Abstract
Objective
To evaluate the clinicopathologic characteristics of head and neck solitary fibrous tumors and features that may predict tumor recurrence.
Study Design
Retrospective review.
Setting
University of California–Los Angeles Medical Center.
Methods
A single-center retrospective study was conducted on pathologically confirmed cases of head and neck solitary fibrous tumors between 1996 and 2021. Patient demographics, clinical course, and histopathologic features were evaluated. Recurrence-free survival was estimated via Kaplan-Meier analysis.
Results
A total of 52 patients were reviewed. The average patient age was 54.7 years (range, 15-89). The most common subsite was the orbit (53.8%, n = 28), but other involved areas included the nasopharynx, paranasal sinuses, and scalp. The median tumor size was 2.95 cm (range, 1.3-11.2). Strong STAT6 (100%) and CD34 (97.9%) expression was observed on immunohistochemistry. Almost all patients were initially managed with wide local excision; 82% of patients (n = 14) had positive margins on pathologic review; and 15% (n = 4) had recurrence at a median 28.5 months (range, 10-113). White patient race was the only significant predictor of tumor recurrence. Patient age (≥55 years), tumor size (≥4), high mitotic rate, and disease subsite were not associated with recurrence.
Conclusion
Head and neck solitary fibrous tumors demonstrate a significantly larger local recurrence rate as compared with their rate of metastasis. They can recur many years following initial therapy, warranting long-term surveillance and follow-up to assess for tumor recurrence.
Solitary fibrous tumors (SFTs) are rare mesenchymal neoplasms that were initially recognized as pleural tumors but are now known to occur at any site, including the head and neck.1,2 Approximately 20% of SFTs occur in the head and neck, most commonly in the oral cavity, but can also arise in the nose and paranasal sinuses, meninges, orbit, major salivary glands, larynx, and thyroid.3-8 Clinical presentation in the head and neck varies by anatomic location and compressive symptoms due to indolent growth. 9
The histopathology of SFTs is variable but typically demonstrates either fibroblastic or cellular spindle-cell morphology. 10 SFTs were initially considered distinct from hemangiopericytomas after Klemperer and Coleman first recognized SFT as its own entity in 1931. 11 With the finding of an intrachromosomal inversion resulting in NAB2-STAT6 gene fusion, tumors previously diagnosed as hemangiopericytomas became recognized as a cellular variant of the classically fibroblastic SFT. 12 As there is often wide variation in the histomorphology of SFTs, immunohistochemical markers play an important role in the diagnosis of SFTs, which typically stain positive for CD34, CD99, STAT-6, and Bcl-2 and negative for EMA and S100. 13
Although SFTs are generally benign and follow an indolent course, there are case reports of lesions with malignant features, often defined by markedly increased cellularity and/or the presence of ≥4 mitoses per 10 high-power fields (hpf).10,14-16 In the review by Cox et al of 142 head and neck SFTs (HNSFTs), 10 tumors exhibited malignant features based on high cellularity and mitotic rate (≥4 mitoses per 10 hpf). 17 Despite these atypical features, not all cases that exhibit malignant microscopic features behave aggressively. In a prospective study by Gholami et al of 219 SFTs, malignant classification on histopathology was not associated with an increased risk of recurrence or death. 16
Due to SFTs’ behavioral variability, several studies have examined their clinicopathologic features to predict tumor aggression.12,18 Recent studies suggest that the presence of a TERT promoter mutation (telomerase reverse transcriptase) resulting in its overexpression may be associated with a shorter disease-free survival.19,20 Another study demonstrated that CD34-negative tumors were more likely to arise in the head and neck area and present as metastatic disease as compared with their CD34-positive counterparts. 21 Other studies have expanded beyond single prognostic markers and described models that estimate the risk of metastasis based on multiple factors, such as patient age, tumor size, mitotic activity, and presence of tumor necrosis.12,18,22,23
Because individual features of SFTs, such as high mitotic rate or the presence of necrosis, do not reliably predict tumor behavior, the World Health Organization’s most recent classification opts to use risk stratification models to determine their prognosis. 24 Although these risk assessment models may serve as a guide for assessing risk of metastasis, recent evidence supports clinical variability in SFT behavior depending on factors including, but not limited to, tumor origin.21,25 Herein, we examine the surgical outcomes for HNSFTs to determine risk factors for recurrence specific to the head and neck.
