Abstract
Background:
Cytokines are crucial mediators in cell-to-cell communication, immune regulation, and the pathophysiology of rhinologic diseases. Distinguishing cytokine profiles can elucidate inflammation types [eg, T-helper type 1 (Th1), Th2, and Th17] and guide therapeutic options. Various methods for cytokine sampling in the sinonasal cavity exist. This review aimed to provide an in-depth review of these methods, including their advantages, limitations, and analytical techniques.
Methods:
A narrative literature review was conducted using Medical Subject Headings (MeSH) keywords in PubMed and Google Scholar. Key MeSH terms include “cytokines,” “mucus,” “interleukins,” “biomarkers,” “sinonasal disease,” “chemokines,” “paranasal sinuses,” and “sampling.”
Results:
There are many different methods of cytokine sampling in the sinonasal cavity. Common methods include nasal lavage, forced exhalation, collection of nasal epithelial lining fluid, nasopharyngeal swab, cytobrush, sponge, suction, and tissue biopsy. Each method used has advantages and limitations, such as type of sample collected, location specificity, invasiveness, sample dilution and volume variability, and utility for downstream analytical assays.
Conclusions:
Advancements in cytokine sampling and pharmacological targeting in sinonasal disease are instrumental for the diagnosis and management of patients with sinonasal disease. Understanding these methods’ strengths and weaknesses can improve patient care and guide treatment development.
Introduction
Chronic rhinosinusitis (CRS) and allergic rhinitis (AR) are 2 of the most common diseases in the United States, with a prevalence of 12% and 14%, respectively.1,2 Though there are set clinical criteria for diagnosing CRS and AR, no “gold standard” diagnostic laboratory test has been established.2,3 The recent adoption of biologics targeting inflammatory interleukins (ILs) in select patient populations with CRS marks a significant milestone. 4 Cytokines are central to the pathophysiology of many rhinologic diseases, influencing the progression and chronicity of conditions such as AR, CRS, and nasal polyps (NP), making cytokines logical targets for treatment.5,6 Though the use of biologics has been revolutionary in the fields of allergy and rhinology, studies investigating the utility of blood count-based inflammatory markers in CRS as a predictor of response to biologics provided underwhelming associations. 7 Other studies exploring the associations between phenotype and endotype showed some incongruence, highlighting the need for objective laboratory findings in disease-affected samples in the diagnosis of CRS. 8
Given the crucial role of inflammation in rhinological diseases, understanding the molecular mediators involved, particularly cytokines, can provide deeper insights into disease mechanisms, potential diagnostic markers, and response prediction to biologics. 9 As advancements are made in elucidating endotypes of rhinologic diseases such as CRS and AR, developing and identifying accurate and reliable cytokine sampling techniques of the nasal cavity and paranasal sinuses is paramount in advancements toward personalized therapy and treatment algorithms in patients. Additionally, the development of noninvasive sampling techniques offers the advantage of feasible collection in low-acuity settings, such as an outpatient clinic, which provides an alternative to more invasive, resource-intensive settings like an operating room. 10
