Abstract
According to the latest classification of vascular lesions, cavernous hemangioma is considered a venous or arteriovenous malformation. The literature has not described the association between vascular malformation of the middle turbinate and angiofibroma of the nasal septum. A 26-year-old female patient with profuse left-sided bleeding and nasal obstruction is presented. She was bleeding from a vascular malformation of the left middle turbinate, and from an angiofibroma, attached to the posterior edge of the nasal septum. The patient also had hypoplasia of the left maxillary sinus and a lack of the septum in the sphenoid sinus. Despite the embolization of the supplier, the patient lost 1.5 L of blood. Although necessary, preoperative embolization is frequently insufficient to prevent significant blood loss during the removal of vascular lesions of the nasal cavity. Vascular malformations of the sinonasal region should be considered in cases of frequent and profuse nosebleeds in patients with hypertrophy of the middle turbinate associated with anatomical variations.
Introduction
Bleeding from the nasal cavity can be a traumatic experience for both the patient and the physician. Among the main causes of nasal bleeding are vascular anomalies, which can be divided into vascular tumors and vascular malformations.1-3 Among benign tumors originating from blood vessels, hemangioma occupies a special place. Hemangioma, comprising about 20% of benign tumors of the nasal cavity and paranasal sinuses, was previously classified into capillary, cavernous, and mixed.1-3 Although capillary hemangioma is a relatively-rare lesion in the nasal cavity, numerous case series have different localizations.1,3,4 According to the current classification, the lesion previously presented as a cavernous hemangioma no longer exists as a separate clinical entity, but is considered a venous or arteriovenous malformation.5-7 This is a very rare lesion in the nasal cavity and sinuses, mainly localized in the maxillary sinus, and much less often on the nasal septum, olfactory cleft, and the nasal turbinates.5-13 An especially-rare vascular malformation is formerly known as “intraosseous cavernous hemangioma.”10-13 The middle turbinate is a very rare primary localization of this lesion, and so far, only three cases have been described in the English literature.11-13 The association of vascular malformation of the middle turbinate with angiofibroma of the nasal septum has not been described in the literature. We present the case of a female patient with life-threatening nosebleeds that were the result of this association.
Case Report
A 26-year-old female patient came for an outpatient examination due to occasional profuse bleeding from the left side of the nasal cavity and difficulty breathing through the nose. The bleeding first appeared a year and a half ago and became more frequent and abundant over time. She was treated for perennial allergic rhinitis, and the laboratory findings, except for mild anemia, were normal. In the medical record, there was no information about hereditary diseases, genetic abnormalities, or similar symptoms in the family. The hematological profile, apart from anemia, was normal. Bleeding and coagulation times were normal, as were blood coagulation factor concentrations. The patient was not taking any medication that affected blood clotting.
Endoscopic examination showed a very hypertrophic left middle turbinate, which pushes the nasal septum to the right side. When touching the mucous membrane with the endoscope, there was scanty bleeding from dilated blood vessels. In the posterior part of the left nasal cavity, a dark purple soft-tissue mass was observed, attached to the back edge of the nasal septum and protruding into the nasopharynx. A computed tomography (CT) scan with contrast injection showed a very enlarged left middle turbinate with a rarefied honeycomb-like bone mass as well as a soft-tissue mass about 10 mm in diameter, which partially fills the left half of the nasopharynx and leads to its asymmetry (Figure 1a and b). Hypoplasia of the left maxillary sinus and septal deviation to the right side were noticeable, as well as the lack of a septum in the oval-shaped sphenoid sinus. Contrast binding was very intensive on the area of the middle turbinate and the mass filling the nasopharynx (Figure 1a and b). After admission for hospital treatment, a digital subtraction angiography (DSA) was performed. The findings indicated the presence of two vascular lesions in the projection of the nasopharynx and the left middle turbinate, supplied mostly from the internal maxillary artery, while the anterior part of the middle turbinate received vascularization from the ophthalmic artery (Figure 2a).
