Abstract
This case report describes rare concomitant allergic fungal rhinosinusitis (AFRS) and chronic granulomatous invasive fungal sinusitis (CGIFS) in a 34-year-old woman with acute lymphoblastic leukemia and graft-versus-host disease (GVHD) post bone marrow transplantation. Initially presenting with rhinorrhea and nasal obstruction, the patient was diagnosed with AFRS in the right maxillary sinus, followed by a postoperative course of CGIFS in the left nasal cavity, showcasing the unique occurrence. She was not immunocompromised during diagnosis. CGIFS may have occurred because of surgery; however, voriconazole led to significant improvement. This case highlights noninvasive and invasive fungal infections in patients with chronic rhinosinusitis and a history of GVHD and underscores the complexity of diagnosing and managing such cases.
Introduction
Fungal sinusitis is classified into invasive and noninvasive types, where allergic fungal rhinosinusitis (AFRS) is a type of noninvasive mycosis, and chronic granulomatous invasive fungal sinusitis (CGIFS) is a type of invasive mycosis. 1
AFRS is frequently reported in South America, whereas its prevalence is low in continental regions with wet climates. Patients with AFRS have an allergy to fungi. CGIFS cases are frequently found in dry climate areas, including India, Sudan, Pakistan, and Saudi Arabia. 1 Invasive mycosis often develops in immunocompromised patients, whereas CGIFS frequently occurs in immunocompetent, healthy young adults. 1 Aspergillus is detected in many patients with CGIFS. 1
Graft-versus-host disease (GVHD) after transplantation causes organ damage. Acute GVHD mainly causes skin, gastrointestinal, and liver lesions, whereas chronic GVHD causes skin, eye conjunctiva, respiratory tract, liver, and gastrointestinal tract lesions. Regarding head and neck lesions, the diagnostic criteria for GVHD proposed by the multicenter study and the National Institutes of Health (NIH) consensus development project 2 mainly describe oral and cervical lesions, and no criteria for rhinosinusitis are mentioned.
Case Report
A 34-year-old woman with a history of acute lymphoblastic leukemia and bone marrow transplantation developed granulomatous inflammation due to GVHD. She also had intravascular fasciitis and meningioma and later developed rhinorrhea and nasal obstruction. After that, she visited the local otolaryngology clinic and received conservative treatment. The following year, erosions were found in the left inferior turbinate, and she was referred to our department for suspected granulomatosis with polyangiitis. Endoscopy revealed a scarring mucosa, which was assumed to have changed after GVHD, in a wide area of the epipharynx, along with mild erosion in the left inferior turbinate. After a biopsy, no findings suggestive of a relapse of leukemia, vasculitis, or malignant lymphoma were observed. Furthermore, she was diagnosed with granuloma. No abnormalities were revealed during imaging, blood tests, or urinalysis. Therefore, the possibility of antineutrophil cytoplasmic antibody-associated vasculitis was ruled out. She was presented to our department again at 34 because rhinorrhea and nasal obstruction worsened. Endoscopy revealed erosions, a granuloma-like mass in the left nasal septum, inferior turbinate (Figures 1A), and mucopurulent rhinorrhea in the right middle nasal meatus. Computed tomography showed a soft tissue shadow with a pale, high-density area in the right maxillary sinus; however, bone destruction or infiltrative shadows around the area were not observed (Figure 1B). Blood tests revealed an eosinophil of 4% [248 cells (<0.5 × 109/L)] with a slightly elevated total IgE level [610 IU/ml (<500 IU/ml)]. She tested positive for specific IgE antibodies against multiple fungi (Alternaria, Aspergillus, Cladosporium, Penicillium, Mucor, and Candida). In addition, β-D glucan levels increased slightly [27 pg/ml (<11 pg/ml)], whereas Aspergillus antigen levels were not elevated.
