Abstract
Objective
To evaluate the safety and efficacy of intravenous tranexamic acid (TXA) administration in anterior cervical discectomy fusion (ACDF) for the treatment of cervical spondylosis in the elderly.
Methods
Data from elderly patients who underwent ACDF between January 2020 and January 2023 were retrospectively reviewed. Patients who received 1 g intravenous TXA administration before skin incision (TXA group) were compared with patients who did not receive TXA (controls). Total and hidden blood loss were calculated, and the following outcomes were recorded: haemoglobin and haematocrit drop, operation time, drainage duration, drain volume, length of hospitalization, coagulation changes, and incidence of complications.
Results
A total of 114 patients were included (TXA group, n = 53 and controls, n = 61). Total blood loss, hidden blood loss, and postoperative drainage volume, haemoglobin and haematocrit drop were significantly lower in the TXA group than the control group. There were no significant differences in operation time, intraoperative blood loss, drainage duration, length of hospitalization, or coagulation function between the two groups. The incidence of complications did not differ significantly between the two groups during 3 months of follow-up.
Conclusions
Intravenous TXA is effective in reducing perioperative blood loss in elderly patients undergoing ACDF without changing the coagulation function or increasing the risk of complications.
Keywords
Introduction
Cervical spondylosis is a common progressive degenerative disease caused by intervertebral disc herniation, facet joint arthritis, and ossification of the posterior longitudinal ligament in the aging spine, resulting in spinal cord compression and leading to a series of clinical symptoms, such as hypaesthesia, soreness, numbness, and muscle weakness.1,2 Anterior cervical discectomy and fusion (ACDF), first proposed by Smith and Robinson in 1958, 3 is a standard treatment procedure for symptomatic cervical spondylosis when conservative management has failed. 4 ACDF has been reported to have good long-term clinical outcomes due to complete decompression, solid fusion, and cervical lordosis recovery.5,6 Minimal trauma, less intraoperative blood loss, and rapid postoperative recovery are also advantages of ACDF. However, in clinical practice, patients who undergo ACDF tend to have lower postoperative haemoglobin levels than anticipated after surgery, particularly in the elderly population.
The concept of hidden blood loss was first proposed by Sehat et al. 7 in 2000, and has since received increasing attention. Blood infiltrating into the soft tissue, remaining in the dead space, and lost due to haemolysis is often ignored by orthopaedic surgeons. 8 Hidden blood loss may exacerbate the postoperative drop in haemoglobin, leading to postoperative anaemia, which may result in a series of complications, particularly in elderly patients. 9 Even in minimally invasive spine procedures, such as percutaneous endoscopic transforaminal discectomy and percutaneous kyphoplasty, where surgical blood loss is almost zero, there is still a lot of hidden blood loss.10,11
Tranexamic acid (TXA) is a synthetic derivative of the amino acid lysine that competitively inhibits the conversion of plasminogen to plasmin, thus preventing fibrin degradation and dissolution of the formed clot. 12 TXA has been proven to reduce perioperative bleeding and the need for blood transfusion, and is commonly used in orthopaedic surgery.13,14 However, to the best of our knowledge, few studies have reported the efficacy and safety of TXA administration in ACDF. Therefore, the aim of the present retrospective study was to evaluate the efficacy of intravenous TXA in reducing total blood loss and hidden blood loss in elderly patients undergoing ACDF, and to examine the safety of TXA by assessing its effects on coagulation function and incidence of thrombotic complications.
