Abstract
Background
Total knee arthroplasty is often complicated by perioperative blood loss. Tourniquets reduce visible bleeding; however, prolonged use may increase hidden blood loss. Intra-articular tranexamic acid may enhance hemostasis without increasing the risk.
Methods
We retrospectively analyzed 85 unilateral primary total knee arthroplasties for osteoarthritis (September 2022 to March 2025). All patients received 1 g intravenous tranexamic acid before incision and were nonrandomly assigned to either of three groups based on evolving institutional protocols and surgeon discretion during the study period as follows: (a) Group A (full-duration tourniquet, n = 28); (b) Group B (half-duration tourniquet, n = 27); and (c) Group C (half-duration tourniquet plus 2 g intra-articular tranexamic acid, n = 30). The primary outcome was postoperative day 3 hemoglobin level; secondary outcomes were calculated blood loss, transfusion, pain scores, early Hospital for Special Surgery knee score (function), and perioperative complications.
Results
A total of eighty-five unilateral primary total knee arthroplasties were analyzed (Group A, n = 28; Group B, n = 27; and Group C, n = 30). Baseline variables were comparable across the three groups. On postoperative day 3, hemoglobin level was highest in Group C (10.97 ± 0.69 g/dL) compared with those in Groups A (9.89 ± 0.80) and B (10.05 ± 0.58) (overall p < 0.001; Groups C vs. A/B, both p < 0.001). Total blood loss decreased stepwise (Group A, 1403 ± 136 mL; Group B, 1163 ± 124 mL; and Group C, 770 ± 121 mL; all pairwise p < 0.001); a similar pattern was observed for hidden blood loss (Group A, 1388 ± 137 mL; Group B, 929 ± 128 mL; and Group C, 529 ± 121 mL; all pairwise p < 0.001). Intraoperative blood loss was lowest in Group A (median 15 mL) compared with those in Groups B (220 mL) and C (230 mL) (p < 0.001). Transfusion rates were 28.6% in Group A, 14.8% in Group B, and 3.3% in Group C (overall p = 0.024). Postoperative pain, as indicated by visual analog scale scores, was lowest in Group C (scores on postoperative days 1 and 3: 4.95 ± 0.85 and 3.57 ± 0.61, respectively) compared with those in Groups B (scores on postoperative days 1 and 3: 6.02 ± 1.22 and 4.63 ± 0.87, respectively) and A (scores on postoperative days 1 and 3: 7.86 ± 0.85 and 6.54 ± 1.00, respectively) (both time points p < 0.001). Based on a comparison of the Hospital Special Surgery knee scores of the three groups, Group C demonstrated superior early functional outcomes (scores: 62.33 ± 2.71 at postoperative 1 week and 83.73 ± 3.14 at postoperative 1 month) compared with Groups B (scores: 56.81 ± 3.11 at postoperative 1 week and 79.11 ± 3.64 at postoperative 1 month) and A (47.86 ± 3.39 at postoperative 1 week and 72.36 ± 3.72 at postoperative 1 month) (all p < 0.01). However, the 6-month Hospital for Special Surgery knee scores of the three groups were comparable (Group A: 89.93 ± 2.42; Group B, 89.96 ± 2.53; and Group C, 91.13 ± 2.06; p = 0.09). Overall complication rates did not differ between the three groups (Group A, 7.1%; Group B, 7.4%; Group C, 3.3%; p = 0.734).
Conclusion
In total knee arthroplasty patients, the combination of a half-duration tourniquet with intra-articular tranexamic acid may provide superior perioperative blood management compared with a full-duration tourniquet or half-duration tourniquet alone. This strategy may reduce total and hidden postoperative blood loss, reduce hemoglobin loss, and lower transfusion rates. Additionally, it may reduce postoperative pain and promote earlier recovery of knee function. In addition, this treatment combination did not cause a significant increase in complications, including thrombosis or infection.
Introduction
Total knee arthroplasty (TKA) is considered the gold-standard procedure for end-stage knee diseases, such as knee osteoarthritis (KOA). With longer life expectancy and rising KOA prevalence, an increasing number of TKA procedures are being performed. 1 However, substantial perioperative blood loss is common in TKA. Total blood loss (TBL) during and after surgery may exceed 1000 mL in primary TKA. 2 Excessive blood loss can cause postoperative anemia, raise transfusion needs, impair wound healing, and slow down recovery. 3 Therefore, effective control of bleeding during and after TKA has become a key clinical focus and aligns with the Enhanced Recovery After Surgery (ERAS) principle. 4
A tourniquet is a common method used for limiting blood loss in the field of orthopedics. Inflation temporarily blocks limb blood flow, which reduces visible intraoperative bleeding and provides a dry, clear field. It may also shorten the operative time and improve the cement interface. 5 However, no consensus exists on the timing and duration of use. 6 Several studies have shown that full-duration use reduces intraoperative bleeding. It does not reduce the TBL or transfusion requirement and is linked to more postoperative pain, limb swelling, and higher deep-vein thrombosis (DVT) risk. 7 In contrast, shorter use promotes early recovery and lowers the complication risk. 8 Based on this evidence, a half-duration/restrictive strategy has been proposed, with brief inflation only during key steps, such as implant insertion and closure, to maintain a clear and safe operative field. 9 Controlled studies have suggested that compared with full-duration use, this approach further reduces perioperative TBL and eases early pain, consistent with the ERAS principles. 10 Notably, the mechanisms of hidden blood loss (HBL) are complex, and only shortening the tourniquet time may not completely resolve this problem.