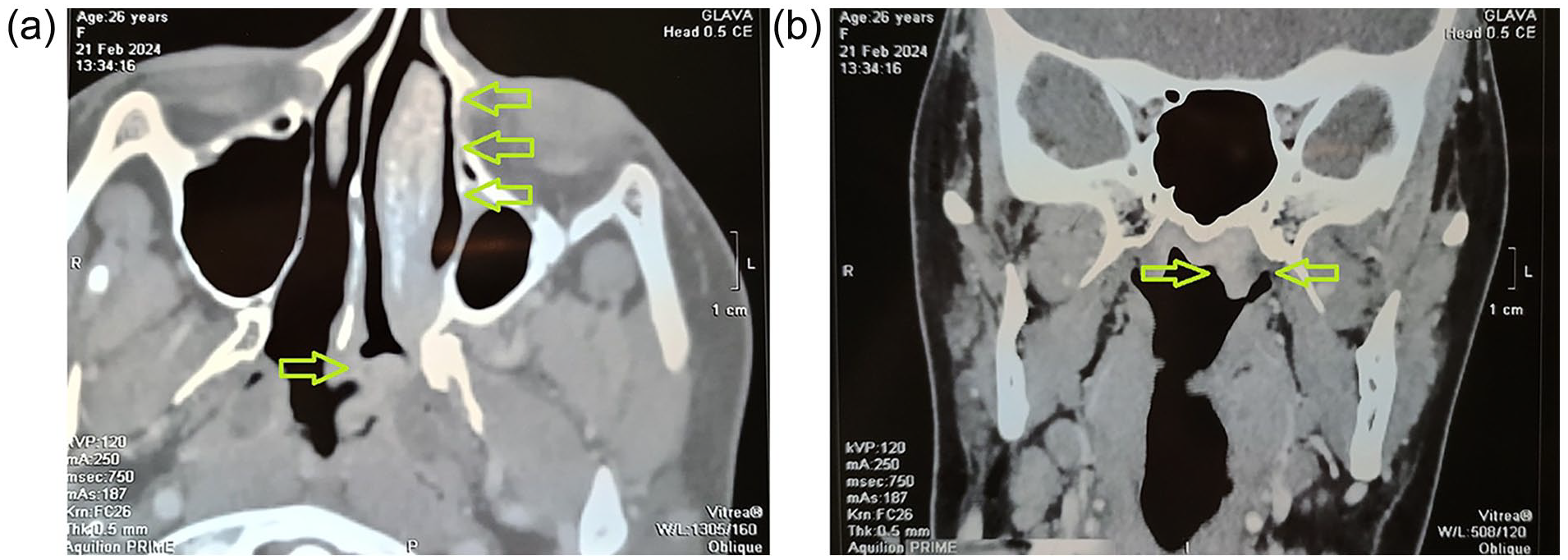
(a) Axial plane CT scan of the paranasal sinuses with contrast injection showing a very enlarged left middle turbinate with a rarefied honeycomb-like bone mass (yellow arrowheads) as well as a soft-tissue mass about 10 mm in diameter, attached to the posterior edge of the nasal septum (yellow arrowhead), which partially fills the left half of the nasopharynx and leads to its asymmetry. Note the hypoplasia of the left maxillary sinus and septal deviation to the right side. (b) The coronal plane of the CT scan shows a soft-tissue mass that partially fills the left half of the nasopharynx (yellow arrowheads) and leads to its asymmetry. Note the lack of a septum in the oval-shaped sphenoid sinus.
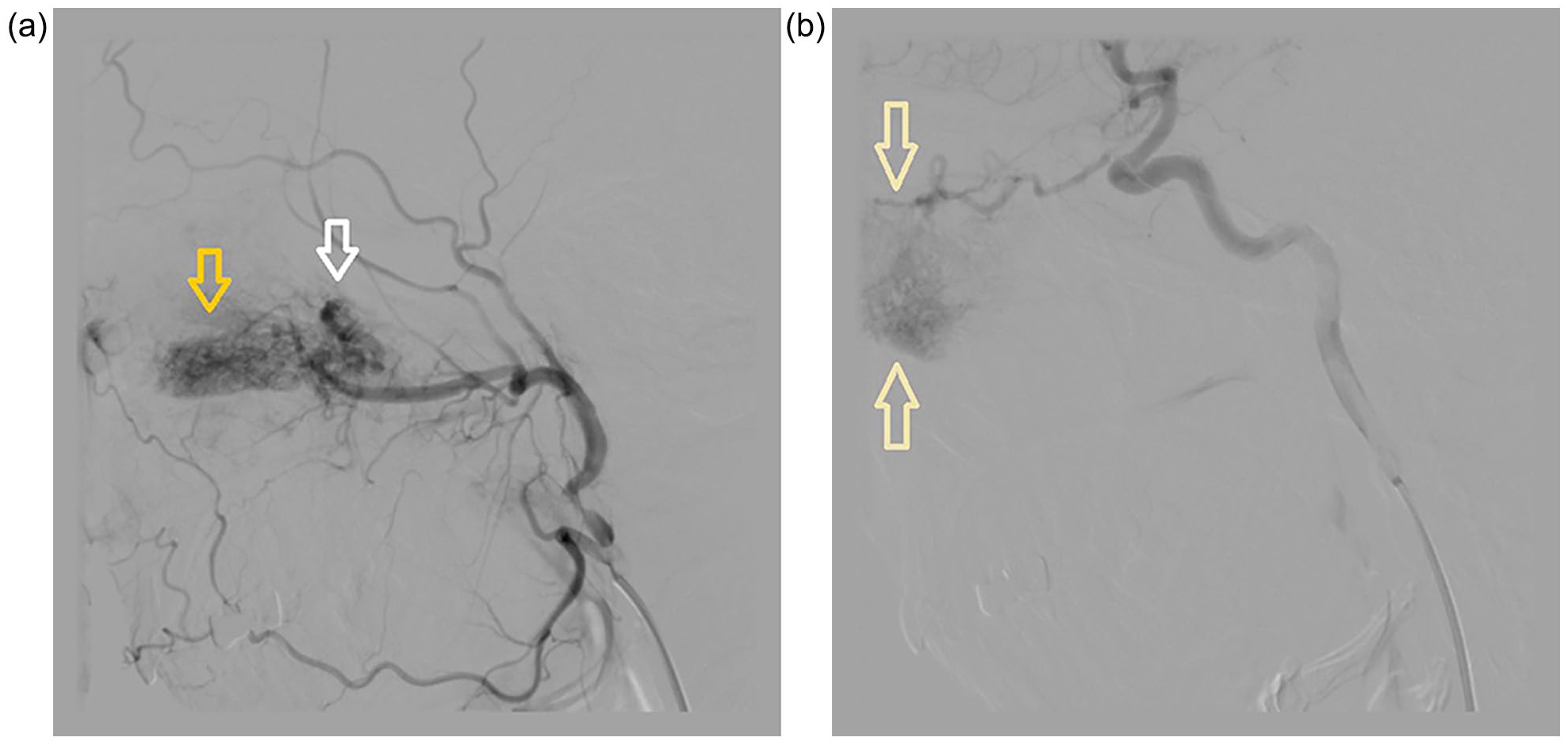
(a) A digital subtraction angiography (DSA) showing the presence of vascular lesions in the projection of the nasopharynx (white arrowhead) and the left middle turbinate (yellow arrowhead), supplied mostly from the internal maxillary artery, while anterior part of the middle turbinate received vascularization from the ophthalmic artery. (b) After the embolization of lesions vascularized from the left maxillary artery, the circulation was reduced in about 90% of the vascularization area of the internal maxillary artery, but about 10% of the vascularization remained from the ophthalmic artery in the anterior part of the middle turbinate (yellow arrowheads).
After the DSA, an embolization of lesions vascularized from the left maxillary artery was performed. The radiologist estimated that in this way the circulation was reduced in about 90% of the vascularization area of the internal maxillary artery, but he noted that about 10% of the vascularization remained from the ophthalmic artery (Figure 2b). Despite the certain risk of bleeding, the next day, endoscopic resection of the left middle turbinate and the mass from the nasopharynx was performed under general anesthesia. This was accompanied by profuse bleeding, mainly from the front-upper parts of the left nasal cavity. The bleeding was stopped with a bipolar electrode, and then, the upper parts of the nasal cavity were firmly covered with gel foam. The left nasal cavity is tamponed with cotton gauze soaked in Vaseline ointment. It was estimated that the patient lost about 1.5 L of blood during the intervention. So, blood and fluid replacement was performed during the following days. In addition, she received tranexamic acid in a dose of 1.0 g and antibiotic cefuroxime in a dose of 1.5 g every 12 h. On the fifth day after surgery, the nasal packing was removed and the patient was discharged home in good general condition, without nasal bleeding.
