Abstract
Introduction
Antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) is a necrotizing vasculitis that predominantly affects small to medium vessels and is associated with ANCA specific for myeloperoxidase (MPO) or proteinase 3 (PR3). 1 According to the pathological biopsy, AAV consists of 3 categories: granulomatosis with polyangiitis (GPA), microscopic polyangiitis, and eosinophilic granulomatosis with polyangiitis. 2 Necrotizing or granulomatous lesions can affect the ear, nose, and throat tract and cause symptoms of Otitis media, chronic rhinitis, sinusitis, or laryngitis. 3 In terms of ear symptoms, 30% to 50% of GPA patients experience Otitis media, resulting in hearing loss. 4
Therefore, the Japan Otological Society proposed the concept of Otitis media with antineutrophil cytoplasmic antibody-associated vasculitis (OMAAV) to describe intractable Otitis media or hearing loss due to effusion and granulation in the middle ear, with or without the manifestation of other systems in AAV patients. 5 According to the current research, a number of OMAAV patients’ hearing loss is severe, which seriously affects their quality of life. 6 However, if ENT and rheumatology doctors can recognize them earlier and provide appropriate treatment for them, their hearing loss can be partly reversible and has little effect on their daily life. 7 Based on studies about OMAAV patients’ hearing recovery, researchers in Japan have found that patients with facial paralysis and hypertrophic pachymeningitis usually have more severe hearing loss and poorer prognosis than those who do not have these conditions.8,9 This systematic review and meta-analysis aims to look into the factors that contribute to poor hearing outcomes in OMAAV patients. Moreover, this study provides the basis for hearing protection.
Methods
Search Strategy and Study Eligibility
The preferred reporting items for systematic reviews and meta-analyses reporting standards were used for conducting this systematic review. 10 The International Prospective Register of Systematic Reviews (PROSPERO) received the review protocol and registered it. And the registration number is CRD42022380125.
A literature search was conducted in the PubMed, MEDLINE, EMBASE, Cochrane, Scopus, and Web of Science databases by 2 independent investigators. There were no restrictions on the publication date, country, ethnicity, or age. The MeSH search of keywords should be the first place, including ANCA-AAV, hearing, and prognosis. Each word searched in MeSH is connected with “OR,” and keywords are connected with “AND.” An example of a search strategy for a specific database has been exhibited at the end of the article. We chose studies based on titles, abstracts, and full texts.
Inclusion and Exclusion Criteria
Inclusion criteria were as follows: (1) published in English until December 1, 2022; (2) observational studies such as case–control study or retrospective cohort study; (3) records of the hearing outcomes after treatment or hearing improvements; (4) examined the association between hearing outcomes and relevant factors such as facial palsy, hypertrophic pachymeningitis, etc.
The exclusion criteria were as follows: (1) duplicate studies; (2) published not in English; (2) reviews, case reports, histopathological studies, and guidelines; (3) studies without hearing outcomes or improvements; and (4) “OR” and “95% CI” unavailable directly or indirectly in studies.
Data Extraction
All data were extracted by 2 independent investigators (M. Y. Xie and Z. Y. Wu). Disagreements were discussed with the third investigator until a consensus was reached. The accuracy of the selected studies and the extracted data was verified by a third investigator (S. Wang). First, we extracted data about the general information of the studies (eg, first author, year of publication, study design, total events). In various studies, researchers discussed different factors affecting hearing outcomes, based on which we identified 8 possible factors: #1 age; #2 the period from onset to diagnosis; #3 the status of ANCA; #4 facial palsy; #5 hypertrophic pachymeningitis; #6 rituximab for treatment; #7 intravenous cyclophosphamide; and #8 disease relapse. Once the factors were included in the study, the data were extracted along with the quantitative data regarding the hearing outcomes.
Quality Assessment and Risk of Bias
Using the Newcastle–Ottawa Scale (NOS), a metric created expressly for evaluating the quality of observational epidemiological studies, 2 researchers (Q. Tang and R. Y. Xue) rigorously evaluated and scored the quality of all qualifying studies. 11 Both case–control and cohort studies can use different versions of the NOS tool. Through star ratings for group selection, group comparability, and the determination of either the exposure being evaluated for case–control studies or the result of interest for cohort studies, the NOS evaluates research bias. This assessment of quality will be undertaken by the first author and checked independently for completeness and accuracy by a second author. Any differences in the assessment of quality will be resolved via discussion between these authors and if they remain unresolved upon referral to a third author.
