Abstract
Nasal hemangiomas most commonly occur in the anterior part of the nose, including the Little’s area of the nasal septum, the anterior tip of the inferior nasal turbinate, and the vestibule. They rarely develop in the posterior part of the inferior nasal turbinate and meatus. Here, we report a case of cavernous hemangioma at the posterior aspect of the left inferior meatus in a 79-year-old man who presented with epistaxis and progressive nasal obstruction for 4 months after blowing his nose forcefully and review the relevant literature. An endoscopic evaluation revealed a dark red mass in the posterior part of the left inferior meatus that did not extend to the posterior choana or nasopharynx. The tumor was removed with endoscopic surgery under general anesthesia. No recurrence was observed during a 2-year follow-up. Our experience suggests that forceful nose blowing is a traumatic stimulus that can lead to hemangiomas arising in the posterior nasal cavity in rare instances.
Introduction
Nasal hemangiomas mostly occur anteriorly on the nasal septum and inferior turbinate and very rarely develop posteriorly.1-3 They can present with symptoms, including unilateral nasal obstruction and epistaxis, progress relatively rapidly, and exhibit an expanding pattern. Therefore, if nasal hemangiomas are large or occur posteriorly, a careful approach is required because differentiation from malignant diseases is necessary, and they pose a risk of a large amount of bleeding. Here, we describe the case of a patient who had progressive nasal obstruction accompanied by epistaxis after forceful nose blowing 4 months before the presentation. A mass, which was subsequently histologically confirmed to be a cavernous hemangioma, was observed posterior to the inferior meatus. The patient was treated with endoscopic surgery. To the best of our knowledge, this is the first report of a nasal hemangioma of this etiology, developing in a very rare location.
Case Presentation
A 79-year-old man visited our hospital with chief complaints of left-sided nasal obstruction that had started 4 months before the presentation. At that time, he had blown his nose forcefully and experienced epistaxis along with yellowish mucus discharge. Subsequently, the bleeding on the left side stopped; however, it frequently manifested as blood-tinged nasal discharge and nasal obstruction occurred, which reportedly worsened.
