Abstract
Solitary fibrous tumors (SFTs) are rare mesenchymal neoplasms that were initially identified in the pleura. SFTs in the nasal or paranasal sinuses are especially rare. Most SFTs exhibit indolent behavior, with a low local recurrence rate. A 39-year-old man complained of bilateral nasal congestion, hyposmia, and occasional right eye tears six months prior to hospitalization. Based on MRI and CT imaging, a total gross surgical resection was achieved. Subsequently, postsurgical histopathological examinations were conducted. Under the microscope, pathological mitotic bodies were visible (<5 mitoses per 2 mm2). The immunohistochemical staining results revealed that tumor cells were positive for CD34, BCL-2, STAT-6, and Ki-67 (<5%) but negative for EMA, S-100, PR, GFAP, and SMA. Based on these findings, the patient was diagnosed with SFT.
Introduction
Solitary fibrous tumors (SFTs) are a heterogenous group of rare spindle cell tumors, with approximately 80% of them being benign. 1 SFTs were first discovered in pleura. 2 Furthermore, the first case of SFTs in the central nervous system (CNS) was reported in 1996. 3 SFTs derive from almost every anatomical location of the human body, mainly in the pleura, pelvis, and retroperitoneum.4,5 The annual incidence rate of extrameningeal SFTs is 0.2 per 100,000 persons. 6 SFTs account for less than 2% of all soft tissue tumors. 7 Previously published series indicate that SFTs of the head and neck account for 15%−20% of all SFTs. 8 We searched the PubMed database and found nine English articles depicting SFTs in the nasal cavity and paranasal sinuses over the last decade. To date, no more than 50 cases of SFT have been reported. Almost all reported cases were confined to the nasal cavity or paranasal sinuses, with only a few cases extending to the orbit and cranial cavity. We only retrieved one case of orbital and intracranial invasion in a 56-year-old female SFT patient. This article, published in 2007, described that the tumor filled the left nasal cavity, upper two-thirds of the right nasal cavity, bilateral ethmoid and frontal sinuses, and infiltrated the left orbit and basi-frontal area. 9 Here, we present the case of a 39-year-old male SFT patient. The tumor originated from the right nasal septum near the crista galli, invading the paranasal sinuses widely and spreading to the right orbit and anterior cranial fossa.
Case Report
On April 6, 2022, a 39-year-old man suffering from bilateral nasal congestion, hyposmia, and occasional right eye tears for six months was admitted to the outpatient clinic without other nasal or neurologic symptoms. There is no relevant past medical history.
