Abstract
Carcinoid tumors, a subset of neuroendocrine tumors, are exceptionally rare in the nose and paranasal sinuses. Owing to their indolent nature, nonspecific clinical symptoms, and variable radiological features, treatment of carcinoid tumors remains a challenge. In this report, we present a case involving a 49 year-old female patient who presented with a carcinoid tumor originating in the sphenoid sinuses. The patient underwent a surgical resection of the tumor and has been followed up in the outpatient clinic, recurrence-free. In addition, we performed an English literature search of cases of carcinoid tumor of the nose and paranasal sinuses from January 2000 through January 2024 in the MEDLINE, EMBASE, and Scopus databases. Furthermore, the clinical characteristics and interventions of carcinoid tumor of the nose and paranasal sinuses were discussed, which might enhance the early diagnosis and provide appropriate management strategies.
Introduction
Carcinoid tumors are rare, indolent neuroendocrine tumors originating from enterochromaffin cells primarily found in the gastrointestinal tract and lungs.1-3 However, they can occasionally develop in the head and neck, either as a primary tumor or more commonly as a metastasis. Although the larynx and middle ear are commonly-affected sites for head and neck carcinoids,4-6 only a limited number of cases have been reported in the nasal cavity and paranasal sinuses.7-16 This rarity complicates their clinical understanding and management.
