Abstract
Background:
Sinonasal renal cell-like adenocarcinoma (SNRCLA) is a rare, low-grade malignant neoplasm originating in the nasal cavity and paranasal sinuses. This report provides a detailed account of the clinical and pathological characteristics of a single case of SNRCLA, highlights its differential diagnosis, and includes a review of relevant literature.
Case description:
A 13-year-old male presented with a mass in the right nasal cavity. Histopathological analysis demonstrated tumor morphology analogous to clear cell renal cell carcinoma, characterized by abundant clear cytoplasm, acinar formations, and a prominent capillary-rich stroma. Immunohistochemical analysis revealed positive staining for cytokeratin pan, cytokeratin 7, vimentin, carbonic anhydrase IX, and SRY-box 10, with partial positivity for S-100. Markers such as paired box 8, atriopeptidase, renal cell carcinoma marker, melanosome, thyroglobulin, and thyroid transcription factor-1were negative. Fluorescence in situ hybridization analysis identified the presence of an EWSR1 gene rearrangement. The patient underwent surgical resection followed by adjuvant radiotherapy. Follow-up evaluations conducted as of November 2024 revealed no evidence of tumor recurrence, and the patient remained in good general health.
Conclusion:
SNRCLA is a rare neoplasm characterized by its low incidence rate and the necessity for exclusion-based diagnosis, requiring differentiation from other clear cell tumors. Immunohistochemistry plays a key role in establishing the diagnosis and distinguishing SNRCLA from histologically-similar entities. This case represents the first documentation of EWSR1 gene rearrangement in SNRCLA, thereby contributing novel insights into its molecular profile.
Introduction
Sinonasal renal cell-like adenocarcinoma (SNRCLA) is a rare form of adenocarcinoma originating in the nasal cavity. It is categorized as a distinct subtype of non-intestinal-type adenocarcinoma of the nasal cavity and paranasal sinuses, necessitating differentiation from other clear cell subtypes originating in these regions, as well as from metastatic tumors according to the 2017 WHO classification of head and neck tumors. 1 This study presents a case of SNRCLA in an adolescent and includes a review of the relevant literature to analyze its clinicopathological features, immunophenotype, molecular changes, and biological behavior. The objective was to enhance an understanding of this uncommon tumor, thereby minimizing the risk of misdiagnosis or inappropriate treatment.
Case Description
A 13-year-old male patient was hospitalized for recurrent bleeding from the right nasal cavity for 2 weeks, with no identifiable cause. The condition was accompanied by intermittent nasal congestion and a reduced sense of smell, without associated symptoms such as nasal or facial pain, dizziness, fever, or rhinorrhea. A contrast-enhanced computed tomography scan revealed a soft tissue mass in the right nasal cavity, measu-ring approximately 38 mm × 19 mm, with significant heterogeneous enhancement and evidence of bone thinning in the ethmoid sinus and nasal septum due to absorption (as depicted in Figure 1). The lesion was primarily considered to be a benign space-occupying mass, and differential diagnoses included hemangioma and other conditions. No additional space-occupying lesions were identified. Endoscopic sinus tumor resection was performed, during which the mass was noted to have an ulcerated surface, a tendency to bleed, and poor demarcation from the surrounding tissues. This study was conducted with approval from the hospital (Approval Number: 2024-GN011).