Before surgery, we performed computed tomography (CT) and magnetic resonance imaging (MRI) to enhancement of the paranasal sinuses. The CT scan detected that the right ethmoid sinuses and middle meatus were filled with high-density soft tissue. The nasal septum deviates to the left, and soft tissue density shadows can be seen in the right frontal sinus, ethmoid sinuses, and left maxillary sinuses. The bony nasal septum, right inferior turbinate, and right maxillary sinus wall were absorbed and destroyed. The right orbit was also infiltrated by the tumor (Figure 1(A)). It is obvious that the tumor compressed the right maxillary sinus and invaded the skull from the coronal CT scan (Figure 1(B)). The sagittal CT scan also validated the tumor extending to the anterior cranial fossa from the image of the bone defect (red arrow) (Figure 1(C)). MRI images further confirmed the findings of the CT (Figure 1(D to (I)). The lesions in the right ethmoid sinuses and middle meatus displayed heterogenous intensity in the T1- and T2-weighted MRI images. MRI imaging indicated that the mass was evidently enhanced with multiple cystic degeneration signal intensities inside. Tumor size was evaluated by MRI, measuring 60×31×42 mm in size. Hyperintensity on T2-weighted MRI images can be explored in the right frontal sinuses, right ethmoid sinuses, and left maxillary sinuses. CT and MRI revealed that the mass had widely invaded the paranasal sinuses and extended to the anterior cranial fossa. Radiological examinations: A high-density lesion in the right ethmoid sinuses and middle nasal meatus. The nasal septum deviates to the left, and soft tissue density shadows can be seen in the right frontal sinus, ethmoid sinuses, and left maxillary sinuses. An (axial CT scan): The bony nasal septum, the right inferior turbinate, and the right maxillary sinus wall are absorbed and destroyed. The right orbit is also infiltrated by the tumor; B (coronal CT scan): The tumor compresses the right maxillary sinus and invades the skull; C (sagittal CT scan): The significant bone defect (red arrow) evidently validates that the lesion has spread to the anterior cranial fossa; D–I (axial, coronal, and sagittal MRI scans): A high-density lesion in the right ethmoid sinuses, middle nasal meatus, right frontal sinus, and left maxillary sinuses, extending to right orbit and anterior cranial fossa. The lesions in the right ethmoid sinuses and middle meatus depict heterogenous signal intensities in the T1- and T2-weighted MRI images. Hyperintensity on T2-weighted MRI images can be explored in the right frontal sinuses, right ethmoid sinuses, and left maxillary sinuses. CT: computed tomography; MRI: magnetic resonance imaging.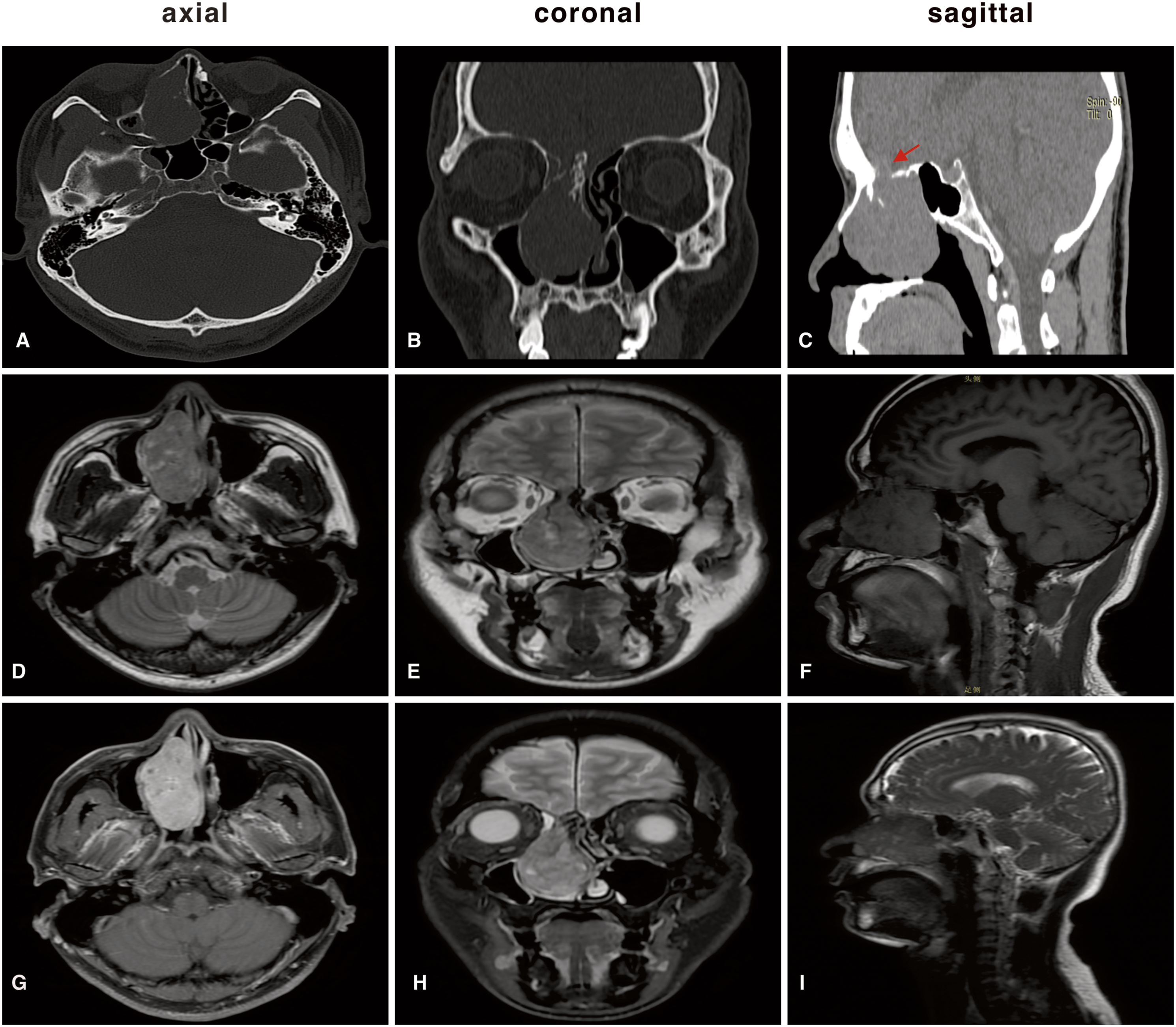
In surgery, the tumor had an abnormally rich blood supply with a tough gray-red appearance, measuring 8×6×3 cm in size. The mass originated from the right nasal septum near the crista galli, infiltrating the paranasal sinuses, invading the right orbit, and extending to the anterior cranial fossa with strong adhesion to the endocranium and posterior wall of the frontal sinus.