The patient’s medical history was as follows: He had a previous diagnosis of high blood pressure, for which he was taking medication; he is currently controlling it through lifestyle changes and regular exercise. In addition, he was taking antiplatelet agents for carotid calcification to prevent stroke, as prescribed by a neurologist. The patient had undergone bilateral Caldwell-Luc surgery and partial resection of the left inferior turbinate 20 years prior and bilateral endoscopic revision sinus surgery 8 years prior. Furthermore, he had undergone ventilation tube insertion several times because of otitis media with effusion of the left ear. At that time, a perforation of the left tympanic membrane was noted.
Endoscopic findings revealed a dark red mass encompassing the posterior part of the left inferior meatus that did not extend to the posterior choana or nasopharynx. The tumor surface was covered with blood clots, and necrotic tissue was observed (Figure 1).
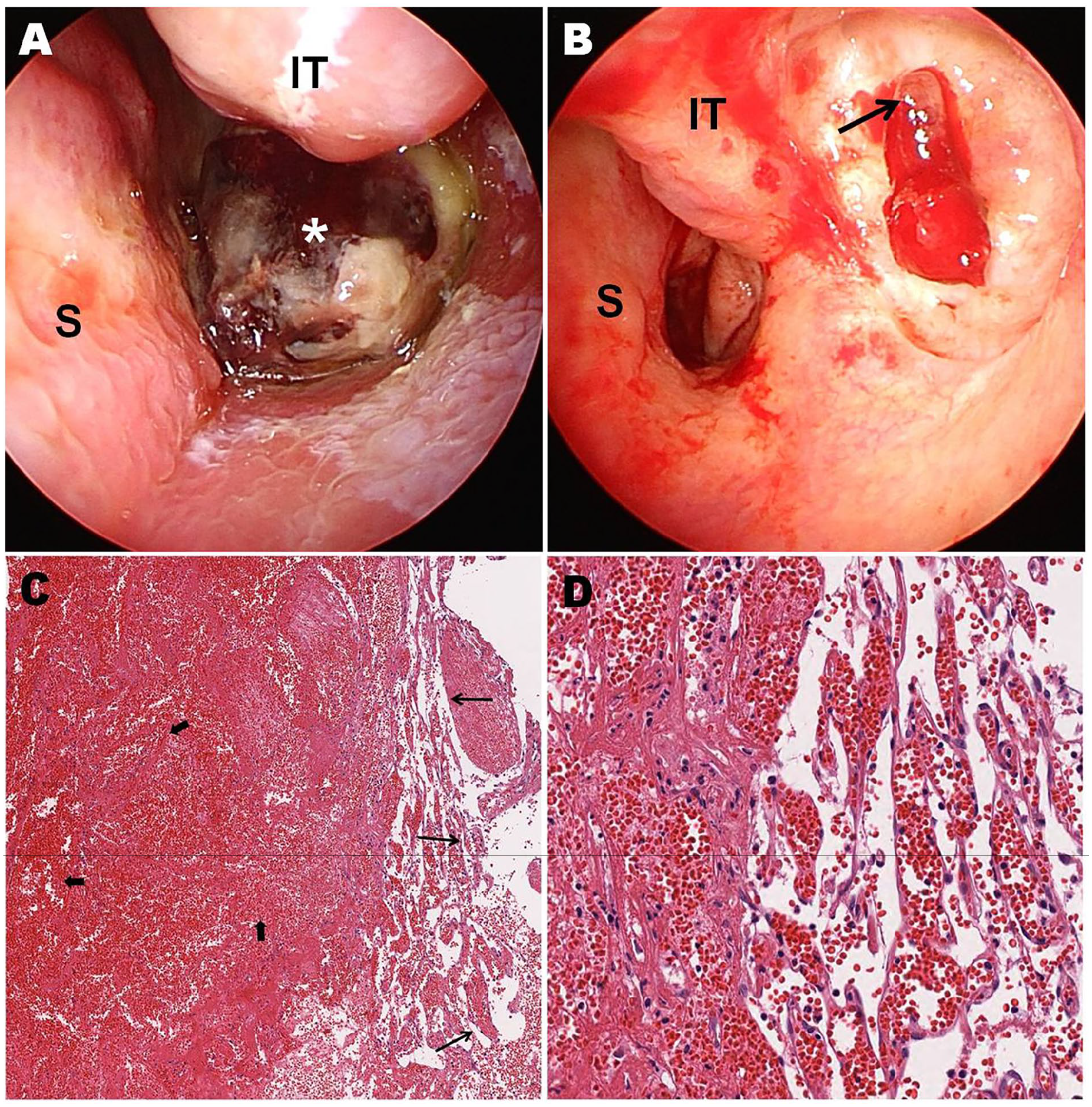
Endoscopic images. (A) A dark red polypoid mass lesion (asterisk) partially covered with necrotic material blocking the inferior meatus of the left nasal cavity. (B) The origin of the tumor (arrow) arising from the posterior lateral wall of the inferior meatus after the removal of the mass (S: nasal septum, IT: inferior turbinate partially resected from previous surgery). Histopathologic images show dilated thin-walled blood vessels with cavernous vascular space (thin arrows) and marked degenerative collection of collapsed dilated blood vessels (thick arrows) in the background of diffuse hemorrhagic tissue, consistent with cavernous hemangioma [hematoxylin and eosin stain, (C) 70× magnification, (D) 100× magnification].
Imaging evaluation using computed tomography (CT) and magnetic resonance imaging (MRI) was performed (Figure 2). CT showed a 2.5 cm × 2.7 cm × 2.5 cm mass that appeared to have originated in the posterior third of the left inferior turbinate, with an expanding appearance, sclerosis, and destruction of the surrounding bone. A comparison with past images revealed that surrounding bony changes, including a defect in the lateral wall of the left nasal cavity, existed even before the revision endoscopic surgery 8 years prior. MRI showed an intermediate-intensity signal from the lesion with a hyperintense peripheral rim on T1-weighted images. On T2-weighted imaging, the mass revealed heterogeneous signal intensity and a low-intensity signal in the outer part of the lesion. Post-contrast MR images showed central area enhancement of the tumor.