Statistical Analysis
Outcomes are presented as the odds ratios (ORs) with the corresponding 95% confidence intervals (CIs). Continuity variables will be expressed using the mean and standard deviation. Studies that are not presented with ORs need to be recounted and calculated, or we seek the help of the original data from the corresponding author. The Higgins I2 test was used to test the heterogeneity. An I2 greater than 50% indicated considerable heterogeneity, and we used the random effect model. Forest plots were used to display the effect size of each study and the pooled outcome effect. By analyzing the differences between the 2 effect models, sensitivity analysis was carried out to evaluate the impact of specific studies. A P value of .05 or less was regarded as significant. R studio 4.2.1 (2022-06-23) was used to conduct the statistical analyses. For the 4 risk factors involved, it is meaningless to conduct the report bias analysis due to the small number of studies each included, so publication bias was not included in this study.
Results
Description of the Included Studies
There are 780 studies being searched. After removing duplicate studies and studies with incomplete basic information, 551 studies were identified. Next, we read article titles and abstracts to screen for research, leaving 17 studies eligible for full-text screening. Out of the 17 studies, 11 studies were case series or case reports, while the full texts of 2 studies had no data on hearing improvement records. Finally, 4 studies were selected for statistical analysis, as depicted in Figure 1.4,12–14
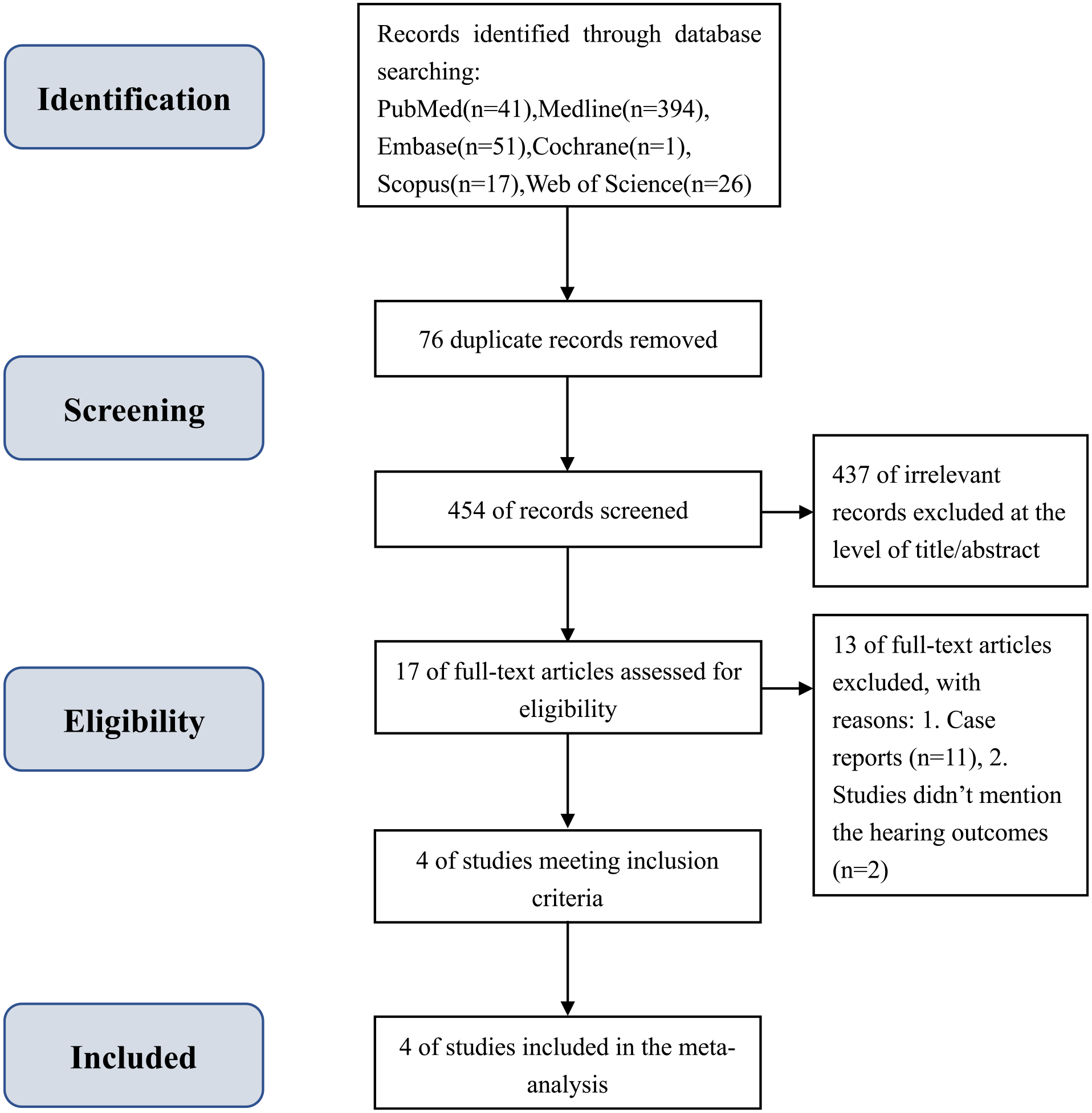
Preferred reporting items for systematic reviews and meta-analyses search flow diagram exhibiting study selection process.
Study Characteristics and Quality Assessment
The characteristics of the eligible studies are summarized in Table 1. Three studies were case–control studies, and 1 was a retrospective cohort study. All studies were performed in Japan because the concept of OMAAV was proposed by the Japan Otological Society, and they are devoted to describing ear involvement in AAV patients. The risk of bias assessment is displayed in Table 2, and they are high-quality studies.
Studies’ Characteristics.
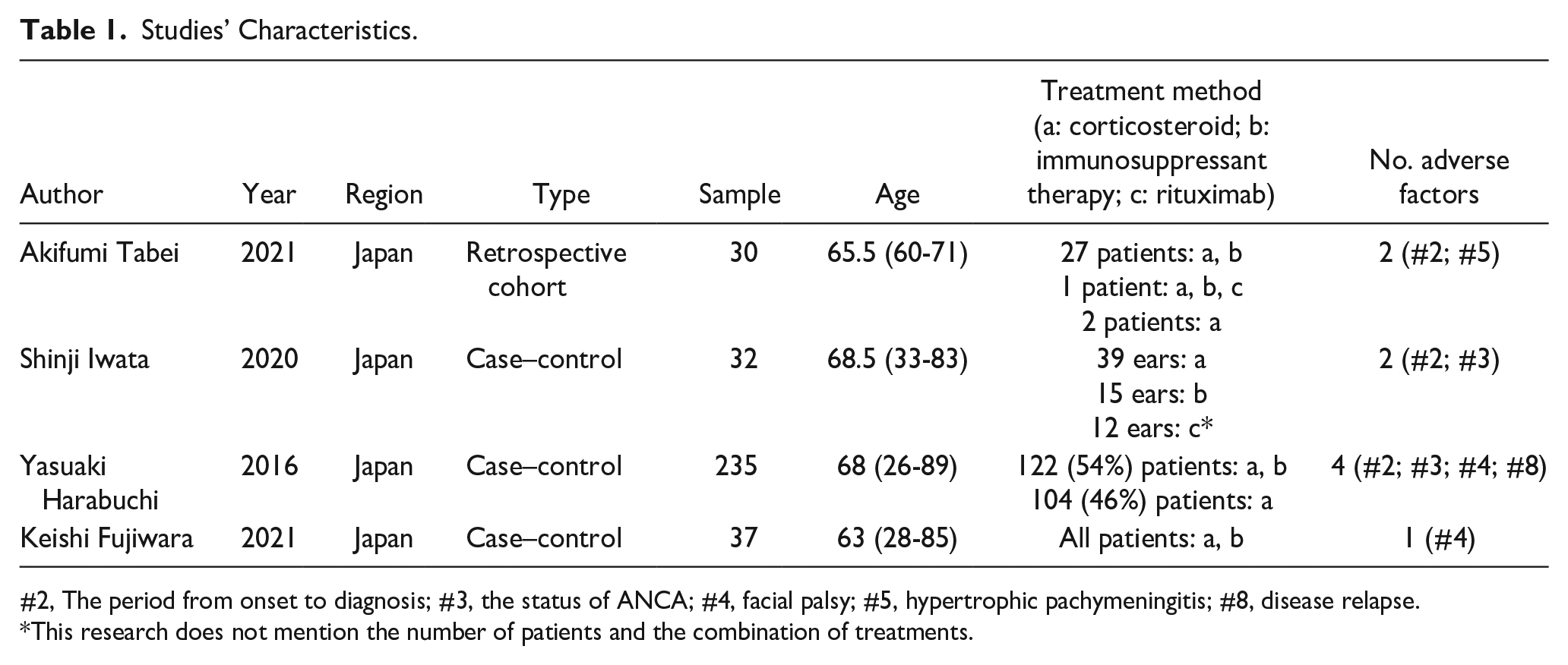
#2, The period from onset to diagnosis; #3, the status of ANCA; #4, facial palsy; #5, hypertrophic pachymeningitis; #8, disease relapse.
