Abstract
Objectives
We assessed the effect of tranexamic acid on postoperative bleeding, and any adverse effects, in patients undergoing head-and-neck surgery.
Methods
We searched databases (PubMed, SCOPUS, Embase, the Web of Science, Google Scholar, and the Cochrane database) from their dates of inception to August 31, 2021. We analyzed studies that compared bleeding-related morbidity between perioperative tranexamic acid and placebo (control) groups. We subanalyzed tranexamic acid administration methods.
Results
The extent of postoperative bleeding (standardized mean difference [SMD] = –0.7817, [–1.4237; –0.1398], P = 0.0170, I2 = 92.2%) was significantly less in the treatment group. However, there were no significant between-group differences in terms of operative time (SMD = –0.0463 [–0.2147; 0.1221]; P = 0.5897, I2 = 0.0% [0.0%; 32.9%]); intraoperative blood loss (SMD = –0.7711 [–1.6274; 0.0852], P = 0.0776, I2 = 94.4%); drain removal timing (SMD = –0.3382 [–0.9547; 0.2782], P = 0.2822, I2 = 81.7%); or the amount of infused perioperative fluid (SMD = –0.0622 [–0.2615; 0.1372], P = 0.5410, I2 = 35.5%). Also, there were no significant between-group differences in laboratory findings (serum bilirubin, creatinine, and urea levels; and the coagulation profiles) between the tranexamic acid and control groups. Topical application was associated with a shorter postoperative drain tube dwell time than was systemic administration.
Conclusion
Perioperative tranexamic acid significantly reduced postoperative bleeding in patients undergoing head-and-neck surgery. Also, topical administration might be more effective in postoperative bleeding and postoperative drain tube dwell time.
Introduction
The head and neck contain many blood vessels; surgical intervention can cause bleeding triggering a need for surgical drainage and blood transfusions. 1 Also, the induction of general anesthesia prior to head-and-neck surgery reduces capillary resistance, thus increasing intraoperative bleeding during surgery. 2 This can make it difficult to identify anatomical structures and landmarks, increasing the risk of perioperative complications, the duration of surgery, and sometimes rendering surgery incomplete. A postoperative hematoma may develop, increasing the risks of poor wound healing and infection. Various methods are used to reduce intraoperative bleeding and thus improve visualization of the surgical area; blood pressure can be lowered by various anesthetics and vasoconstrictors.
Tranexamic acid is an anti-fibrinolytic drug that binds plasminogen during the coagulation cascade. This interferes with fibrinolysis by stabilizing clots, reducing the risk of further bleeding.3,4 Tranexamic acid is administered to reduce bleeding during surgery. However, whether tranexamic acid reduces perioperative bleeding during head-and-neck surgery remains controversial.1,5-9 Postoperative bleeding is closely associated with postoperative morbidity, late discharge, and slow recovery from head-and-neck surgery. We explored the utility of tranexamic acid as a bleeding control agent in patients undergoing head-and-neck surgery.
Patients and Methods
Search and Study Selection
We searched PubMed, SCOPUS, Embase, the Web of Science, Google Scholar, and the Cochrane database; we retrieved all relevant, randomized controlled trials described in English prior to August 2021. The search terms were: “head and neck,” “surgery,” “tranexamic acid,” “anti-fibrinolytic agents,” “blood loss,” “bleeding,” “hemorrhage,” “operative time,” and “thromboembolism.” Two authors independently reviewed the titles and abstracts of all studies and excluded those that were irrelevant. If an abstract alone was not informative, the full text was checked. We sought to contact the authors if data were missing or incomplete. The exclusion criteria were any procedure other than head-and-neck surgery (e.g., abdominal surgery); a repeat report based on the same data; and a lack of quantifiable data or an inability to derive such data from the report. The selection flow chart is presented in Figure 1. The study was registered in the Open Science Framework (https://osf.io/wx7ac/). Flow diagram of study selection.