Methods
A single-center retrospective study was conducted on pathologically confirmed cases of HNSFTs between 1996 and 2021. This study was approved by the Institutional Review Board of the University of California, Los Angeles. Patients with an initial diagnosis of hemangiopericytoma and SFT of the head and neck were included. Patients’ demographic, clinical, pathologic, immunohistochemical, and follow-up data were reviewed. Demographic details consisted of patient sex, age at surgery, and race/ethnicity. Tumor size was preferably obtained from radiographic measurements such as computed tomography and magnetic resonance imaging. Otherwise, they were obtained from pathologic specimen size in its largest dimension. Immunohistochemical markers were obtained, and the 11 most common markers were included for analysis ( Figure 1 ). High mitotic figures, increased cellularity, and nuclear pleomorphism were noted ( Figure 2 ). Modality of treatment and, if applicable, adjuvant therapy were also noted. Outcome was indicated as recurrence, disease-free survival, last known disease-free follow-up, or death. Descriptive statistics were mean, median, range, and 95% CIs for the collected variables.
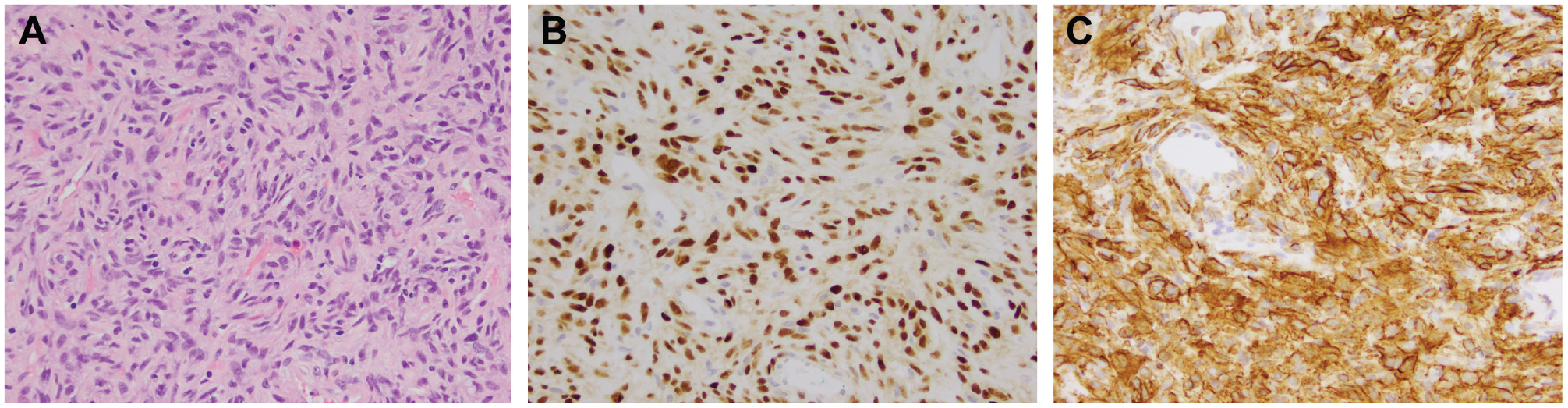
(a) Photomicrograph stained with hematoxylin and eosin demonstrates spindled ovoid cells arranged with no discernable architecture or histologic evidence of malignancy. Tumor cells show positive staining for (b) nuclear STAT 6 and (c) diffuse CD34 expression.