This report presents a case of a 49 year-old woman diagnosed with a primary typical carcinoid tumor in the sphenoid sinus, which was treated successfully with surgery. Additionally, a comprehensive review of the English literature on carcinoid tumors in the nasal cavity and paranasal sinuses is conducted, aiming to improve the understanding of their clinical characteristics and management.
Case Report
A 49 year-old female patient presented with a 10 year history of chronic nasal congestion accompanied by left-sided watery rhinorrhea. The patient had no significant medical history, and pituitary hormone levels were within normal limits. Notably, nasal endoscopy revealed a reddish mass with a smooth surface in the left choana and nasopharynx.
A prior paranasal sinus magnetic resonance imaging (MRI) at another hospital showed a homogeneous mass in the left sphenoid sinus, left choanal region, and nasopharynx, along with an enlarged sella suggestive of an empty sella (Figure 1A). After admission, a paranasal sinus high-resolution computed tomography (CT) scan was performed, revealing a soft tissue mass located in the left sphenoid sinus. The mass extended from the sphenoid sinus into the left nasal cavity, choana, and nasopharynx, accompanied by partial bony defects at the sellar floor and the junction of the sellar floor with the middle clivus (Figure 1B).
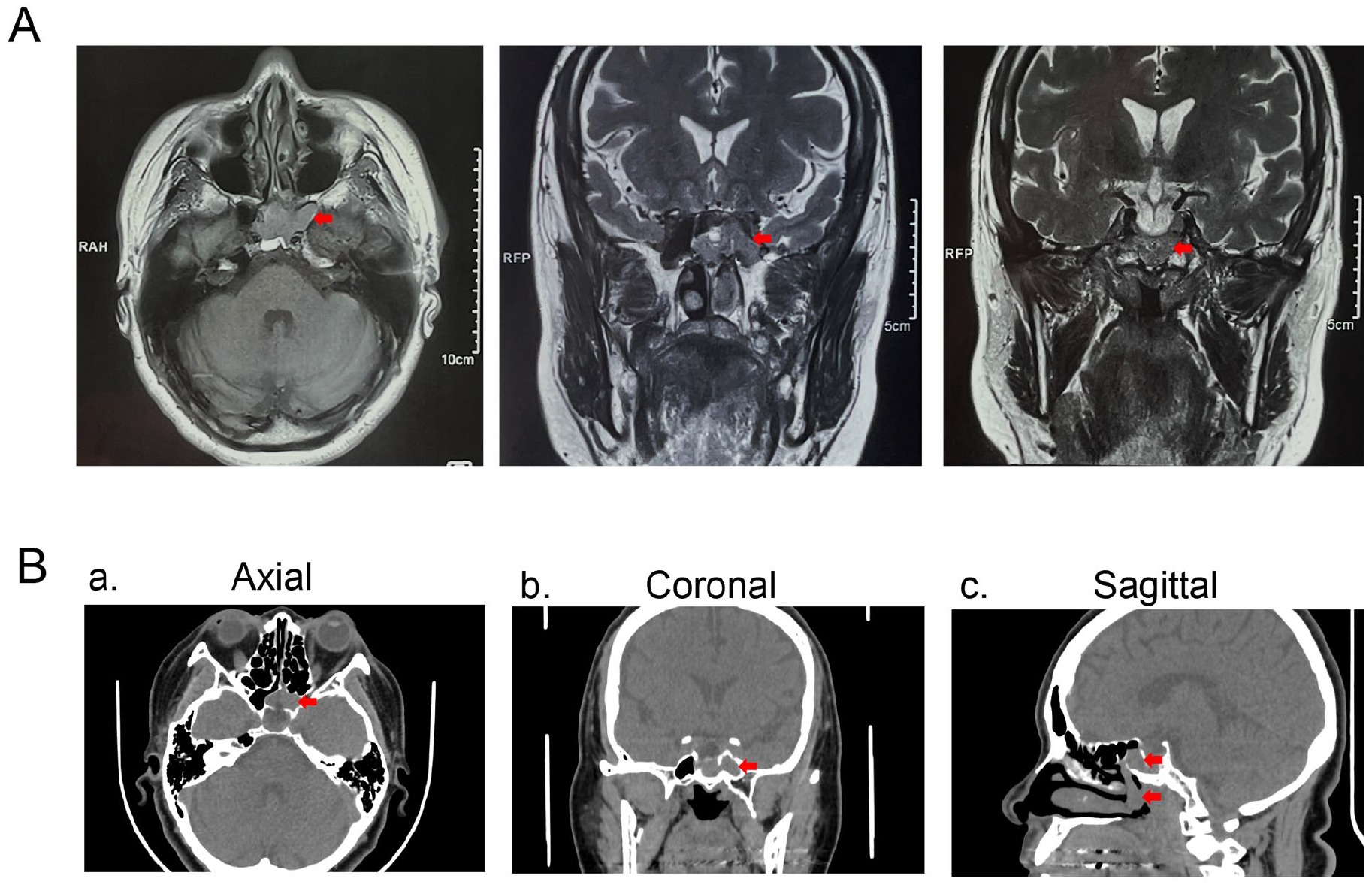