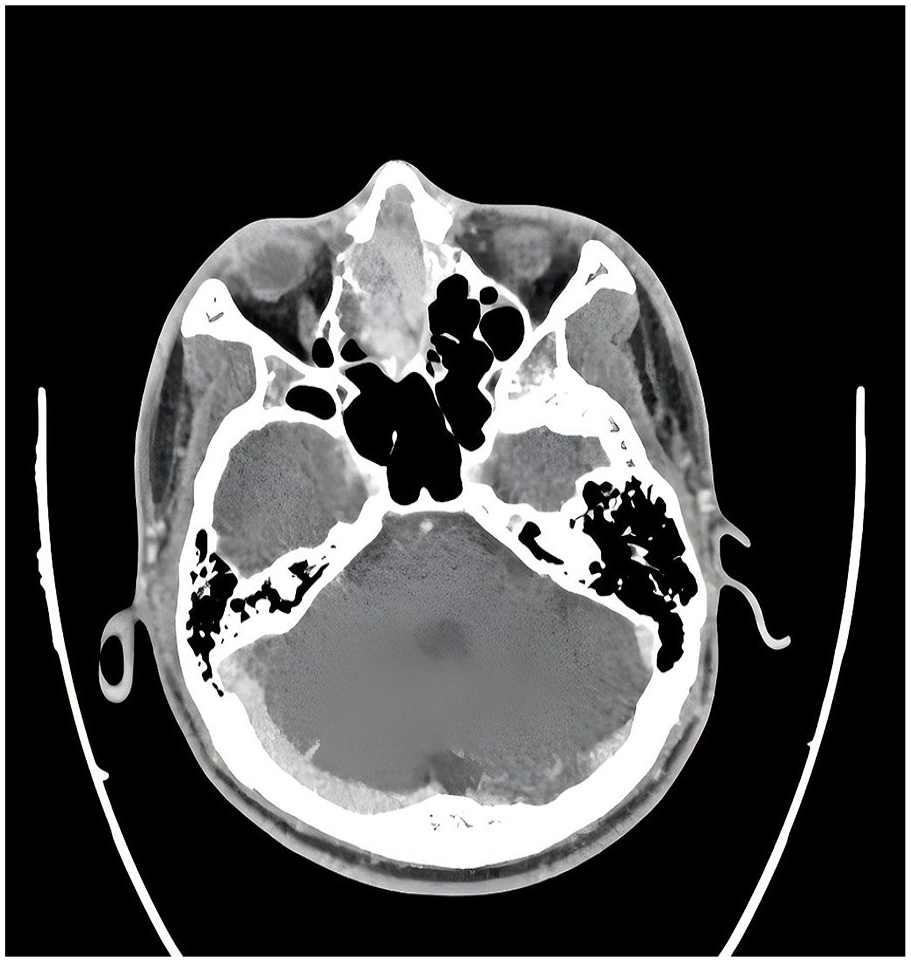
CT scan demonstrating a mass in the right nasal cavity. CT, computed tomography.
Pathological Examination
Macroscopic Findings
The specimen consisted of multiple grayish-white and gray-brown tissue fragments, measuring approximately 5 × 3.5 × 2 cm in total. The cut surfaces were solid, firm in texture, and displayed grayish-white and grayish-red coloration.
Microscopic Findings
Histologically, the tumor tissue was arranged in small nests and acinar formations, with occasional acinar dilation containing minimal eosinophilic secretions resembling thyroid colloid (as depicted in Figure 2). Tumor cells exhibited abundant clear cytoplasm, mildly-atypical nuclei, and an absence of significant pathological mitotic figures (as depicted in Figure 3). The stroma was abundant in capillaries, with areas of hemorrhage but no evidence of necrosis.