Data Extraction and Risk of Bias
Data were extracted using a standard form.10-12 We recorded patient numbers, the grading scales used, intra- and postoperative outcomes, incidences/percentages of adverse effects, and the p-values from comparisons between the tranexamic acid and control groups. The outcomes were operative time;1,5-8 intraoperative blood loss;1,5,7,9 intraoperative volume replacement;1,5,8 postoperative blood loss (or drainage amount);1,5-7,9 drainage duration (days to postoperative drain removal);1,6,7 postoperative laboratory findings (including coagulation profiles;1,5,7,8 and the occurrence of any of postoperative flap necrosis, an oro-cutaneous fistula, or a thrombotic accident (incidences/percentages).1,5,6,8 The results were compared between patients receiving perioperative tranexamic acid either intravenously or topically, and controls given saline or other drugs. The risk of bias was evaluated using the Cochrane tool. 13
Statistical Analysis
The meta-analyses were conducted using “R” statistical software (R Foundation for Statistical Computing, Vienna, Austria). When the original data were presented as continuous variables, standardized mean differences (SMDs) were derived to calculate effect sizes because the measures of surgical time, perioperative blood loss, intraoperative volume replacement, postoperative laboratory findings, and coagulation profiles were not standardized. In all other cases, incidences are presented as odds ratios (ORs). Sensitivity analyses were performed to estimate the effects of each study on the overall results.
Results
Summary of the Included Studies and the Risk of Bias Assessment.
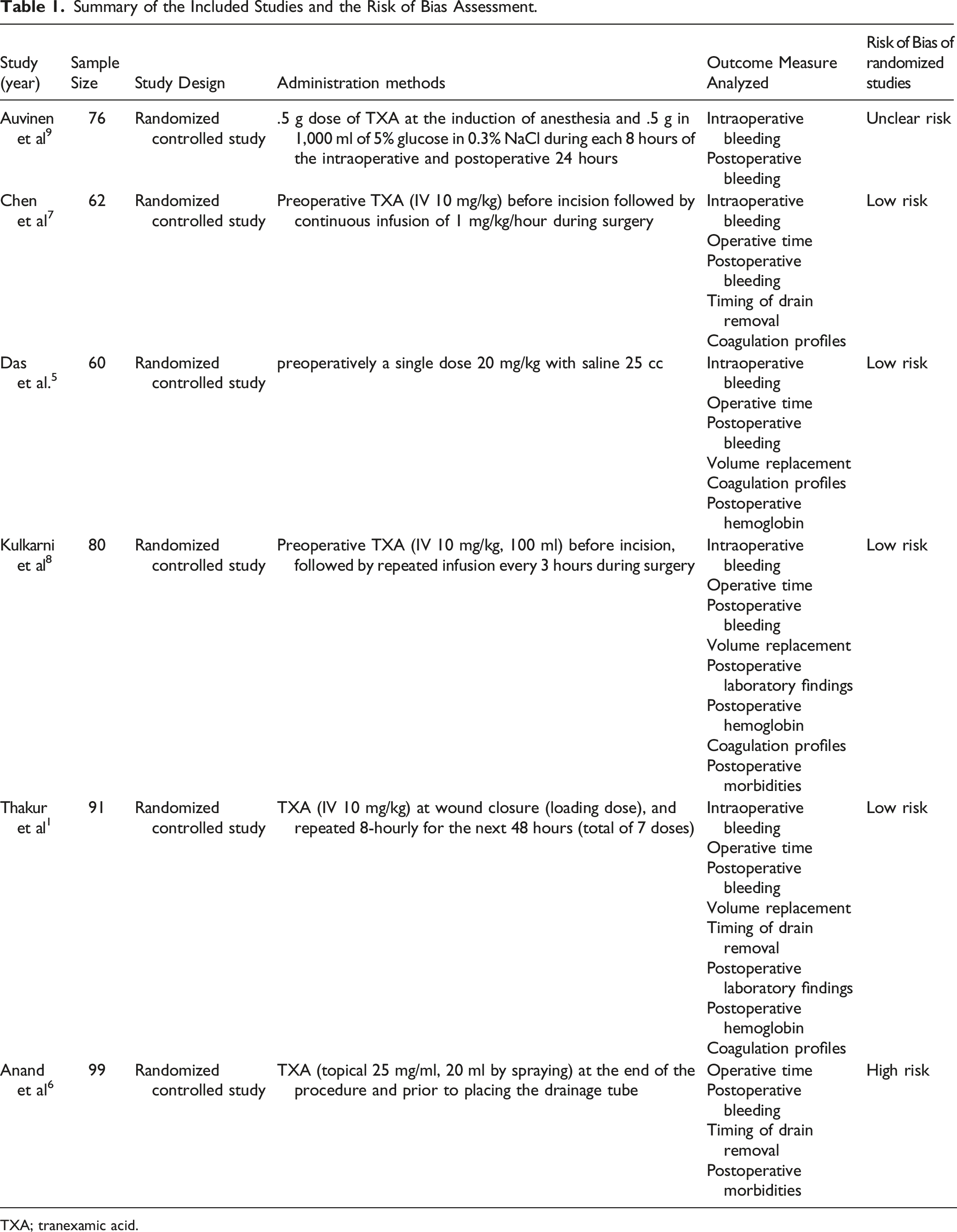
TXA; tranexamic acid.