AR is a condition that is strongly associated with a T-helper type 2 (Th2) inflammatory response characterized by an increase in levels of IL-4, IL-5, IL-10, and eosinophil cationic protein (ECP), the latter of which is a hallmark biomarker for eosinophil activation and recruitment.11,12 IL-2 levels have been associated with an increased risk of NP, while platelet-derived growth factor-BB is associated with a decreased risk. 5 CRS is an idiopathic disease that is shown to encompass numerous phenotypes and inflammatory profiles. CRS is clinically-commonly differentiated into 2 subtypes: CRS without NP (CRSsNP) and CRS with NP (CRSwNP). Initial studies on biomarkers within CRS attributed CRSsNP to the Th1 inflammatory endotype and CRSwNP to the eosinophilic-predominant Th2 inflammatory endotype. 13 Further studies that aimed to cluster endotypes from inflammatory markers found 10 clusters that could be sorted into 4 main categories based on their cytokine profiles. The first is the eosinophilic/Th2/antibody-driven subtype associated with ECP, IL-5, IgE, SE-IgE, and albumin. The second was neutrophilic/pro-inflammatory characterized by IL-1β, IL-6, IL-8, and MPO. The third profile, Th17/Th22, was associated with IL-17A, IL-22, and TNF-α. The final is a Th1 profile associated with IFN-γ. 14 Another study highlights that CRSwNP often involves type 2 inflammation, which is linked to severe cases, asthma comorbidity, and recurrence. Treatment strategies focus on precision medicine, including biologics targeting Th2 cytokines (IL-4, IL-5, and IL-13). 15 Biologics, such as dupilumab (targeting IL-4 and IL-13), mepolizumab, benralizumab, reslizumab (targeting IL-5), and omalizumab (targeting IgE), have shown efficacy in reducing polyp size, improving nasal symptoms, and enhancing quality of life. 16
In 2023, the EPOS/EUFOREA updated guidelines in a statement on the utilization of biologics in CRSwNP based on eosinophilia as a proxy for type 2 inflammation. 4 Other research worked on developing a novel diagnostic model to identify inflammatory endotypes in CRSwNP using cytokine analysis, particularly IL-5 levels. The authors utilized this model to develop a simple clinical scoring system, establishing a preliminary endotype classification algorithm to personalize treatment. 17 As this area of precision medicine is new, evaluating the costs and benefits of the numerous sampling methods available to assess cytokine levels is critical, which may lead to better treatment of AR and CRS.
Blood (serum and plasma), cerebrospinal fluid, urine, saliva, mucus, and tissue are all common biological specimens that have shown utility in elucidating cytokine levels and profiles in medicine. 18 In the setting of rhinologic disease, the sinonasal cavity offers a local niche that is optimal for studying immune responses and mucosal immunity in the disease-affected area and the area most likely to benefit from biologic treatment.
This review aimed to evaluate the various cytokine sampling methods used in the nasal cavity and paranasal sinuses. We highlight their advantages, limitations, and applicability in clinical and research settings. A concise overview of the different techniques is presented in Table 1. Additionally, we provide a broad overview of cytokine profile interpretation and analysis methods, emphasizing their relevance in characterizing inflammatory endotypes and guiding personalized therapeutic approaches for rhinologic diseases. By synthesizing current evidence, this review aimed to provide groundwork for future directions in personalized medicine, including cytokine profiling, to help with endotype-based disease stratification in conjunction with clinical findings and targeted treatment strategies in sinonasal pathologies.
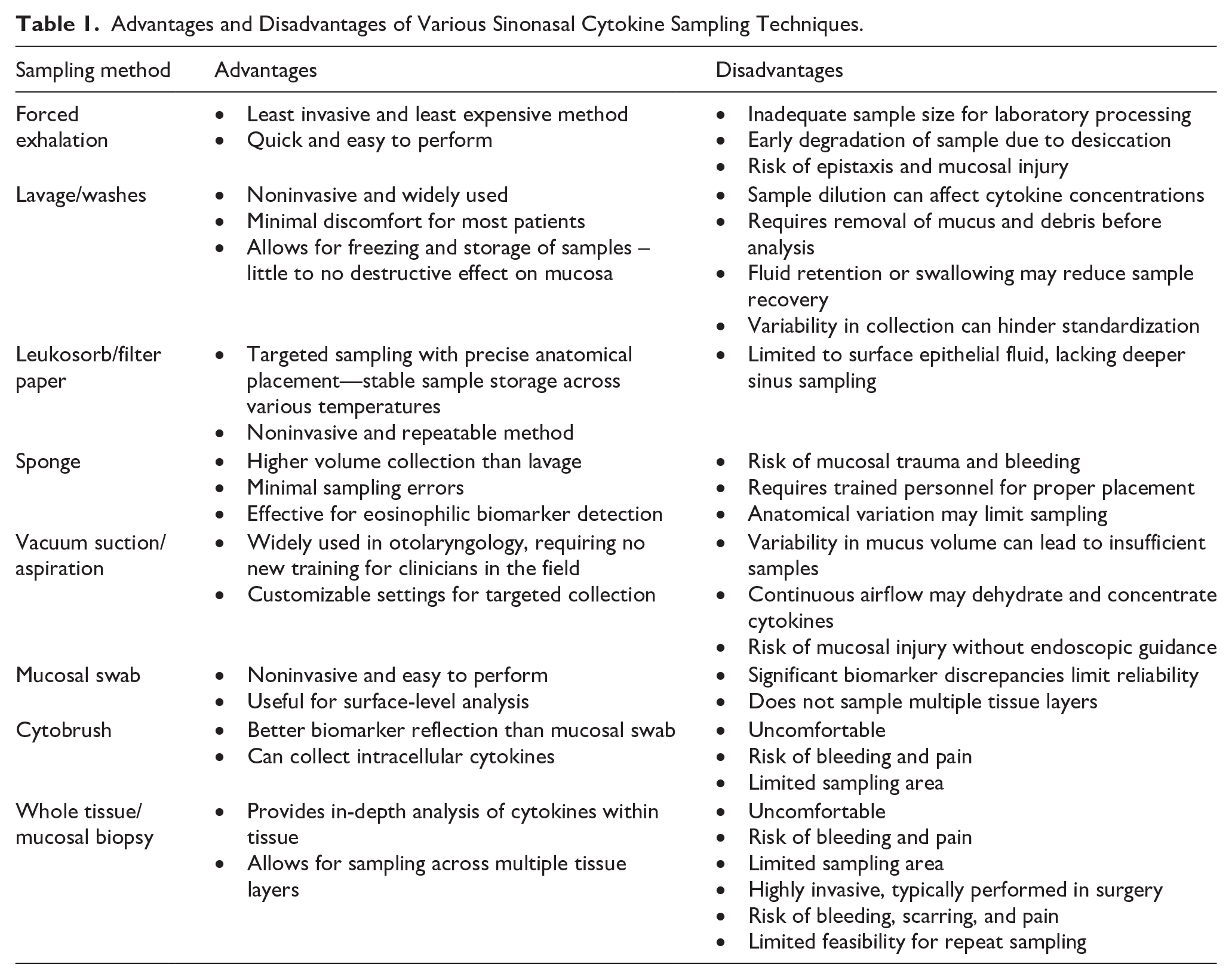
Advantages and Disadvantages of Various Sinonasal Cytokine Sampling Techniques.
Literature Search Strategy