Hematoxylin-eosin (H&E) staining and immunohistochemical (IHC) were performed on the resected specimens. The histologic features of the resected surgical specimens are illustrated in Figure 2. Under the microscope, the background displayed red-stained collagen fibers with hyaline degeneration. Numbers of spindle-shaped or oval tumor cells can be seen with staghorn-shaped vessels (green arrow), which is a typical appearance in SFTs (Figure 2(A)). Examining high-power fields (HPF) demonstrated that tumor cells demonstrated mild to moderate atypia. The nuclear membrane of tumor cells is clear, the chromatin is uniform or granular, and the nucleoli are visible (Figure 2(B)). Pathological mitotic bodies can be seen in tumor cells (green arrow), and the mitotic count was less than 5 per 2 mm2 (Figure 2(C)). Immunohistochemically, tumor cells were diffusely positive for hematopoietic progenitor cell antigen (CD34) (Figure 2(D)), B-cell lymphoma (BCL-2) (Figure 2(E)), and signal transducer and activator of transcription 6 (STAT6) (Figure 2(F)). The proliferation labeling index of the Ki67 antibody was less than 5% positive in the tumor cells (Figure 2(G)). The expression of epithelial membrane antigen (EMA) (Figure 2(H)), S-100 protein (S-100) (Figure 2(I)), progesterone receptor (PR) (Figure 2(J)), glial fibrillary acidic protein (GFAP) (Figure 2(K)), and smooth muscle actin (SMA) (Figure 2L) were negative. Histopathologic analysis of the specimens. A: Microscopic examinations depict that the background