Perioperative Outcomes
The extent of postoperative bleeding (drainage amount) (SMD = –.7817 [–1.4237; –.1398], P = .0170, I2 = 92.2%) was significantly lower in the treatment group (Figure 2). However, there were no significant between-group differences in terms of operative time (SMD = –.0463 [–.2147; .1221], P = .5897, I2 = .0% [.0%; 32.9%]); intraoperative blood loss (SMD = –.7711 [–1.6274; .0852], P = .0776, I2 = 94.4%); drain removal timing (SMD = –.3382 [–.9547; .2782], Pp = .2822, I2 = 81.7%); or the amount of infused perioperative fluid (SMD = –.0622 [–.2615; .1372], P = .5410, I2 = 35.5%). Significant inter-study heterogeneity (I2 > 50) was evident, with the exceptions of the operative time and amount of infused perioperative fluid. Tranexamic acid was administered either systemically (intravenously) or topically. We thus performed subgroup analyses. The extents of postoperative bleeding and the operative times did not differ between the two subgroups, but topical administration tended to better reduce postoperative bleeding (SMD = –.6924 [–1.3923; .0076] vs. –1.3789 [–1.8191; –.9387]; P = .1036) and significantly reduce the drain tube dwell time (–.0842 [–.4113; .2428] vs. –1.0973 [–1.5212; –.6734]; P = .0004). However, only one study compared the two routes of administration;
6
therefore, the conclusion should be interpreted with caution. Surgical outcomes between tranexamic acid group and control. Operative time (A), intraoperative blood loss (B), extent of postoperative bleeding (C), amount of perioperative fluid infused (D), and drain removal timing (E).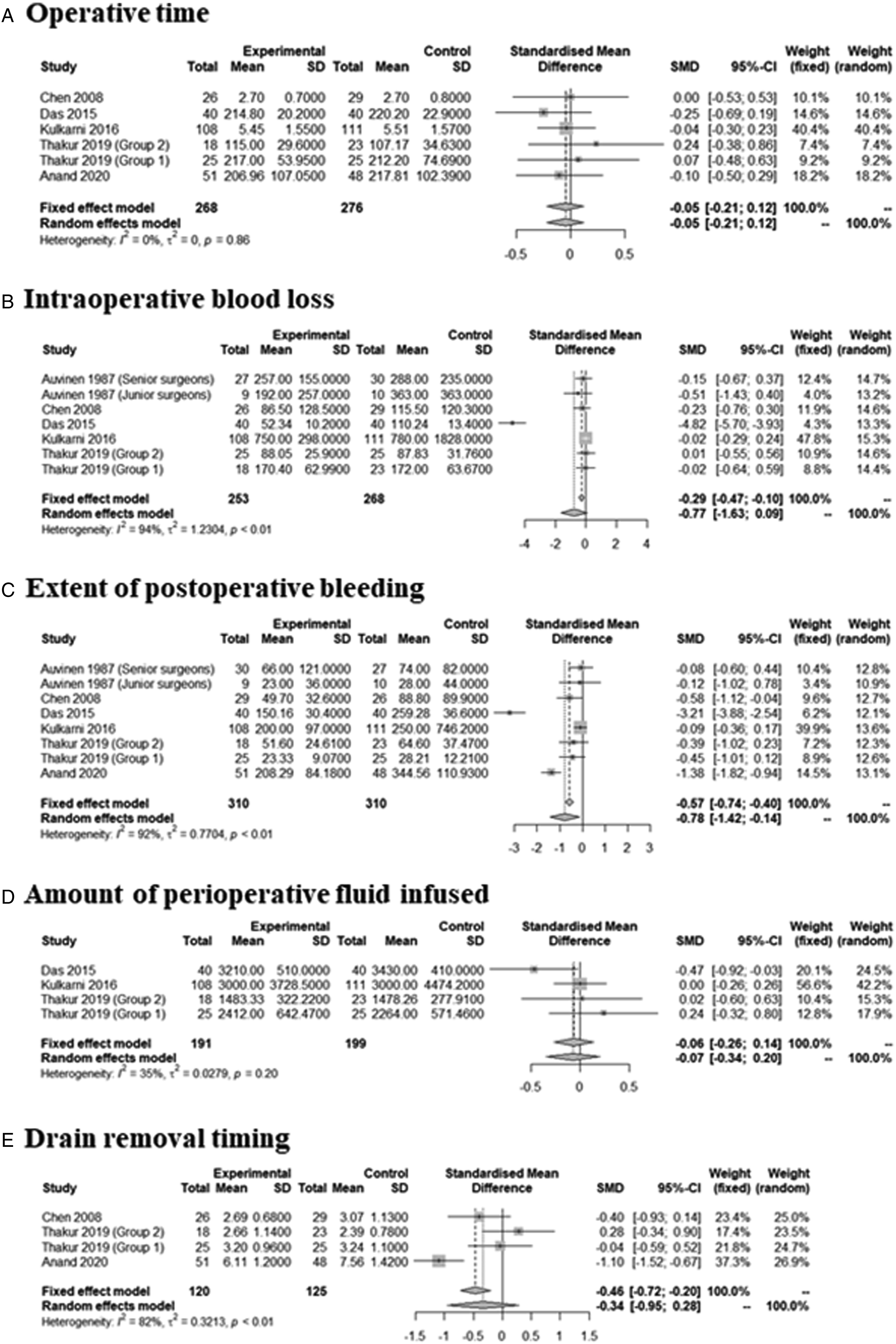
Peri- and PostOperative Adverse Effects
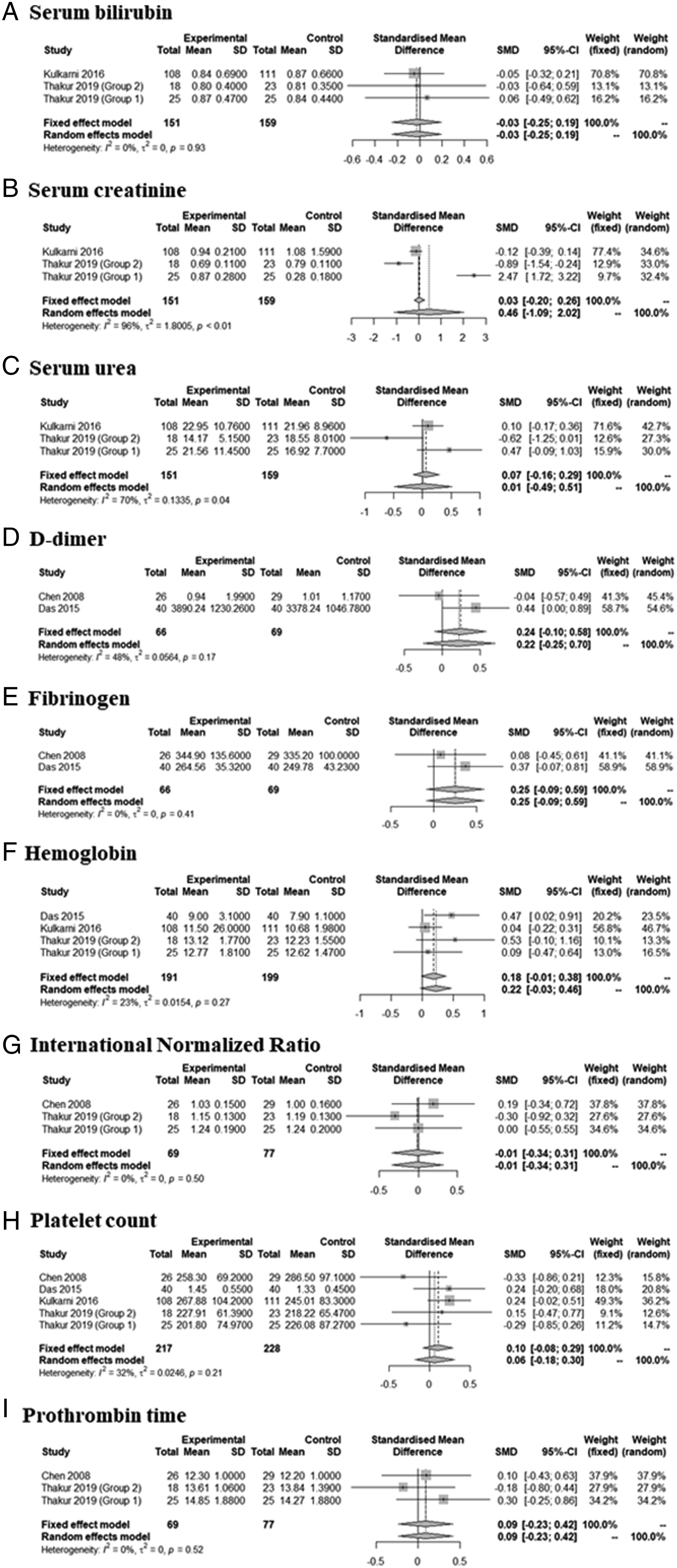
The incidences of adverse events, including perioperative blood transfusion (OR = 0.2043; 95% CI [0.0180; 2.3240], I2 = 89.8%), postoperative skin flap necrosis (OR = 0.8015; 95% CI [0.2364; 2.7173], I2 = 0.0%), and oro-cutaneous fistulation (OR = 0.5913; 95% CI [0.1384; 2.5254], I2 = 0.0%), did not differ between the two groups (Figure 3). There was no significant inter-study heterogeneity (I2 < 50) in terms of any outcome