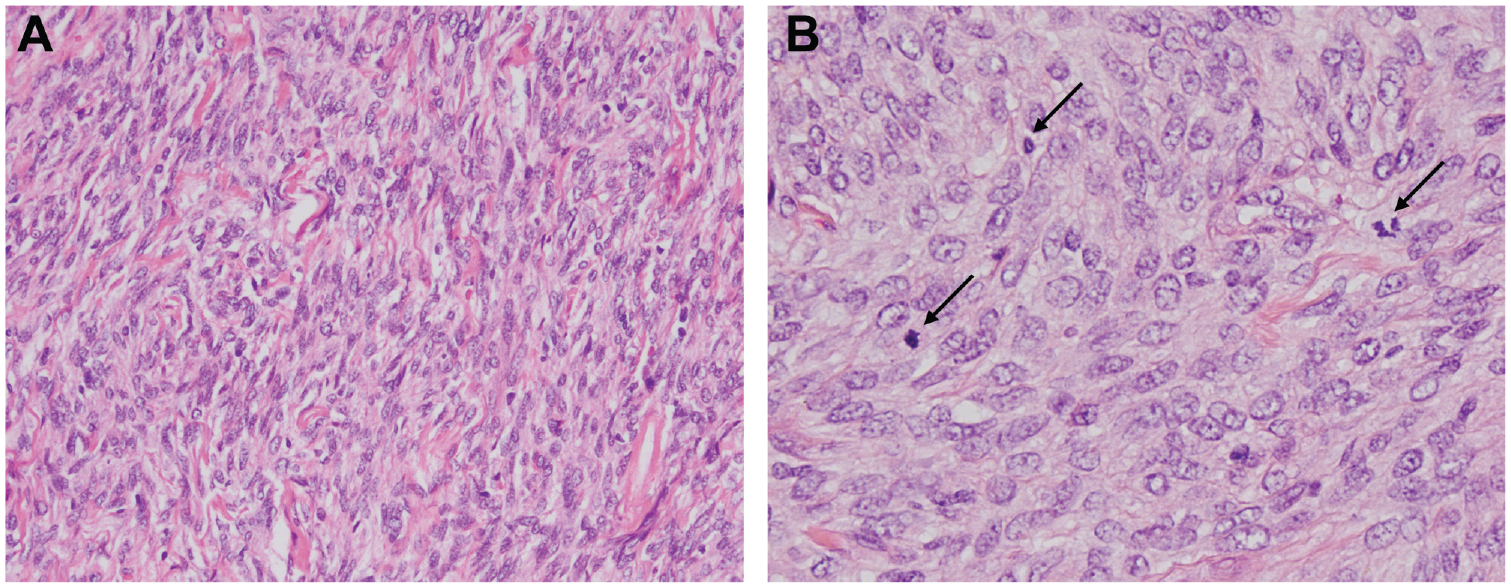
(a) Photomicrograph stained with hematoxylin and eosin demonstrates hypercellular population of spindle cells with pleomorphic and vesicular nuclei. (b) High-power view shows increased mitoses (black arrows).
For analysis of recurrence, defined as the development of tumor at the site of origin after treatment was completed, patients must have undergone en bloc excision of the tumor with curative intent. To exclude persistent disease due to incomplete initial management, patients who underwent tumor debulking or had a known tumor left behind were excluded. Patients who underwent previous excision with no surgical record were excluded, regardless of future recurrence status. Patients must have had at least 3 months of follow-up after surgery. Recurrence-free survival was estimated with Kaplan-Meier analysis. A log-rank test was used to assess differences by groups. Patients were censored accordingly at the date of last clinical follow-up, loss to follow-up, or death. All statistical analysis was conducted with SPSS Statistics Software version 28 (IBM).
Results
Our search identified 52 patients treated for HNSFTs at our institution ( Table 1 ). This cohort consisted of 23 males and 29 females resulting in a slight female predilection (55.8%). The average age was 54.7 and ranged from 15 to 89 years. Patients were predominantly described as White (n = 37, 71.2%), followed by Asian/other (n = 8, 15.4%), Hispanic/Latino (n = 4, 7.7%), and Black (n = 3, 5.8%).
Clinicopathologic Characteristics of Patients With Solitary Fibrous Tumor of the Head and Neck.

Clinical Features
Reported symptoms of orbital SFT included pain, ptosis, proptosis, optic neuropathy, strabismus, diplopia, exophthalmos, decreased visual acuity, and facial numbness. One patient with a supraglottic tumor experienced dysphonia. Most patients complained of a palpable mass prior to diagnosis.