Radiographic findings of a primary carcinoid tumor of the sphenoid sinus. (A) Contrast-enhanced MRI showing the left sphenoid tumor (arrow) with extension toward the nasopharynx. (B) Paranasal sinus CT scans showing an enhancing mass (arrow) in the left sphenoid sinus with bulging toward the left nasal cavity, choana, and nasopharynx. (a) Axial view, (b) coronal view, (c) sagittal view. CT, computed tomography; MRI, magnetic resonance imaging
Surgical resection was performed using an endoscopic ipsilateral mononostril transsphenoidal approach, aided by a navigation. The tumor had a smooth, dark-red surface without a discernible envelope and extended into the left nasal cavity, posterior nostril, and nasopharynx through the enlarged natural ostium of the sphenoid sinus (Figure 2A). After debulking, the tumor base was found at the base of the sella and adjacent middle clivus, with partial bone deficiency. The tumor was excised with clear margins, and reconstruction was performed using abdominal fat and a middle turbinate mucosal flap (Figure 2B).
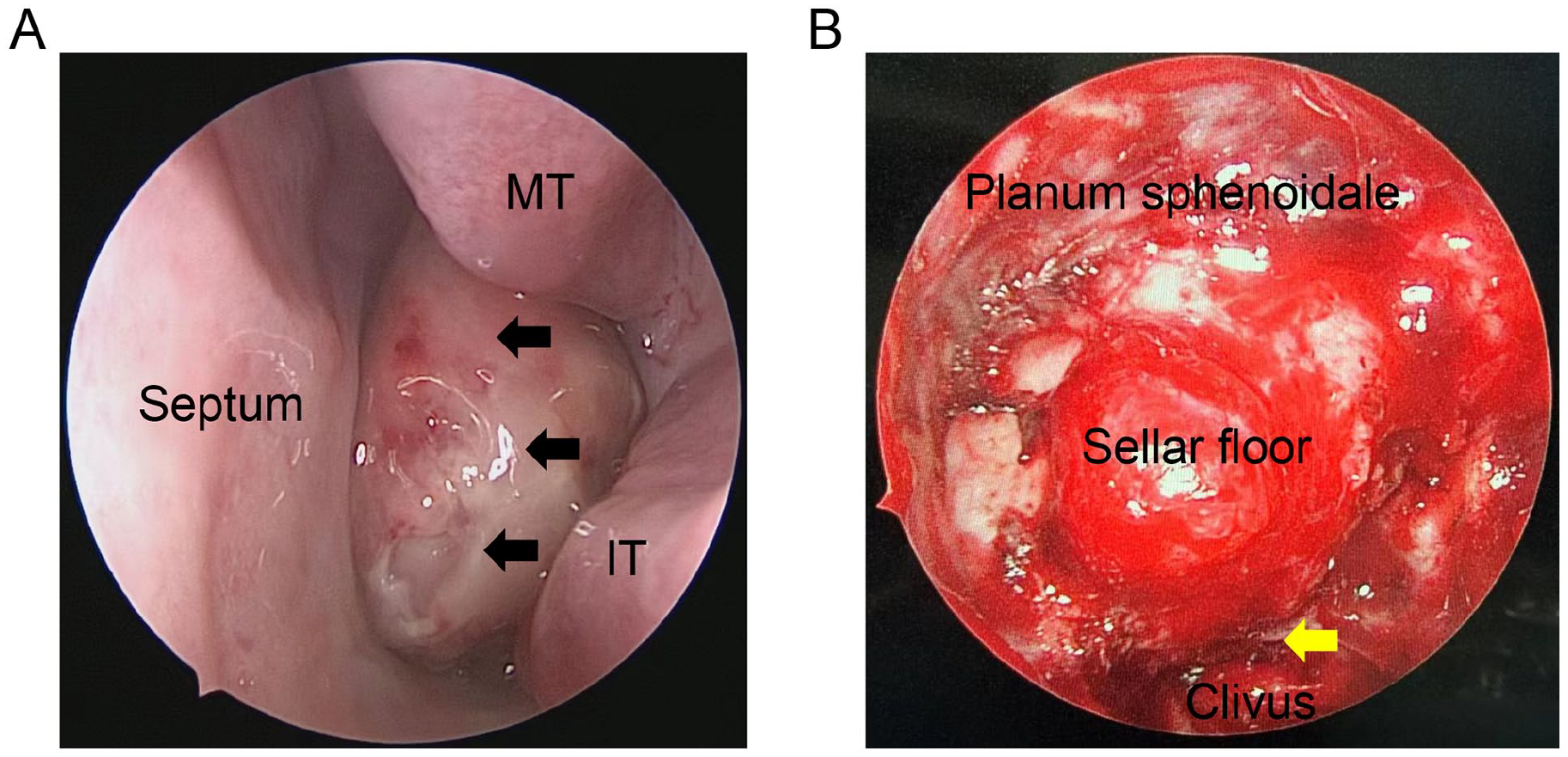
Nasal endoscopic examination showing a reddish mass (arrow) filling the left nasal cavity (A) and showing that tumor was excised with clear margins in the left sphenoid sinus (B). Yellow arrow indicates the attachment of carcinoid tumor.
Histopathological evaluation confirmed a well-differentiated neuroendocrine tumor composed of small cells with regular, well-rounded nuclei and the absence of mitotic, apoptotic, or necrotic features (Figure 3A). Carcinoid tumors produce abundant secretory granules with intense expression of neuroendocrine markers, such as chromogranin A (CgA), CD56, cytokeratin, and synaptophysin. 