Abstract
Introduction
Epistaxis is a common problem, with a lifetime prevalence as high as 60% of people in the United States, with 6% seeking medical attention. 1 Clinical practice guidelines from the American Academy of Otolaryngology—Head and Neck Surgery from 2020 recommend that identification of risk factors includes family history of bleeding disorder, use of anticoagulant or antiplatelet medications, or intranasal drug use, and examination with anterior rhinoscopy. Examination using nasal endoscopy remains controversial, and it is currently recommended that patients with recurrent nasal bleeding despite prior treatment with packing or cautery, recurrent unilateral nasal bleeding, or concern for unrecognized pathology contributing to epistaxis undergo nasal endoscopic examination. However, this recommendation has medium level of evidence and is largely based on observational studies. 1 Nasal and paranasal tumors may need to be considered in this cohort of patients.
There is a long list of benign nasal tumors that may present in this manner. Prior studies have found that inverted papilloma, for example, presents as epistaxis in 17% of cases.2,3 More vascular tumors may present as epistaxis even more frequently; in the case of lobular capillary hemangioma, 95% of patients had epistaxis at presentation. 4
Although malignancy is a rare cause of epistaxis, it is important to evaluate for given the severity of disease. Nasopharyngeal carcinoma (NPC) is more common in individuals of southeast Asian descent, and thereby, may be a more common cause of epistaxis in this population.5,6 Although cervical lymphadenopathy is the most common presenting symptom of NPC, there is some evidence that symptoms such as epistaxis may occur earlier in the course of the disease.7,8
Given the low incidence of nasal and nasopharyngeal neoplasm, it is difficult to ascertain the incidence of epistaxis as a presenting symptom. In this study, we aimed to identify the incidence of neoplastic disease in patients referred for epistaxis, with secondary objectives of identifying need for further workup including imaging or biopsy.
Patients and Methods
A retrospective cohort study was completed. Institutional Review Board approval was obtained at Tufts Medical Center (#STUDY00001236). Informed consent was waived by the Tufts Medical Center Institutional Review Board. We reviewed charts of 2827 adult patients who presented to a tertiary academic medical center’s otolaryngology department over an 11 year period (2010-2021) with a chief complaint of epistaxis. The primary outcome was identification of a nasal or nasopharyngeal neoplasm. If a mass was identified, it was recorded as either benign or malignant, as well as if a more specific diagnosis was described in the physician’s note.
Specific inclusion and exclusion criteria were followed. Patients were excluded if younger than 18 years of age, if the visit was telehealth, and if the epistaxis was described as postoperative bleeding. Most important, all patients included in this study underwent rigid or flexible nasal endoscopy, as is the standard practice of patients presenting with epistaxis at Tufts Medical Center department of otolaryngology. Other secondary outcomes, along with known risk factors for nasal malignancy or epistaxis were noted. These include need for further workup including imaging or biopsy. Demographics such as patient age, sex, and race were examined. Smoking status was categorized as current smoker, former smoker, or never smoker as noted in the electronic medical record. Use of anticoagulant and antiplatelet medication such as warfarin (coumadin), clopidogrel (plavix), apixaban (eliquis), enoxaparin (lovenox), ticagrelor (brilinta), rivaroxaban (xarelto), heparin, aspirin/dipyridamole (aggrenox), or fondaparinux (arixtra) by the patients was recorded. Comorbidities that could play a role in the pathophysiology of epistaxis were recorded including hypertension, allergic rhinitis, and vasculitis. Family history of NPC was also noted among patients, including immediate and extended family. The treatment of the patient’s epistaxis was also recorded as a surrogate to better elucidate the severity of the epistaxis. Treatment was divided into 4 distinct categories. Category 1 was conservative treatment including topical medication and pressure, patient education of these measures, and prevention of epistaxis (petroleum jelly, nasal saline humidification). Category 2 involved patients receiving office cautery or absorbable packing. Category 3 encompassed patients receiving nonabsorbable packing. Finally, category 4 involved surgical intervention in the operating room to stop the epistaxis. Further workup of epistaxis including imaging or biopsy was recorded as secondary outcomes.
Once all data were collected, the cohort of patients after inclusion and exclusion criteria was 1164. All data points were organized in a protected spreadsheet. Statistical analysis was conducted using Python 3.7.4 in a Jupyter Notebook. SciPy, NumPy, pandas, and Matplotlib were Python packages used for data analysis.9-13 Univariate analysis with odds ratios (ORs) was carried out to determine associations between presence of nasal masses and demographic factors, smoking status, family history, medical comorbidities, workup, or treatment. A chi-squared test was used to determine if ORs were statistically significant (P < .05). When the expected value of any of the cells in a contingency table was below 5, Fisher’s exact test was used to determine significance of ORs.
