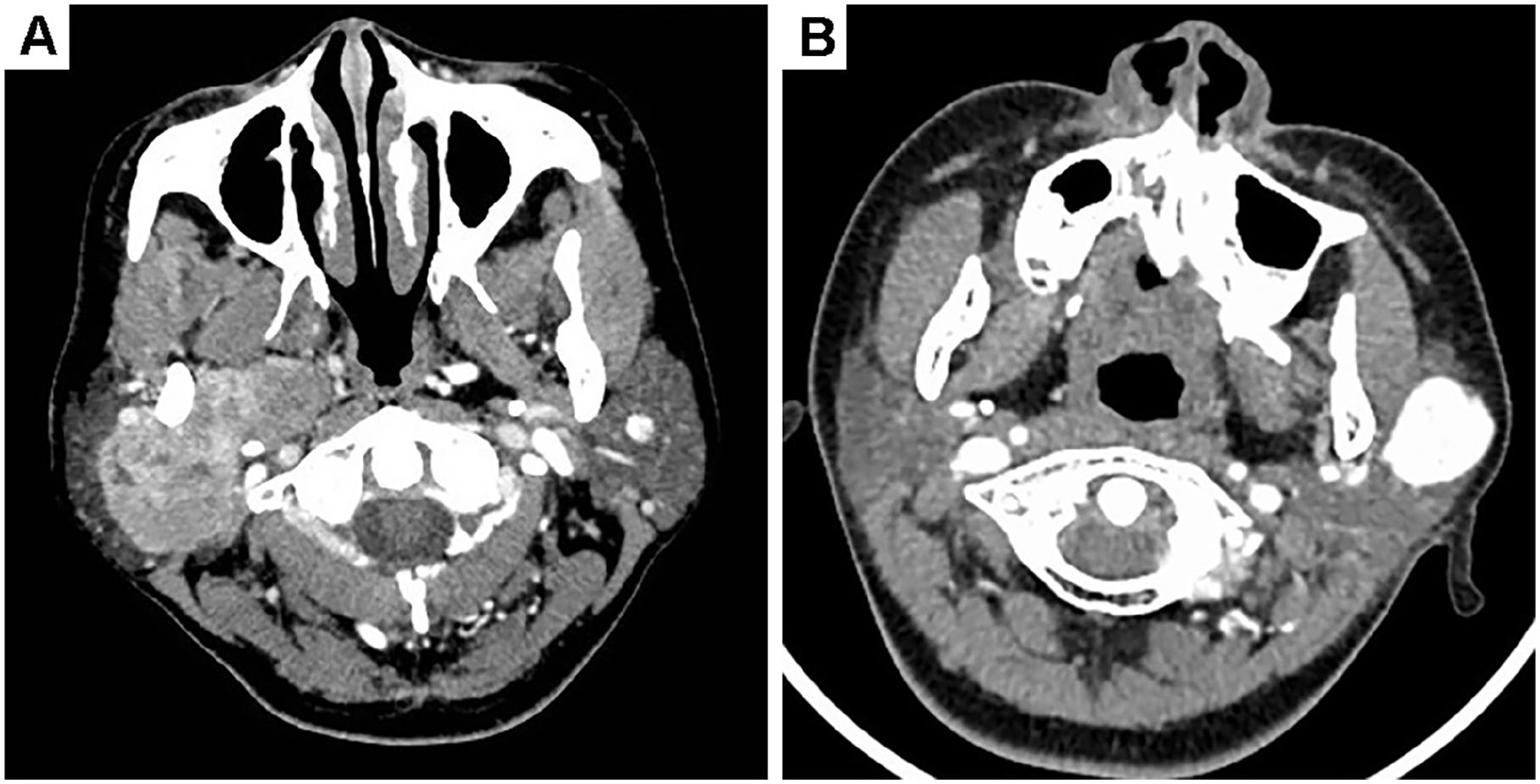
The cervical CT scan before operation. (A) CT scan of Case 1. (B) CT scan of Case 2.