17 Further analysis through immunohistochemistry revealed positive staining for CD56, CgA, cytokeratin 8/18, insulinoma-associated protein 1 (INSM1), phosphoenolpyruvate carboxy kinase, somatostatin receptor, and synaptophysin; low positive expression for Ki67; and negative expression for Pit-1, adrenocorticotropic hormone (ACTH), follicle-stimulating hormone, growth hormone, luteinizing hormone, sex-determining region Y-box 2 confirming the diagnosis of a primary typical carcinoid tumor of the sinus (Figure 3B).
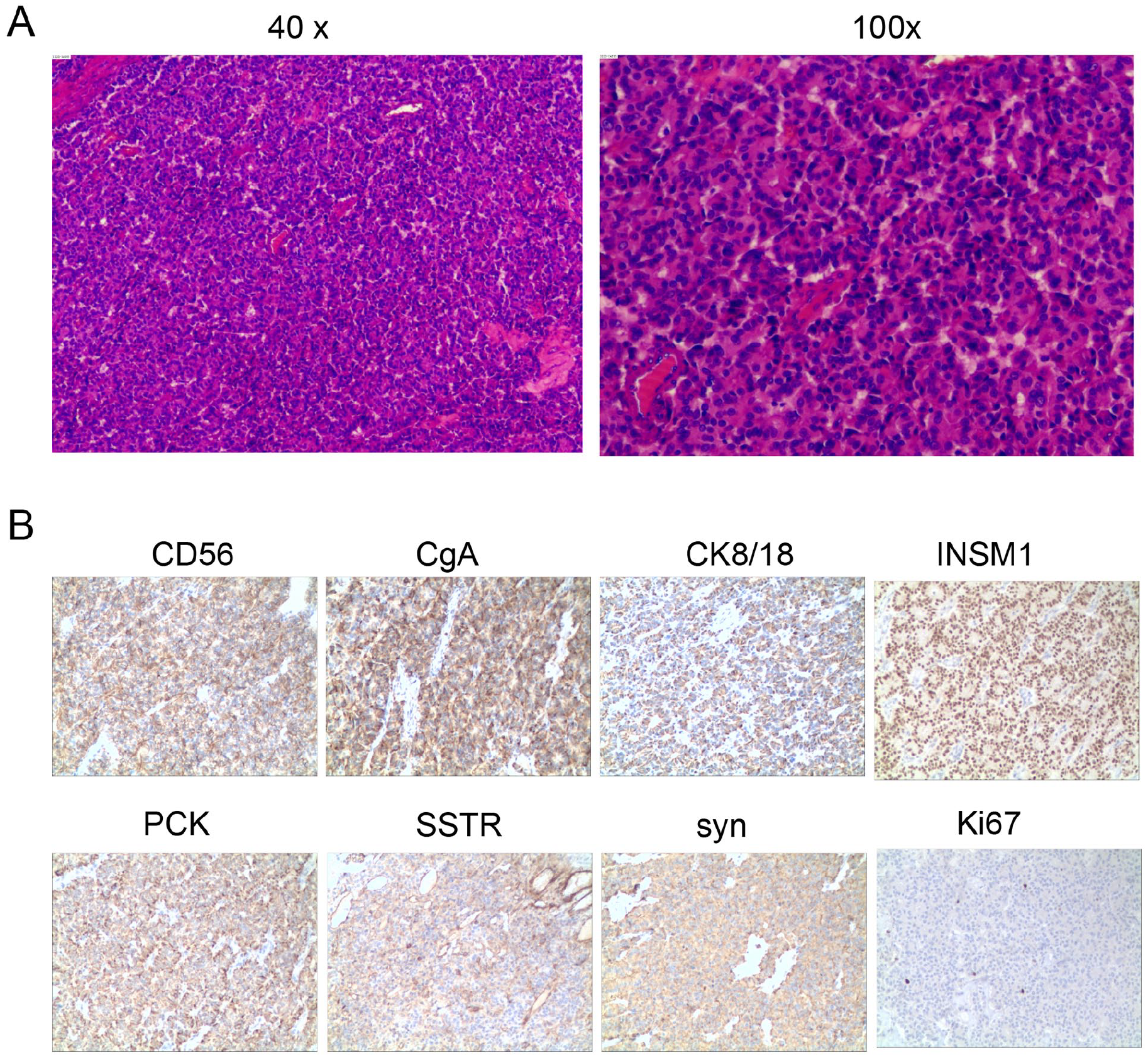
Histopathologic findings of a typical carcinoid tumor. (A) Hematoxylin-eosin staining showing a predominant population of uniform plasmacytoid tumor cells with little cellular pleomorphism and rare mitoses (original magnification 40× and 100×). (B) Immunochemistry staining showing that tumor cells were positive for CD56, CgA, CK8/18, INSM1, PCK, SSTR, and syn, and expressed low levels of Ki67. CgA, chromogranin A; INSM1, insulinoma-associated protein 1; PCK, phosphoenolpyruvate carboxy kinase; CK8/18, cytokeratin 8/18; SSTR, somatostatin receptor; syn, synaptophysin.
Following surgery, the patient had an uneventful postoperative recovery and was discharged on the fifth postoperative day. Further investigations revealed no signs of carcinoid syndrome or distant metastasis. The patient declined radiotherapy, chemotherapy, and other related treatments but has been tumor-free during a 1 year follow-up (Figure 4).