except for perioperative blood transfusion. There was no thrombotic accident. Tranexamic acid did not significantly affect the laboratory findings: serum bilirubin (SMD = –.0285 [–.2514; .1944], P = 0.8022, I2 = .0%); serum creatinine (SMD = .4626 [–1.0932; 2.0184], P = .5601, I2 = 96.0%); serum urea (SMD = .0137 [–.4854; .5127], P = .9571, I2 = 69.5%); the coagulation profile (D-dimer SMD = .2430 [–.0972; 0.5831], P = .1615, I2 = 47.6%;), fibrinogen (SMD = 0.2517 [–.0877; .5912], P = .1461, I2 = .0%); Hb (.1848 [–.0148; .3845], P = 0.0696, I2 = 23.2%); the international normalized ratio (–.0114 [–.3376; .3147], P = .9453, I2 = .0%); the platelet count (.1026 [–.0842; .2894], P = .2816, I2 = 31.8%); or the prothrombin time (.0913 [–.2350; .4175], P = .5834, I2 = .0%)(Figure 4). There was no significant inter-study heterogeneity (I2 < 50) except in terms of the serum creatinine and urea levels. Surgery-related adverse effects between tranexamic acid group and control. Extent of perioperative blood transfusion (A), skin flap necrosis (B), and oro-cutaneous