At surgery, the mass was found in the deep lobe of the parotid gland extending into the right parapharyngeal space, with no obvious abnormalities in the superficial lobe. The tumor had a complete capsule and slight adhesion to the surrounding tissues, and a local excision was performed including total parotidectomy (Figure 2). The intraoperative frozen section showed a spindle cell mesenchymal tissue-derived tumor. The patient had manifestations of facial paralysis after surgery, and recovered about 5 months later.
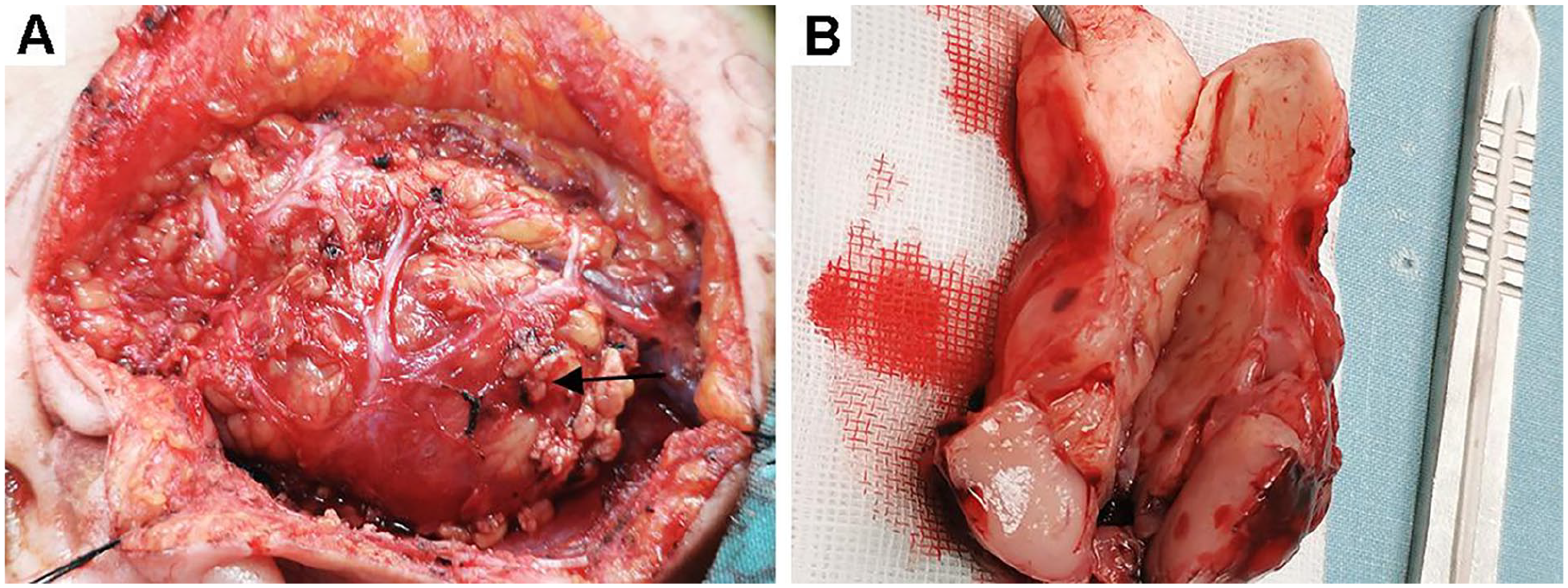
The solitary fibrous tumor of deep lobe of the parotid gland. (A) The deep lobe tumor of the parotid gland. (B) Macroscopic appearance of the tumor.
The final pathology of the mass showed a spindle cell mesenchymal tissue tumor with nuclear division visible, about 3 to 4 mitoses/10 HPF (high-power fields). The immunohistochemistry revealed strongly positive for CD34, CD99, Vimentin, Bcl-2 and STAT6; weakly positive for SMA, Ki-67 and P53; negative for CK, EMA, GFAP, S100, SOX-10, Desmin, P63, calponin and CD117. According to these, the patient was confirmed the diagnosis of SFT of the right parotid gland. The patient was regularly followed up after the operation, and no obvious recurrence changes were observed.