Patients and methods
Study population and design
This retrospective study involved consecutive elderly patients who underwent ACDF at the Affiliated Hospital of Jiaxing University between January 2020 and January 2023. The inclusion criteria were as follows: (1) age >65 years; and (2) degenerative cervical spondylosis myelopathy or radiculopathy, and no significant improvement after conservative therapy for 3 months. The exclusion criteria were as follows: (1) history of cervical spine trauma and/or previous cervical surgery; (2) incomplete clinical data and/or follow-up less than 3 months; (3) coagulation dysfunction, and/or a history of thromboembolism or antiplatelet therapy within 6 months; (4) allergy to TXA; (5) patients with severe liver, kidney and/or cardiovascular disease; and/or (6) history of myocardial or cerebral infarction during the previous 6 months. Included patients were divided into two groups based on surgeon preference as they adapted the use of TXA: patients who received a single intravenous dose of 1 g TXA in 100 ml normal saline 15 min prior to skin incision (TXA group) and patients who did not receive TXA (controls). All patients underwent ACDF with the same surgical technique and all surgeries were performed by the same surgeons (Jiang Y, Huang ZH, Zhu JY). A 24 h drainage of <30 ml was considered the criterion for removal of the drainage tube. All patients wore a neck brace for rehabilitation 24 h after surgery and were followed-up for at least 3 months. The study was approved by the Ethics Committee of Affiliated Hospital of Jiaxing University (approval No. 2022-LY-489), and due to the retrospective study design, was exempt from the requirement of formal informed consent. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 15
Data collection
The demographic and clinical data of all patients were collected, including age, sex, height, weight, body mass index (BMI), fusion segment, American Society of Anesthesiologists (ASA) classification, duration of symptoms, comorbidity (hypertension, diabetes mellitus), operative time, intraoperative blood loss, drainage volume, drainage duration and length of hospitalization. In all patients, a complete blood count and coagulation function, including haematocrit (Hct), haemoglobin, activated partial thromboplastin time (APTT), prothrombin time (PT), and international normalized ratio (INR) was recorded before the operation and repeated on postoperative day 1 and 3. If the postoperative haemoglobin level was <70 g/L, the patient received a blood transfusion to maintain the haemoglobin level. Deep vein thrombosis (DVT) was detected by routine ultrasound examination 1 week postoperatively and at 3 months of follow-up, or at any time during the onset of clinical symptoms. Pulmonary embolism was diagnosed based on clinical symptoms and an enhanced chest computed tomography scan.
Calculation of blood loss
Intraoperative blood loss was calculated through the final volume in the suction containers minus the irrigated volume and the dry weight of the soaked sponges and gauze pieces used during the surgery. The patient’s blood volume was calculated according to the Nadler formula: 16 Patient’s blood volume (L) = k1 × height (m)3 + k2 × weight (kg) + k3, (where male: k1 = 0.3669, k2 = 0.03219, k3 = 0.6041; and female: k1 = 0.3561, k2 = 0.03308, k3 = 0.1833). Total blood loss was calculated according to the Gross formula: 17 Total blood loss = patient’s blood volume × ([preoperative Hct–postoperative Hct]/average Hct), where average Hct = the mean of preoperative and postoperative Hct. Hidden blood loss = total blood loss–surgical bleeding–drainage volume.
Outcome measurement
The primary outcomes were total blood loss and hidden blood loss. The secondary outcomes were perioperative haemoglobin and Hct drop, operation time, drainage duration, drain volume, length of hospitalization, coagulation changes, and incidence of complications.
Statistical analyses
The simple size was calculated by G*Power software, version 3.1.9.7 (UCLA Statistical Consulting Group, Los Angeles, CA, USA) and was estimated as 1-β at the power of 80%, α error of 0.05 and enrolment ratio was kept as 1. Based on the primary outcome of total blood loss determined by a preliminary study and considering a 5% dropout rate, 49 patients were required for each subgroup. All data were collected and analysed using IBM SPSS software, version 20.0 (IBM, Armonk, NY, USA). Normally distributed continuous variables are presented as mean ± SD and were analysed using a two-tailed independent samples t-test. Categorical variables are presented as n (%) prevalence and were compared using χ2-test or Fisher’s exact test, as necessary. P < 0.05 was considered to be statistically significant.
Results
Between January 2020 and January 2023, 146 consecutive elderly patients underwent ACDF at the Affiliated Hospital of Jiaxing University, of whom, 32 did not meet the eligibility criteria for this study. Thus, a total of 114 elderly patients who underwent ACDF and met the eligibility criteria were enrolled into this study: 53 patients in the TXA group and 61 patients in the control group (Figure 1). There were no statistically significant between-group differences in demographic and baseline clinical characteristics, such as biological sex, age, BMI, fusion segment, ASA classification, duration of symptoms or comorbidity (Table 1).

Flow chart showing enrolment of patients into the study. TXA, tranexamic acid.
Demographic and clinical data of elderly patients who underwent anterior cervical discectomy fusion to treat cervical spondylosis who either received preoperative tranexamic acid (TXA group) or controls who did not receive TXA.

Data presented as n, n (%) prevalence, or mean ± SD.
ASA, American Society of Anesthesiologists; BMI, body mass index.
No statistically significant between-group differences (P > 0.05; two-tailed independent samples t-test, χ2-test or Fisher’s exact test).