Results
A total of 1164 patients presented with epistaxis and met inclusion criteria. Thirty-six nasal masses were found among patients presenting with epistaxis (3.09%, P < .001), comprised primarily of benign etiology (2.66%) including nasal polyposis, lobular capillary hemangioma, cavernous hemangioma, papilloma, and thornwaldt cyst. Asian race correlated with finding of mass lesion (75%, P = .007). Five malignancies were found (0.43%) including NPC (2 patients), extranodal NK T-cell lymphoma (2 patients), and nasal adenocarcinoma (1 patient). Of the 5 patients with a malignant mass, 4 (80%) were Asian and 1 (20%) was white (Figure 1). Of note, there was no sinonasal squamous cell carcinoma found in this patient population.
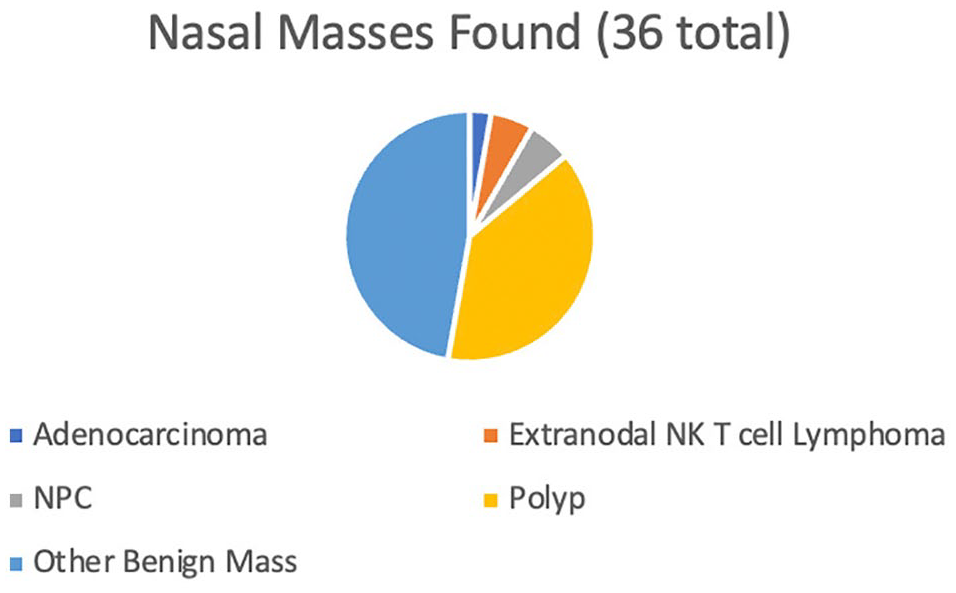
Nasal Masses Found.
In patients presenting with epistaxis, no statistically significant correlation was identified between patient characteristics including sex, medical comorbidities, anticoagulation, or smoking status and diagnosis of nasal neoplasm. Also, there was no statistical significance found between treatment category (as a marker for epistaxis severity) and diagnosis of nasal neoplasm. There was a statistical significance between CT scans and biopsies, and diagnosis of nasal neoplasm (Table 1).
Comparing Variables of Interest in Mass and Control Populations.
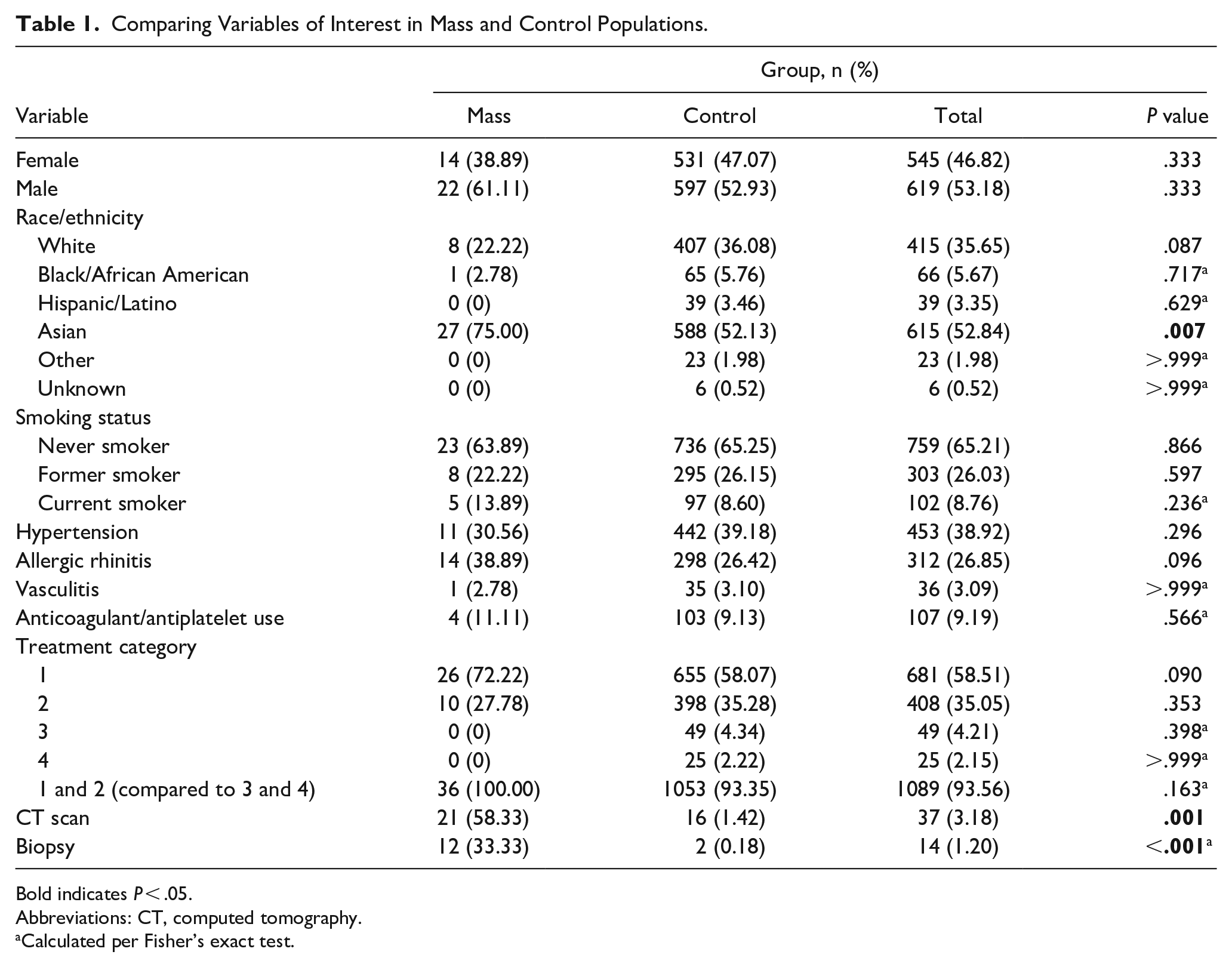
Bold indicates P < .05.
Abbreviations: CT, computed tomography.
Calculated per Fisher’s exact test.
Discussion
Benign and malignant nasal masses are relatively rare findings, making the study of their presentation difficult. Much of the existing literature revolves around case reports. Our goal was to elucidate the incidence of epistaxis as a presenting symptom of nasal mass requiring further workup. Furthermore, we aimed to quantify the percentage of patients who present to the adult otolaryngology clinic with epistaxis and were ultimately diagnosed with neoplastic disease.