Case 2
A 16-year-old female patient was admitted to hospital due to a left cervical mass for over 1 year. On physical examination, there was a well-defined, painless mass in the left parotid gland. Preoperative ultrasonography of the parotid gland showed a solid hypo-echoic mass measuring about 30*25*20 mm with an irregular shape. Enhanced CT of the neck revealed a 22*28 mm round mass in the superficial lobe of the left parotid gland, with significant enhancement (Figure 1B).
During the operation, we found the mass was located in the superficial lobe of the parotid gland, and the facial nerves were well protected. The patient underwent left superficial parotidectomy. The intraoperative frozen section showed a spindle cell myoepithelioma. The patient recovered well without any manifest facial nerve palsy.
The microscopic examination showed short spindle and ovoid cells arranged in a collagenous matrix with nuclear division visible, about 3 to 5 mitoses/10 HPF. The immunohistochemistry revealed strongly positive for CD34, CD99, Vimentin and STAT6; weakly positive for SMA and Ki-67; negative for CK, EMA, Desmin, S100, MyoD1 and P63 (Figure 3). On the basis of our findings, the tumor was diagnosed as SFT of the left parotid gland. The patient was regularly re-examined after the operation, and no obvious recurrence changes were observed.
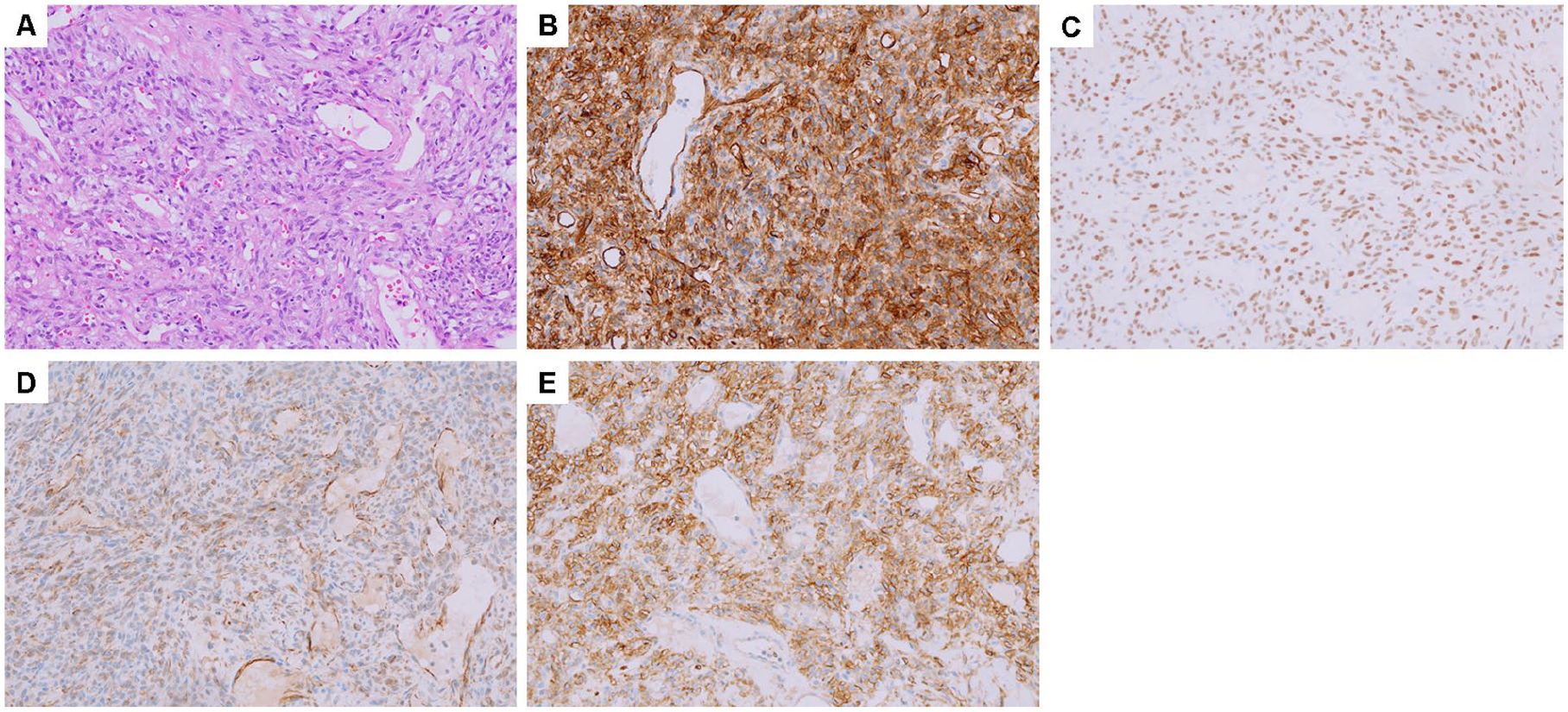
The pathological results of solitary fibrous tumor for parotid gland. (A) Hematoxylin and eosin stain. (B) Immunohistochemistry of CD34 showed positive staining. (C) Immunohistochemistry of STAT6 showed positive staining. (D) Immunohistochemistry of Vimentin showed positive staining. (E) Immunohistochemistry of CD99 showed positive staining (original magnification A, B, C, D, E ×20).