is red-stained collagen fibers with hyaline degeneration. Numbers of spindle-shaped or oval tumor cells can be seen with staghorn-shaped vessels (green arrow) (HE 100×); B: Tumor cells are mild to moderate atypia. The nuclear membrane of tumor cells is clear, the chromatin is uniform or granular, and the nucleoli are visible (HE 400×); C: Pathological mitotic bodies in tumor cells (green arrow) are obvious, and the mitotic count was less than 5 per 2 mm2 (HE 400×); D–F: The tumor cells are diffusely positive for CD34 (D), BCL-2 (E), and STAT6 (F) (IHC 400×); G: The expression of Ki-67 is less than 5% positive in tumor cells (IHC 100×); G–L: The tumor cells are negative for EMA(H), S-100(I), PR(J), GFAP(K), and SMA(L) (IHC 100×). HE: hematoxylin and eosin staining; IHC: immunohistochemistry.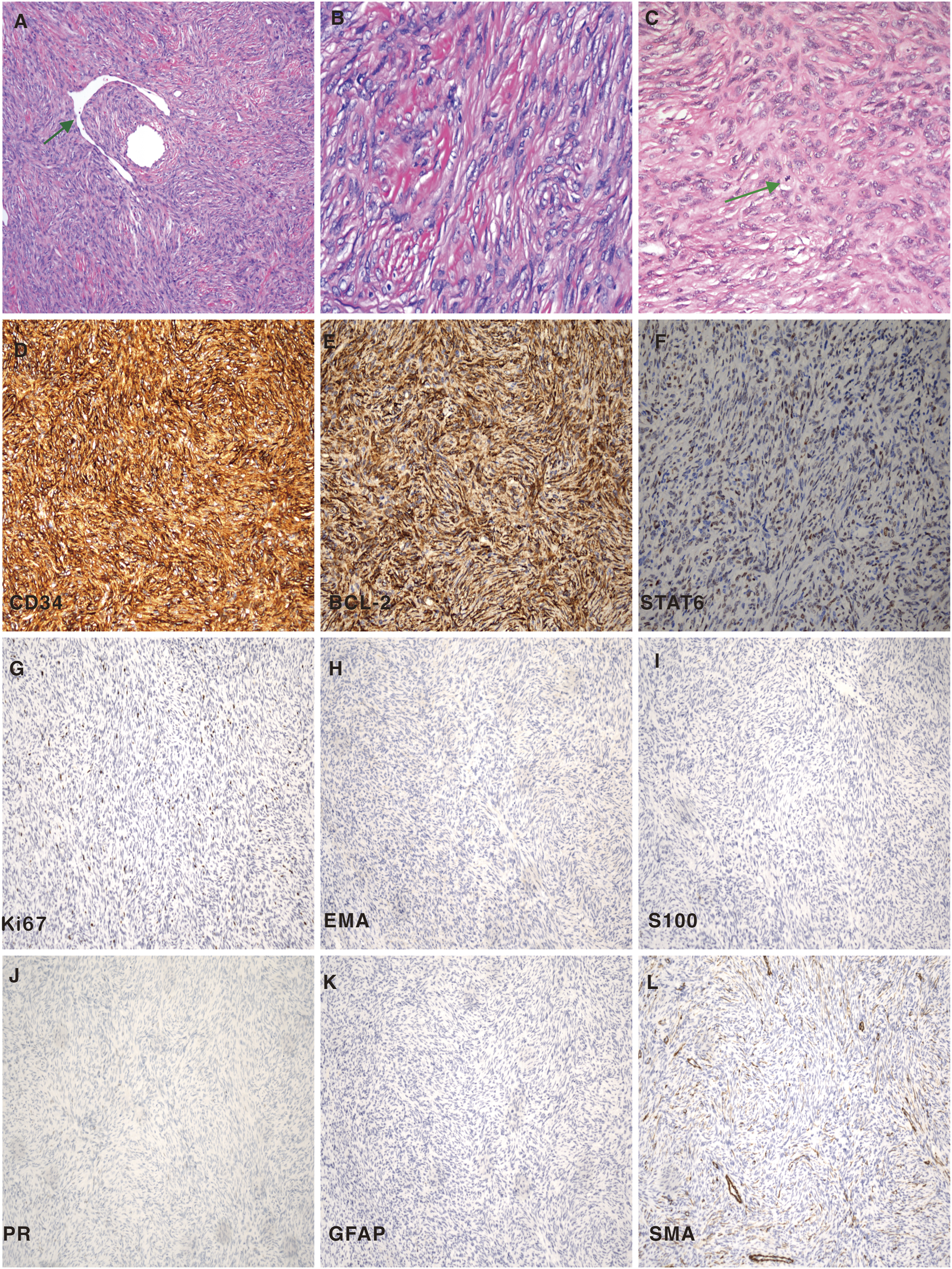
Discussion
The etiology of SFTs remains elusive. However, scientists have discovered that the formation of the NAB2-STAT6 gene fusion causes overexpression of a chimeric transactivation factor, eventually leading to tumor proliferation. SFTs mostly occur in adults in their mid-40s, ranging from 9 to 86 years.
1
It is notoriously difficult to predict the clinical course of SFTs. About 5–20% of SFTs behave clinically aggressive or malignant manner. SFTs in the nasal cavity and paranasal sinuses are commonly indolent and rarely demonstrate clinically malignant behavior. In 2020 WHO classification, the “benign” or “malignant” classic terms for SFTs have been eluded.