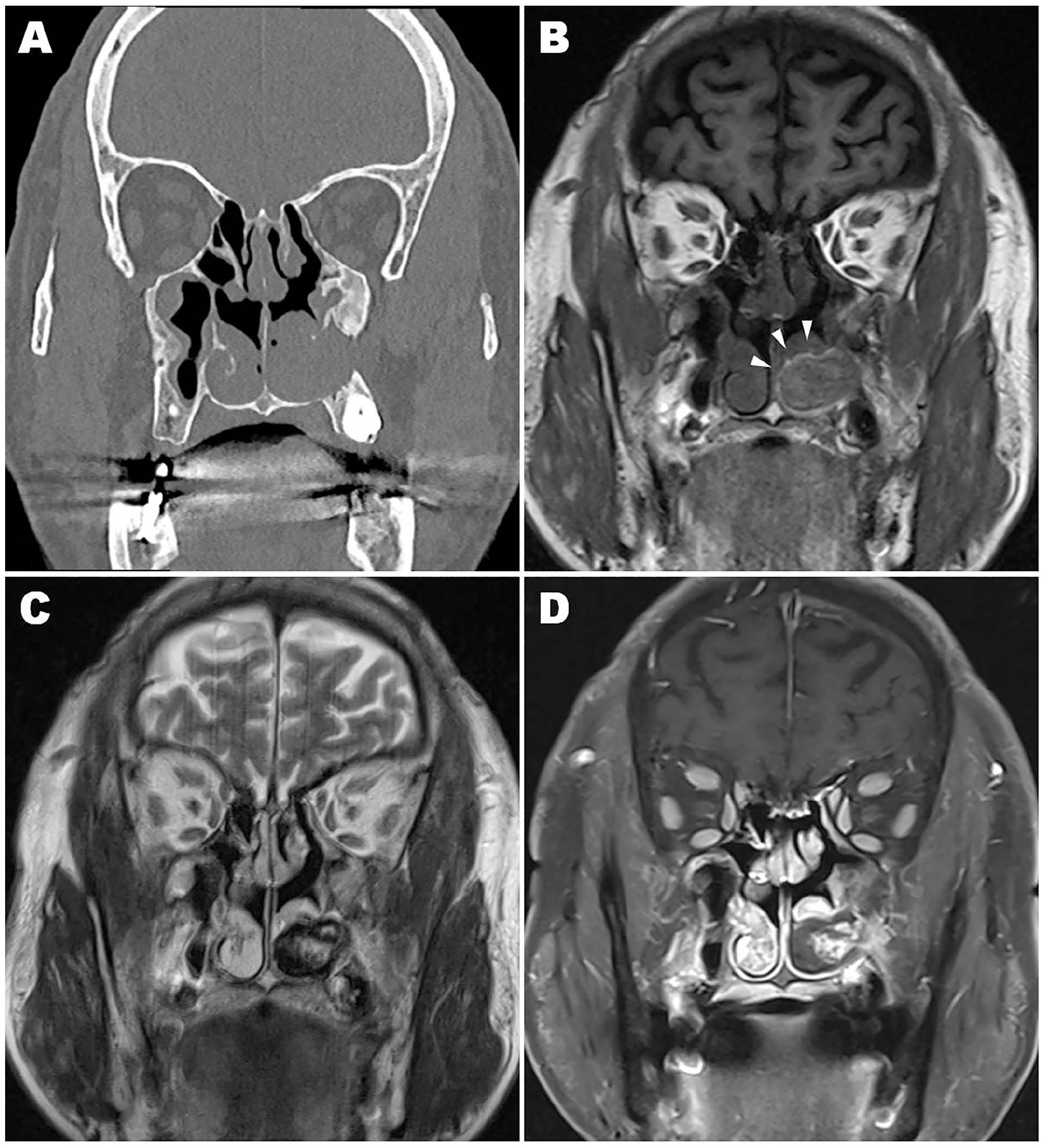
Radiologic images. (A) Coronal computed tomography shows a lesion blocking the inferior meatus of the left nasal cavity. The lateral wall defect was present even before the revision surgery 8 years prior. (B) T1-weighted magnetic resonance images show an intermediate-intensity tumor with a high signal intensity peripheral rim (arrowheads). (C) T2-weighted magnetic resonance images show a heterogeneous tumor with a high signal intensity in the central area and hypo-intensity in the peripheral area. (D) Post-contrast magnetic resonance images show a central enhancement.
Endoscopic surgery was performed under general anesthesia. The mass was not connected to the nasal septum and originated from the lateral wall of the nasal cavity. The mass was carefully removed to avoid unexpected or abrupt bleeding; no adhesion to the surrounding area was observed during removal, and almost no bleeding occurred. The origin of the tumor was the lateral wall of the posterior portion of the inferior meatus (Figure 1B). After removal, hemostasis was performed using bipolar electrocautery.
Histopathological examination revealed dilated thin-walled blood vessels with cavernous vascular space and a marked degenerative collection of collapsed dilated blood vessels in the background of diffuse hemorrhagic tissue, consistent with cavernous hemangioma (Figure 1C and D).
The patient had an uneventful postoperative course; he is currently being followed up for 2 years without any recurrence.