Case 1 was approved by the Research Ethics Committee of Central South University, Changsha, China. Case 2 was approved by the Research Ethics Committee of Shenzhen University, Shenzhen, China. We have obtained Written Informed Consent from the patient (Case 1) and from the patient’s legally authorized representative (Case 2) for the publication of both case reports and any accompanying images.
Discussion
SFT is a mesenchymal spindle cell tumor derived from fibroblasts. It usually occurs in the pleura but may also affect extrapleural sites, with approximately 6% of cases involving the head and neck region. 4 SFT of the parotid gland is a rare fibrous tumor, generally considered to originate from the mesenchymal cells of the parenchyma. Currently, only one reported case has documented a tumor arising from the parotid duct. 5 Since Hanau and Miettinen first reported the localization of the parotid gland in 1995, there have been approximately 43 cases of parotid SFT reported in the literature.6,7 This article retrospectively analyzed the English literature on parotid SFT published from 1995 to 2024 through PUBMED/MEDLINE, EMBASE and CINAHL databases. After excluding articles with limited information or duplicate publications, a total of 32 cases with relatively complete clinical data are summarized in Table 1.2,3,5-34
The Clinical and Pathological Characters, Diagnosis, Treatment and Outcome of the Solitary Fibrous Tumor for Parotid Gland.
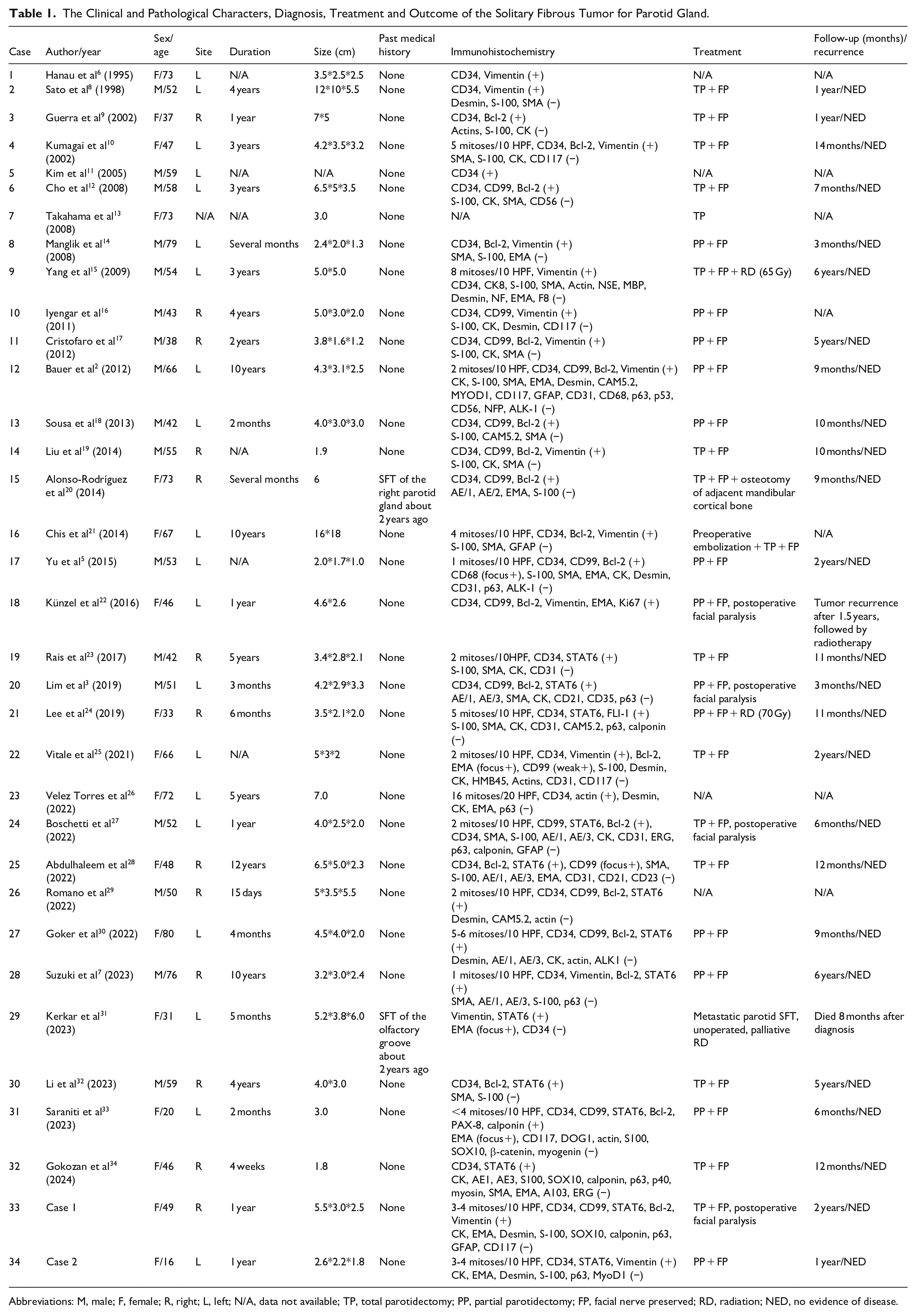
Abbreviations: M, male; F, female; R, right; L, left; N/A, data not available; TP, total parotidectomy; PP, partial parotidectomy; FP, facial nerve preserved; RD, radiation; NED, no evidence of disease.