We found that of patients who presented to an adult otolaryngology clinic with a chief complaint of epistaxis, 3.09% were found to have a nasal mass. This is a statistically significant finding in our cohort and required action by the otolaryngologist. The finding of a statistically significant association between nasal mass and further workup with imaging or biopsy is not surprising. This does, however, highlight the importance of nasal endoscopy in the evaluation of a patient with epistaxis, as has been the standard practice at our tertiary academic center. Without endoscopic identification of the mass lesion, it is likely that no further workup would have been undertaken.
The significant finding of Asian ethnicity being associated with nasal mass overall was an interesting finding and may be indicative of the higher prevalence of NPC and extranodal nasal type NK/T-cell lymphoma in this population.7,14 Four of 5 patients who were identified with malignant disease in our study population had these 2 diagnoses, and 4 out of 5 patients with malignances were of Asian ethnicity. Patients with NPC present with nodal metastasis in 75% to 90% of the cases. 8 The prognosis of patients who present with nodal metastasis (N1 and greater) versus local disease (N0) is worse when examining cancer staging. 15 In NPC, epistaxis may represent an early symptom of local disease that can facilitate diagnosis prior to the development of nodal metastasis. Our study identified 2 cases of NPC out of a total of 1164 patients referred for epistaxis. Both patients were of southeast Asian descent, indicating that in our study, 2 of 1164 patients of southeast Asian descent were ultimately diagnosed with NPC. In a study from 2005, 3 patients who had extranodal NK/T-cell lymphoma that was initially misdiagnosed as benign nasal disease were described. 16 In that series, misdiagnosis delayed treatment by 3 to 12 months, highlighting the importance of early detection and subsequent workup of nasal pathology. Malignant disease must be considered in the differential diagnosis of epistaxis, particularly in Asian populations.