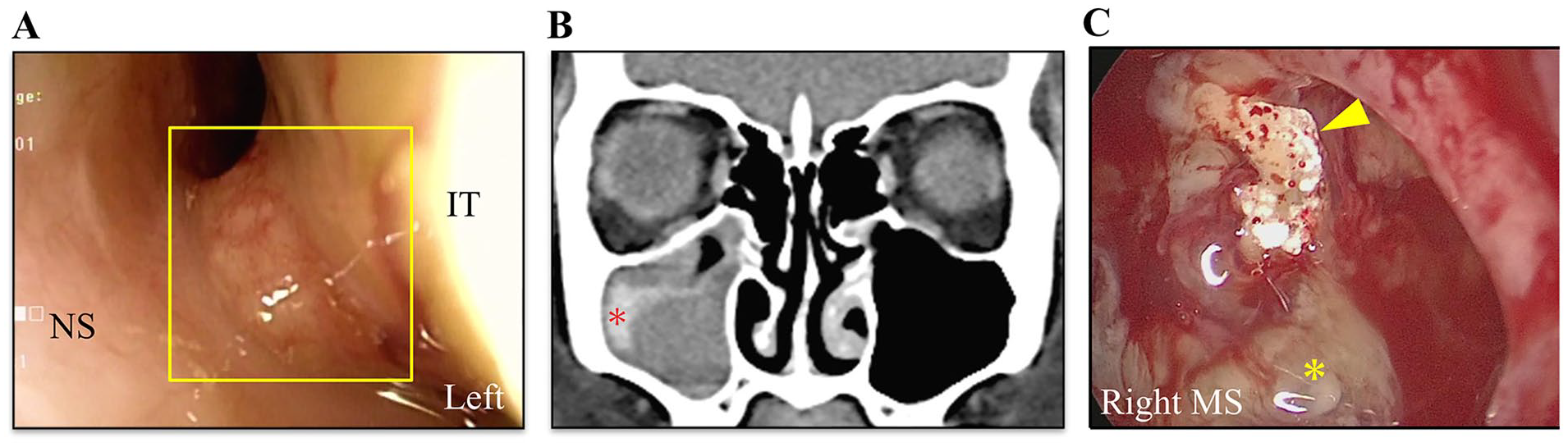
Physical and surgical findings from the second visit. (A) Granuloma-like mass (square) in the left inferior turbinate are found. A diagnosis of inflammatory granuloma associated with mycosis is made after biopsy. (B) CT findings of paranasal sinuses (coronal section) at re-examination (34 years); Soft tissue shadow with a pale high-density area is found in the right maxillary sinus (*). However, bone destruction or infiltrative shadows around the area are not observed. (C) Intraoperative findings (3 months later after presentation); mucin (*) and a fungus ball (arrowhead) are accumulated in the right maxillary sinus. CT, computed tomography; NS, nasal septum; IT, inferior turbinate; MS, maxillary sinus.
After a biopsy of the left inferior turbinate mucosa, the patient was diagnosed with inflammatory granuloma and concomitant mycosis. However, we confirmed that she did not have invasive mycosis. Histopathological identification of fungal species was difficult. Three months later, she underwent right-sided endoscopic sinus surgery (ESS), left-sided submucosal turbinectomy, and septoplasty. Mucin and a fungus ball were found in the right maxillary sinus (Figure 1C), and the causative organism was identified as Aspergillus fumigatus in the mucin and fungus ball cultures. Pathological examination revealed the presence of the eosinophilic inflammation and the fungi with Y-shaped mycelium in the mucin but no fungi were found in the mucosa of the right maxillary sinus, leading to right-sided AFRS diagnosis. Pathological diagnosis of the left inferior turbinate mucosa was granuloma and concomitant mild mycosis. No vascular wall infiltration was observed, and fungal species identification was difficult. The left inferior turbinate lesion with no invasive fungal infection was determined.
Postoperatively, mild mucosal edema in the right maxillary sinus persisted, but nasal symptoms and left nasal findings improved, and the patient was followed up on an outpatient basis. However, 6 months later, pain developed in the nasal vestibulum and erosion appeared in the left nasal vestibulum and anterior inferior turbinate. The erosion in the left nasal mucosa gradually worsened (Figure 2A), and a rash appeared on the nose dorsum (Figure 2B). Contrast-enhanced magnetic resonance imaging showed inflammatory signals from the left nasal vestibulum to the nasal septum, left inferior turbinate, nose dorsum, and high signal intensity area suggestive of inflammation in the right maxillary sinus (Figure 2C). The biopsy performed on the left nasal mucosa and skin of the nose dorsum confirmed granuloma formation in the blood vessel and intravascular infiltration of fungi, resulting in a left-sided CGIFS diagnosis. Voriconazole (VRCZ) 400 mg was orally administered. A colony was formed in the culture of biopsy specimens; however, the species was unidentified. Furthermore, biopsy specimens were sent to the Medical Mycology Research Center at Chiba University. Genetic testing showed that CGIFS was caused by a fungus of the Dothideomycetes class (the family Pleosporales, genus Didymellaceae). VRCZ treatment for 6 months administered orally improved her condition (the rash in the left dorsum of the nose disappeared; however, scarring remained in the areas from the left nasal vestibulum to the nasal cavity). No relapse was observed after 3 years.