A review was performed using PubMed and Google Scholar from December 2 to 18, 2024. Searches combined Medical Subject Headings and text words related to sinonasal cytokine sampling, including terms “cytokines,” “chemokines,” “sinonasal disease,” and methods like “lavage,” “leukosorb,” “sponge,” “swab,” “cytobrush,” “biopsy,” and “suction.” Relevant articles were selected based on methodological focus, clinical applicability, and sample utility. Reference lists of key studies were also reviewed for additional sources.
Methods of Nasal Cytokine Sampling
Forced Exhalation
Forced exhalation, colloquially known as “blowing your nose,” is the simplest, least invasive, least expensive, and least timely-sampling method. This technique involves closing the mouth and manually obstructing the nasal valves unilaterally while expulsing nasal secretions through the contralateral nostril. Unfortunately, sample sizes from this method have been shown to provide an inadequate sample size for laboratory processing. Secretions are also typically desiccated due to increased exposure to the lumen of the nasal passages, leading to early degradation of the sample and a reduction in viable cells available for query. 19 Furthermore, this method can induce epistaxis and mucosal injury. 20
Lavage/Washes
Nasal lavage (NL) involves propelling a liquid, typically saline, into the nostrils while the patient’s neck is extended at a 45° angle. The saline remains in the nostrils for a predetermined time before the subject leans forward and the sample drains from the nostrils or is gently expelled into a collecting basin. 21
NL has been widely studied and utilized as one of the least invasive and most efficacious methods of sinonasal sampling. Minimal resources are required for this technique, making sample collection relatively inexpensive. Tolerability varies, but due to the common practice of nasal saline irrigation, most patients experience minimal discomfort. This technique has also been shown to have little to no destructive effect on the native mucosa. 19 Studies have explored the effects of storing, processing, and shelf life of NL samples on the extracted cytokine profiles. The effluent from this method has the advantage of being able to freeze, which helps preserve and store the sample. 22
One of the most common limitations when employing the NL technique is sample dilution. One study found that up to 35% of samples had analyte concentrations below thresholds for highly-sensitive assays. 23 NL also requires the removal of large mucus clumps and cellular debris through centrifugation and filtration before the sample can be stored or analyzed, making it a time-intensive process in the laboratory. 22 Moreover, NL can be limited by fluid retention in the nasal cavity and paranasal sinuses or swallowed by the subject after exiting the nasal choanae, thus affecting cytokine concentrations and end sample volume. 24 Variability in effluent collection may limit accurate dilution factor calculations for many samples. 19
Leukosorb/Filter Paper
Leukosorb (Pall Scientific, Port Washington, NY, USA) is a specialized absorbent paper that can be cut into strips to extract nasal epithelial lining fluid (NELF). This is carried out by spraying the nostrils with sterile normal saline, inserting test strips into the nasal cavity, and clamping the nostrils for 2 minutes. Fluid retained on the strips can be stored and analyzed for various inflammatory cytokines. Strips have also been shown to capture upper respiratory microbes.25,26 This method offers a noninvasive and practical approach to collecting localized biological samples for diagnostic and research purposes.