Most patients (51/52) were treated with primary tumor resection with curative intent. One patient with an orbital tumor opted to decompress the optic nerve for improvement of visual acuity without full resection. Notably, 14 (26.9%) patients required repeat excisions and/or adjuvant therapy for disease persistence, many of whom presented to our institution after multiple previous attempts at tumor removal. Of these, 2 patients required 5 surgical procedures for full tumor resection. Four (7.7%) patients underwent incomplete resection and were thus excluded from recurrence analysis. Reasons for incomplete excision included tumor infiltration into sensitive structures such as nerve and the carotid artery, potential interruption of the pial circulation, and dense adherence to the pharyngeal wall. Three (5.8%) patients underwent preoperative angioembolization, and an additional 3 (5.8%) patients underwent postsurgical adjuvant radiotherapy.
Three patients were clinically diagnosed with malignant SFTs, defined by the tumors’ histopathologic features or multiple episodes of recurrence. One patient was diagnosed with malignant SFT of the orbit after multiple cases of locoregional recurrence. This patient underwent 3 orbitotomies and had plans in favor of repeat excision before being lost to follow-up. Another patient’s sinonasal tumor exhibited high mitotic index and recurred 3 times following 3 excisions. This patient then underwent debulking followed by adjuvant radiotherapy. Subsequent magnetic resonance imaging showed no evidence of disease. Finally, in the last patient diagnosed with malignant SFT that originated from the maxilla, the tissue exhibited increased cellularity with pleomorphic cells, hyperchromatic nuclei, and high mitotic activity. This patient underwent 4 resections, followed by adjuvant radiotherapy and a maxillectomy, after which margins were confirmed negative with no evidence of recurrent disease at last follow-up.
One patient had metastatic SFT in which a primary periorbital tumor metastasized to the lungs, confirmed via fine-needle aspiration of the thorax. Plans were in favor of adjuvant chemotherapy and radiation with possible surgery, but the patient was lost to follow-up thereafter.
Histopathologic Features
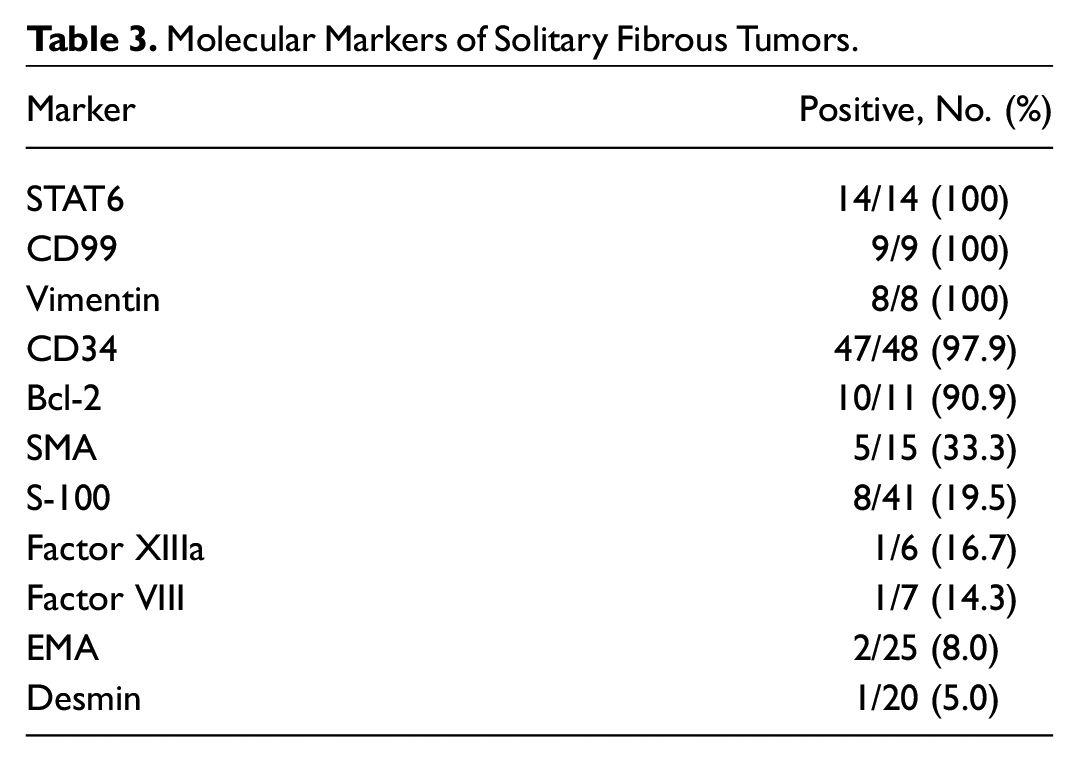
The tumor size ranged from 1.3 to 11.2 cm (median, 2.95; Table 2 ). The most common tumor sites were the orbit (53.8%), sinonasal cavity (15.4%), and parotid gland (11.5%). Nearly all tumors were positive for CD34 (97.9%), and all tumors were positive for STAT6 (100%; Table 3 ). Other common tumor markers included Bcl-2 (90.9%), vimentin (100%), and CD99 (100%). Less common tumors markers were S-100 (19.5%) and SMA (33.3%).