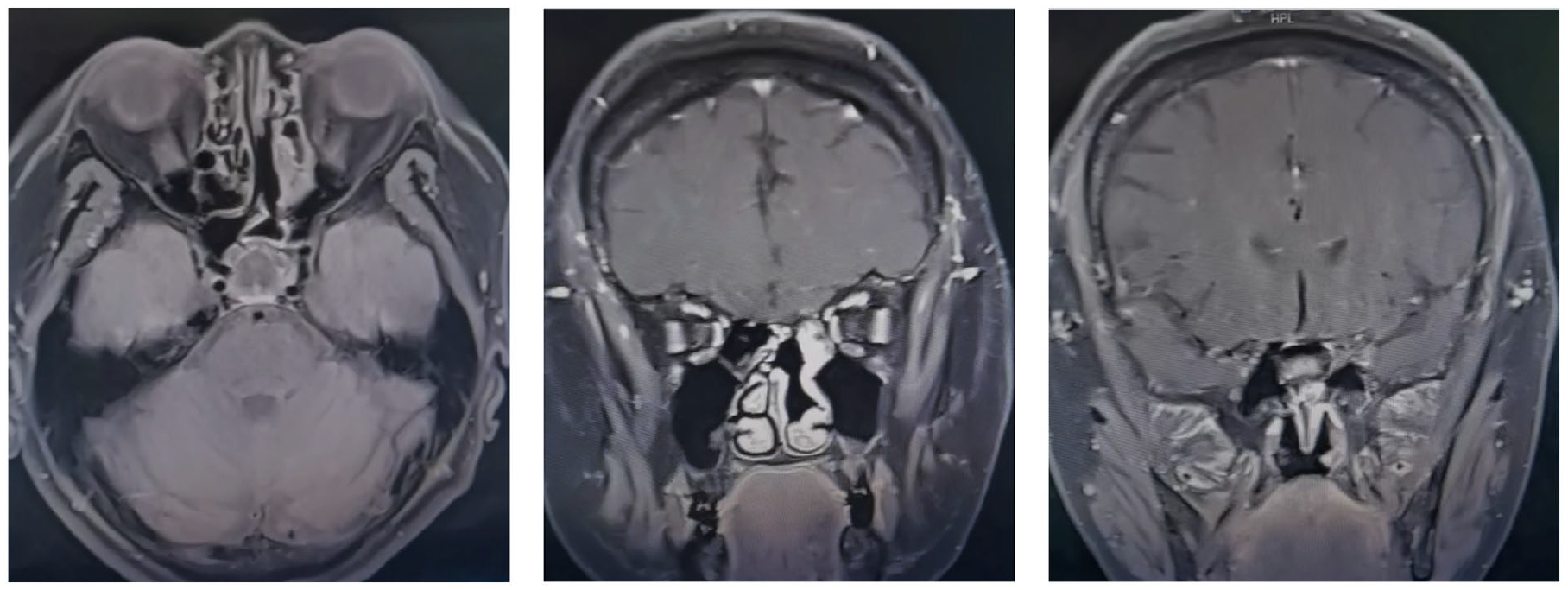
MRI scans performed at 1 year postoperatively showing no relapses at the left sphenoid sinus and nasopharynx. MRI, magnetic resonance imaging.
Discussion
According to the World Health Organization classification, epithelial neuroendocrine tumors in the nose and paranasal sinuses are categorized as neuroendocrine neoplasm grade 1 (typical carcinoid tumor); neuroendocrine neoplasm grade 2 (atypical carcinoid tumor); and neuroendocrine neoplasm grade 3 (small cell carcinoma). 18 Primary carcinoid tumors in the sinonasal cavity are extremely rare and account for <0.5% of all sinonasal tumors.19,20 The majority of these tumors are typical carcinoids and occur in the fourth to sixth decade of life, with a slight male predominance.7-13,15,16 To our knowledge, there is no previous report reviewing the existing cases of carcinoid tumors of the nose and sinus.
Here, we report a case of primary carcinoid tumor originating in the left sphenoid sinus, manifesting as nasal congestion and persistent left watery rhinorrhea. Radiographic imaging revealed a mass centered in the sphenoid sinus and extending into the nasopharynx. Additionally, the sella was enlarged, implying an empty sella. The tumor was removed with clear margins, and reconstruction of the sellar floor was performed using abdominal fat and a free middle turbinate mucosal flap. Histopathological analysis confirmed the presence of a well-differentiated carcinoid tumor. The patient declined additional treatment modalities and maintained tumor-free status during a 1 year postoperative follow-up.