Leukosorb strips can be precisely directed to the anatomical region of interest, allowing targeted endonasal sample collection. Their utility has also been demonstrated in endoscopic application to show consistent recovery of cytokines without losses through dilution, as seen in techniques like NL. 27 Retained samples demonstrate stability when stored at a range of temperatures (room temperature, 37°C, and −80°C) in preserving the integrity of cytokines and viral particles for subsequent analysis. 26 Furthermore, this method is noninvasive and repeatable, with studies showing day-to-day consistency in the makeup of intra-nare sampling, suggesting that this methodology does not disrupt the integrity of the nasal mucosa like other noninvasive methods. 26 . This technique is compatible with a variety of platforms commonly utilized to quantify cytokines. 28
The use of Leukosorb strips is associated with potential limitations. Mechanical trauma to the strip itself during insertion may introduce artifacts, which could affect sample integrity, although this is not well-characterized. Moreover, this method is confined to the surface epithelial fluid and lacks the specific sampling advantages of more invasive methods that can reach deeper sinuses. Depending on desired sampling location, such as within the sinuses versus in the nasal passages, use of this sampling method may require a trained professional for insertion. Furthermore, depending on how the strips are cut and their surface area, sampling location is limited to a finite region, rather than sampling from the larger sinonasal cavities, as would be completed in a wash or lavage. Finally, while the concentration of samples collected via this method is a benefit for evaluating low-abundance cytokines, the relatively-low amount of sample eluted per person can limit the number and type of assays conducted.
Sponge
The use of an absorbent material, such as a sponge, has increased in popularity in the past few decades to extract mucus from the sinonasal cavity.25,29 Sponges can be manufactured using various materials such as polyurethane, polyvinyl alcohol, and cellulose. Using instrumentation, the sponge can be attached to the head of a thin, long, hollow tube made of plastic for ease of application or placed in the target area, typically between a turbinate and septum.
Sponges have minimal sampling errors and extract a significantly-higher concentration of nasal secretions than NL, leading to more consistent measurements across collected samples.30,31 Sponges were also shown to have the highest percentage of samples above detectable limits compared with forced exhalation, NL, and suction. However, to our knowledge, concentrations of mediators collected via sponge have not been directly compared with NELF.
Sponges are typically-soft and malleable material but still have risks of mucosal trauma and bleeding associated with insertion and removal. Any rigid surgical instrumentation inserted into the nasal cavity to place the sponge in the desired location also has associated risks. To minimize risks, sponges must be placed by otolaryngologists or other trained staff, which poses a disadvantage for large-scale collections where rapid sampling from numerous individuals is required, such as in larger clinical or epidemiological studies. Due to its direct contact nature, this technique has limitations in the sampling location. Unlike NL, sponges cannot directly sample mucus in the paranasal sinuses but have great efficacy sampling locations such as the inferior/middle turbinates and bulla ethmoidalis. 32 In a similar vein, anatomical variation can limit sampling, for example, in severe septal deviation, mucosal hypertrophy, and nasal polyposis.33,34
Vacuum Suction/Aspiration Techniques
Suction is a technique that involves using a rigid or flexible extended vacuum device connected to tubing that empties into a collecting basin to extract mucus from the nasal cavity. These devices often have customizable settings for vacuum strength and attachment types that help minimize mucosal damage and reach obscure areas.