Radiographic Size of Solitary Fibrous Tumors in Its Greatest Dimensions.
Molecular Markers of Solitary Fibrous Tumors.
Outcomes
Following exclusion criteria, 26 cases were analyzed for recurrence. True recurrence was reported in 4 (15.4%) patients ( Table 4 ). Three of these patients experienced 1 recurrence after initial management and underwent reexcision. One patient experienced 2 recurrences, requiring 2 excisions with clear margins. None of these patients required adjuvant therapy or experienced malignant tumor transformation.
Recurrence of Solitary Fibrous Tumors Among Patients With Follow-up Data.
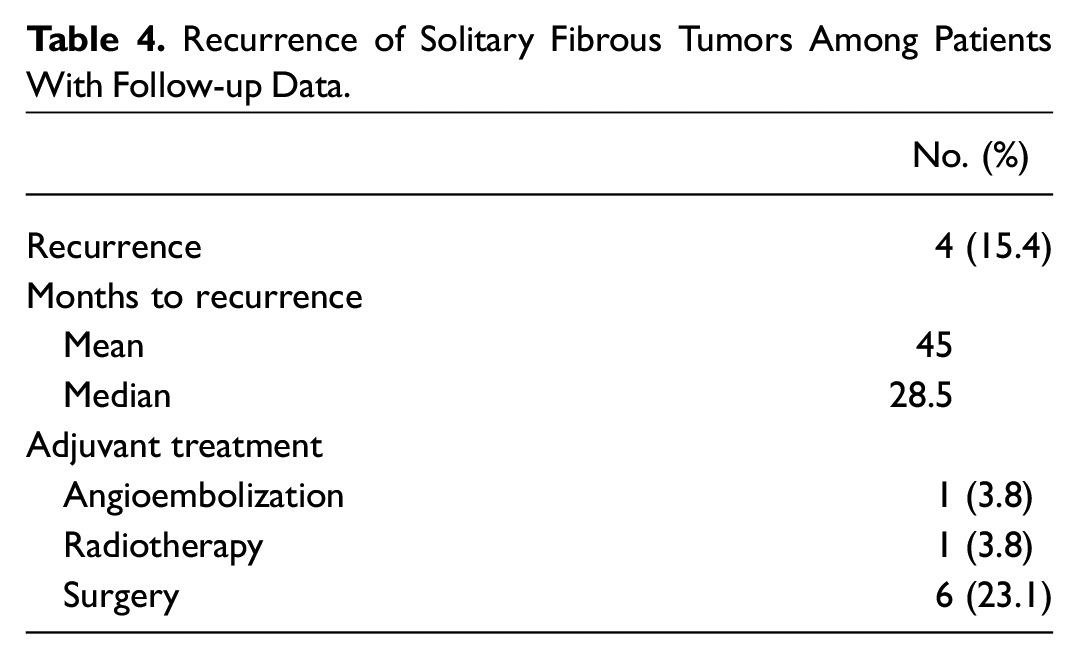
The last known disease-free period for the cohort was a median 31 months (range, 3-167). Median time to recurrence was 28.5 months (range, 10-113; Figure 3 ). Log-rank testing showed that the following were not associated with shorter recurrence-free survival: age (tested categorically with ≥55 years; P = .191), sex (P = .374), and tumor size (≥4 cm; P = .89). Differences among groups regarding tumor site was also insignificant (P = .754). However, White race was significantly associated with higher recurrence rates than Hispanic, Black, or Asian/other (P < .001). As only 1 variable was statistically significant, a multivariate analysis was not performed.