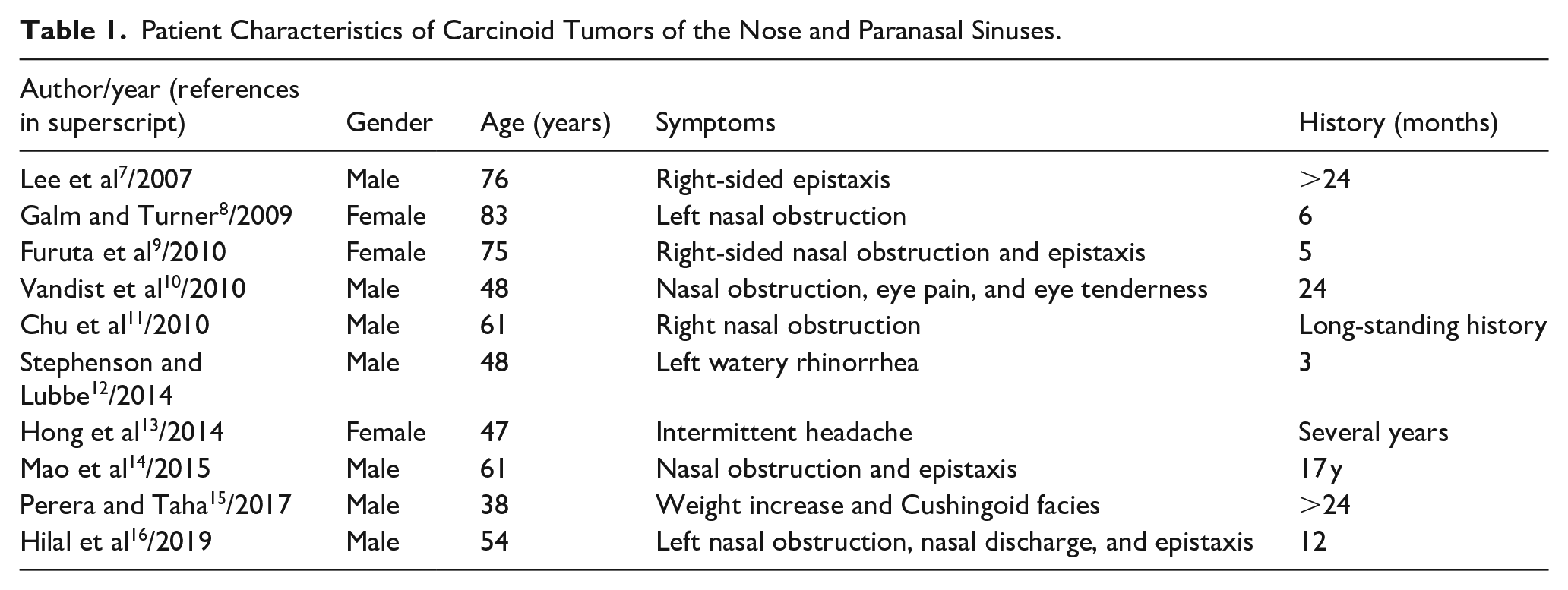
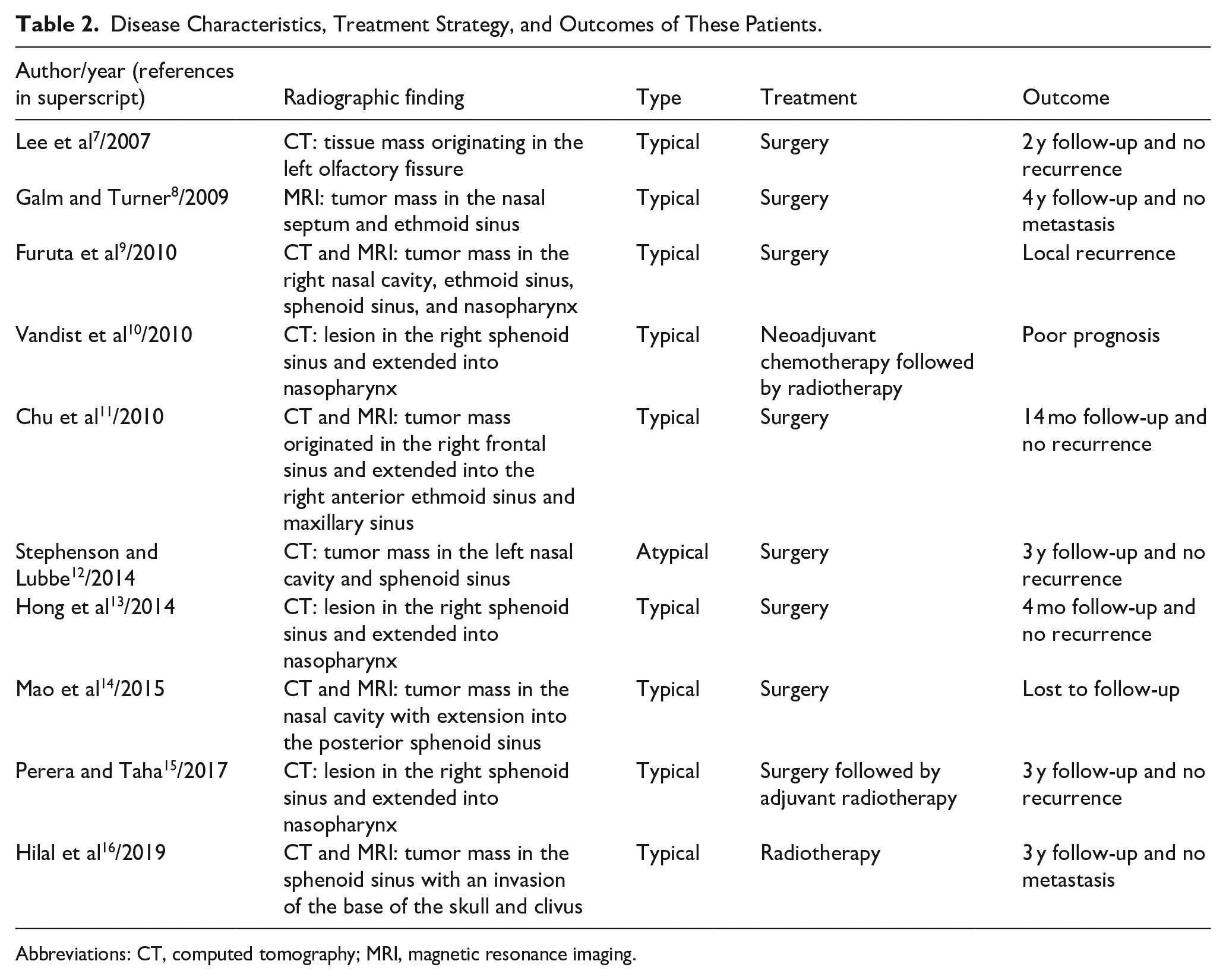
We also performed an English literature search for cases of carcinoid tumor of the nose and paranasal sinuses, and only 10 cases were collected.7-16 The clinical and demographic characteristics, including age, gender, symptoms, radiographic findings, treatments, and outcomes of these subjects are summarized in Tables 1 and 2. Of the 10 patients included, a higher proportion of male patients (70%) were affected, and the majority of individuals were elderly, with ages ranging from 38 to 83 years. Nasal obstruction was the most common symptom (60%), followed by epistaxis (40%). Only 1 case exhibited weight gain and Cushingoid facies, along with ectopic ACTH syndrome. 15 There are 7 cases of carcinoid tumors of the paranasal sinuses (5 originating in the sphenoid sinus, 1 in the ethmoid sinus, and 1 in the frontal sinus) and 3 cases of carcinoid tumors of the nasal cavity (1 in the olfactory fissure, 1 in the nasal septum, and 1 in the nasal cavity). The sphenoid sinus was the most frequently affected, often accompanied by bone destruction, followed by the ethmoid sinuses, while the maxillary sinus was the least affected. Histologically, carcinoid tumors were categorized into typical and atypical subtypes. 21 Typical carcinoids are well-differentiated lesions and can be distinguished from moderately-differentiated atypical carcinoid tumors by the absence of mitotic activity, pleomorphism, and necrosis. Although a single case of sinonasal atypical carcinoid was reported, its clinical presentation was nonspecific. 15 Notably, clinical manifestations were similar between the 2 subtypes, and complete surgical removal with clear margins yielded favorable postoperative outcomes in most cases.22-24 Additionally, Hilal et al treated a 54 year-old patient with definitive intensity-modulated radiation therapy, with no recurrence after 3 years of follow-up. 16 However, Vandist et al documented the case of a 48 year-old patient who received neoadjuvant chemotherapy followed by consolidation radiotherapy, resulting in an unfavorable prognosis. 