This research does not mention the number of patients and the combination of treatments.
Risk of Bias Assessment.
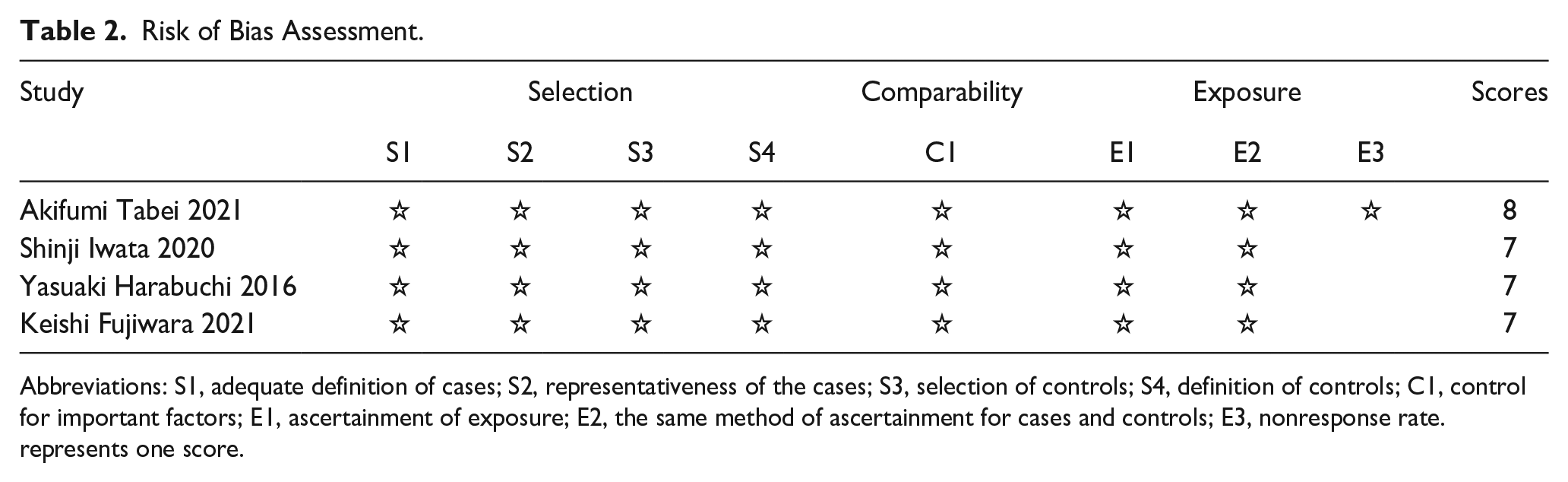
Abbreviations: S1, adequate definition of cases; S2, representativeness of the cases; S3, selection of controls; S4, definition of controls; C1, control for important factors; E1, ascertainment of exposure; E2, the same method of ascertainment for cases and controls; E3, nonresponse rate. represents one score.