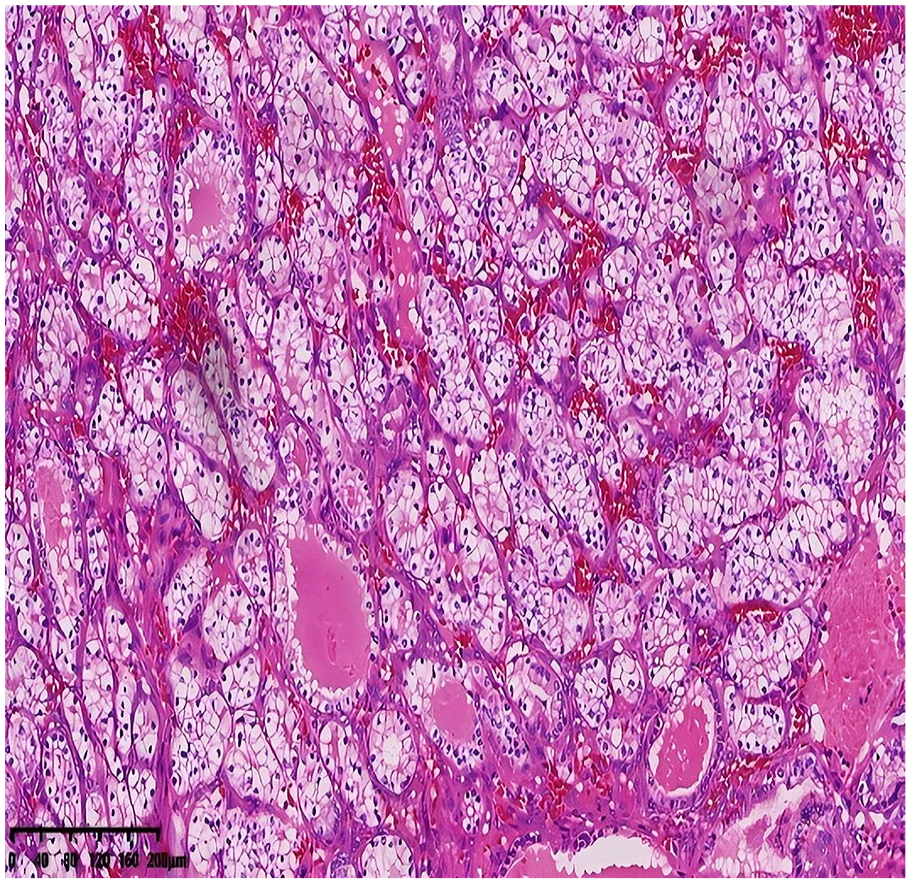
Histological image revealing tumor tissue with a solid acinar structure, including individual acini containing pink secretions (H&E staining, ×100 magnification).
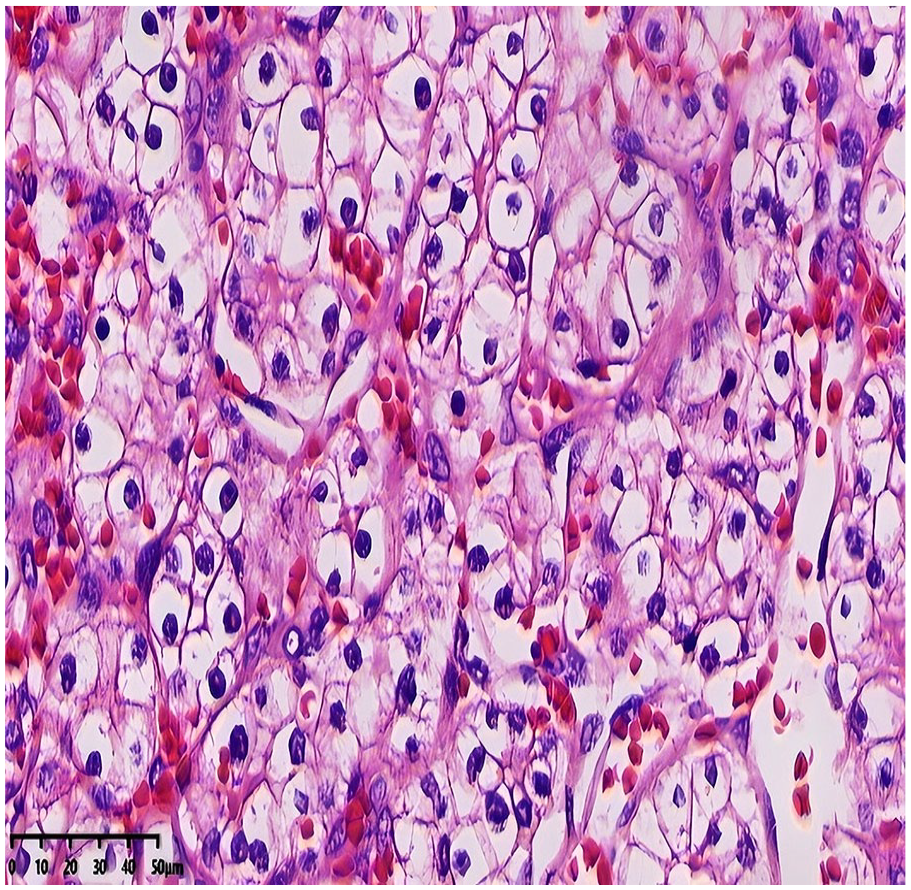
High-magnification histological view displaying low-grade nuclei, with no evidence of necrosis or mitotic figures (H&E staining, ×400 magnification).