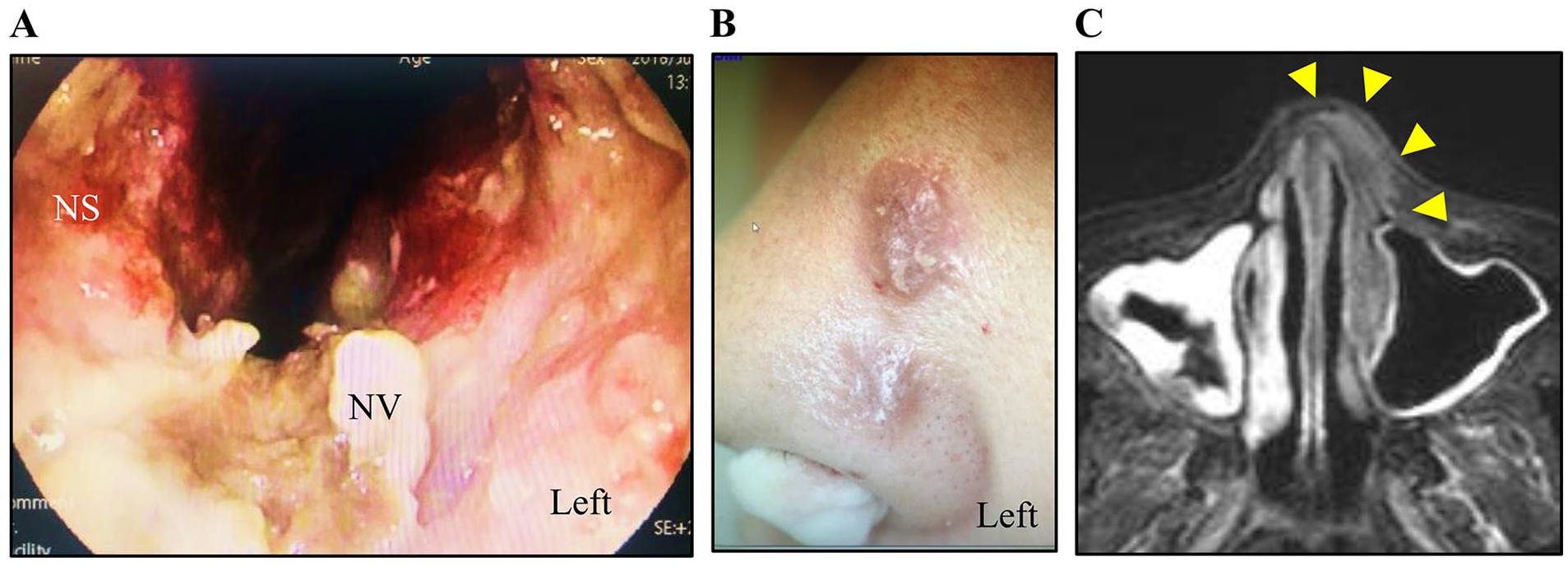
Clinical findings a after 8 months postoperatively. (A) Findings of the left nasal cavity 8 months after surgery; erosions in the nasal vestibulum and inferior turbinate have gradually worsened. (B) Findings of the left dorsum of the nose 8 months after surgery; rash accompanied by pigmentation and protrusion appears. (C) Contrast-enhanced MRI findings 8 months after surgery (Gd-enhanced, horizontal section); it shows inflammatory signals (arrowhead) with a pale shadow is found in the areas from the left nasal vestibulum to the nasal septum, left inferior turbinate, and dorsum of the nose as well as a mild signal abnormality suggestive of inflammation in the right maxillary sinus. MRI, magnetic resonance imaging; NS, nasal septum; NV, nasal vestibule.