There was no association found between smoking and nasal mass. In a meta-analysis from 2004, analyzing the epidemiological evidence of association between smoking and cancer, a positive dose-response relationship was suggested between cigarette consumption and sinonasal cancers. 17 It also suggested the strongest relationship between smoking and sinonasal squamous cell carcinomas. In our cohort, we had no diagnosis of squamous cell carcinoma. Therefore, the lack of association with smoking and nasal mass is likely reflective of mostly benign masses being found and the small number of malignancies identified. A larger study population may have found an association between nasal mass and smoking.
Interestingly, no patients in our cohort were found to have sinonasal squamous cell carcinoma, as this type of malignancy represents 51.6% of all sinonasal cancers. 18 A larger cohort of patients with epistaxis may have revealed more patients with sinonasal squamous cell carcinoma. In addition, this could suggest that sinonasal squamous cell carcinoma is less likely to present with epistaxis, but further research would be needed to determine if this is true.
Furthermore, lack of association between epistaxis severity (using treatment category as a surrogate for severity) and nasal mass suggests that it would not be appropriate to assume risk of nasal mass based on epistaxis severity. While there is no standard definition of severe epistaxis, various studies use bleeding duration >30 minutes over 24 hours, history of hospitalization for epistaxis, or prior need for blood transfusions as indicators that require prompt management. 1 Current literature examining epistaxis severity and nasal masses focuses on juvenile nasopharyngeal angiofibroma. This is a rare tumor mostly presenting in adolescent male patients and can present with unilateral, unprovoked, and profuse epistaxis in 60% to 76% of cases.19,20 Thus, it is recommended that these patients undergo nasal endoscopy. No data currently exist on association between epistaxis severity and nasal mass in adult patients. In our study, using treatment of epistaxis as an indicator for epistaxis severity, we found no association between epistaxis severity and risk of nasal mass. However, it should be noted that all these patients were seen as outpatients, and thus certain instances of epistaxis occurring in an inpatient were not included in this study. Still, it bears noting that practitioners should not be reassured by epistaxis that is quick to resolve with conservative measures. In our study, severity of bleeding was not a marker for severity of illness.
Limitations in this study include the small number of patients with both epistaxis and nasal masses. A larger study with data such as smoking status, epistaxis risk factors, and epistaxis severity may have more significance. This is inherent to the design in that mass lesions (and neoplastic disease) are a rare cause of epistaxis, but still a diagnosis that must not be missed. This study was also limited to adult patients, and therefore cannot be extrapolated to patients younger than 18 years who present with epistaxis. Only adult patients were included in this study because our main goal was to evaluate the incidence of mass, and more specifically malignant masses in patients presenting with epistaxis. As a large number of patients with nasal/nasopharyngeal masses were older adults, we chose to focus on the adult population.
Other presenting symptoms of nasal mass, such as nasal congestion, lymphadenopathy, facial numbness or hypoesthesia, anosmia, and hemoptysis were not analyzed in this study. Not fully evaluating the clinical status and overall health of these patients is a drawback of the study. An examination of epistaxis with other associated symptoms of nasal mass may increase the incidence of nasal mass found. In addition to other presenting symptoms, a better understanding of the exposures known to increase risk of nasopharyngeal malignancy or mass could have been significant in our cohort. One study showed a correlation between NPC and occupational exposures such as chemical vapors, exhausts/smokes, and acids/alkalis. 21 Unfortunately, these data were not readily available for this study, and further research into this topic could better elucidate the correlation between certain occupations and exposures with nasopharyngeal masses.
Another limitation is that this study did not examine if patients with nasal mass could have been identified on anterior rhinoscopy. If this had been described, it could have led to a smaller incidence of patients with nasal mass who would have only been detected on rigid or flexible nasal endoscopy. In addition, this study is limited to patients at a tertiary care facility in a metropolitan area and may not be generalizable to rural areas. Further research could analyze the cost-effectiveness of using nasal endoscopy on every patient who presents with epistaxis.
Conclusion
Epistaxis is a common cause for referral to an otolaryngologist. Although the incidence of malignancy in our patient population was low, nasal lesions warranting further workup were identified in 36 patients (3.09%). Findings of neoplasm were more common in patients of Asian ethnicity, which may reflect the increased prevalence of NPC in this population. Other considerations that should be taken into account include the risks associated with nasal endoscopy, such as epistaxis and discomfort, while minor, should also be weighed against the benefit of finding an actionable nasal mass. Other risk factors for nasal mass and an overall examination of the patient should be considered when working up a patient with epistaxis. Considering these other clinical factors, and the data presented here, otolaryngologic evaluation and nasal endoscopy should be considered in cases of epistaxis, as has been the standard practice at our otolaryngology outpatient tertiary care center.
Footnotes
Authors’ Note
Poster Presentation: COSM 2022 Triological Society (Submission 2776); Dallas, TX; April 27 to May 1, 2022.
Data Availability Statement
Data are available and can be shared upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