Parotid SFT typically shows no age or gender predilection. The average age of patients reported in the literature is 53.1 years (20-80 years).2,3,5-34 Case 2 is the youngest patient reported in the literature so far. Clinically, the symptoms of SFT depend on the location of the tumor and the affected organs. Patients with SFT in the superficial lobe of the parotid gland usually have no obvious clinical symptoms and generally present as a palpable, slowly growing, painless mass. If the parotid SFT extends into the parapharyngeal space, it may also be accompanied by sleep apnea. 8 Only 1 case of malignant parotid SFT had symptoms of facial paralysis before surgery. 26 In rare cases, the large SFT may cause hypoglycemia, arthritis and osteoarthropathy due to the secretion of insulin-like growth factor. 35 The 2 SFT patients reported in this article have no obvious clinical manifestations and no facial paralysis before surgery.
SFT usually presents as an isolated, well-defined and homogeneous mass in CT scan, and shows significant heterogeneous enhancement. It is usually manifested in the MRI examination as low signal intensity on T1 and T2 weighted images, and a mild high signal area on T2 weighted images, showing significant gadolinium enhancement. The combination of these features leads to a “chocolate chip” appearance, which is helpful for differential diagnosis. In addition, if there are multiple SFT lesions and high levels of fluorodeoxyglucose metabolism in PET-CT scans, SFT malignancy should be considered. 22 The 2 patients reported in this article underwent CT scans before surgery, and Case 1 showed progressive enhancement, while Case 2 showed a significantly enhanced lesion. Unfortunately, due to economic and other reasons, both patients in this article did not undergo MRI scans.