Tranexamic acid (TXA) is an established antifibrinolytic drug. By blocking plasminogen activation on fibrin, TXA stabilizes clots and reduces blood loss and transfusion risk. 11 In TKA, TXA is safe and effective when used intravenously, as an intra-articular injection, or as periarticular infiltration. It is especially useful for controlling postoperative HBL.12,13 Higher local doses raise drug levels at the surgical site, suppress fibrin breakdown, and reduce joint bleeding, without increasing thrombotic events such as DVT. 14 TXA is also linked to less postoperative inflammation and drainage; some studies have reported lower inflammatory marker levels and better functional recovery. 15 Based on this strong evidence, TXA has been included in standardized perioperative care for joint replacement. 16 Therefore, combining TXA with an optimized tourniquet strategy may further improve bleeding control and recovery in TKA.
Tourniquet use and TXA each offer advantages for perioperative blood management (PBM) in TKA; however, clinical evidence to guide an optimal combined strategy that simultaneously balances surgical field exposure, blood-loss control, and postoperative recovery remains limited. The aim of this study was to compare three perioperative blood-management strategies: (a) full-duration tourniquet; (b) half-duration tourniquet; and (C) half-duration tourniquet combined with local TXA and evaluate their effects on perioperative blood loss control and early postoperative functional recovery. We hypothesized that compared with full-duration or half-duration tourniquet use alone, use of a half-duration tourniquet plus local TXA strategy further reduces postoperative blood loss and promotes early rehabilitation. We expect that our findings will provide additional clinical evidence to support PBM and ERAS principles in TKA.
Methods
Study design and participants
This was a single-center retrospective cohort study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 17 Patients with end-stage KOA who underwent primary unilateral TKA at our hospital from September 2022 to March 2025 were included. In total, 98 cases were identified. Five patients with final preoperative hemoglobin (Hb) level <12.0 g/dL and eight with traumatic or rheumatoid arthritis were excluded, resulting in a final sample of 85 KOA patients. All patients received 1 g intravenous (IV) TXA before incision and were nonrandomly assigned to either of three groups based on evolving institutional protocols and surgeon discretion during the study period: (a) Group A (full-duration tourniquet, n = 28); (b) Group B (half-duration tourniquet, n = 27); and (c) Group C (half-duration tourniquet plus intra-articular TXA 2 g, n = 30). Baseline variables, including sex, age, body mass index (BMI), preoperative visual analog scale (VAS) score, 18 preoperative Hospital for Special Surgery Knee Score (HSS), 19 and preoperative Hb—did not differ between the three groups (all p > 0.05). The groups were comparable. Baseline characteristics of the three groups were as follows:
Inclusion and exclusion criteria
The following inclusion criteria were applied: (a) age, 60–80 years; (b) primary KOA and scheduled for primary unilateral TKA; (c) No anticoagulant or antiplatelet use within 7 days preoperatively and last preoperative Hb level ≥12.0 g/dL; (d) American Society of Anesthesiologists (ASA) class I–II; and (e) Complete records, imaging, and follow-up, with ≥6 months of follow-up.
The following exclusion criteria were applied: (a) age <60 or >80 years; (b) history of DVT or PE, known coagulopathy or hypercoagulable state, or inability to stop anticoagulant/antiplatelet therapy due to comorbidities; (c) nonprimary knee disease (e.g. rheumatoid, post-traumatic, or post-infectious arthritis; gout; and psoriatic arthritis); (d) severe knee deformity or marked ligament insufficiency requiring a constrained or hinged implant; (e) prior major ipsilateral knee surgery that could affect outcomes (e.g. revision TKA, high tibial osteotomy, and unicompartmental arthroplasty); (f) simultaneous bilateral TKA or staged bilateral TKA with an interval <3 months; (g) active infection or poor skin at the incision site; (h) ASA class ≥III or severe cardiac, cerebrovascular, hepatic, or renal dysfunction; (i) known allergy or contraindication to TXA (e.g. prior seizures, disseminated intravascular coagulation, active thromboembolism, and severe renal impairment); and (j) inability to complete at least 6 months of follow-up.
Surgical procedure and perioperative management
This retrospective study applied no experimental changes to the clinical care protocol. Data were obtained from medical records and the anesthesia and laboratory information systems. All TKAs were performed under general anesthesia by senior surgeons from the same team, using a medial parapatellar approach and a cemented, stabilized-type prosthesis of the same brand (Zimmer Biomet, USA). A pneumatic tourniquet was used intraoperatively with a pressure of 60 kPa. ERAS-based care was used as follows: administration of 1 g IV cefazolin 30 min before incision, no routine drains, knee motion started on postoperative day (POD) 1, and removal of elastic bandage at 24 h. Hemostasis protocols were as follows. Group A: the tourniquet was inflated before the incision and released after skin closure; Group B: torniquet was inflated before implant insertion and released after closure; Group C: same as for Group B plus 2 g intra-articular TXA after skin closure (single injection diluted in normal saline). All groups received 1 g IV TXA 20–30 min before the incision, diluted in 100 mL normal saline as baseline antifibrinolytic therapy. No additional postoperative IV TXA was administered during hospitalization or the follow-up period. Postoperatively, anticoagulation followed institutional protocols. First, 4100 IU nadroparin calcium was subcutaneously administered once daily during hospitalization; then, 10 mg rivaroxaban was administered once daily for 4 weeks after discharge. Lower limbs were monitored; duplex ultrasound was performed for suspected thrombosis (e.g. swelling and pain) to screen for DVT. A restrictive transfusion strategy was used: transfusion was indicated at Hb <7.0 g/dL or at 7.0–8.0 g/dL in the presence of symptoms or cardiovascular disease. Transfusion decisions were made by the attending anesthesiologist and surgeon based on the restrictive threshold together with clinical signs of inadequate oxygen delivery (e.g. hemodynamic instability and symptomatic anemia) and comorbid cardiovascular disease. Intraoperative and postoperative transfusions were recorded, including occurrence and timing of the first transfusion.