Total blood loss and hidden blood loss on postoperative day 1 were comparable between the two groups: 292.06 ± 111.11 ml and 187.39 ± 109.67 ml, respectively, in the TXA group compared with 332.11 ± 134.30 ml and 217.27 ± 136.44 ml, respectively, in the control group. However, on postoperative day 3, total blood loss and hidden blood loss were significantly lower in the TXA group (352.35 ±125.73 ml and 226.56 ± 117.32 ml, respectively) than in the control group (432.56 ± 128.84 ml and 295.46 ± 118.11 ml, respectively; P < 0.05). Compared with the control group, TXA was found to have decreased total blood loss by 19.6% and hidden blood loss by 23.3%, but there was no significant change in the percentage of hidden blood loss in total blood loss. There were no statistically significant between-group differences in operation time, intraoperative blood loss, drainage duration, length of hospitalization or incidence of complications (Table 2). Postoperative 24 h drainage volume and total postoperative drainage volume were also statistically significantly lower in the TXA group than in the control group (P < 0.05; Table 2). No patient in either group required postoperative blood transfusion.
Clinical data of elderly patients who underwent anterior cervical discectomy fusion to treat cervical spondylosis who either received preoperative tranexamic acid (TXA group) or controls who did not receive TXA.

Data presented as n (%) prevalence or mean ± SD.
POD, postoperative day.
NS, no statistically significant between-group difference (P > 0.05; two-tailed independent samples t-test, χ2-test or Fisher’s exact test).
There were no statistically significant between-group differences in PT, APTT, or INR at the preoperative, postoperative day 1 or postoperative day 3 time-points (Figure 2). In addition, there were no statistically significant between-group differences in haemoglobin and Hct on the preoperative, and postoperative day 1 and 3 time-points (Figure 3). The postoperative drop in haemoglobin and Hct were significantly lower in the TXA group than in the control group (8.02 ± 3.14 g/L versus 10.07 ± 3.31 g/L and 3.09 ± 1.08% versus 3.90 ± 1.28%, respectively, P < 0.05).
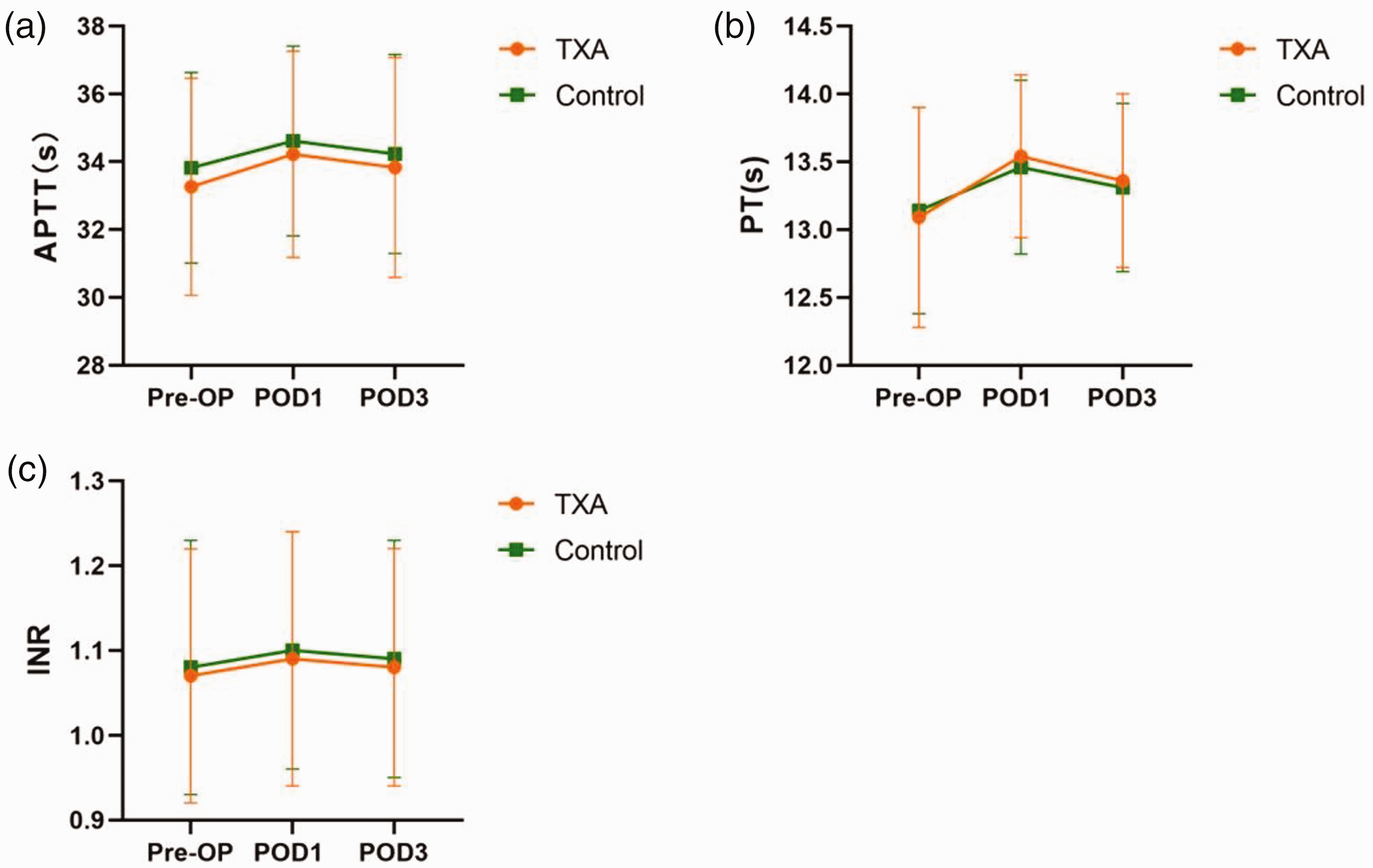
Line graphs of data from elderly patients who underwent anterior cervical discectomy fusion to treat cervical spondylosis who received preoperative tranexamic acid (TXA) or controls who did not receive TXA, showing: (a) activated partial thromboplastin time (APTT); (b) prothrombin time (PT); and (c) international normalized ratio (INR) preoperatively (Pre-OP), postoperative day 1 (POD1) and postoperative day 3 (POD3). Data presented as mean ± SD.