Discussion
AFRS and CGIFS occur in immunocompetent, healthy young adults. Two previous reports have described cases of AFRS complications after CGIFS treatment 1 and 2 cases in which patients with AFRS were treated with high-dose steroids and converted to CGIFS. 3 Our patient underwent transplantation for leukemia <20 years ago and discontinued an immunosuppressant <10 years ago. Therefore, no immunodeficiency appeared to be involved in the CGIFS complication. However, she also developed CGIFS contralateral to AFRS. There are two likely causes for AFRS onset and concomitant CGIFS: GVHD and surgery. GVHD causes various damage to organs in the body. Rhinosinusitis frequently occurs in patients with chronic GVHD and is excluded from GVHD diagnostic criteria proposed by the NIH consensus development project, but it frequently occurs in patients with chronic GVHD, and the relapse rate is high. 4 In these patients, super-microstructures of the airway, such as the mucous layer and ciliary epithelium, are impaired after immunosuppression, resulting in mucosal surface degeneration and decreased mucus clearance, 4 being vulnerable to respiratory infections. In patients with GVHD, Th2 cells are activated, and eosinophils, IL4, IL5, and IL13 are increased. 5 Therefore, they experience eosinophilic fasciitis, enteritis, and pneumonia. Herein, type-2 inflammation activation caused by GVHD may be associated with AFRS onset. Although the presence of nasal sinus mucosal damage immediately after transplantation was unknown, the patient experienced tongue granuloma, intravascular fasciitis of the lower extremities, and meningioma. She also presented with scarring findings observed in the epipharynx, indicating systemic inflammation. Therefore, she may have experienced severe rhinosinus mucositis. Six months after surgery, inflammation spread to surgical areas, particularly the nasal septum and left inferior turbinate. Impaired physical barrier function, caused by cartilage and bone defects and mucosal resection, may induce parasitic fungi infiltration. Alarifi et al 1 reported that 3 of 7 patients with CGIFS had a history of ipsilateral ESS. Hence, surgical history is crucial, particularly in patients with fungal sinusitis.
Herein, the causative organism of CGIFS was a fungus of the Dothideomycetes. CGIFS cases are frequently found in dry climatic areas. Aspergillus is detected in many patients with CGIFS. No study has reported the harmful effects of Didymellaceae on human health. In this case, the mucin and fungal ball of the right maxillary sinus were cultured, and A. fumigatus was identified. However, 6 months later, when CGIFS developed, a fungus of the Dothideomycetes was identified from the left inferior turbinate culture, suggesting that right maxillary sinusitis and left rhinosinusitis are inflammatory diseases induced by another causative microorganism. The specimens were sent to the Medical Mycology Research Center using a United Nations-certified container. In addition, the preoperative pathology results showed that the left inferior nasolacrimal mucosa was parasitized by a fungus, but no culture test was performed, and it is unclear whether it was Dothideomycetes.
CGIFS symptoms persist, and initial symptoms mainly consist of nonspecific symptoms such as nasal obstruction, leading to diagnostic delay. CGIFS is frequently found secondary to exophthalmos diagnosis. Sharif et al proposed 3 risk factors for CGIFS: chronic fungal rhinosinusitis (≥12 weeks), diabetes, and residence in rural areas. 6 Herein, erosion developed in the left nasal cavity 4 years before the CGIFS diagnosis, possibly due to fungi invasion. The patient had no history of diabetes but lived in a rural area. Generally, CGIFS treatment, based on the treatment strategy of chronic invasive fungal rhinosinusitis, consists of surgery and administration of antifungal agents. VRCZ is a strong antifungal drug, 1 and the patient responded well. CGIFS prognosis may be more favorable than other types of chronic invasive fungal rhinosinusitis; however, delayed detection of CGIFS may cause intraorbital or intracranial progression. In patients with chronic fungal rhinosinusitis, including AFRS, close attention to their condition is needed to assess CGIFS risk and detect their visual symptoms.
Identifying the causative microorganisms of deep mycosis is challenging due to the vast number of fungal species. Further investigations are needed to understand the relationship between fungi, patients’ conditions, and their characteristics that harm human health.
Footnotes
Acknowledgements
We would like to express my sincere gratitude to specially appointed professor, Katsuhiko Kamei, Medical Mycology Research Center, Chiba University, for his support and assistance. His invaluable feedback and guidance were crucial in developing new insights in this field. We would also like to thank Crimson Interactive Pvt. Ltd. (Ulatus)—www.ulatus.jp for their assistance in article translation and editing. We also would like to thank Enago (![]() ) for English language editing.
) for English language editing.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Institutional Review Board
Ethical approval was obtained from the institutional review board of the Tokai University Hospital along with the approval number (22J007) for this case report. We have obtained a written consent from the patient.