Immunohistochemical Findings
Tumor cells demonstrated positivity for cytokeratin pan (CKpan), cytokeratin 7 (CK7), SRY-box 10 (SOX10) (Figure 4), and vimentin, with carbonic anhydrase IX (CAIX) showing diffuse membranous positivity (as depicted in Figure 5). Partial positivity was noted for S-100, while melanosome (HMB45), melanin A (Melan-A), calponin, DOG-1, p40, p63, paired box 8 (PAX8), renal cell carcinoma (RCC), atriopeptidase (CD10), thyroglobulin (TG), and thyroid transcription factor-1 (TTF-1) were negative. The Ki-67 proliferation index was approximately 10%.
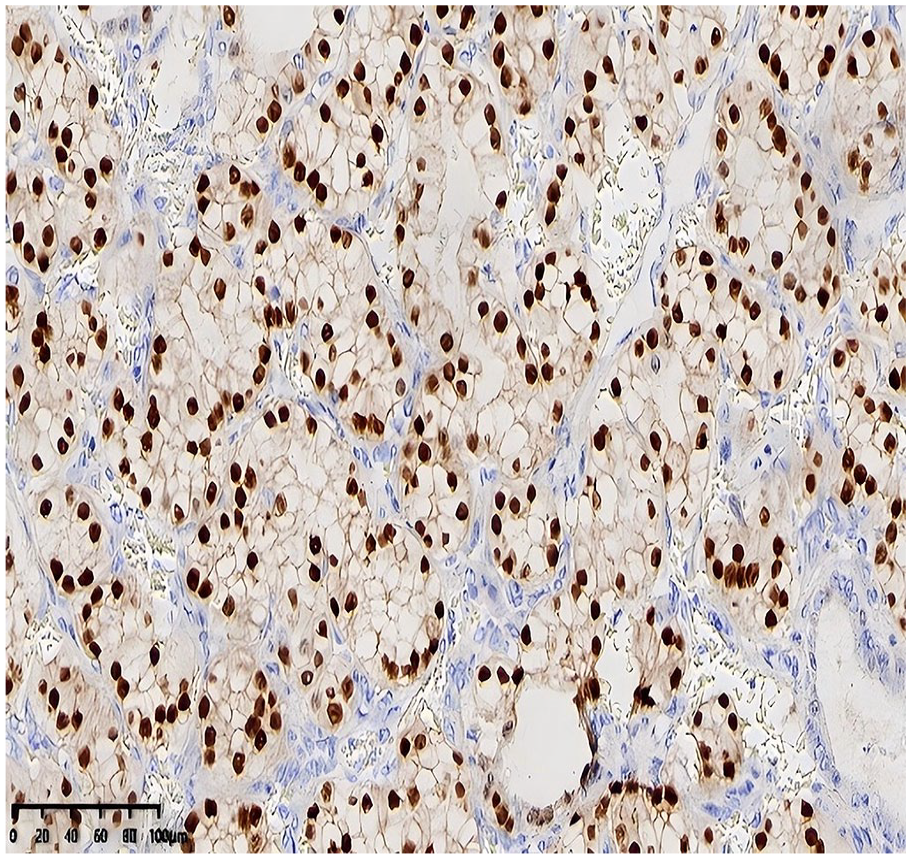
Immunohistochemical staining demonstrating nuclear positivity for SOX10, detected using the Enhance Labeled Polymer System (EnVision method). SOX10, SRY-box 10.
