Abstract
Keywords
Introduction
Human papillomavirus (HPV)’s oncogenic role is constantly scrutinized in head and neck cancers. Despite the presence of HPV markers in up to 25% of sinonasal tumors, 1 the significance of HPV in these cancers remains unclear.2-4 The diagnosis of HPV-related multiphenotypic sinonasal carcinoma (HMSC), as first described by Bishop et al in 2013, adds to this complexity. 5 Patients typically present with large tumors causing progressive nasal obstruction, a reflection of the indolent growth of HMSC.6-8 Differentiating HMSC pathologically is challenging due to its shared histological features with the solid and cribriform variant of adenoid cystic carcinomas (AdCC) and basaloid squamous cell carcinomas (B-SCC).9-11 Furthermore, literature on HMSC’s preoperative radiological assessment and surgical management remains scarce. In this case series, we present 3 cases of HMSC that were successfully managed through endoscopic surgical approaches. The objective of the study herein is to describe the unique characteristics and endoscopic surgical treatment of this rare tumor.
Cases
The HMSC cases were retrieved from the pathological database of our center, which collects information on histological diagnosis and date of surgery from all patients treated at the CHU de Québec—Université Laval. Ethical approval was obtained from the Research Ethics Committee of the CHU de Québec-Université Laval for case series studies (Project number: 2024-7354). All patients were individually met and gave their written consent to participate in the study.
Patient 1
A 72-year-old male presented in our clinic with a 6 month history of nasal obstruction, epistaxis, and weight loss. The patient was an active smoker. On physical examination, he had a polypoid nasal mass obstructing both nasal vestibules and a bulging right nasal bone with intact overlying skin. Both vision and extraocular movements were normal. A computed tomography (CT) scan showed a massive bilateral nasal cavity lesion (Figure 1a-c). The vomer was completely eroded, medial maxillary walls were lateralized, and the cribriform plate was indissociable from the tumor. The mass was closely related to the right internal carotid artery in the sphenoid sinus. Magnetic resonance imaging (MRI) revealed a lobulated mass measuring 8.9 cm × 6.4 cm × 8.7 cm without intracranial, palatal, nasopharyngeal, carotid, or perineural invasion. The tumor was slightly hyperintense on T2 and hypointense on T1 sequences with moderate gadolinium enhancement. Preoperative positron emission tomography (PET) scan showed no regional or distant disease.