Microscopically, the lesion in the middle turbinate consisted of highly-dilated vascular spaces interspersed with bony substances (Figure 3a). There were mostly-venous spaces lined with thinned endothelium without cellular atypia, although arterioles and arterial capillaries were also observed (Figure 3b). Histology pointed to the presence of an arteriovenous malformation but with absolute venous domination. The analysis of the mass from the nasopharynx indicated the presence of irregular highly-dilated blood vessels that disturbed smooth muscle organization, without elastic fibers and a bizarre appearance, immersed in a dense fibrous stroma (Figure 3c). The finding was suggestive of an angiofibroma, although we performed an immunohistochemical (IHC) analysis to make a differential diagnosis. Human anti-CD34 antibodies (Elabscience, Houston, Texas, USA) were used to detect CD34 molecules and IHC staining was highly positive for CD34, suggesting the diagnosis of angiofibroma (Figure 3d). Accordingly, we checked the patient’s endocrine status by measuring the blood concentrations of sex hormones and it was normal.
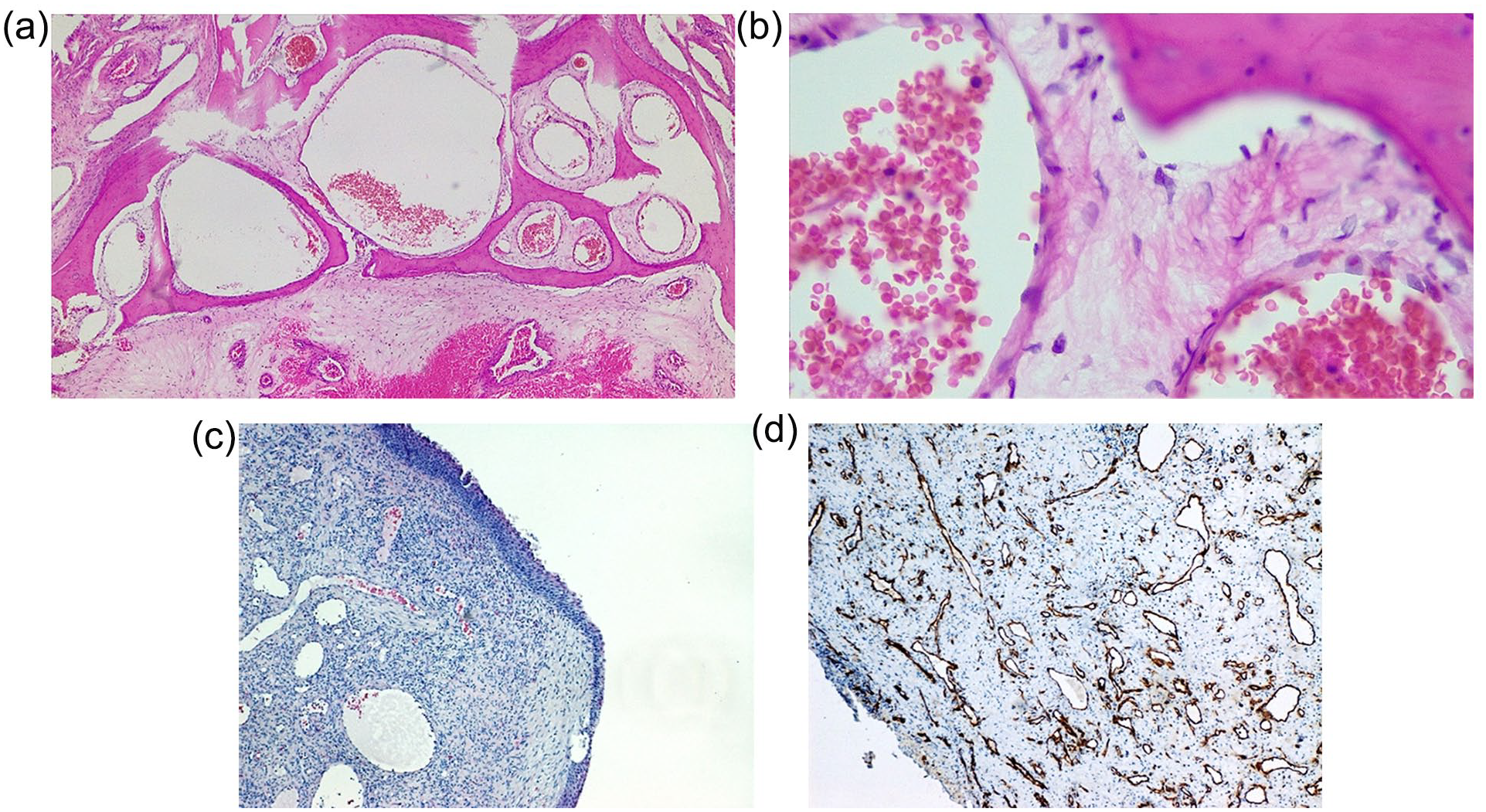
(a) Microscopically, the lesion in the middle turbinate consists of highly-dilated vascular spaces interspersed with bony substances (hematoxylin & eosin staining, 200× magnification). (b) There are mostly-venous spaces lined with thinned endothelium without cellular atypia (hematoxylin & eosin staining, 400× magnification). (c) Microphotograph of the mass from the nasopharynx showing the presence of irregular highly-dilated blood vessels that disturbed smooth muscle organization, without elastic fibers and a bizarre appearance, immersed in a dense fibrous stroma (hematoxylin & eosin staining, 200× magnification). (d) Immunohistochemical staining is highly positive for CD34, suggesting the diagnosis of angiofibroma (200× magnification).