fistulation (C). Laboratory findings between tranexamic acid group and control. Serum bilirubin (A), serum creatinine (B), serum urea (C), D-dimer (D), fibrinogen (E), and hemoglobin (F), INR (G), platelet count (H), and prothrombin time (I).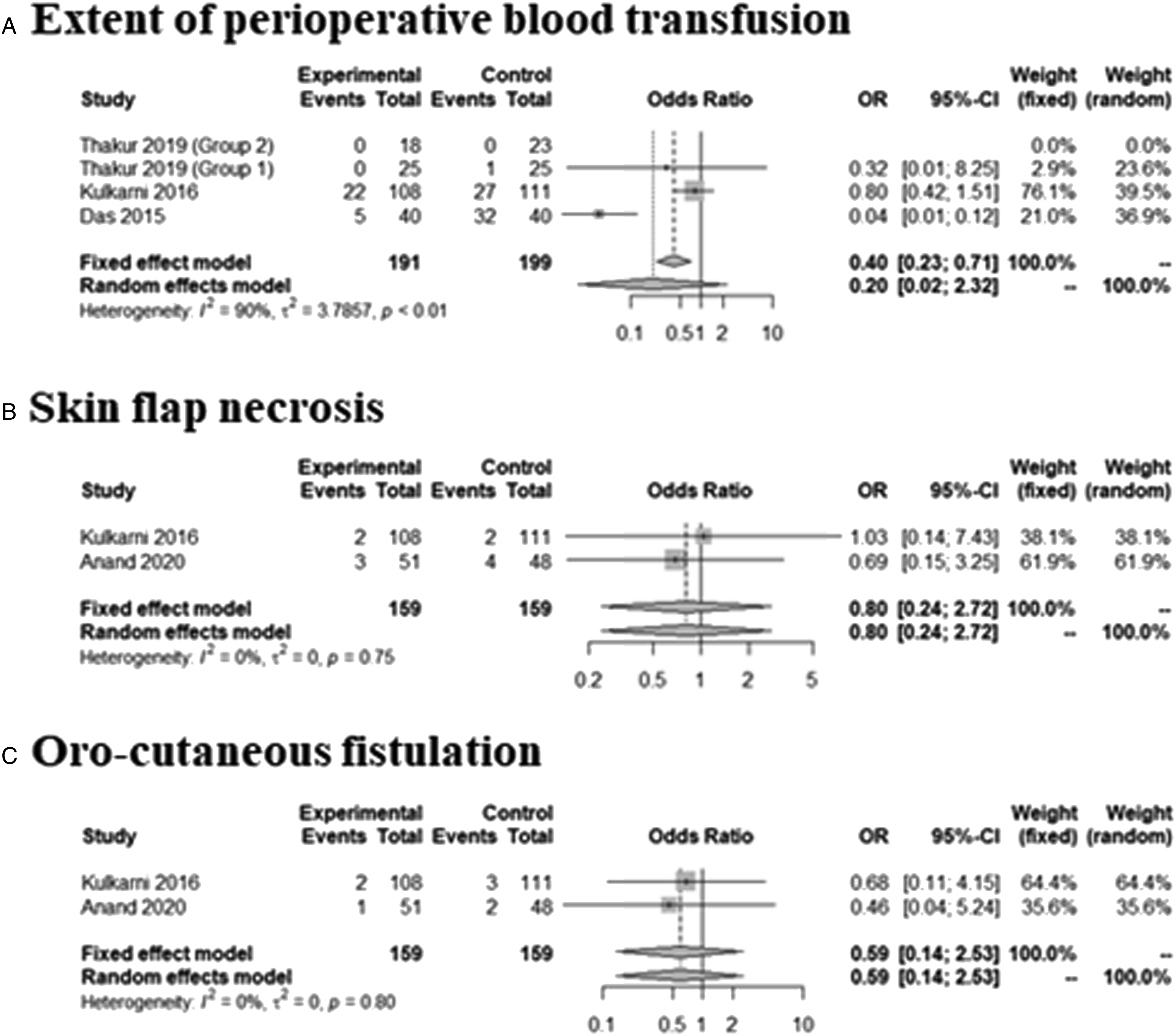
Sensitivity Analyses
Sensitivity analyzes were performed by evaluating differences in pooled estimates by repeating the meta-analysis each time omitting one other study. All results were consistent with the above results.
Discussion
Intraoperative bleeding can cause significant blood loss, decrease the surgical field of view, and increase the surgical time. 14 Tranexamic acid is a synthetic derivative of lysine that blocks the lysine-binding site of plasminogen, thus inhibiting activation and fibrin binding, and fibrinolysis. 15 Several studies have explored the utility of tranexamic acid during head-and-neck surgeries.1,5-7 Although several reviews and meta-analyses have shown that tranexamic acid aids nasal surgery,16-18 no meta-analysis has yet focused on head-and-neck surgery.