Line graphs of data from elderly patients who underwent anterior cervical discectomy fusion to treat cervical spondylosis who either received preoperative tranexamic acid (TXA group) or controls who did not receive TXA, showing: (a) haematocrit (Hct) levels; and (b) haemoglobin (Hb) levels preoperatively (Pre-OP), postoperative day 1 (POD1) and postoperative day 3 (POD3). Data presented as mean ± SD.
Discussion
Reducing perioperative blood loss is a crucial issue in surgery because massive blood loss leads to postoperative anaemia and increases the need for blood transfusion. Allogeneic blood transfusion is associated with a risk of infection, anaphylactic reaction, haemolytic transfusion reaction, prolonged length of hospital stay, and delayed functional recovery, particularly in elderly patients.18,19 Hidden blood loss accounts for a significant portion of the total blood loss, and has attracted attention from spinal surgeons, including Smorgick et al., 20 who reported that hidden blood accounted for 42% of total blood loss during posterior spinal decompression and fusion. Wen et al. 21 reported a mean hidden blood loss of 261 ml, which was 50% of the total blood loss following anterior cervical fusion surgery, and ASA classification and gender were independent risk factors. In the present study, cervical spondylosis in elderly patients undergoing ACDF was found to be significantly associated with perioperative hidden blood loss. The mean perioperative hidden blood loss in control patients was 295.46 ± 118.11 ml, which accounted for 68.15% of the total blood loss. The present results of hidden blood loss were higher than those reported by Wen et al., 21 possibly because the present study comprised an elderly population. Loose muscle space in elderly patients provides a larger space for hidden blood loss, and decline in body function also increases the risk of hidden blood loss. Therefore, hidden blood loss is a problem that requires urgent attention and cannot be ignored, particularly in elderly patients. Conversely, the present study showed lower blood loss than that reported by Cai et al., 22 which may have been due to shorter operation time in the present study resulting in less tissue trauma and reduced blood loss. In addition, the experience and habits of the surgical team also play an important role in the variation in results. Hidden blood loss in ACDF is mainly due to the haemolytic reaction, blood infiltration into the soft tissue, and residual blood in the dead space created by discectomy and tissue separation. Several studies have confirmed that age is an independent risk factor for hidden blood loss,23–25 and elderly patients are less tolerant to postoperative anaemia due to perioperative massive blood loss, which results in poor physical condition. Therefore, reducing perioperative blood loss during orthopaedic surgery in elderly patients is particularly important.