Data collection
Data were retrospectively extracted from electronic medical records, anesthesia records, and the laboratory system; patients with complete data who had been followed up for ≥6 months were included. The primary endpoint was the POD3 Hb level. Secondary endpoints were as follows: (a) TBL, calculated using the Gross method, with estimated blood volume (BV) derived using Nadler’s formula; (b) HBL, defined as TBL − intraoperative blood loss (IBL); (c) IBL; (d) transfusion rate; (e) VAS scores on POD1 and POD3; (f) HSS (function) at 1 week, 1 month, and 6 months postoperatively; and (g) complications, including DVT/PE, wound complications, and 30-day readmission or death.
Blood loss assessment
Estimated BV (L) was calculated using Nadler’s formula:
20
2. TBL (mL) was estimated using the Gross method:
21
where 3. HBL (mL) was defined as HBL =TBL − IBL. Because TBL is transfusion-corrected, the transfused volume is not added again when calculating HBL.
Data processing
Data were analyzed using Statistical Package for Social Sciences (SPSS) software (version 27.0, IBM Corp., Armonk, NY, USA). For continuous data, normality was tested using the Shapiro–Wilk test. Normally distributed data were reported as mean ± SD (x̄ ± s); skewed data were reported as median (interquartile range (IQR) values). Categorical variables were presented as counts (percentages). For between-group comparisons, methods were chosen according to data type. If data were normal with equal variances, one-way analysis of variance (ANOVA) was performed, followed by least significant difference (LSD) for pairwise tests when the ANOVA results were significant. If data were normal with unequal variances, Welch’s ANOVA was used, followed by Games–Howell when the ANOVA results were significant. Repeated-measures data were analyzed using repeated-measures ANOVA. Sphericity was checked using Mauchly’s test, and degrees of freedom were corrected when needed. Time, group, and interaction effects were assessed. Within-group comparisons were performed using paired t-tests or the Wilcoxon signed-rank test with Bonferroni adjustment. Non-normal continuous data were compared using the Kruskal–Wallis test; if the results were significant, pairwise Mann–Whitney U tests with Bonferroni adjustment were performed. Categorical variables were compared using chi-square test or Fisher’s exact test. All tests were two-sided. The significance level was set at α = 0.05, and p < 0.05 was considered statistically significant.
Results
Patient characteristics
From September 2022 to March 2025, 98 primary TKA patients were identified; after exclusions, 85 were included. All patients completed at least 6 months of follow-up. Baseline characteristics are presented in Table 1. No significant differences were observed among the three groups in terms of sex, age, BMI, preoperative VAS score, preoperative HSS, or preoperative Hb levels (p-values in Table 1). The proportion of female patients was 67.9% in Group A, 66.7% in Group B, and 63.3% in Group C (χ2 = 0.143, p = 0.931).
Preoperative baseline characteristics of the three groups (mean ± SD unless stated otherwise).

Values are presented as mean ± SD for continuous variables and count (female) for sex. Between-group comparisons were performed using one-way ANOVA for continuous variables (reported as F) and chi-square test for sex (reported as χ2).
BMI: body mass index; Hb: hemoglobin; VAS: visual analog scale; HSS: Hospital for Special Surgery knee score; A: full-duration tourniquet; B: half-full-duration tourniquet; C: half-full-duration tourniquet plus intra-articular tranexamic acid (2 g); ANOVA: analysis of variance.
Changes in Hb levels
Preoperative Hb levels were similar in all three groups (Group A, 13.55 ± 1.15 g/dL; Group B, 13.19 ± 0.92 g/dL; and Group C, 13.24 ± 0.97 g/dL; F = 0.996, p = 0.374). By POD3, Hb levels had fallen significantly in all groups (all p < 0.001), with significant between-group differences (F = 20.369, p < 0.001). The mean POD3 Hb level in Group C was 10.97 ± 0.69 g/dL. The average POD3 Hb level of Group C was significantly higher than those of Groups A (9.89 ± 0.80 g/dL, p < 0.001) and B (10.05 ± 0.58 g/dL, p < 0.001). The difference in the mean POD3 Hb levels between Groups A and B was not significant (p = 0.07). Together, these data suggest that a half-duration tourniquet plus local TXA offers a clear advantage in terms of limiting HBL and reducing the postoperative reduction in Hb levels. Detailed data are presented in Table 2.
Comparison of the hemoglobin levels of the three groups at two time points (x̄ ± s, g/dL).

Data are presented as mean ± SD values. Overall comparison was performed using one-way ANOVA (F). Superscript letters indicate between-group differences evaluated using post-hoc LSD tests (α = 0.05); groups with the same letter were not significantly different from each other, while groups with different letters were significantly different from each other. Exact pairwise p-values for POD3 Hb: A vs. B, p = 0.07; A vs. C, p < 0.001; and B vs. C, p < 0.001.
Hb: hemoglobin; POD3: postoperative day 3. A: full-duration tourniquet; B: half-full-duration tourniquet; C: half-full-duration tourniquet plus intra-articular tranexamic acid (2 g); ANOVA: analysis of variance; LSD: least significant difference.