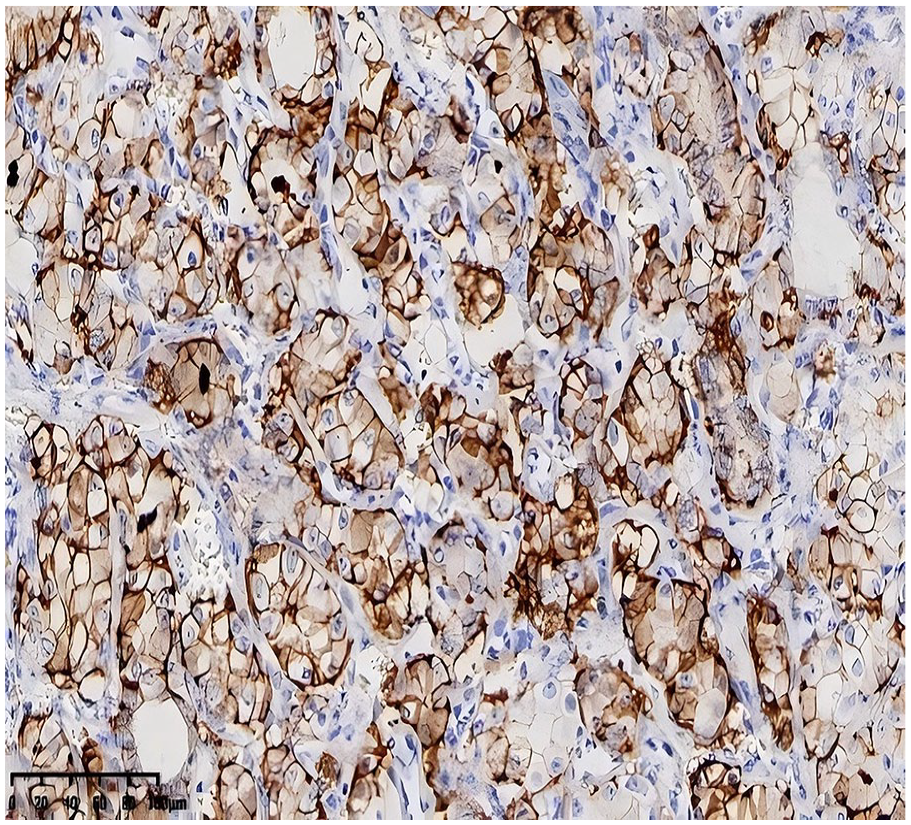
Membranous positivity for CAIX observed on immunohistochemistry, detected using the EnVision method. CAIX, carbonic anhydrase IX.
Molecular Findings
Fluorescence in situ hybridization (FISH) testing revealed positivity for Ewing sarcoma breakpoint region 1 (EWSR1) gene rearrangement (as depicted in Figure 6) and negativity for Epstein-Barr virus-encoded RNA (EBER).