Discussion
The pathogenesis of hemangiomas remains unclear. However, traumatic/inflammatory irritation, hormonal stimuli, underlying vascular malformations, viral oncogenes, angiogenic growth factors, and hemodynamic instability have been suggested as possible etiologies.4-6
In the nasal cavity, hemangiomas are mostly observed in the anterior part, including Little’s area of the nasal septum, the anterior end of the inferior turbinate, and the vestibule. The anterior part of the nasal cavity is thought to be rich in vascular supply and is more exposed to habitual picking and nasal packing, making it more susceptible to irritation. Hemangiomas rarely occur in the posterior nasal cavity.7,8
Histologically, hemangiomas are classified mainly based on the size of the dominant blood vessel.1-3 Capillary hemangioma, which is the most common, is characterized by capillary-sized vessels, submucosal vascular proliferation, and capillary lobules. Cavernous hemangiomas show dilated blood-filled vascular channels and larger endothelial-lined vascular spaces. Mixed hemangiomas show a mixture of the aforementioned findings. No differences are observed in clinical manifestations according to histological classification.
Reports of hemangiomas arising in the posterior third of the inferior turbinate and meatus are rare.3-15 We thoroughly searched the PubMed database for related studies and identified 14 cases of hemangiomas arising in the posterior third of the inferior turbinate and meatus. Of these cases, 3 were related to previous surgeries or treatments, 3 were considered to be related to pregnancy, and 7 were idiopathic. The main symptoms in almost all patients were nasal obstruction and epistaxis, and the disease usually persisted for several months. Histologically, they were primarily capillary hemangiomas (Table 1).
Reported Cases of Hemangiomas Arising from the Posterior Area of the Inferior Turbinate and Meatus.
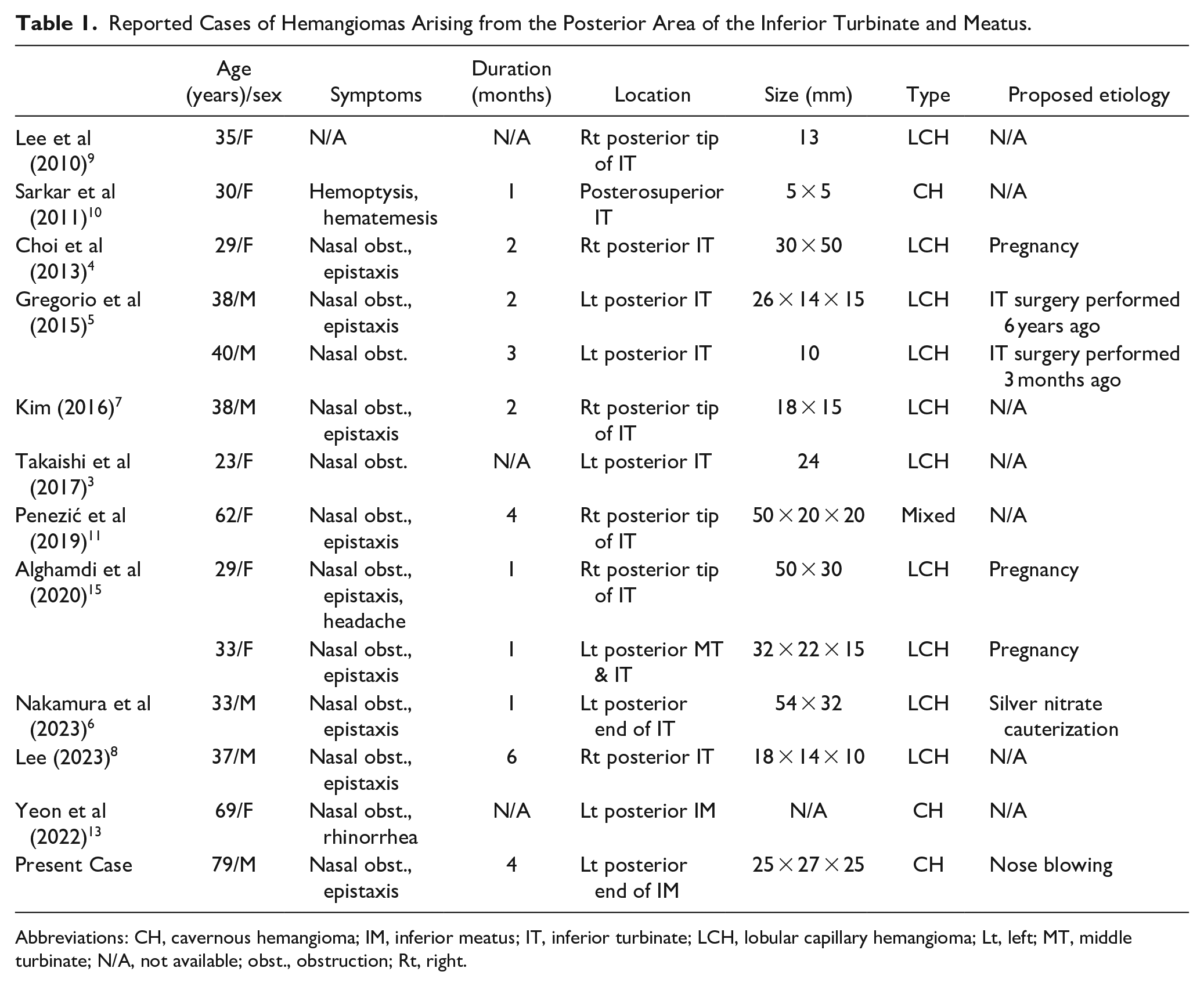
Abbreviations: CH, cavernous hemangioma; IM, inferior meatus; IT, inferior turbinate; LCH, lobular capillary hemangioma; Lt, left; MT, middle turbinate; N/A, not available; obst., obstruction; Rt, right.