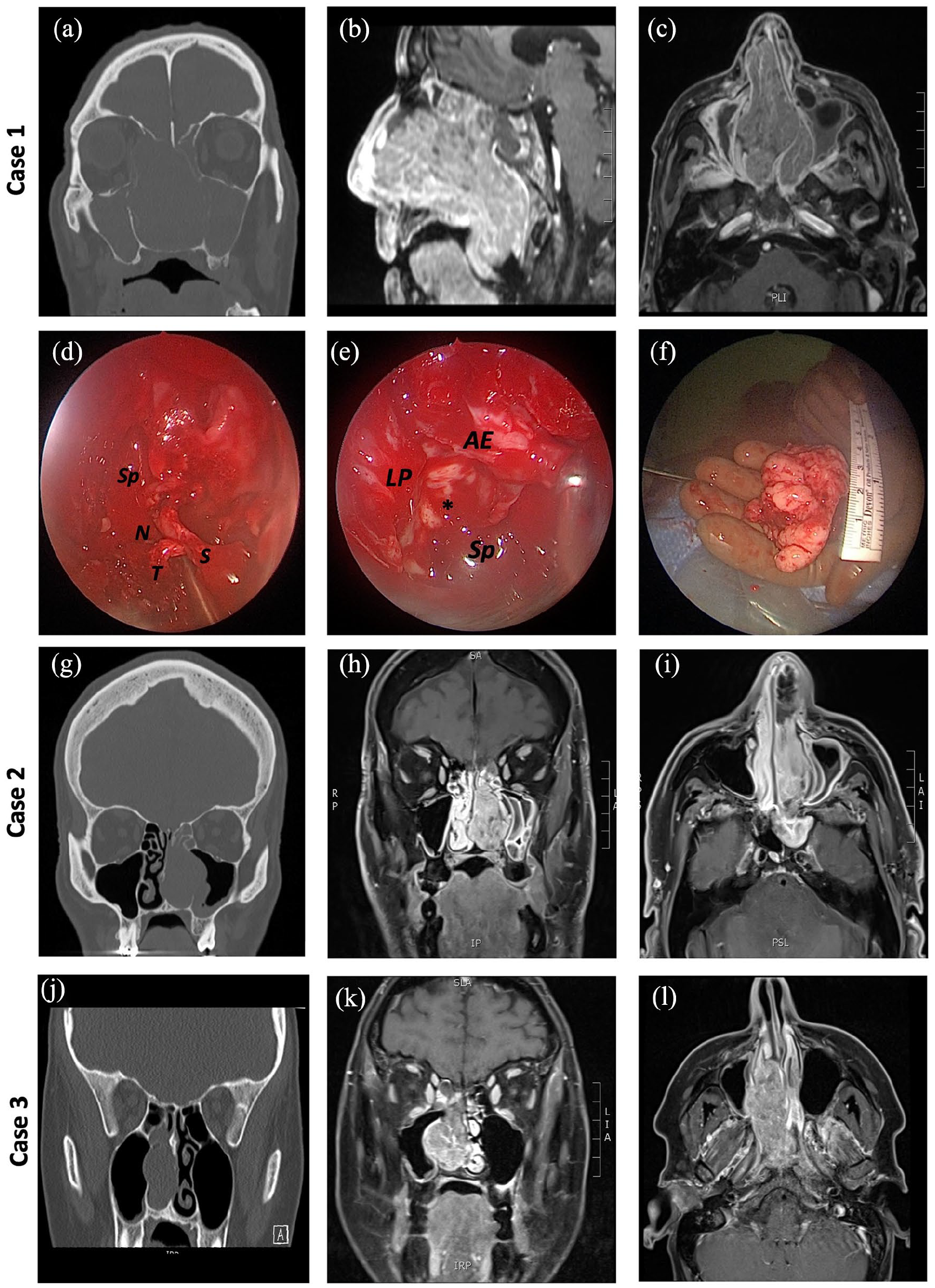
Radiological imaging and surgical pictures. Case 1 (a) Coronal CT imaging of the invasive tumor, (b) sagittal and (c) axial T1 nasopharyngeal MRI with gadolinium and fat saturation, (d) posterior septal attachment of the tumor during surgery, (e) right lamina papyracea (LP), anterior ethmoidal artery (AE), and optic-carotid recess (*). Septum (S), sphenoid sinus (Sp), tumor (T), nasopharynx (N), (f) nasopharyngeal part of the tumor, removed. Case 2 (g) coronal CT imaging, (h) coronal and (i) axial T1 nasopharyngeal MRI with gadolinium and fat saturation. Case 3 (j) coronal CT imaging, (k) coronal and (l) axial T1 nasopharyngeal MRI with gadolinium and fat saturation. CT, computed tomography; MRI, magnetic resonance imaging.
In-office incisional biopsy suggested an HMSC, with focally positive SOX-10 immunostaining and HPV-18 DNA confirmation with polymerase chain reaction (PCR). The DNA analysis was negative for other HPV subtypes (types 16, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68). The tumor was staged cT4N0M0 and a multidisciplinary tumor board committee recommended surgery plus adjuvant radiotherapy. Consent was obtained with the patient for a combined endoscopic and possible open approach.
The surgery proceeded with bilateral endoscopic tumor debulking. Intraoperatively, intranasal structures were markedly thinned, but all the anterior nasal cavity mucosa remained exempt from tumor invasion. Ethmoid cells were depressed by the tumor. The right optic canal and internal carotid artery were exposed in the sphenoid sinus (Figure 1d-f). Although the lesion seemed extensively infiltrative on preoperative imaging, all anatomical boundaries were maintained. The mass originated from the posterior septum and a posterior septectomy was performed. Multiple mucosal margins were sampled peripheral to this region. Once completely detached, the tumor was removed via the oropharynx. All intraoperative frozen section margins were negative. The dehiscence of the posterior cribriform plate and sphenoid roof was repaired with a multiple layer technique.