Meta-Analysis
Due to the limited number of included studies and patients, as well as the different types of studies, we were unable to explore all 7 exposure factors mentioned above. In the future, more original studies need to be carried out by scholars in related fields. Based on the extracted data, we investigated the effects of exposure to 4 factors: facial palsy, hypertrophic pachymeningitis, ANCA-negative status, and the period from onset to diagnosis on the prognosis of hearing outcomes. In the 4 included articles, all patients had their hearing collected at diagnosis and after treatment. The average hearing threshold of 0.5, 1, and 2 kHz was adopted to represent the hearing condition, and the time of hearing outcomes collected was not explicitly mentioned in 3 articles.4,13,14 Only 1 article represented hearing thresholds at 5 frequencies of 0.25, 0.5, 1, 2, and 4 kHz, and explicitly collected Pure-tone Audiometry (PTA) results before treatment, 1, 2, 4, 8, 12, 24, 36, and 48 weeks after treatment, and during remission. 12 The studies with hearing data will follow the definition that the good prognosis group should achieve a hearing level <40 dB at remission or gain hearing improvement ≥30 dB, and the others will be categorized into the poor prognosis group.
Facial palsy
All eligible studies reported the occurrence of peripheral facial palsy (Figure 2). In total, 297 events were included in the poor hearing prognosis, and 312 events were included in the good hearing prognosis. There was significantly increased odds of peripheral facial palsy in the poor hearing prognosis group (OR 1.51; 95% CI 1.07-2.15; I2 = 0%; P = .02).
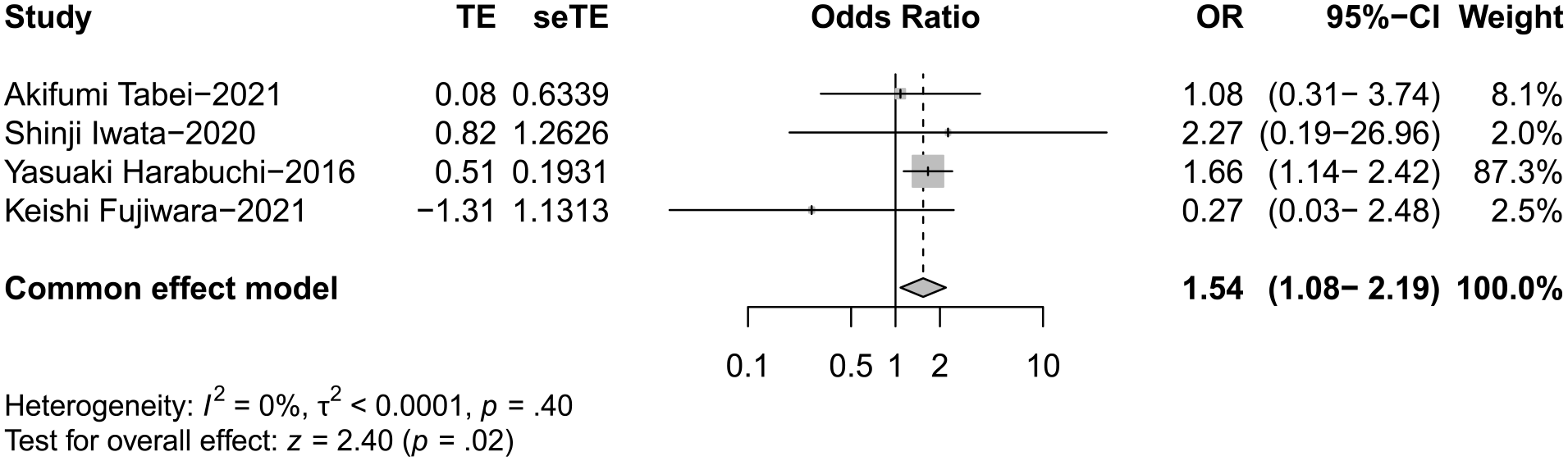
Forest plot showing the odds of facial palsy in OMAAV patients. OMAAV, Otitis media with antineutrophil cytoplasmic antibody-associated vasculitis.
Hypertrophic pachymeningitis
Three studies evaluated hypertrophic pachymeningitis for 558 events. In all, 90 of 277 events with poor hearing outcomes were proven to have hypertrophic pachymeningitis by MRI scanning. There was a statistically significant difference in the incidence of hypertrophic pachymeningitis between the 2 groups (OR 1.73; 95% CI 1.18-2.53; I2 = 24%; P = .005) (Figure 3).