Suction has historically been an indispensable and extensively-utilized instrument in the armamentarium of otolaryngologists and surgeons across various specialties. As suction/aspiration is widely utilized and trained for in otolaryngology training, using this technique to collect samples for research would not require mastery of new techniques, but rather application of established techniques for new goals. This technique has also been shown to provide high concentrations of immune mediators when compared to the sponge technique. 19
Like sponges, suction instrumentation has a risk of injury when instruments are inserted into the nasal cavity. It requires trained users to extract mucus from desired anatomical locations while minimizing mucosal trauma, potentially hindering large-scale sample collection. Unfortunately, due to the vast variability in mucus secretions throughout the day, hydration, or disease status, some of the volume of mucus collected can be insufficient for analysis, up to half of collected specimens in 1 study. 19 Continuous airflow used in this technique may also dehydrate the viable samples, artificially inflating cytokine and biomarker levels by concentrating levels in collected samples. 19
Mucosal Swab
Mucosal swabs are sterile instruments commonly used to collect focal surface-level samples from mucosal surfaces. These swabs typically consist of an absorbent applicator tip made of cotton or a synthetic material such as rayon attached to a shaft. Mucosal swabs are gentle and versatile tools that collect mucus, mucosal cells, and microorganisms for analysis. However, 1 study found that mucosal swab sampling showed significant discrepancies in many biomarkers, limiting its reliability for precise CRS endotyping. This is likely due to the mucosal swabs’ inability to sample across multiple tissue layers or limiting sampling to localized regions in the sinonasal cavity. 10
Cytobrush
A cytobrush is typically used to collect cell samples from focal areas with bristles on the tip of the shaft. These bristles are designed to exfoliate and gather cell samples for cytologic analysis. The cytobrush, however, causes significantly-more discomfort in patients than swab and NL techniques. 35 In a study comparing cytokine levels when using a mucosal swab and cytobrush against a whole tissue biopsy, cytobrush sampling more closely reflected tissue patterns for most biomarkers than the swab. 10
Whole Tissue/Mucosal Biopsy
Whole tissue biopsy is a technique that involves the procurement of whole epithelial cells, such as ciliated and basal cells, from a region of the nasal cavity or paranasal sinuses using a curette and/or forceps. Numerous locations provide adequate tissue for sampling. The inferior turbinate is anatomically one of the most accessible structures within the nasal cavity, making it one of the most common sites of biopsy. 36 In functional endoscopic sinus surgery, a common procedure offered to patients with nasal airway obstruction, anterior ethmoid cells, such as ethmoid bulla, are routinely removed as part of the operation. 37 Due to the en bloc nature of the technique, tissue biopsy also allows for the analysis of layers from bone to epithelial layer. Unlike mucosal sampling methods, such as NELF, NL, and nasopharyngeal swabs, from which sampling primarily extracellular cytokines in the secretions of mucosa, biopsy and cytobrush techniques allow for examination of intracellular cytokines within the mucosal cells.