Here, the patient clearly remembered that he experienced a nosebleed when he blew his nose forcefully 4 months before the presentation. Blood-tinged nasal discharge frequently occurred afterward; in addition, nasal congestion occurred, which gradually worsened thereafter. Hemangiomas are known to develop in the anterior part of the nose not only due to the rich vascular supply but also because of habitual nose picking or frequent nasal packing. Our case shows that forceful nose-blowing may act as a traumatic stimulus capable of activating the development of hemangiomas in the posterior aspect of the nose. However, it remains unclear whether nose blowing caused the development of the hemangioma or stimulated a preexisting asymptomatic hemangioma that had occurred due to other causes, including previous surgery.
In addition, the patient’s previous diagnosis of hypertension, which is reportedly associated with the development of hemangiomas, and the use of antiplatelet medications (UCLID tablets, Yuyu Pharma Inc.; ticlopidine hydrochloride 250 mg; Ginkgo biloba dried extract 80 mg) may have contributed to the development of the hemangioma.1,4
The appearance of hemangiomas in the nasal cavity varies depending on their histological type, the degree of hemorrhage, fibrosis, or neovascularization.1,6,14 In CT, capillary hemangiomas characteristically appear as well-defined masses without internal calcification and with homogeneous enhancement. They appear as low or isointense signal intensity lesions on T1-weighted images and as lesions with mixed signals in the center surrounded by low-intensity signals in the periphery, which histologically corresponds to thrombi, on T2-weighted images. In CT, cavernous hemangiomas appear as larger and heterogeneous masses, and enhancement can exhibit a centripetal or multifocal pattern. The reported appearance of cavernous hemangiomas varies slightly among studies. In some reports, it appeared as homogeneous isointense masses without enhancement on T1-weighted images and as slightly high signal intensity masses on T2-weighted images. In other reports, it appeared as masses with high signal intensity with central enhancement on T1-weighted images and as masses with low signal intensity on T2-weighted images.
Here, on T1-weighted images, the lesion had an intermediate signal intensity, with a high signal intensity peripheral rim and central enhancement. On T2-weighted images, the tumor showed heterogeneous signal intensity, with low and high signal intensity in the outer part and center, respectively.
Conclusion
We endoscopically treated a cavernous hemangioma arising from the posterior lateral wall of the left inferior meatus in a 79-year-old man who visited our hospital because of epistaxis and progressive nasal obstruction after forceful nose blowing. The patient was followed up for 2 years without any signs of recurrence. This unique etiology and location of the hemangioma has not been previously reported. Our experience indicates that forceful nose blowing—similar to habitual nose picking, which is considered a cause of anterior nasal hemangioma—is a traumatic stimulus that can lead to the development of hemangiomas in the posterior nasal cavity in rare instances.
Footnotes
Authors’ Contributions
Y.W.L.: conceptualization, investigation, writing – original draft. J.O.K.: data management & curation, writing – original draft. J.C.: supervision, validation, writing – reviewing & editing, final approval of the manuscript.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a VHS Medical Center Research Grant, Republic of Korea (grant number: VHSMC 22012).
Ethical Approval
This study was approved by the institutional review board of Veterans Health Service Daejeon Hospital.
Statement of Informed Consent
Written informed consent was obtained from the patient for publication of this report and any accompanying images.