5
Furthermore, a novel risk stratification scheme for SFTs incorporates the patient’s age (below 55 or 55 years old and above), tumor size (<5 to ≥15 cm in increments of 5 cm), mitotic count (0, 1 to 3, or ≥4/10 high power fields), and tumor necrosis (<10% or ≥10%) has been recommended to predict prognosis.5,10 Details of the risk stratification of SFTs for the development of metastasis are illustrated in Figure 3. According to the risk stratification scheme, different risk factors for each patient correspond to different scores. The total score of the four risk factors was calculated. Then, the patient is classified into three risk classes: low risk: 0 to 3 points, intermediate risk: 4 to 5 points, and high risk: 6 to 7 points. The total score for the patient in our case (age: <55 years old, tumor size: 5≤10 cm, mitotic count: 1 to 3/10HPF, necrosis <10%) is 2 points. Therefore, the patient in our case should be identified as a low-risk class with the development of metastasis. Risk stratification of SFTs for the development of metastasis. SFTs: solitary fibrous tumors.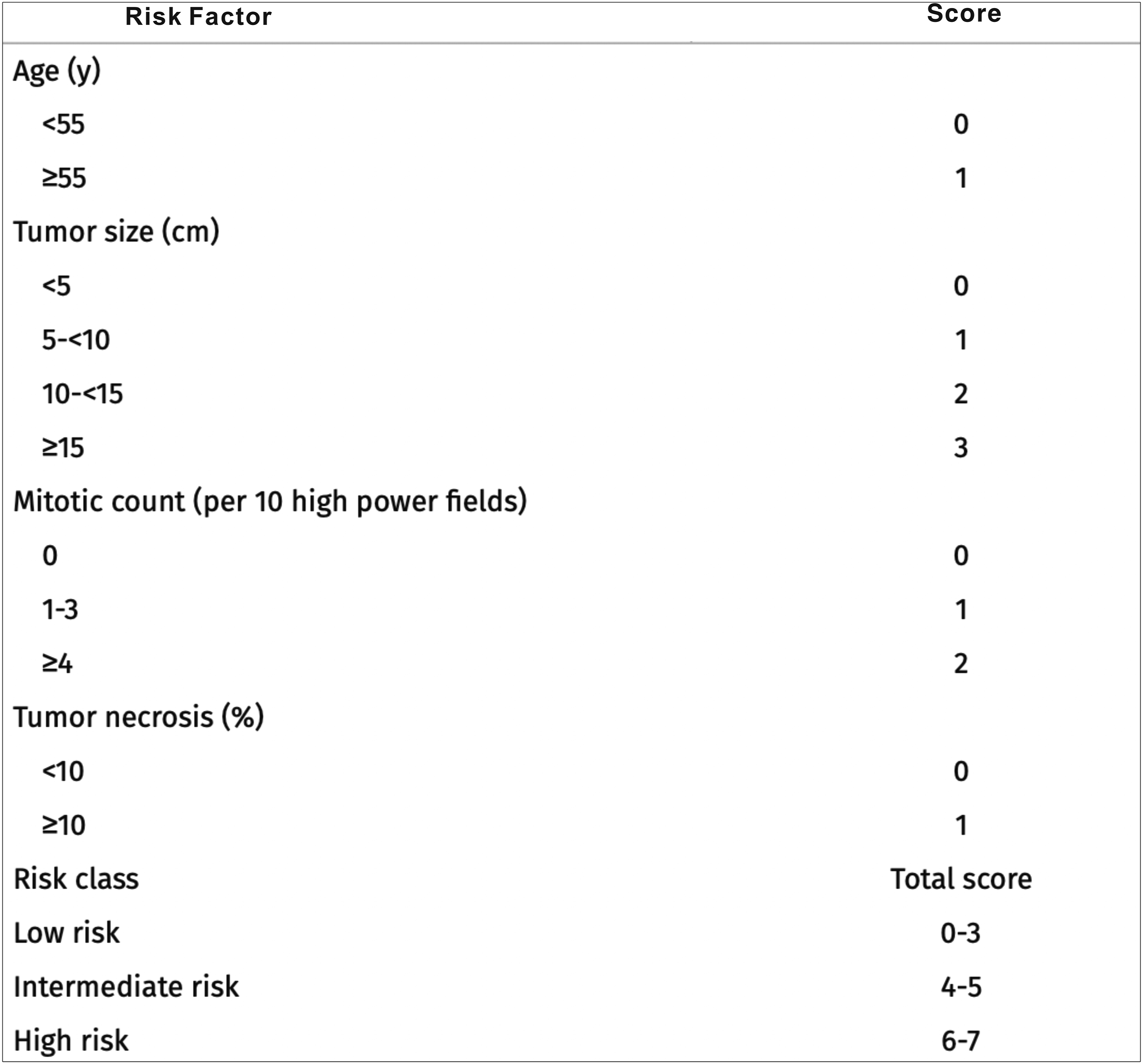
Progressive nasal obstruction is the most common clinical symptom of SFTs in the nasal cavity and paranasal sinuses. 11 In addition, symptoms of epistaxis, rhinorrhea, anosmia, headache, facial pain, exophthalmos, and visual disorders have been reported due to compression of the orbit patients. Studies indicate that SFTs in the CNS present mostly tentorium, posterior fossa, ventricular system, and spine symptoms. 12 Although the patient with SFT in our case had tumor intracranial and right orbital invasion, the man was only troubled with bilateral nasal obstruction, hyposmia, and occasional right eye tears for six months.