HMSC diagnosis with vascular invasion was confirmed in the final pathology report (Figure 2a). Histopathology showed areas with squamous differentiation and others with a biphasic luminal-abluminal AdCC-like aspect. Immunohistochemistry (IHC) was diffusely positive for p16 in both components, and p40, p63, CK7, CD117, and smooth muscle actin highlighted the biphasic luminal-abluminal aspect in AdCC-like areas. Adjuvant postoperative radiotherapy began 10 weeks after surgery since the patient required pre-irradiation dental extractions. The total 60 Gy treatment was not completed due to a left retinal detachment after 27 Gy. Both endoscopic and radiological follow-up, at 9 and 7 months postoperatively, respectively, showed no signs of residual or recurrent tumor.
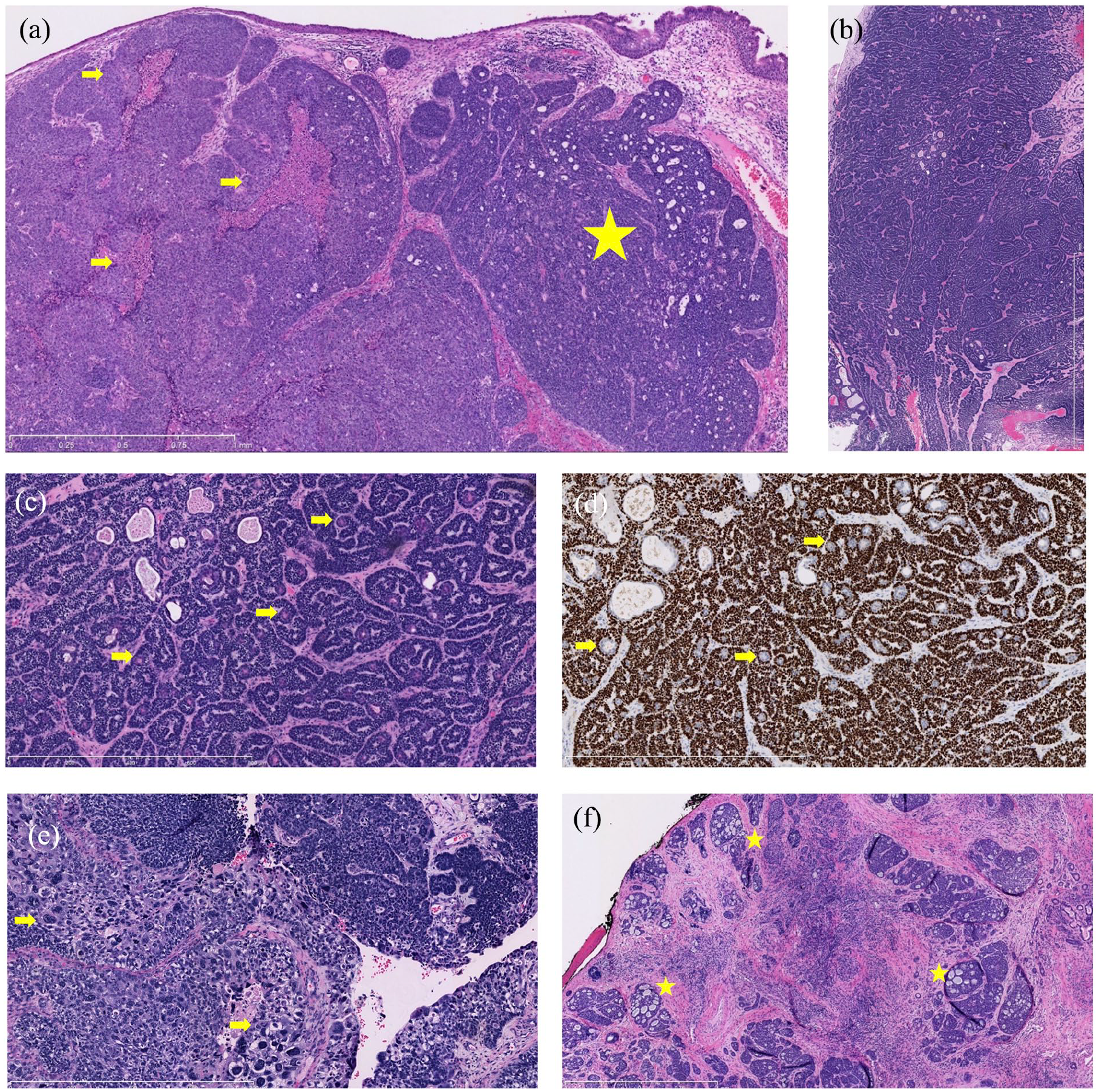
Histologic sections of the 3 tumors. Case 1 (a) Tumor showing a basaloid adenoid cystic carcinoma-like portion on the right side with lumen and pseudo-cribriform aspect (star) and a nonkeratinizing squamous cell carcinoma aspect with pinkish areas of necrosis (bracket) on the left side revealing its multiphenotypic nature (5×). Case 2 (b) (0.5×) and (c) (10×) Lesion almost entirely composed of an adenoid cystic carcinoma-like aspect with basaloid cells and pink eosinophilic luminal cell components, (d) that are negative on the p40 immunostain (arrows; 10×). SOX10 positivity (not shown) does not favor salivary gland tumor as it is also positive in HMSC. Case 3 (e) Original biopsy signed out as poorly differentiated squamous cell carcinoma with a basaloid squamous cell carcinoma component. The highly atypical cells seen (arrow, lower portion) are described in cases of HMSC (10×), (f) the resection specimen showed a pseudo-cribriform adenoid cystic carcinoma-like morphology more developed than in the biopsy (stars; 5×). HMSC, human papillomavirus-related multiphenotypic sinonasal carcinoma.
Patient 2