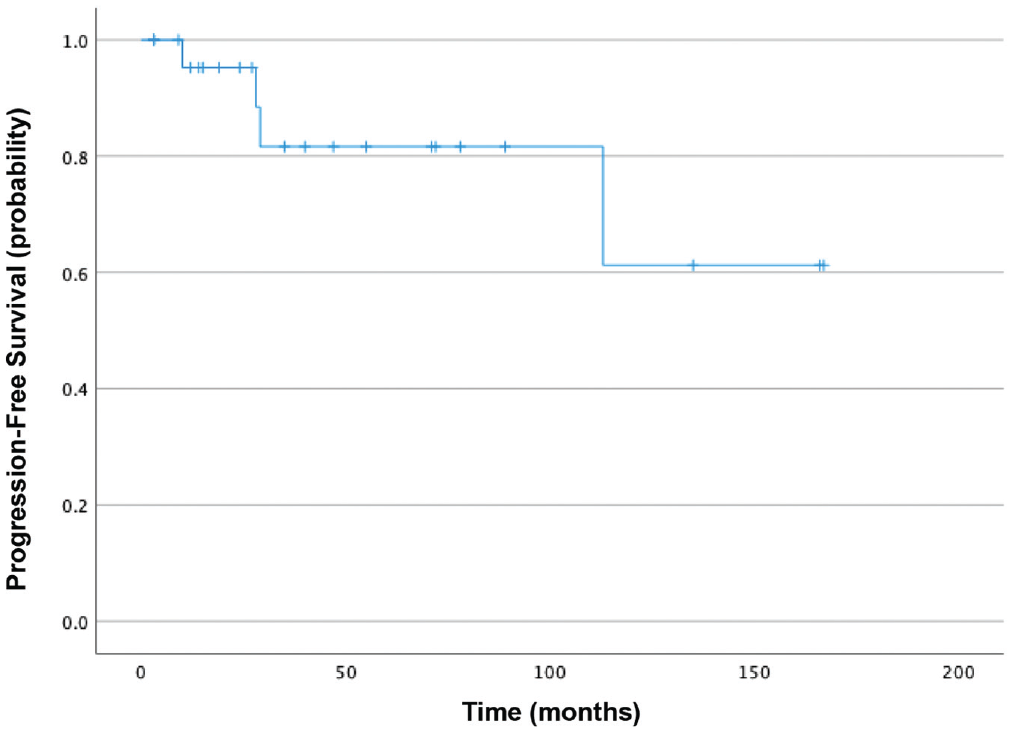
Progression-free survival.
Of the 26 patients analyzed for recurrence, 17 had data regarding marginal analysis on biopsy after initial surgery. Of these 17 patients, 14 (82%) had margins positive for tumor, while only 3 (18%) were confirmed to have negative tumor margins. All 3 patients with negative tumor margins on histopathology did not experience recurrence.
In addition, 13 patients had data regarding mitotic figures. Among these patients, 3 had mitotic figures ≥4 per 10 hpf. No significant associations were found between low and high number of mitotic figures (stratified at ≥4/10 hpf) and tumor recurrence (P = .627).
Discussion
Although SFTs as a whole are well known and highly classified, just 600 HNSFTs have been reported in the literature since 1991. 25 Our study in this patient cohort provides insight into HNSFTs’ clinicopathologic characteristics and prognostic associations.