Operative time and tourniquet time
There were significant between-group differences in the operative times of the three groups (F = 89.18, p < 0.001). Group A had the shortest average operative time (82.21 ± 1.77 min), which was shorter than those of Groups B (88.04 ± 2.41 min) and C (88.43 ± 1.65 min) (both p < 0.001). No between-group difference was found for Group B vs. Group C (p = 0.448). The between-group difference for the average tourniquet time was more marked (F = 3144.47, p < 0.001). Group A had the longest tourniquet time (82.21 ± 1.77 min), longer than those for Groups B (34.93 ± 2.97 min) and C (35.00 ± 2.83 min) (both p < 0.001). The average tourniquet times of Groups B and C were comparable (p = 0.914). The half-duration strategy reduced the total tourniquet time by approximately 57%. Despite the added local TXA step, Group C did not demonstrate a longer operative time than Group B (p = 0.448). These data indicate that, compared with a full-duration tourniquet, a half-duration tourniquet markedly shortens the inflation time but is associated with a longer overall operative time. Thus, adding local TXA does not prolong the procedure duration compared with half-duration alone. Detailed data are presented in Table 3.
Comparison of the operative time and tourniquet time between the three groups (x̄ ± s, min).
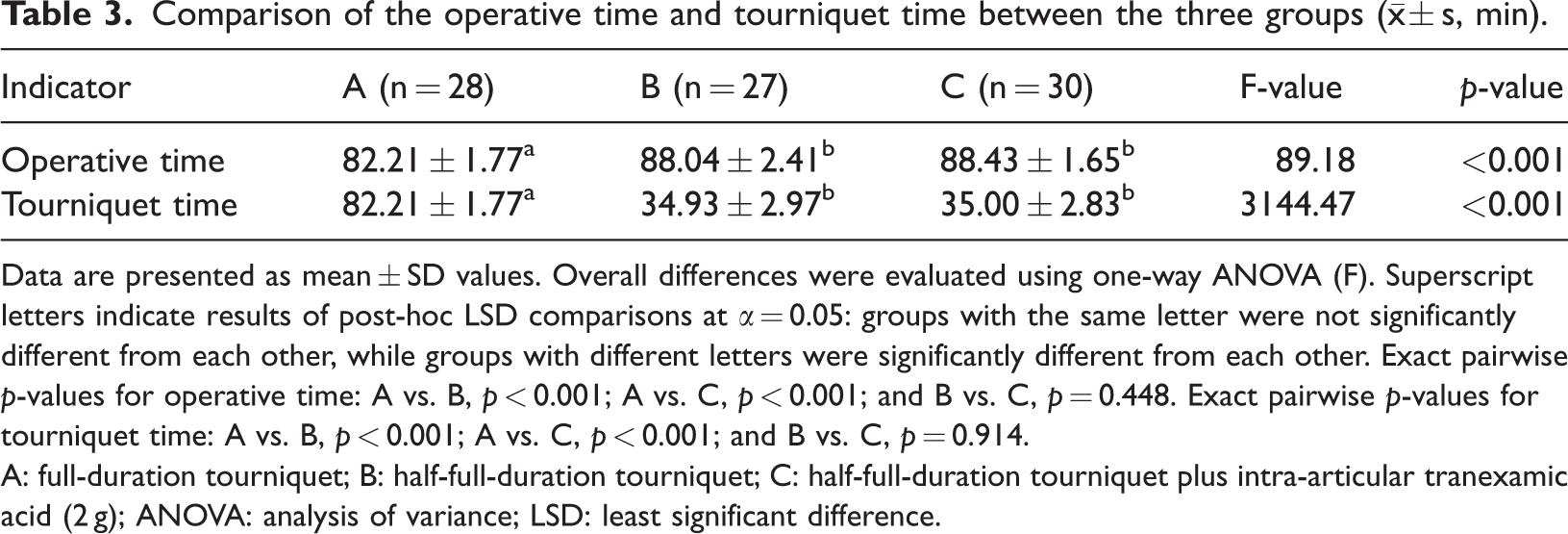
Data are presented as mean ± SD values. Overall differences were evaluated using one-way ANOVA (F). Superscript letters indicate results of post-hoc LSD comparisons at α = 0.05: groups with the same letter were not significantly different from each other, while groups with different letters were significantly different from each other. Exact pairwise p-values for operative time: A vs. B, p < 0.001; A vs. C, p < 0.001; and B vs. C, p = 0.448. Exact pairwise p-values for tourniquet time: A vs. B, p < 0.001; A vs. C, p < 0.001; and B vs. C, p = 0.914.
A: full-duration tourniquet; B: half-full-duration tourniquet; C: half-full-duration tourniquet plus intra-articular tranexamic acid (2 g); ANOVA: analysis of variance; LSD: least significant difference.
Blood loss
Differences in visible IBL and postoperative TBL and HBL were significant among the three groups (Table 4). Group A had lesser IBL than the other two groups (median values: Group A = 15 mL, Group B = 220 mL, and Group C = 230 mL; Kruskal–Wallis H = 57.414, p < 0.001). This indicates that effective control was achieved with a full-duration tourniquet. Groups B and C had similar median values (p = 0.091). Group A had the highest postoperative TBL (1403 ± 136 mL), followed by Group B (1163 ± 124 mL), and Group C (770 ± 121 mL). The group effect was significant (ANOVA, F = 183.683, p < 0.001), and all pairwise comparisons were significant (all p < 0.001). HBL (TBL − IBL) followed the same pattern, Group A (1388 ± 137 mL) had the highest HBL, followed by Groups B (929 ± 128 mL) and C (529 ± 121 mL) (F = 322.538, all pairwise p < 0.001). These findings show that shortening the tourniquet time and applying TXA both help reduce HBL, with the combined approach being most effective. Differences in the POD3 Hb levels mirrored the TBL pattern; Hb levels were higher in Group C than in Groups A and B, as described above. Thus, a full-duration tourniquet reduces intraoperative bleeding; however, substantial postoperative HBL offsets this benefit. Use of a half-duration tourniquet lowers TBL and HBL compared with the use of a full-duration tourniquet. Local administration of TXA in combination with a half-duration tourniquet further reduces the perioperative TBL by approximately one-third compared with the use of a half-duration tourniquet alone, with significant differences between both groups.