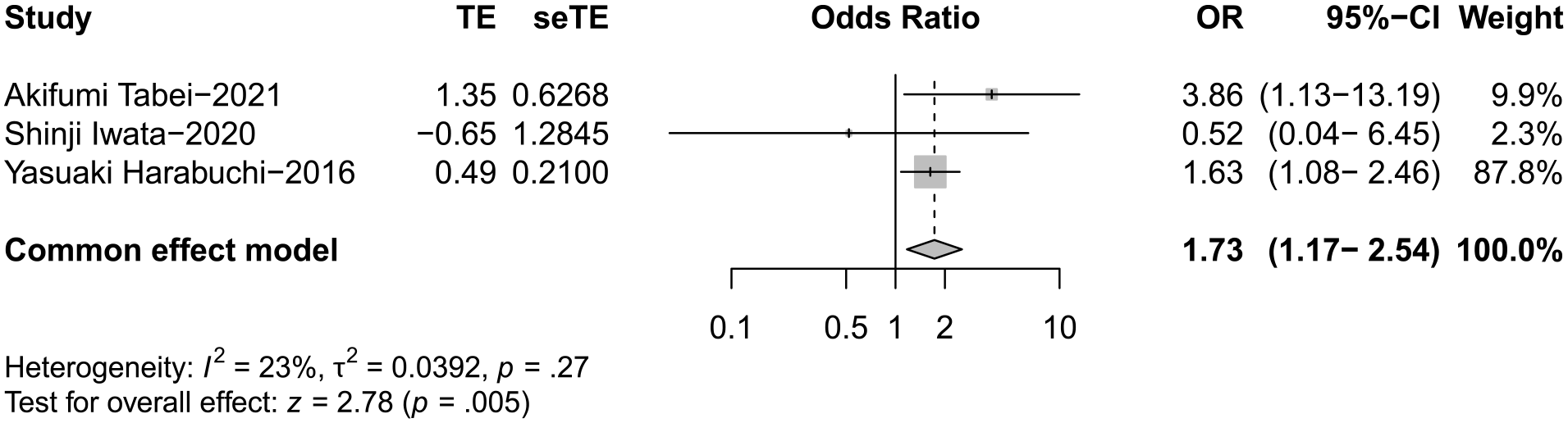
Forest plot showing the odds of hypertrophic pachymeningitis in OMAAV patients. OMAAV, Otitis media with antineutrophil cytoplasmic antibody-associated vasculitis.
ANCA negativity
Three studies with a total studied population of 246 poor prognosis patients and 300 controls were implemented to identify a significant association between ANCA negativity and poor hearing outcomes (OR 1.75; 95% CI 1.11-2.77; I2 = 33; P = .02) (Figure 4), which means that patients with poor prognosis were 1.75 times more likely to be ANCA negative than those with good prognosis.
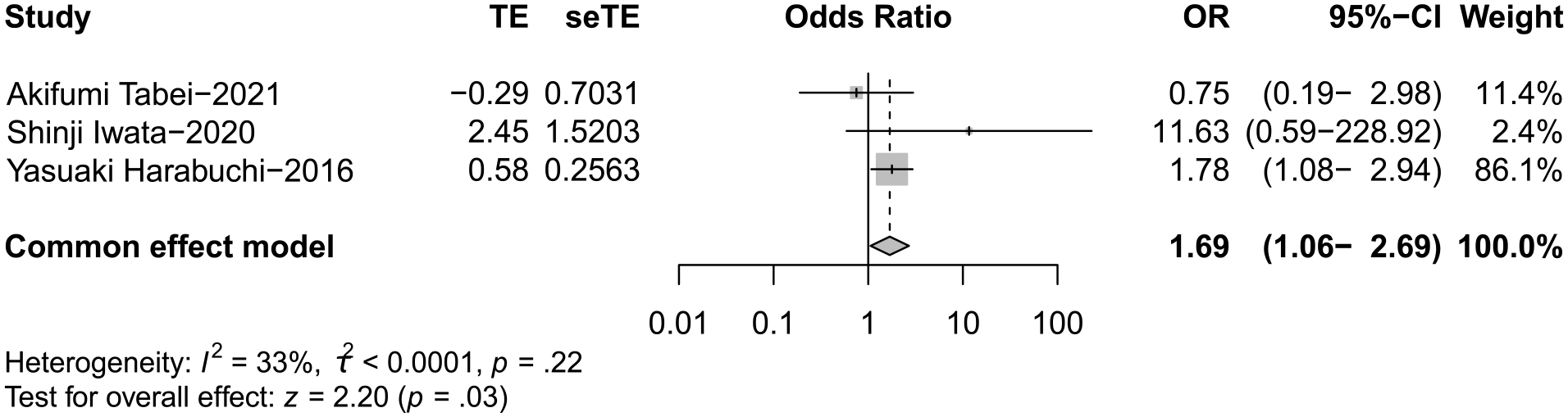
Forest plot showing the odds of ANCA negativity in OMAAV patients. ANCA, antineutrophil cytoplasmic antibody; OMAAV, Otitis media with antineutrophil cytoplasmic antibody-associated vasculitis.