10 Accordingly, complete surgical resection is recommended as the primary treatment approach for both types of carcinoid tumors. In advanced or recurrent cases, adjuvant therapies, including radiation therapy, are considered.
Patient Characteristics of Carcinoid Tumors of the Nose and Paranasal Sinuses.
Disease Characteristics, Treatment Strategy, and Outcomes of These Patients.
Abbreviations: CT, computed tomography; MRI, magnetic resonance imaging.
The diagnosis of carcinoid tumors of the nose and paranasal sinuses requires a combination of clinical, radiological, and pathological evaluations. 21 The clinical presentation of this tumor can be nonspecific and depends on the location and function of the primary tumor, as well as the secretion of specific hormones.7-13,15,16 Due to their slow-growing nature, nasal obstruction and bleeding are the most frequently-observed symptoms in cases of carcinoid tumors in the nasal region. The diagnosis is often delayed due to the presence of nonspecific symptoms. Carcinoid syndrome, characterized by the presence of episodic flushing, wheezing, diarrhea, and right heart failure, can develop following metastasis to the liver or lungs, where functional lesions can release vasoactive chemicals into the systemic circulation. 25 However, it is important to note that carcinoid syndrome is relatively uncommon, reported in only ~10% of individuals with carcinoid tumors. In the present study, none of these patients exhibited any symptoms suggestive of carcinoid syndrome. An estimated 4% of cases of ACTH-dependent Cushing’s syndrome are caused by lung carcinoid tumors, which are aggressive and release ectopic ACTH. 26 Based on literature research, only 1 case was reported to present with weight increase and Cushingoid facies, along with ectopic ACTH syndrome. 15 Accordingly, unilateral progressive nasal obstruction and bleeding in the elderly may indicate a carcinoid tumor of the nose or paranasal sinuses.