Two months after the surgical treatment, the magnetic resonance imaging showed the absence of the left middle turbinate and nasopharyngeal lesion (Figure 4). Eight months after the surgery, the patient feels well and denies profuse nosebleeds.
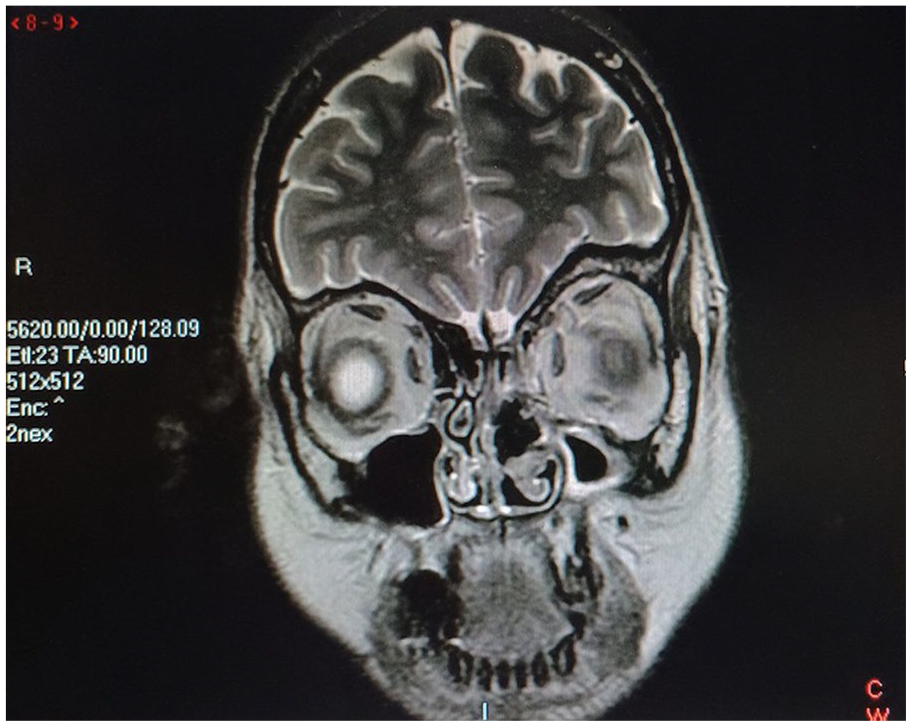
Postoperatively, the magnetic resonance imaging showed the absence of the left middle turbinate and nasopharyngeal lesion.
Discussion
The case we have just described clearly shows that detailed diagnostics and preoperative embolization in some cases are not enough to prevent intense bleeding from the surgical field. During the surgical intervention, the vascular lesion bled intensively from the branches that supplied the lesion from the ophthalmic artery. To prevent endocranial and ophthalmologic complications, embolization of branches of the internal carotid artery is avoided by radiologists. However, even the remaining 10% of vascularization was enough for the patient to lose a large amount of blood during the intervention. Based on the DSA findings and histological analysis, we detected an arteriovenous malformation in the region of the middle turbinate. Based on the DSA findings, the detailed, elongated and often tortuous arteries that feed the lesion are shown in the most detail, and venous drainage through dilated veins is detected.