The mass in our case appeared hyper-signal intensity on the CT scan because of the rich fusiform cells of SFTs. The literature reveals that SFTs are usually homogeneously isointense to gray matter on T1-weighted MRI images and generally appear heterogeneously isointense or hypointense on T2-weighted MRI images. Hypointense on T2-weighted MRI imaging is considered a high prediction of SFTs.13,14 In our case, T2-weighted MRI images represented hyperintense in the right frontal sinuses, right ethmoid sinuses, and left maxillary sinuses.
Histopathologically, the tumor specimen expressed CD34 (+), BCL-2 (+), STAT6 (+), Ki-67 (<5%+), EMA (–), S-100 (–), PR (–), GFAP (–), CD68 (–), and SMA (–) and characterized by mitotic count < 5/2 mm2, high cell density, and staghorn-shaped vessel.
Most cases of SFTs are positive for CD34 and vimentin but negative for S-100 (adjunctive markers are BCL-2 and CD99). Immunoreactivity is diffusely expressed in SFTs. These immunoreactivity features can differentiate SFTs from other tumors. The expression of EMA, S-100, PR, GFAP, and SMA are sometimes useful for the differential diagnosis of SFTs from tumors with muscle, epithelial, or neural origin. For example, it can distinguish between meningioma (usually Vimentin+/EMA+/CD34−/S100−) and chordoma (Vimentin+/EMA+/Cytokeratin+/S100+). The positive expression rate of STAT6 in SFTs is 97.5%. 15 Detection of the expression of STAT6 is considered a surrogate for the presence of NAB2-STAT6 gene fusion to support the SFTs diagnosis. 16 The sensitivity of STAT6 in SFTs is up to 98%, and the specificity of STAT6 in SFTs is 87%.17,18 The CD34-positive expression rate in SFTs is 88.8%.15,19 The positive expression of CD34 is notoriously nonspecific and can be found in a wide range of soft tissue tumors (such as low-grade fibroblastic and myofibroblastic tumors). 20 The positive expression rate of BCL-2 is 88.8% in SFTs. Considering that the positive immunostaining of BCL-2 is associated with spindle cellularity, it is also expressed in some fibroblastic spindle cell sarcomas, spindle components of DD-LPS, and synovial sarcoma. 5 The proportion of positive Ki-67 >5% in SFTs is associated with tumor recurrence. 21
Most SFTs exhibit indolent behavior, with a low local recurrence rate, and can be cured with complete resection. Adequate surgical resection, when feasible, is recommended for all patients with SFTs. 22 Applying adjuvant radiotherapy has been proven to have longer recurrence-free intervals, but its effect on overall survival remains controversial.23,24
In conclusion, the patient in our case suffered from bilateral nasal congestion, hyposmia, and occasional right-eye tears. Before the operation, the paranasal sinus CT and MRI revealed that the mass had invaded the paranasal sinuses widely and extended to the right orbit and anterior cranial fossa. During surgery, the tumor derived from the upper right nasal septum near the crista galli. Histopathological examinations supported the diagnosis of SFT. Close and long-term follow-up for the patient should be considered.
Footnotes
Authors’ Note
Fan Yang, Liu-Qing Zhou, Jing-Cai Chen, Shan Chen, Di-Wei Zhou, Gang Zhong, and Yan-Jun Wang contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bethune Charitable Foundation (grant number BQETY-SSPC (8)-E 01) and Health Commission of Hubei Province Scientific research project (grant number WJ2021M250).
Statement of Informed Consent
This article has obtained written informed consent of patients for their anonymized information to be published in this article.
Data Availability Statements
All data generated or analyzed during this research are included in this published article. Data supporting the findings of this study are available from the corresponding author upon reasonable request.