Tranexamic acid has been given topically or intravenously to patients at risk of hemorrhage or who require hemostasis during surgery. The intravenous dose is 10–15 mg/kg; if necessary, this can be repeated once or twice at 3-h intervals. 19 The maximum hemostatic effect was evident at 1 g; higher doses were no better in most adults. 14 Doses less than 10 mg/kg ineffectively or less effectively controlled bleeding. 20 Topical application has the advantage that the area of concern is directly targeted.
We analyzed intraoperative blood loss; the extents of peri- and postoperative bleeding; the amount of perioperative fluid infused; the time to drain removal; laboratory data (including the coagulation profile); and the incidences of perioperative blood transfusion, skin flap necrosis, and oro-cutaneous fistulation. The extent of postoperative drainage was less in the treatment group. However, there were no significant between-group differences in operative time, intraoperative blood loss, drain removal timing, or the amount of perioperative fluid infused. These results are similar to those of previous studies. 7 Major bleeding, such as intraoperative bleeding, can be controlled by the operator's attention, such as electrocautery. However, postoperative bleeding (drain) is caused by minor vessel damage, which is difficult for the operator to control. 1 Since the use of tranexamic acid can reduce postoperative bleeding, it is likely to be clinically useful. Drain removal timing differs from postoperative bleeding (drain amount) because the criteria for removal depend on the clinician or institution. Therefore, of the two indicators, the postoperative bleeding amount is considered a more objective evaluation indicator.
When intravenous and topical tranexamic acid were compared, some differences were evident. Systemic administration is claimed to exert a local effect because the acid is active principally at the site of injury (not elsewhere in the body); the wound is the only site of fibrinolytic activity. 8 However, we confirmed that intravenous tranexamic acid had no advantage over the topical route in reducing postoperative bleeding or the drain tube dwell time.1,5-7 The topical route may better reduce bleeding (although statistical significance was not attained, given the small number of patients) and the drain tube dwell time. In patients undergoing head-and-neck surgery, a drain tube is frequently used to remove excess blood or fluid and reduce dead space. 21 The tube dwell time is closely related to the length of hospital stay. 22 Therefore, earlier drain removal is associated with faster recovery.6,19 Local tranexamic acid application obviously affords a high concentration in the wound. 6 Topical tranexamic acid reduced surgical bleeding during endoscopic sinus surgery, and seroma and hematoma formation in patients undergoing breast surgery.17,20 These results are consistent with our analysis. However, more clinical trials are needed.
Although tranexamic acid is relatively inexpensive and safe, the side effects include nausea and/or vomiting, orthostatic hypotension (rare), and deep venous thromboembolism (one case, thus very rare). However, recent studies found that tranexamic acid did not in fact cause thromboembolic events.1,23,24 However, given the concern, D-dimer levels after clotting were measured. 7 The levels before and after tranexamic acid treatment did not differ, in line with the results of a previous study. 24 One work reported that very high tranexamic acid doses, or long-term use, might be associated with thromboembolic events. 25 However, perioperative doses of 10–20 mg/kg were safe.23,24
Our study has certain limitations. First, we included patients undergoing resection at various sites, for whom different reconstruction flaps were placed; the extent of head-and-neck drainage may thus have varied. However, the intervention and control groups were all well-matched in such terms; our analyses should not have been affected. 6 Second, we found only six randomized, controlled prospective studies; the sample size was not large. In addition, the studies varied somewhat in terms of tranexamic acid dose timing, amount, and measurement method. A large, randomized, controlled clinical study is required. Third, drainage after head-and-neck surgery removes both blood and serous discharge; tranexamic acid does not affect the level of the latter. This may confound the results. 7
As a result, perioperative tranexamic acid was effective in reducing postoperative bleeding and topical administration might be more effective, but additional research is needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) (2022R1F1A1066232, 2019M3A9H2032424, 2019M3E5D5064110), and by the Ministry of Trade, Industry & Energy (MOTIE, Korea) (20012378).