A 65-year-old male was referred for a 3 month history of nasal obstruction and recurrent left epistaxis. The patient was known to have dyslipidemia and second-hand exposure to tobacco smoke. Nasal endoscopy showed a left friable polypoid mass. CT imaging confirmed a 6.5 cm × 2.5 cm × 8 cm left nasal cavity mass, with a displacement of the nasal septum and extension into the posterior ethmoid cells. On MRI, the mass appeared isointense on T1 with homogeneous gadolinium enhancement and was of intermediate bright intensity on T2. There was no orbital, intracranial, or perineural invasion (Figure 1g-i). No distant metastases were identified on PET. An AdCC was suspected after analysis of an in-office incisional biopsy. The lesion was clinically staged as a T2N0M0 tumor. The tumor board committee recommended surgery with adjuvant radiation therapy.
The patient underwent endoscopic tumor removal including left maxillectomy, ethmoidectomy, sphenoidotomy, as well as a posterior septectomy. The tumor was pedicled on the posterior edges of both inferior and middle turbinates and the mucosa surrounding the sphenopalatine foramen. Careful dissection was performed to remove all the tumor’s attachments. Margins surrounding the lesion were sampled.
Final pathology showed HMSC without perineural, vascular, or osseous invasion and clear margins (Figure 2b-d). Histopathology was AdCC-like showing basaloid cells with a dual population of luminal and abluminal cells highlighted by IHC positive for CD117, Epithelial Membrane Antigen (EMA), Carcinoembryonic Antigen (CEA), and Cytokeratin 7 in luminal epithelial cells and S-100, p40, p63, CD10, and Cytokeratin 5/6 in abluminal myoepithelial cells with focal positivity for Glial Fibrillary Acidic Protein (GFAP) and Muscle-specific actin. The morphology and IHC results suggested AdCC. However, p16 was strongly and diffusely positive and PCR probes testing (Cobas 4800 HPV test, Roche Diagnostics, Switzerland) confirmed the presence of non-HPV16 and non-HPV18 high-risk HPV DNA (HPV types 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and/or 68) supporting the diagnosis of HMSC. The patient underwent adjuvant radiation therapy of 56 Gy in 28 fractions and was free from recurrence on both imaging and endoscopic examination 17 months following treatment.