Our study supports clinicopathologic features of HNSFTs noted in prior studies. Our cohort had a slight proclivity for female patients (55%) while prior studies found no difference between sexes.17,26 We confirmed this wide age range (15-89 years), similar median age (54.7 years), and tumor size (2.95 cm). Corroborating the findings of Smith et al that orbital SFTs represent a significant portion of HNSFTs, the most common subsite that we noted was indeed the orbit (55.8%). 26 We also found strong expression of CD34 (97.9%) and STAT6 (100%) in these tumors, as had prior studies that established the latter as a marker for HNSFTs, carrying >95% diagnostic sensitivity.27-31
Importantly, our cohort confirms the behavioral tendency of HNSFTs to locally recur rather than metastasize. Our cohort experienced a recurrence rate of 15.4%, similar to prior studies showing rates of 15% (2/13), 32 8% (1/12), 33 and as high as 40% (19/48). 26 In contrast, just 1 patient in our cohort had metastatic disease to the lungs. Reports of HNSFT metastasis are rare, with only a few published in the literature.26,34 In the largest meta-analysis of HNSFT cases (n = 587), Stanisce et al found only 2 patients who experienced metastatic disease, in which pulmonary lesions were found shortly after surgery. 25 We note that the lungs were the most common site of distant metastasis of SFT in all reports. 35 Whether this behavior is significantly associated with HNSFTs, however, remains to be elucidated.
Herein, we report one of the few studies on HNSFT’s time to recurrence. Our median time to recurrence was 28.5 months. In previous studies, Baněčková et al and Smith et al showed a median time to recurrence of 36 and 19 months, respectively.26,36 The most clinically important finding is that recurrence may present years following initial therapy. Our latest time to recurrence was 113 months. In the literature, recurrence of HNSFT has been reported as late as 228 months, nearly 20 years from initial treatment. 26 We therefore emphasize the importance of long-term follow-up and surveillance to assess for recurrence of HNSFTs.
Our study found a high rate of positive margins on surgical pathology (82%). Only 3 patients were confirmed to have negative tumor margins. Notably no patient with negative margins on histopathologic review experienced local recurrence. To highlight the importance of achieving clear surgical margins, a previous meta-analysis demonstrated that the presence of positive margins was the sole factor predictive of time to recurrence in HNSFTs. 25
Studies of extracranial HNSFTs have reported difficulties obtaining en bloc resection due to either a thin friable capsule or adherent attachments to critical structures, 37 explaining the high rate of positive margins. Further exemplifying this surgical challenge is our finding that nearly 30% of our cohort required either multiple operations or adjuvant therapies before obtaining anatomically clear margins. Indeed, 2 patients required 5 surgical procedures due to incomplete resection. We therefore echo the conclusion from Cox et al that HNSFTs may be difficult to fully excise due to the anatomic complexity of their surroundings and that lack of full excision likely plays a role in their tendency to locally recur. 17
Finally, when testing for factors prognostic of shorter recurrence-free survival, we found patient race to be statistically significant, with White patients experiencing the highest rates of recurrence. Published analyses of race in SFTs did not previously find this to be a significant prognostic factor. 38 Furthermore, to our knowledge, no previous study has explored the link between patient ethnicity and tumor behavior in HNSFTs. However, we caution the interpretation of this finding given our small cohort of 26 patients included in recurrence analysis, as our finding may be underpowered. In contrast to the risk assessment model of SFTs by Demicco et al, 18 we did not find patient age (≥55 years) or mitotic figures (≥4/10 hpf) to predict tumor aggression. In addition, this group’s tumor size stratification of ≥15 cm was not used, given that HNSFTs rarely grow to achieve this size. Like other publications, we caution the use of their model for use in prognosticating HNSFTs. Not only are head and neck tumors significantly smaller than pleuropulmonary or abdominopelvic tumors, but our study showed differing behaviors exhibited by head and neck vs intrathoracic SFTs, the most salient being the former’s propensity to recur rather than metastasize. The Demicco et al model applies size cutoffs too high to be relevant to HNSFTs, and the model focuses on SFTs’ metastatic potential rather than their potential for local recurrence.
Conclusion
HNSFTs are a rare occurrence, with only 600 previously reported in the literature. Our analysis of 52 patients validates that HNSFTs demonstrate a significant local recurrence rate while rarely metastasizing. Importantly, they can recur many years following initial therapy. Therefore, early- and long-term surveillance and follow-up are warranted to assess for recurrence. Finally, our analysis supports that surgical excision with clear margins should remain the mainstay of treatment. Because our study further validated HNSFTs’ unique presentation and tumor behavior as compared with SFTs of other sites, we believe a site-specific risk assessment model for HNSFT recurrence is warranted.