Period from onset to diagnosis
Only 2 studies met the inclusion criteria because the time intervals of each study record were inconsistent and could not be uniformly extracted. There was no significant difference between the 2 groups (SEM ± SD 2.54; 95% CI −1.56-6.64; I2 = 98%; P = .22) (Figure 5).
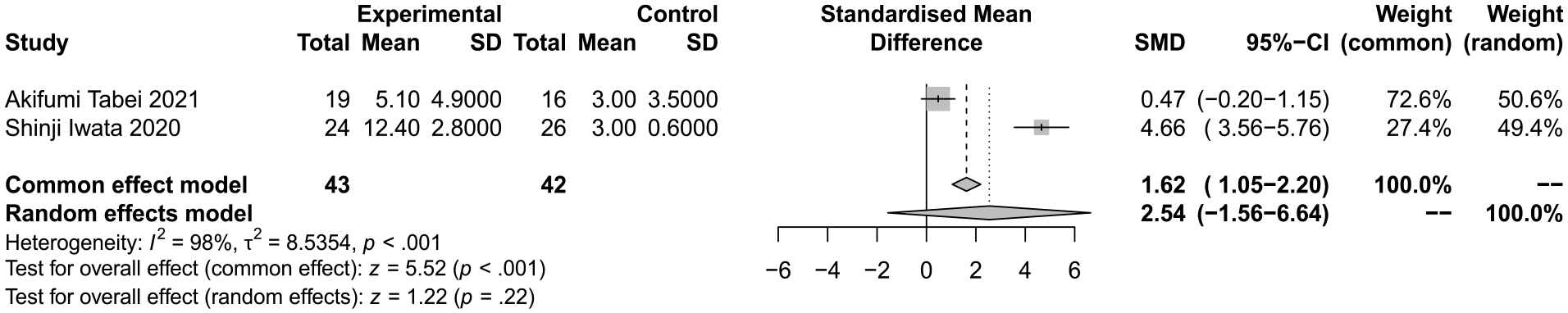
Forest plot showing the odds of period from onset to diagnosis in OMAAV patients. OMAAV, Otitis media with antineutrophil cytoplasmic antibody-associated vasculitis.
Sensitivity Analysis
In terms of the heterogeneity in the meta-analysis results, sensitivity analysis should be conducted for the period from onset to diagnosis. Due to the quantity of studies, we chose to compare the difference between using the random effect model and the fixed effect model to pool the effect. We can see the different results in Figure 5, and it turns out to be unstable.
Discussion
Our meta-analysis included 344 patients in 4 studies. From the results of each factor odds for poor hearing prognosis, we found that people with poor hearing prognosis were inclined to have facial palsy, hypertrophic pachymeningitis, and ANCA negativity. No obvious relation can be drawn between the hearing prognosis and the period from onset to diagnosis, for which the underlying reason is the bias against the quantity of the studies and the consistency of the data extracted from the studies.