Patient 3
A 46-year-old female was referred to our center following an excisional biopsy of a right nasal cavity mass. The patient was known for hypothyroidism and had a 3 month history of nasal obstruction. On endoscopic evaluation, a remnant of the tumor’s attachment was seen on the right posterior septum. Preoperative imaging showed a right nasal cavity mass pushing on the left maxillary wall and ethmoid cells (Figure 1j-l). On MRI, the tumor measured 5.4 cm × 2.6 cm × 3.5 cm and appeared hypo to isointense in T1 and iso to hyperintense and heterogenous in T2-weigted images. A heterogenous enhancement was seen with gadolinium. No intracranial, orbital, or perineural invasion was noted. The initial pathological report suggested a B-SCC (Figure 2e). After a revision of the pathological specimen, a diagnosis of HMSC was suggested due to the biphasic AdCC-like areas with strong p16 expression, correlating with HPV-16 DNA confirmation on PCR. Other HPV-subtype DNA PCR analyses were negative (6, 11, 18, 26, 31, 33, 35, 39, 40, 42, 45, 51, 52, 53, 54, 55, 56, 58, 59, 61, 62, 64, 66, 67, 68, 69, 70, 71, 72, 73, 81, 82, 83, 84).
A complementary endoscopic surgical resection was performed with posterior septectomy, right ethmoidectomy, sphenoidotomy, and perilesional mucosal margin sampling. The diagnosis of HMSC with perineural invasion and negative margins was confirmed on final pathology analysis. The tumor showed predominant areas with squamous differentiation and larger pseudo-cribriform AdCC-like areas than in the biopsy (Figure 2f). IHC for p63 and cytokeratin 5/6 and 7 confirmed the biphasic luminal-abluminal cells in these AdCC-like areas. The tumor was staged as a T2N0M0 of the nasal cavity. The patient completed a treatment of adjuvant radiotherapy for a total of 60 Gy in 30 fractions and has been free of disease on both endoscopic and MRI imaging 4 years following treatment.
Discussion
To date, at least 93 HMSC cases have been reported in the English literature.12-19 All patients described herein presented with nasal obstruction and 2 presented with recurrent epistaxis, both symptoms being the most reported with HMSC.6,18 On endoscopy, HMSC often appears as well defined, pedicled or polypoid, friable masses,7,10,20-23 frequently originating from the posterior nasal cavity, 24 as in our cases. Due to HMSC’s slow growth, 24 patients frequently present with large tumors at diagnosis, with over 25% classified as T3 or T4 stages.7,18 However, formal intracranial extension is uncommon.16,25 In our case series, no intracranial invasion was noted, although 1 patient did present skull base dehiscence.
Radiologically, HMSC is characterized on CT scans as an expansile soft tissue mass, with compressive changes of nasal structures, well-defined margins, and heterogenous enhancement without bony invasion.10,20,21,26,27 These findings were noticeable features in all presented cases. MRI typically shows HMSC as iso to hyperintense on T2-weighted and heterogeneously enhanced on T1-weighted images after gadolinium administration.15,28 The first case presented in our series has one of the largest tumor volumes reported in English literature to date, which surpasses the mean HMSC radiological diameter of 4.6 cm. 15 Adamane et al 28 and Abdeljelil et al 29 also reported voluminous HMSC tumors, with wider longitudinal measures, but smaller total volumes.