The clinical diagnosis of SFT is difficult, and diagnosis mainly relies on pathological histology and immunohistochemistry. Generally, SFT is composed of uniformly arranged, bipolarly differentiated spindle-shaped cells, located in highly collagenized stroma. 6 CD34, Bcl-2, Vimentin and CD99 are highly expressed in SFT and are relatively specific indicators for diagnosis. However, CK, EMA, CAM5.2, SMA, Desmin, GFAP, CD117 and S-100 protein are generally negative. 5 CD34 is an important and sensitive marker for diagnosing SFT, but it is not positive in all cases. Generally, it indicates a malignant SFT when there are more than 6 mitoses in every 10 high-power fields (>6/10 HPF) under the microscope. It is worth noting that malignant SFT often has a lower CD34 immunoreactivity. 18 Recent studies have found that there is a phenomenon of NAB2 and STAT6 gene fusion in the 12q13 region of SFT, and this gene fusion induces STAT6 nuclear expression.36,37 The nuclear expression of STAT6 can be detected in more than 95% of SFTs, while it is usually expressed at a low level in other mesenchymal tumors. 38 Currently, immunohistochemistry of STAT6 has been used as an alternative diagnostic marker for SFT, with high sensitivity and specificity, even in malignant cases. 3 In this article, the immunohistochemical indicators of Case 1 were all positive for CD34, Vimentin, Bcl-2, CD99 and STAT6, while Case 2 was positive for STAT6, CD34, CD99 and Vimentin. Both of these 2 cases met the diagnostic criteria for SFT.
Among the cases of primary parotid SFT, only 2 cases of malignant SFT have been reported.15,26 At present, surgical resection remains the main treatment method for most primary SFTs in the parotid gland. Up to now, only 2 cases of local recurrence after surgery have been reported in the literature.20,22 Since SFT is usually a tumor with abundant blood supply, preoperative embolization can be considered to reduce the risk of bleeding for larger tumors.18,21 Some scholars have reported good effects of postoperative chemotherapy and radiotherapy for cases with malignant features and/or positive resection margins. 18 However, further evidence is needed to confirm the role of adjuvant therapy.15,24,31 Since the histological characteristics of SFT cannot reliably predict the clinical behavior of the tumor. Demicco et al 39 proposed a risk stratification model using clinical and pathological features to predict the risk of recurrence and metastasis of SFT, including age (≥55 years old), tumor size exceeding 5 cm, presence of necrosis and increased mitotic activity (≥4 mitoses/10 HPF), and divided the tumors into low-, medium- and high-risk groups. The 2 patients in this article were classified as low-risk groups according to the risk stratification model, and no recurrences were observed during the follow-up after surgery.
Conclusion
The symptoms of parotid SFT are mostly painless masses in the parotid gland region. CT and MRI can provide limited diagnostic information. Immunohistochemistry is still necessary for the diagnosis of SFT. The preferred treatment is to perform surgery with negative surgical margins.
Footnotes
Author Note
Dr Weiyu Zhu takes responsibility for the integrity of the content of the paper.
Ethical Considerations
Ethics approval was obtained from the ethics committee of The Second Xiangya Hospital, Central South University, and South China Hospital, Shenzhen University. The procedures performed in this study were in accordance with the World Medical Association Declaration of Helsinki (version 2002) and the additional requirements. The informed consents of the patients were signed.
Author Contributions
Material preparation, data collection and analysis were performed by Shuang Wang, Biao Xing, Zhenzhang Lu, Danhui Yin, Xinming Yang and Weiyu Zhu. The first draft of the manuscript was written by Shuang Wang and Weiyu Zhu, and all the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Shenzhen Science and Technology Program (grant number: JCYJ20230807141100001) and Natural Science Foundation of Hunan Province (grant number: 2022JJ40712).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author at