Comparison of blood loss (mL) among the three groups.

Superscript letters indicate the results of between-group comparisons performed using LSD or Mann–Whitney U tests, α = 0.05; groups with the same letter were not significantly different from each other, while groups with different letters were significantly different from each other. Total and hidden blood loss were analyzed using one-way ANOVA; intraoperative blood loss was analyzed using the Kruskal–Wallis test.
Pairwise p-values for TBL: A vs. B <0.001, A vs. C <0.001, B vs. C <0.001; IBL: A vs. B <0.001, A vs. C <0.001, B vs. C = 0.091; HBL: A vs. B <0.001, A vs. C <0.001, B vs. C <0.001.
TBL: total blood loss; IBL: intraoperative blood loss; HBL: hidden blood loss; A: full-duration tourniquet; B: half-full-duration tourniquet; C: half-full-duration tourniquet plus intra-articular tranexamic acid (2 g); ANOVA: analysis of variance; LSD: least significant difference.
Transfusion
A total of thirteen perioperative allogeneic transfusions were recorded, including 8 in Group A (28.6%), 4 in Group B (14.8%), and 1 in Group C (3.3%). The overall difference in transfusion rates among the groups was significant (Fisher–Freeman–Halton exact test, p_exact = 0.024) (Table 5). Pairwise tests showed a higher rate in Group A than in Group C (Fisher’s exact test, p = 0.011), while differences between Groups A and B and Groups B and C were not significant (p = 0.329 and 0.179, respectively). Regarding timing, in Group A, 6 of 8 transfusions occurred intraoperatively or within 24 h, and 2 occurred on PODs 2–3; in Group B, 3 of 4 transfusions occurred intraoperatively or within 24 h and 1 on PODs 2–3; the single event in Group C occurred intraoperatively or within 24 h postoperatively (Table 6). Thus, most transfusions occurred intraoperatively or during the early postoperative period. Combined hemostatic strategy (Group C) was associated with a lower need for transfusion. Lower transfusion exposure may reduce transfusion-related complications and medical costs. Although the preoperative Hb levels of all included patients were ≥12 g/dL, the higher TBL and HBL—particularly in the full-duration tourniquet group—were associated with greater early postoperative reduction in Hb levels, which explains the observed transfusion events.
Comparison of the transfusion rates (%) among the three groups.

Superscript letters indicate the results of between-group comparisons; groups with the same letter were not significantly different from each other, while groups with different letters were significantly different from each other. The overall comparison was performed used the Fisher–Freeman–Halton exact test (two-sided; exact p = 0.024).
Letters were assigned based on three 2 × 2 Fisher’s exact tests with Bonferroni correction (α_adj = 0.017): A vs. B, p = 0.329; A vs. C, p = 0.011; and B vs. C, p = 0.179.
A: full-duration tourniquet; B: half-full-duration tourniquet; C: half-full-duration tourniquet plus intra-articular tranexamic acid (2 g).
Time of first transfusion across postoperative days in the three groups.
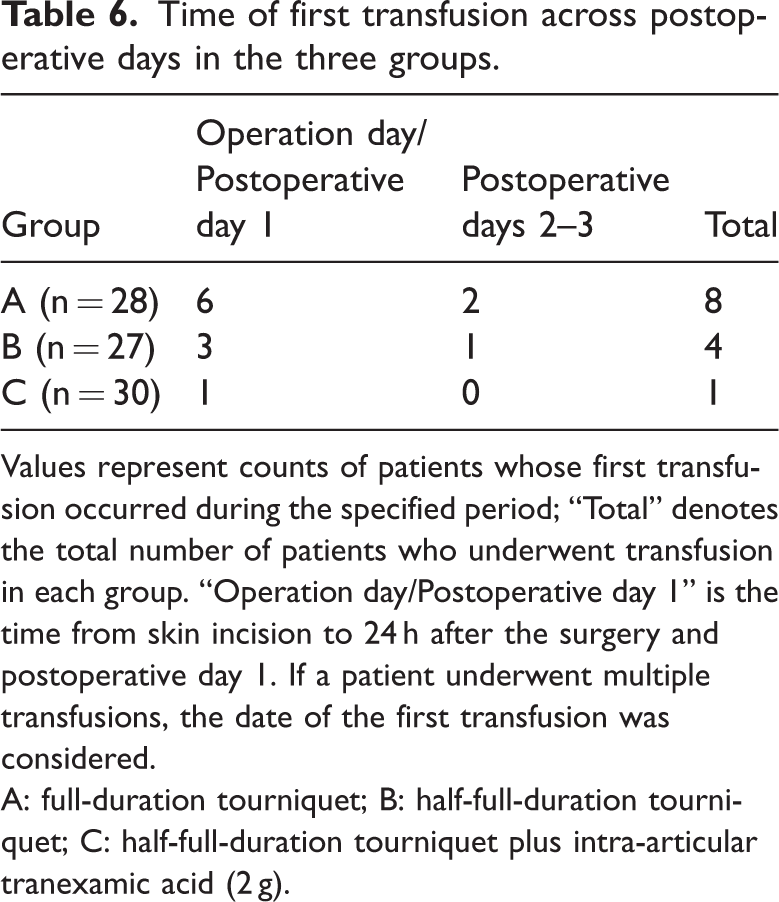
Values represent counts of patients whose first transfusion occurred during the specified period; “Total” denotes the total number of patients who underwent transfusion in each group. “Operation day/Postoperative day 1” is the time from skin incision to 24 h after the surgery and postoperative day 1. If a patient underwent multiple transfusions, the date of the first transfusion was considered.