As expected, these techniques are the most invasive sampling methods, and almost exclusively occurs either as part of clinical research studies, as part of a necessary surgical procedures, or as part of a workup for another condition due to the high risk or epistaxis, scarring (more specifically for surgical tissue removal), and pain. Due to the focal nature of these techniques, biopsies can sample a specific area of the sinonasal cavity, limiting the breadth of sampling. The use of local anesthetic, topical and/or injected, and vasoconstrictors is common practice when performing these types of procedures in the nasal cavity. 38 Local anesthetics such as lidocaine have been shown to possess anti-inflammatory properties that alter T-cell proliferation and chemokine levels. 39 Also, quantifying changes in cytokine levels is constrained by the limited feasibility of obtaining repeat biopsies from patients. Alternatives such as sponge, NL, NELF, and swab methods have shown efficacy in previous studies, with repeat sampling showing significant correlations between mucus secretions and native tissue biopsy, further decreasing the need to utilize this aggressive technique for sampling.10,31,40
Analysis and Interpretation of Samples
A range of analytical techniques are used to quantify cytokine levels in the sinonasal cavity, each offering unique advantages for profiling inflammatory markers. Enzyme-linked immunosorbent assay is a widely-employed method known for its high sensitivity and specificity, enabling the detection and quantification of cytokines either individually (single-plex) or in groups (multiplex format). 41 Multiplex bead-based assays use color-coded antibody-coated beads to quantify multiple cytokines simultaneously, providing a detailed cytokine profile from small sample volumes. 28 These methods can be used to detect both secreted cytokines in mucosal samples and intracellular cytokines from digested/dissociated tissue samples. Western blotting detects intracellular cytokines in tissue or cell lysates by separating proteins via gel electrophoresis, transferring them to a membrane, and using specific antibodies for identification, including posttranslational modifications. 42 Immunohistochemistry can visualize intracellular cytokines in tissue biopsies while preserving spatial context, using antibody-based enzyme or fluorescent labeling to assess cytokine distribution in inflammatory and immune responses. 43
Flow cytometry, particularly intracellular cytokine staining, identifies and quantifies cytokine-producing cells by staining fixed and permeabilized cells with fluorescently-labeled antibodies.44,45 Mass spectrometry techniques, including multiple reaction monitoring, are valuable for their ability to quantify cytokines in complex biological matrices with high specificity without relying on antibodies, making them significantly-more cost-effective.46,47
Transcriptional analysis, such as RNA-sequencing or real-time quantitative PCR, can be used to quantify cytokine mRNA levels in cellular samples, serving as a proxy of cytokine production and allowing simultaneous assessment of multiple cytokines. 48
Challenges and Limitations of Sinonasal Cytokine Sampling
Though the quantification of biomarkers collected from each of the sampling techniques described here may be a good proxy for true disease state and spectrum of inflammation within a patients’ nasal cavity, it has the potential to inaccurately reflect true cellular and inflammatory processes within the mucosa. 10 Patient nasal cavity inflammation and secretion volume vary widely due to time of day, seasonal changes, and environmental exposures. Circadian rhythms can affect congestion and mucosal secretion levels, while seasonal changes, especially in spring and fall, can increase inflammation and symptoms. 49 Environmental conditions, including humidity and pollution, also play a role.50,51 These variations can confound analytics and interpretation of samples collected, masking a patient’s true endotype. To mitigate these limitations, strategies such as repeat sampling, preconditioning the sinonasal cavity (eg, saline spray), and conducting patient histories that capture exogenous exposures can help improve consistency and accuracy in cytokine measurements and/or allow for statistical adjustment to account for factors such as exposure and seasonality.
Conclusions and Future Directions in Cytokine Sampling
Cytokine sampling in the sinonasal cavity offers critical insights into inflammatory rhinologic disease states. Techniques like NL, sponges, swabs, Leukosorb sampling of NELF, and biopsies involve varying levels of invasiveness and sample quality. These methods advance diagnostics and support biologic therapies targeting specific cytokines. Wider and more accessible application of cytokine sampling may also help identify exposures that lead to health disparities due to environmental factors.50,51 Future directions include refining noninvasive techniques and leveraging cytokine data for personalized medicine, enabling health care providers to offer tailored treatments that address less invasive and more holistic patient care.
Footnotes
Author Contributions
Y.M., E.H.B., and M.E.R. developed the manuscript concept. Y.M., M.L., E.H.B., L.O.B., and M.E.R. completed literature searches. Y.M., M.L., E.H.B., and M.E.R. drafted the manuscript and provided critical edits. All authors approved the final version.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors were supported by the following National Institute of Health grants during the completion of this manuscript: K01ES032837 and T32DC005360.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