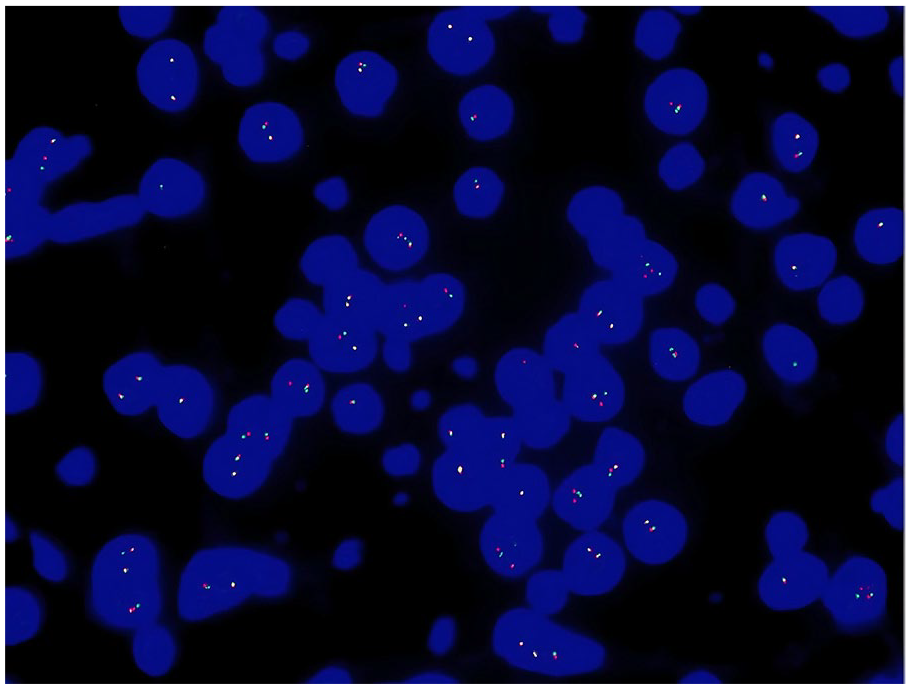
EWSR1 (22q12) gene rearrangement, with a positive proportion accounting for 27% (threshold for positivity >10%). EWSR1, Ewing sarcoma breakpoint region 1.
Pathological Diagnosis
Primary SNRCLA associated with EWSR1 gene rearra-ngement.
Treatment and Follow-Up
The patient underwent surgical intervention followed by 28 sessions of radiotherapy. At the 14 month follow-up in November 2024, no evidence of recurrence or metastasis was observed, and the patient remained in good general health.
Discussion
Adenocarcinomas of the nasal cavity, sinuses, and skull base are categorized into salivary gland-type adenocarcinomas and non-salivary gland-type adenocarcinomas based on the 2017 edition of the WHO Classification of Head and Neck Tumors. The latter group is further divided into intestinal-type adenocarcinomas (ITACs) and non-ITACs, with non-ITACs classified into low-grade and high-grade subtypes. SNRCLA is recognized as a distinct subtype of low-grade non-ITACs. 2 The descriptive term “renal cell-like” was first introduced by Zur et al in 2002. 3 Currently, only case reports are available in the literature, both domestically and internationally, and the etiology, clinical progression, optimal treatment, and prognosis of the tumor remain inadequately defined.4-6 Additionally, it has been reported that SNRCLA may occur in association with von Hippel-Lindau syndrome.7,8
A summary by Tokarz et al included 15 cases (9 females and 6 males) with an age range of 22 to 89 years. 6 The primary clinical manifestations were epistaxis and nasal congestion. Similarly, Yuan et al summarized 5 cases (1 female and 4 males), with an age range of 29 to 52 years, noting local recurrence in 3 cases following surgical resection. 5 The patient in this case, aged 13 years, represents the youngest reported to date.
Histological Features
Microscopically, SNRCLA exhibits characteristics reminiscent of low-grade clear cell RCC. The tumor is composed of solid, acinar, microcystic, or papillary formations, with tumor cells appearing polygonal and of larger volume. The cytoplasm is abundant and transparent, with a low nuclear-to-cytoplasmic ratio, indistinct nucleoli, infrequent mitoses, and areas of hemorrhage. Necrosis and neural invasion are typically absent. 4
Immunohistochemical Profile
Tumor cells often express markers associated with nasal mucosal epithelium, including DOG-1, SOX10, and CAIX, as well as strong positivity for CKpan and CK7. Some cases express S-100 and vimentin. Negative staining has been reported for markers such as HCK, p63, PAX8, RCC, CD10, SMA, and TTF-1.
Genetic Findings
As of right now, no specific genetic changes have been consistently associated with SNRCLA. Wu et al reported negative results for EWSR1-ATF1 rearrangements via FISH testing. 9 Yuan et al found no EWSR1 or ETS Variant Gene 6 (ETV6) rearrangements in 5 cases. 5 Notably, this case represents the first report of EWSR1 gene rearrangement identified through FISH testing.