At present, there have been many case reports of facial palsy in AAV, and the incidence of facial paralysis is higher in patients with OMAAV.8,15 In terms of pathogenesis, the middle ear is the most likely candidate for the lesion site on the facial nerve, and vasculitis in the vasa nervorum of the facial nerve is one of the possible reasons, and patients with GPA proved it in a histopathologic study or recent case report.16,17 In addition, dehiscence of the facial canal in the tympanic cavity results in inflammatory granulation spreading to the canal, leading to peripheral facial nerve palsy. 18 The side affected by facial palsy is always consistently the same as that with more severe Otitis media. 12 In most cases, facial palsy tends to occur with hypertrophic pachymeningitis together, and the most characteristic features of OMAAV are the frequent occurrences of facial palsy and hypertrophic pachymeningitis irrespective of geographic area.19,20 However, the jury is still out on cause and effect. It is worth that the middle ear, facial nerve, and dura mater anatomically adjoin each other, which means that they interact with each other, making it more difficult to find the cause. Both of them displayed an unfavorable clinical course that threatened hearing and daily life.4,12,14 Veins of the middle ear communicate with the veins of the dura mater, and small-vessel vasculitis, and/or inflammatory mediators in the middle ear may directly spread to the dura mater, leading to hypertrophic pachymeningitis. 21 On the other hand, some scholars believe that facial paralysis is caused by the intracranial pathway due to hypertrophic pachymeningitis. 18 Severe headache is known to be the most common symptom. 22 We should pay more attention to their clinical symptoms in our clinical work. Above all, based on this analysis, peripheral facial palsy and hypertrophic pachymeningitis are common in OMAAV patients and tend to predict severe hearing loss, but the underlying mechanism needs to be further explored.
As mentioned above, AAV is a vasculitis characterized by ANCA, but in the course of the clinical work, the negativity of both PR3-ANCA and MPO-ANCA is not rare. 21 Our meta-analysis found that it was statistically significant with poor hearing outcomes. This result is probably ascribed to the difficulty of diagnosis caused by ANCA negativity, leading to the adverse consequences of delayed treatment, which is also consistent with the conclusion of many studies on the association between the period from onset to diagnosis and hearing outcomes.1,12 There are several interpretations for ANCA negativity: the current technology is limited; the titer of ANCA is too low to test; and a third type of ANCA exists. Currently, researchers are working on other methods of detecting ANCA to improve the sensitivity, and whether there exists a new ANCA related to OMAAV also needs to be explored in the future. 23 It remains to be explored in the future whether new types of ANCA exist and are linked to hearing loss, which helps us to understand the underlying mechanism.
The period from onset to diagnosis was not significantly related to poor hearing prognosis in our meta-analysis. This result may be due to the small number of studies included, resulting in high heterogeneity. We have carried out sensitivity analysis, and the results under the 2 effect models are totally different, demonstrating that the results are unstable. However, based on current understanding, the more time it takes to diagnose, the worse the outcome of hearing acquisition may be.12,13 Overall, early diagnosis and treatment are important for the “reversible” sensorineural hearing loss treatment of OMAAV. 24 Undeniably, there are still many confounding factors for the recovery of hearing: the initial level of hearing, the speed and severity of OMAAV progression, patient age, and so on, which we are looking forward to determining.
There were some limitations in this meta-analysis that may lead to unstable results. First, a few studies met the inclusion criteria, and they were all observational studies, which are susceptible to selection and confounding bias. More rigorously designed studies are needed in the future. In terms of data extraction, since only 1 study defined good hearing prognosis, we used the conclusions given by the other studies for analysis if we did not obtain the raw data. With further research, scholars should define hearing improvement. Due to the lack of data, we cannot analyze the effect of rituximab for treatment and intravenous cyclophosphamide on OMAAV patients’ hearing outcomes. Last, the data did not allow us to perform a subgroup analysis depending on the severity of hearing outcomes.
Conclusion
We found that OMAAV patients with facial palsy, hypertrophic pachymeningitis, and ANCA negativity had a significant association with poor hearing prognosis. We did not find any association between the period from onset to diagnosis of OMAAV and poor hearing outcomes. Clinical doctors should pay more attention to OMAAV or AAV patients when they experience manifestations such as facial palsy or severe headache, which provides a clue to recognize the underlying hearing problems. On this basis, we can continue to explore whether the treatment has an impact on hearing so that OMAAV patients can avoid severe hearing loss.
Footnotes
Acknowledgements
The data used in this paper are all extracted from the original paper except the original data provided by Prof. Toru Sakairi that is not publicly available.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National High Level Hospital Clinical Research Funding (Grant No.2022-PUMCH-095).
Ethical Approval
This study was approved by the Ethics Review Committee of Peking Union Medical College Hospital, Chinese Academy of Medical Sciences (I-23PJ345).