Previous CT image descriptions of vascular malformation affecting bone substance, including those in the nasal septum and nasal turbinate, suggest honeycomb-like bony matrix enhancement.8-13 Pathohistological analyses showed the presence of thin-walled venous or mixed anastomosing vascular channels between bony trabeculae.8-13 The lesions described in the nasal cavity are, as in the case of our patient, covered by a healthy mucosal lining. Such a lesion was previously described as a “cavernous hemangioma”. However, a large part of the professional and scientific public accepted the fact that cavernous hemangioma does not exist as a separate clinical entity. The International Society for the Study of Vascular Anomalies (ISSVA) has proposed a classification that divides vascular anomalies into tumors (benign, borderline, and malignant) from vascular malformations.5-7 Malformations can be divided into simple (capillary, venous, lymphatic, arteriovenous), combined, of major named vessels, and associated with other anomalies.5-7 According to that classification, the lesion previously known as “cavernous hemangioma” is not a tumor, but a non-infiltrating, predominantly-venous malformation. However, the bizarreness of our case emphasizes the association with anatomical variations of the sinonasal region and another vascular lesion, which protruded into the nasopharynx and was histologically diagnosed as an angiofibroma. Marked pushing of the nasal septum to the right side, hypoplasia of the left maxillary sinus, and agenesis of the septum within the sphenoid sinus could be explained by the extreme spread of the vascular lesion within the middle turbinate. That lesion disrupted the normal vascularization of the bony walls of the paranasal sinuses and led to disturbances in their development. The lesion diagnosed as angiofibroma was separated from the left middle turbinate and attached to the posterior margin of the nasal septum. Before surgery, it was embolized and removed endoscopically without significant bleeding. Angiofibroma occurs mainly in male adolescents, although it also affects older age groups and women. Although it is classified as a benign vascular tumor, its origin remains controversial.14,15 While some link it to genetic disorders due to the deletion of chromosome 17, including the p53 gene suppressor region and the Her-1/neu oncogene, others consider it a hormone-dependent tumor.14,15 However, there is a part of the scientific public that considers it as a vascular malformation rather than a tumor. Discontinuity of the vascular basement membrane, focal lack of pericytes, irregularity in the structure of the muscle layer of blood vessels, and high level of immunoexpression of laminin α2 in endothelial cells indicate that the blood vessels are in an early-developmental stage and immature. 14 Angiofibroma with primary extranasopharyngeal localization is a separate form of this lesion. 16 It mainly appears on the mucous membrane of nasal cavity structures: nasal septum, inferior, and middle turbinate. 16 It occurs more often in the female population than nasopharyngeal angiofibroma. 16 The intensity of bleeding is usually lower than the angiofibroma of the nasopharynx and, in addition to standard diagnostics, IHC analysis is also recommended. 16 The high level of CD34 expression in the endothelium of blood vessels of angiofibroma determines it more as a vascular malformation. 14 However, the level of immunoreactivity for CD34 is much higher in angiofibroma than in vascular malformation, which could be useful in the differential diagnosis between these two types of lesions. 15 Some vascular malformations and tumors develop in isolation, whereas others develop within the phenotype of a syndrome, including Von-Hippel Lindau, Sturge Weber, Kippel–Trenaunay–Weber, Sneddon, and Rendu–Osler–Weber (Hereditary Hemorrhagic Teleangiectasia) syndrome. 17 However, there are no reports in the literature describing the synchronous presentation of arteriovenous malformation of the middle turbinate and angiofibroma arising from the nasal cavity structures.
Conclusion
This is a unique case of association of vascular malformation of the middle turbinate and angiofibroma with attachment at the posterior edge of the nasal septum. Detailed diagnostics and preoperative embolization are necessary, but frequently insufficient to prevent significant blood loss during the intervention. Vascular malformations of the sinonasal region should be considered in cases of frequent and profuse nosebleeds in patients with hypertrophy of the nasal turbinate associated with other anatomical variations of the sinonasal region.
Footnotes
Acknowledgements
This study was conducted as a part of a scientific project of the Faculty of Medicine of the Military Medical Academy, University of Defence, Belgrade, Serbia (MFVMA02/23-25/).
Authorship
U.G., A.J., B.V.Đ, J.S., M.L., and A.P. provided direction and guidance throughout the preparation of the manuscript. U.G., J.S., M.L., and A.P. performed the literature search and wrote the original manuscript. J.S. and A.P. provided constructive suggestions and significantly revised the manuscript. U.G. and A.P. collected clinical samples. All authors read and approved the final manuscript.
Data Availability Statement
Data will be made available on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient Consent and Ethical Statement
The studies involving human participants were reviewed and approved by the Ethics Committee of our institution (Approval number 21/2022). The patient provided written informed consent to participate in this study. Pathological samples were acquired during the operation.