The EWSR1 gene, located on chromosome 22q12, was first identified in Ewing’s sarcoma. It encodes a multifunctional protein that participates in various cellular processes, including gene expression, meiosis, mitosis, cell signaling, DNA repair, and cellular senescence. Rearrangements involving the EWSR1 gene, which result in fusion with other genes, have been implicated in the development of numerous tumor types, such as Ewing’s sarcoma, clear cell sarcoma of soft tissue, desmoplastic small round cell tumor, and soft tissue myoepithelioma. 10 SNRCLA with EWSR1 gene rearrangement has not been previously reported.
SNRCLA must be distinguished from the following entities:
Salivary gland clear cell carcinoma: The tumor typically exhibits nested, sheet-like, or cord-like patterns, often surrounded by hyalinized stroma, with frequent bone and nerve invasion. Tumor cells are polygonal with clear or eosinophilic cytoplasm, and mitotic figures are rare.
Immunohistochemistry: Positive for CKpan, P63, and P40, indicative of squamous differentiation. It often harbors the EWSR1-ATF1 fusion gene.
Distinction: SNRCLA does not express P63 or P40, differentiating it from CCC.
Myoepithelial Carcinoma
Morphology
The tumor presents as multinodular, with solid or reticular patterns, areas of necrosis, and diverse tumor cell morphologies. Cellular atypia, mitotic activity, and frequent squamous metaplasia are observed.
Immunohistochemistry
Positive for CKpan, CK5/6, P63, S-100, calponin, SMA, and glial fibrillary acidic protein, with Ki-67 proliferation index often exceeding 10%.
Distinction
SNRCLA lacks significant cellular atypia and frequent mitotic figures, and does not express CK5/6, P63, or other markers associated with myoepithelial carcinoma.
Metastatic Clear Cell RCC
Morphology
This tumor closely resembles SNRCLA in histological appearance. In cases of suspected adult SNRCLA, metastatic RCC must first be excluded, as 6% to 15% of RCC cases can metastasize to the head and neck. 11
Imaging and immunohistochemistry
RCC is positive for RCC marker, CD10, and PAX8, whereas SNRCLA does not express these markers.
Metastatic Papillary Thyroid Carcinoma
Relevance in pediatric cases
Papillary thyroid carcinoma (PTC) is the most common endocrine malignancy in children and adolescents, and in younger populations, it is often more invasive and severe.12,13
Morphology
Due to the presence of thyroid follicle-like structures, metastatic PTC must be excluded in pediatric patients.
Immunohistochemistry and molecular markers
PTC expresses TG, TTF-1, and PAX8 and often exhibits the BRAF V600E gene mutation. These markers are absent in SNRCLA. This case of SNRCLA demonstrates unique molecular features, including EWSR1 gene rearrangement, which further distinguishes it from other clear cell tumors in the nasal cavity and underscores the importance of comprehensive pathological evaluation in its diagnosis.
Conclusion
In summary, this report presents a rare case of SNRCLA with EWSR1 gene rearrangement. The clinical and pathological features, as well as immunohistochemical findings, align with the established diagnostic criteria for SNRCLA. This case broadens the understanding of SNRCLA by expanding its known age range and molecular characteristics. These findings help in contributing valuable data to support pathological diagnosis, inform clinical management strategies, and guide follow-up care for this rare tumor.
Footnotes
Authors’ Contributions
M.T.: Formal Analysis, Writing—original draft. H.Y.: Investigation, Writing—original draft. C.Y.: Data curation and Funding acquisition. P.-J.L.: Conceptualization, Formal Analysis. X.-T.Y.: Conceptualization, Writing—review and editing. All authors read and approved the final draft.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Yunnan Digestive Endoscopy Clinical Medical Center Foundation for Health Commission of Yunnan Province (Grant Number: 2022LCZXKF-XH10). The funding body had no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Ethics Approval and Consent to Participate
This study was conducted with approval from the Ethics Committee of The First People’s Hospital of Yunnan Province (Approval Number: 2024-GN011). This study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from the participant’s legal guardians.
Consent for Publication
The patient’s guardians signed a document of informed consent.