A: full-duration tourniquet; B: half-full-duration tourniquet; C: half-full-duration tourniquet plus intra-articular tranexamic acid (2 g).
Pain and functional recovery
Early trends in pain VAS score and HSS (function) differed among the three groups. The preoperative VAS score and HSS did not differ across the groups (p > 0.05), indicating comparable baseline scores. Group A demonstrated the highest scores for postoperative pain, consistent with the longest tourniquet time. In Group A, the VAS score increased from ∼6 preoperatively to ∼8 on POD1 and then returned to ∼6 on POD3. Group B showed a gradual decline, and Group C showed the largest reduction. Between-group differences in VAS scores on PODs 1 and 3 were significant (F = 69.58 and 91.47, respectively, both p < 0.001). The average VAS score of Group C was lower than that in Group B, which was lower than that in Group A (all pairwise p < 0.001). Repeated-measures ANOVA showed significant group effects and a group × time interaction (p < 0.01), indicating different impacts on pain relief; Group C achieved the best control, followed by Group B (Table 7). HSSs (function) increased over time in all three groups (time effect p < 0.01). At 1 week and 1 month postoperatively, the between-group differences were significant (both p < 0.01). Group C had the highest HSS (function), Group B had an intermediate average score, and Group A had the lowest score. At 1 week postoperatively, the HSS rose by ∼5 points in Group A versus ∼15 in Groups B and C. The score of Group C was higher than that of Group B (p < 0.01), and the score of Group B was higher than that of Group A (p < 0.01). At 6 months postoperatively, the HSS did not differ among the groups (p > 0.05), and all patients achieved good functional recovery (Table 8). These results indicate that the combined strategy significantly promoted early functional recovery, with better knee function within 1 month than with a full-duration tourniquet. Although long-term functional recovery was similar across groups, less perioperative pain and anemia may enhance early rehabilitation engagement and satisfaction.22,23
Preoperative and postoperative VAS scores (x̄ ± s, points) among the three groups.

Data are presented as mean ± SD values. Superscript letters indicate between-group comparisons within the same row; groups with the same letter were not significantly different from each other, while groups with different letters were significantly different from each other. Post-hoc comparisons were performed using LSD/Games–Howell with Bonferroni adjustment (α = 0.05). Repeated-measures ANOVA: time effect F = 309.43, p < 0.01; group effect F = 35.72, p < 0.01; time × group interaction F = 369.06, p < 0.01. Within-group time effects: A, F = 308.76; B, F = 220.36; C, F = 77.16; all p < 0.001.
VAS: visual analog scale; POD3: postoperative day 3; POD1: postoperative day 1; A: full-duration tourniquet; B: half-full-duration tourniquet; C: half-full-duration tourniquet plus intra-articular tranexamic acid (2 g); ANOVA: analysis of variance; LSD: least significant difference.
Comparison of the preoperative and postoperative HSSs (x̄ ± s, points) among the three groups.

Data are presented as mean ± SD values. Superscript letters indicate between-group comparisons within the row; groups with the same letter were not significantly different from each other, while groups with different letters were significantly different from each other. Post-hoc comparisons were made using the LSD test (α = 0.05).
Repeated-measures ANOVA: time effect F = 4787.89, p < 0.01; group effect F = 39.73, p < 0.01; time × group interaction F = 640.74, p < 0.01. Within-group time effects: A, F = 1986.15; B, F = 1316.39; C, F = 1379.76; all p < 0.01.
HSS: Hospital for Special Surgery knee score; unit: points. A: full-duration tourniquet; B: half-full-duration tourniquet; C: half-full-duration tourniquet plus intra-articular tranexamic acid (2 g); ANOVA: analysis of variance; LSD: least significant difference.
Postoperative complications
Group A had 2 events (7.1%), Group B had 2 (7.4%), and Group C had 1 (3.3%). The overall difference was not significant (Fisher–Freeman–Halton exact test, p = 0.734) (Table 9). Systemic complications occurred once in each group: one patient developed DVT in Group A, and there was one cardiovascular event in Groups B and C; no incidence of pulmonary embolism was observed. Surgery-related events included poor wound healing (n = 1) in Group B; no wound infection, hematoma, or stiffness occurred in any group. Other events included nerve injury (n = 1) in Group A (common peroneal nerve palsy); no periprosthetic fracture or persistent pain occurred in any group. No 30-day readmission or 30-day mortality occurred in any group.
Postoperative complications in all three groups (n, %)

Data are presented as counts (percentages). The p-value was obtained by performing the two-sided Fisher–Freeman–Halton exact test (3 × 2) for the overall row. Event breakdown: systemic complications included DVT in Group A (1 case) and cardiovascular events in Group B (1 case) and Group C (1 case); no pulmonary embolism observed. Surgery-related complications included delayed wound healing in Group B (1 case); no wound infection, hematoma, or stiffness were reported. Other complications included nerve injury in Group A (1 case). No 30-day readmission or death was observed in any group.
A: full-duration tourniquet; B: half-full-duration tourniquet; C: half-full-duration tourniquet plus intra-articular tranexamic acid (2 g); DVT: deep-vein thrombosis.