In recent years, TXA has been widely used to reduce blood loss in orthopaedic surgery, and its efficacy and safety in countering the risk of perioperative blood loss have been shown. 26 Tsutsumimoto et al. 27 reported that 15 mg/kg intravenous TXA reduced hidden blood loss by 89.8 ml (264.1 ± 75.1 ml versus 353.9 ± 60.8 ml, P < 0.01) in cervical laminoplasty during the first postoperative 40 h. Another study showed that hidden blood loss and total blood loss were significantly lower in patients undergoing percutaneous endoscopic transforaminal lumbar interbody fusion receiving intravenous administration of 1 g TXA than in control patients. 28 The most commonly used delivery route for TXA in spinal surgery is intravenous, but there are no guidelines to clarify the dose of TXA used. Several studies concluded that the dose of TXA in single intravenous application ranges from 15 to 30 mg/kg or 1 to 2 g in spinal surgery.29–32 The present study selected the 1 g TXA dose, because it was the easiest to administer and has been verified previously as providing effective haemostatic control when administered before an operation.19,28,33 Due to haemodynamic parameters stabilising on postoperative day 2 or 3, total blood loss and hidden blood loss were calculated based on Hct values preoperatively and on postoperative day 3. 20 To the best of our knowledge, this is the first study of TXA for postoperative hidden blood loss in elderly patients undergoing ACDF for cervical spondylosis, and the study results showed that a 1 g dose of TXA decreased total blood loss by 19.6% and hidden blood loss by 23.3%, but there was no significant change in the percentage of hidden blood loss in total blood loss. TXA may promote coagulation, quickly act on the surgical site and effectively inhibit the blood clot breakdown, reducing small blood vessel bleeding and muscle tissue oozing, thereby decreasing the volume of hidden blood loss. In the present study, surgical bleeding was comparable between the two groups (66.51 ± 22.07 ml versus 71.97 ± 21.53 ml), which was also seen in the study by Steinle et al., 34 (131.73 ± 58.89 ml versus 138.09 ± 53.21 ml, P = 0.207). Postoperative drainage is an important component of total blood loss, and the total drainage volume was reduced by almost 10% in the TXA group in the present study. However, there was no difference in the time of drain removal, possibly because of the low drainage volume in ACDF. The present results also showed that the postoperative haemoglobin and Hct drops were significantly different between the two groups, but there was no difference in the levels of haemoglobin and Hct on the first and third postoperative days. Large amounts of haemoglobin loss may lead to the development of anaemia, and the administration of TXA might reduce 2 g/L haemoglobin loss in ACDF during the perioperative period. In the present study, the drop in haemoglobin was lower than reported by Steinle et al., 34 possibly because the enrolled patients in their study had greater height and weight and higher blood volume.
Continuous activation of the coagulation process after major orthopaedic surgery may last up to 4 weeks, and the risk of postoperative DVT formation might last up to 3 months. 35 A potentially increased risk of thrombosis is the main concern in the clinical application of TXA. Ko et al. 36 reported a comparable incidence of thromboembolism after using TXA in lumbar fusion surgery of 0.8% versus 1.2% in the control group. Another study concluded that intravenous TXA was not associated with increased risk of thromboembolism. 37 In the present study, two patients in the TXA group experienced intramuscular venous thrombosis during the 3-month follow-up period, while one case of DVT and one case of wound hematoma were found in the control group. Intravenous administration of 1 g TXA in elderly patients undergoing ACDF did not significantly increase the risk of thrombotic complications, and coagulation function did not show any significant difference versus controls during the perioperative period. We speculated that TXA-reduced blood loss relies on inhibition of the fibrinolytic system rather than promotion of coagulation. At the same time, the use of TXA during the perioperative period of ACDF did not shorten the length of hospitalization, which is similar with the results of a previous study. 34 The present results indicate that the administration of TXA in elderly patients is relatively safe.
The present results may be limited by several factors. First, this was a single-centre, retrospective study with a relatively small sample size. Secondly, patients were only followed for 3 months after surgery, therefore, the safety of long-term TXA use cannot be accurately concluded. Thus, further multicentre randomized controlled trials with larger sample sizes are needed to confirm the efficacy and long-term safety of TXA for anterior cervical discectomy fusion.
Conclusion
Intravenous administration 1.0 g of TXA is effective in reducing total blood loss, hidden blood loss, total drain output, and the degree of haemoglobin and Hct drop, without increasing the risk of thrombotic complications, but had no significant effect on intraoperative blood loss, shorter hospitalization, or coagulation function in elderly patients undergoing ACDF.
Footnotes
Acknowledgements
The authors thank all patients for their participation.
Author contributions
Yi Jiang: conceptualization. Zhenqi Lou and Yi Jiang: writing-original draft, and performed the procedures. Jinyu Zhu: writing-review and editing. Zhihai Huang and Jieyang Zhu: methodology. Kanling Jiang and Lan Lai: data curation and data analysis.
Data availability statement
The datasets generated during this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was supported by Jiaxing Science and Technology Project (2021AY30020, 2021AD30165) and Jiaxing Key Discipline of Medicine-Orthopaedics (2023-ZC-012).