Currently, there is no consensus on the optimal treatment for HMSC, but surgery remains the primary therapeutic approach, 27 often supplemented with adjuvant radiotherapy.6,18 Few studies have focused on the surgical management and radiological correlation of these tumors. 6 The remodeling of sinonasal structures as opposed to aggressive invasion appears to be a hallmark of HMSC.26,27 This finding is paramount for surgeons, as resections may be less challenging than expected based on preoperative imaging and that total endoscopic surgery may be undertaken in most cases. This paradox was observed in our cases. Even though aggressive invasion was suspected in preoperative imaging, this behavior was not surgically observed which allowed successful endoscopic management of the 3 cases. Combined endoscopic-assisted30,31 or total endoscopic surgery7,10,20-23,27,32 has been described for HMSC. Of note, one of the largest HMSC tumors previously described was also managed endoscopically despite its voluminous dimensions. 28 Consequently, HMSC radiological appearance and size should not preclude from primary endoscopic removal. Endoscopic surgery should be favored when possible since it improves quality of life 33 and reduces the risk of complications 34 without increasing the positive margin risk.34-36
Moreover, there is a mismatch between clinical behavior of HMSC and its pathological aspects, as this tumor shows high-grade histological features despite a clinically slow evolution. 37 The distinct clinical evolution of HMSC compared to AdCC emphasizes the importance of recognition and testing by pathological teams. HMSC is a challenging diagnosis due to its overlapping histological features with AdCC, adenosquamous carcinoma, or B-SCC.9,10 Initial misdiagnosis is described in the literature and was seen in 2 of our cases. In Case 3, initially diagnosed as a B-SCC, the biphasic AdCC-like aspect in the basal areas was overlooked and not investigated with appropriate luminal and abluminal cell markers. In Case 2, the striking and diffuse AdCC-like morphology on the biopsy along with SOX10 positivity pushed the interpretation toward a diagnosis of AdCC. However, SOX10 expression, initially described in salivary gland tumors (including AdCC), is also observed in HMSC and does not allow to distinguish these entities. Positivity for SOX10 markers with p16 positivity in HMSC but not in pure SCC may help distinguish these 2 diagnoses.9,24,38 Proof of HPV, with PCR or in situ hybridization, is mandatory to confirm HMSC diagnosis.39,40 It helps distinguish it from p16 positive and MYB, MYBL1, or NFIB gene fusion negative AdCC.8,41 Although the presence of myoepithelial and ductal cells evokes a salivary gland origin, squamous differentiation with occasional epithelial atypia/dysplasia in the overlying mucosae is not infrequent in HMSC tumors and might hint to the diagnosis.37,42
The most common HPV serotype found in HMSC is HPV-33, while only a subset is associated with HPV-16, as in the third case described here.6,27 This contrasts with the strong association of HPV-16 and p16-positive oropharyngeal cancer. 43 Many other serotypes, such as HPV-26, 7 HPV-35,14,30 HPV-51, 44 HPV-52, 28 HPV-56,6,19 HPV-68, 45 and HPV-82, 22 have been reported. To our knowledge, we report the second case associated with HPV-18. 13 One limitation of our study is that we have only analyzed HPV DNA instead of RNA, since our center does not have access to RNA analysis. Positivity for HPV DNA does not prove the transcriptional activity of the virus in the tumor and HPV RNA analysis is preferable for this purpose.
HMSC rarely metastasizes.6,18,46 HMSC tends to recur with long disease-free intervals, up to 30 years, making regular lifelong follow-up mandatory.44,47 The median disease-free interval is 37 months, and the local recurrence rate is low before 23 months but could exceed 40% with time.6,15,21,44,47 No local recurrences were observed in our study. However, only one patient reached the 2 year follow-up period, which limits the interpretation of the recurrence parameters.
Conclusion
Diagnosing HMSC presents significant challenges for both clinicians and pathologists. The critical role of IHC and HPV-DNA testing in accurately identifying HMSC cannot be overstated. This case series not only reinforces existing knowledge but also highlights a notable discrepancy between the radiological appearance and the actual surgical invasiveness of HMSC. It is imperative for skull base surgeons to be aware of this pathologic entity, as our findings suggest that complete endoscopic removal is often feasible and should be considered. The growing body of case reports on HMSC will undoubtedly contribute to refining treatment strategies and guiding future optimal management decisions for this complex diagnosis.
Footnotes
Authors’ Note
The first case in this article was presented at the AHNS 11th International Conference on Head and Neck Cancer in Montreal (Canada) from July 8 to 12, 2023.
Data Availability
Raw data can be shared by the corresponding author on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