Discussion
TKA is the standard treatment for end-stage KOA. However, heavy perioperative blood loss is common; TBL during and after surgery may exceed 1000 mL and can delay wound healing, increase infection risk, and hinder early recovery.2,24,25 Efforts to minimize blood loss have led to the development of various strategies, including tourniquet management and antifibrinolytic therapy. Despite these advances, the optimal combination of these methods to improve both blood loss control and recovery remains an area of active research. Recent studies have shown that optimizing blood management can improve patient outcomes. For example, Jiang W et al. 26 demonstrated that combining tourniquet techniques with antifibrinolytic agents significantly reduces perioperative blood loss, while Parikh et al. 27 highlighted the importance of individualized blood-management protocols in improving postoperative recovery. The novelty of this study is that it provides a pragmatic head-to-head comparison of three commonly used perioperative blood-management strategies, while simultaneously evaluating both blood-loss indices (including HBL) and early recovery outcomes, thereby offering ERAS-relevant evidence for strategy selection. The main finding was that combined use of a half-duration tourniquet and 2 g intra-articular TXA outperformed a full-duration tourniquet: it reduced TBL and HBL, limited the reduction in Hb levels, lowered transfusion rates, eased postoperative pain, and improved early function, without increasing postoperative complications.
This favorable effect may result from the combined action of mechanical hemostasis and antifibrinolytic therapy. First, shortening the tourniquet time may prevent ischemia–reperfusion injury, thereby reducing tissue edema and inflammation and lowering postoperative HBL, limb swelling, and pain.28–30 In the present study, HBL, which contributes significantly to postoperative anemia and delayed recovery, was significantly reduced in the combined strategy (Group C). 31 Studies have shown that HBL can lead to substantial reductions in postoperative Hb levels, contributing to symptoms of anemia and delayed recovery. 32 In this study, the combined strategy of a half-duration tourniquet and intra-articular TXA resulted in significantly reduced postoperative pain and swelling, with enhanced functional recovery. These findings align with previous studies that have demonstrated similar benefits from the combination of reduced tourniquet time and TXA.29,33 The reduced pain and swelling are likely attributable to the combined effects of reduced ischemia–reperfusion injury from tourniquet use and suppression of postoperative fibrinolysis by TXA. The faster functional recovery observed in Group C may be attributed to both reduced blood loss and improved recovery environment facilitated by TXA.
Second, local high-dose TXA has been shown to induce high antifibrinolytic levels at the surgical site, suppressing fibrin breakdown and controlling postoperative wound oozing. 14 Similar results have been consistently reported by several studies that describe TXA’s efficacy in reducing both visible and HBLs in TKA.34,35 Importantly, TXA has been shown to be safe; it does not cause any significant increase in thromboembolic complications at doses up to 2 g, further supporting its widespread use in blood-management protocols.36–38 These findings are consistent with the proposed mechanism; compared with Group B, Group C demonstrated further reductions in total and HBLs, better maintenance of postoperative Hb levels, and a transfusion rate of 3.3%. At the same time, the lowest postoperative pain scores and the fastest early knee functional recovery were observed in Group C. These findings suggest that local high-dose TXA suppresses postoperative hyperfibrinolysis and reduces HBL. It may also reduce inflammatory exudation, providing additional benefits in terms of pain relief and faster functional recovery. It should be emphasized that all patients received preoperative IV TXA as baseline antifibrinolytic therapy. In Group C, a local high-dose TXA was added on this basis. This “intravenous plus local” approach is consistent with current evidence, according to which, combined routes are more effective than a single route.34,39 A stronger hemostatic effect was achieved through the synergy of the two routes. Consequently, the Group C regimen demonstrated the best overall benefits in terms of PBM and early recovery.
These findings are supported by recent high-quality studies. Evidence from multiple randomized controlled trials (RCTs) and meta-analyses shows that a full-duration tourniquet does not reduce TBL or transfusion requirement in TKA patients. Transfusion is generally uncommon in primary TKA patients with preoperative Hb levels >12 g/dL when modern PBM principles are strictly applied. However, tourniquet use mainly reduces visible IBL and does not prevent postoperative HBL and hemodilution, which may still cause a clinically relevant early decline in Hb levels and symptomatic anemia in a subset of patients, thereby triggering transfusion despite an initially normal Hb level. It may result in increased postoperative pain through ischemia–reperfusion injury and slow functional recovery, raising the risk of complications.6,40 In their systematic review, Han et al. report that tourniquet use reduces only intraoperative bleeding; it does not lower TBL or transfusion needs and may worsen postoperative pain and joint stiffness. 6 In a randomized controlled trial, Chaiyakit et al. reported similar findings. Shortening the tourniquet time may promote early recovery without increasing TBL. 10 Based on another line of evidence, large-sample studies have reported that combined use of a half-duration tourniquet and antifibrinolytic therapy can significantly improve postoperative anemia. For example, Bayrak et al. analyzed 558 TKA cases and found that TXA use was associated with a smaller postoperative reduction in Hb levels and lower transfusion needs. 41 Additionally, evidence from network meta-analyses indicate that local high-dose TXA (≥2 g) further reduces HBL and does not increase the risk of complications.36–38 Taken together, these findings support the effectiveness and safety of the combined strategy of a half-duration tourniquet plus local TXA administration used in this study.
The benefits and limitations of tourniquets and TXA should be weighed, and use should be tailored according to the patient. The tourniquet provides a bloodless field, reduces visible intraoperative bleeding, and may improve the initial stability of cement fixation.42–44 Reports have shown that TKA with a tourniquet has a lower rate of early prosthetic loosening than procedures that do not use a tourniquet. This finding suggests that tourniquet use helps initial implant fixation. 42 However, the adverse effects of tourniquet use should not be overlooked; prolonged inflation can cause limb ischemic pain, muscle weakness, and limit joint motion and may trigger significant rebound bleeding (HBL).30,45 It is also linked to higher risks of DVT and wound complications. 8 By contrast, TXA achieves hemostasis by inhibiting fibrinolysis. Local use of TXA creates a high drug level at the surgical site while systemic exposure remains limited. It has been shown not to increase the risk of DVT or pulmonary embolism, 37 not to impair wound healing, and not to worsen postoperative pain or functional outcomes.14,38 Many studies and meta-analyses have consistently shown that using TXA during TKA, either intravenously or locally, does not increase the risk of thromboembolic events. 37 TXA can be used safely even in high-risk patients with a history of venous thromboembolism, when used appropriately. 46 Therefore, routine use of TXA in PBM for TKA can reduce reliance on the tourniquet without increasing complications. Notably, local TXA alone does not provide an immediate bloodless field. Some intraoperative bleeding may occur. Thus, many investigators recommend combining a half-duration tourniquet with TXA, as in the present study, to maintain a clear operative field while minimizing postoperative bleeding.
The results of this study have clear clinical significance. Control of postoperative anemia and pain is a key component of the ERAS principles. 47 A large body of evidence has linked moderate-to-severe postoperative anemia to limited early mobility, more complications, and higher readmission risk.48–50 Therefore, modern ERAS and PBM principles emphasize strict adherence to restrictive transfusion strategies and avoidance of unnecessary transfusions. They also recommend the use of comprehensive perioperative measures for optimal prevention and correction of postoperative anemia.51–53 The combined hemostatic regimen significantly reduced the proportion of patients who met the postoperative anemia threshold; the postoperative Hb level in Group C was higher than those in Groups A and B. Transfusion needs were lower in Group C. These results may help patients walk earlier, shorten their hospital stay, and reduce transfusion-related risks and costs. 54 Moreover, no increase in thrombotic complications—such as DVT and pulmonary embolism—was observed with the combined regimen. This is consistent with prior evidence which states that TXA does not increase thromboembolic events. 37 By reducing bleeding and transfusion needs, TXA may lower the overall perioperative complication rates. 54 With respect to pain and function, the combined group reported least postoperative pain and demonstrated fastest early functional recovery. Although the 6-month functional recovery was similar across groups, better early control of pain and anemia significantly improved comfort and satisfaction during recovery.22,23 Taken together, a half-duration tourniquet plus local TXA strategy shown in this study balanced postoperative bleeding control with early recovery and is valuable for perioperative management.
Limitations and future directions
This study has certain limitations, which should be addressed in future work. First, the study used a single-center retrospective design. The sample size was limited, and group sizes differed slightly (28, 27, and 30). This design is prone to selection bias and may limit generalizability. In addition, due to the retrospective design and nonrandom group assignment, the possibility of residual confounding cannot be completely excluded. Some potential interferers may not have been fully captured or adjusted for (e.g. comorbidity severity, perioperative medications, intraoperative hemostatic maneuvers, and rehabilitation adherence), which could have affected blood loss and early recovery outcomes. Second, although the same surgical team performed all the procedures to reduce variability, no subgroup analysis was conducted for potential surgeon-to-surgeon technical differences. Differences in individual surgeons’ habits and technique details may have existed, and patient outcomes—such as blood loss and postoperative recovery—may have been affected. Third, follow-up data were concentrated from the perioperative period to 6 months postoperatively, and longer-term data were not available. Therefore, the long-term effects of this blood-management strategy on implant stability, implant survival, and late knee function could not be determined. Finally, blood loss was estimated using indirect formulas. This approach is convenient but may have introduced errors. However, multiple measures were combined to provide a composite assessment, partly offsetting this limitation. In addition, intraoperative blood pressure may influence blood loss; however, due to incomplete anesthesia records and inconsistent recording frequency, intraoperative blood pressure could not be standardized well and included in the statistical analyses. To further test the reliability of these findings, large prospective multicenter studies should be prioritized. Hemostasis strategies tailored to patient subgroups (e.g. those at high risk of intraoperative bleeding) should also be explored. In future work, follow-up should be extended to evaluate the effects of this optimized blood-management strategy on long-term implant outcomes, overall medical costs, and patient satisfaction.
Conclusion
In TKA, the combination of a half-duration tourniquet with intra-articular TXA may provide superior PBM compared with a full-duration tourniquet or half-duration tourniquet alone. This strategy may reduce total and hidden postoperative blood losses, limit reduction in Hb level, and lower transfusion rates. Additionally, it may reduce postoperative pain and promote earlier recovery of knee function. No significant increase in complications, including thrombosis or infection, was observed.
Footnotes
Acknowledgments
None.
Authors’ contributions
Study conception and design: Fengji Liu, Bingchi He, and Ning Wang
Data curation and analysis as well as manuscript drafting: Fengji Liu, Futeng Wang, and Xingyu Wang
Manuscript writing and revision: Fengji Liu and Shuhan Jia
Supervision and critical revision: Ning Wang
All authors have read and approved the final manuscript.
Availability of data and materials
The datasets are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of the Central Hospital Affiliated to Shenyang Medical College (Approval Number:EC-2024-179(02)). As a retrospective study, the requirement for informed consent was waived. This study was conducted in accordance with the Declaration of Helsinki, adopted in 1975 and revised in 2024. All patient details were deidentified. All patient data were kept confidential and used only for this analysis.
Funding
The study was funded by the Scientific Research Fund of the Department of Science and Technology of Liaoning Province, China, [Grant Number: 2024-MSLH-463].