Depending on their origin site, carcinoid tumors may possess the capacity to secrete vasoactive chemicals, with serotonin (5-hydroxytryptamine) being the most notable, particularly in midgut carcinoid tumors. 27 Urinary 5-hydroxyindole acetic acid (5-HIAA), the breakdown product of serotonin, has long served as the gold standard for diagnosing and monitoring carcinoid tumor patients. 28 While its sensitivity is notably lower (35%), its specificity is nearly 100%, owing to potential influences from drug use and dietary habits on urinary 5-HIAA levels. CgA is a 49 kDa acidic, hydrophilic protein expressed in the chromaffin granules of neuroendocrine cells. In contrast to serum serotonin levels and urinary 5-HIAA, serum CgA levels can be used in the detection of both functioning and nonfunctioning tumors. Despite the fact that its specificity is lower than urinary 5-HIAA levels (86% vs 100%, respectively), its sensitivity is higher (35% vs 68%, respectively). 29 There is a positive correlation between serum CgA levels and tumor burden; therefore, using serum CgA levels along with urinary 5-HIAA levels will yield a higher degree of accuracy in detecting relapses in carcinoid tumor patients.
CT and MRI are recommended for all cases of nose and paranasal sinus tumors to evaluate the degree of local invasion of tumors and assess the presence of obstructive inflammation. Unlike other sinonasal tumors, carcinoid tumors mostly commonly originate in the sphenoid sinus instead of the maxillary sinus. CT typically reveals a well-defined mass, occasionally exhibiting cystic changes or focal calcification in carcinoid tumors. 30 On the other hand, MRI typically reveals homogeneous density or signal intensity, appearing as a slight hypointense lesion on both T1- and T2-weighted imaging, with a pronounced increase on contrast-enhanced MRI. 30 Additional imaging modalities such as positron emission tomography (PET) and CT of the chest may be employed to evaluate the presence of distant metastasis. PET scanning utilizing 18F-labeled fluorodeoxyglucose ( 18 FDG) is a widely-used imaging technique in clinical oncology. However, the uptake of FDG in carcinoid tumors is confined due to their low proliferative activity and high differentiation rate. Consequently, several tracers tailored to the specific characteristics of carcinoid tumors, such as 18F-DOPA, 11C-labeled 5-hydroxytryptophan, and 68Gallium-DOTATATE, have been developed for PET imaging in these tumors.31,32
Histopathological examination is a crucial diagnostic tool for differentiating carcinoid tumors from other tumor subtypes. Histological staining reveals carcinoid tumor cells as round, polyhedral, or spindle-shaped, with granular, eosinophilic, amphophilic, or oncocytic cytoplasm. Nuclei exhibit small, hyperchromatic features, or are intermediate to large, with coarse granular or speckled chromatin. Typical carcinoids show fewer than 2 mitoses/10 high-powered fields (HPF) and lack any presence of necrosis. Atypical carcinoids are characterized by 2-10 mitoses/10 HPF and/or the presence of necrosis. Immunohistochemical markers, such as CgA, synaptophysin, CD56, and INSM1, are widely used to determine neuroendocrine differentiation. 17 Several pieces of evidence suggest that the Ki67 proliferative index is closely correlated with patient survival time in carcinoid tumors, even though the Ki67 proliferative index is not included as a diagnostic tool for prognostic stratification. A higher Ki67 index in metastatic carcinoids indicates a poorer prognosis, 33 thus recommending the Ki67 as a prognostic marker in carcinoid tumors.
Conclusion
A primary typical carcinoid tumor of the nose and paranasal sinus is a rare neoplasm that warrants consideration as part of the differential diagnosis for indolent sinonasal tumors, particularly in patients experiencing nonspecific symptoms, such as nasal obstruction and bleeding. Serum CgA and urinary 5-hydroxyindolacetic acid levels are valuable for diagnosis and relapse detection in carcinoid patients. CT and MRI are recommended for evaluating tumor location and assessing inflammatory lesions. Histopathological evaluation is essential for disease identification. Additionally, the Ki67 index has emerged as a reliable prognostic indicator. Early diagnosis and appropriate management play a key role in achieving favorable outcomes. Complete surgical excision remains the primary treatment of choice, supplemented by targeted adjuvant therapy in advanced cases. Nevertheless, further studies are needed to establish optimal management strategies for these infrequent tumors to enhance understanding and improve patient care.
Footnotes
Ethical Considerations
All procedures conducted in this study involving human participants were in accordance with the ethical standards set by the institution and were approved by the Ethics Committee of Tongji Hospital (TJ-IRB20230848).
Consent to Participate
Written informed consent was obtained from the patients involved in the study.
Author Contributions
Zhe-Zheng Wang: data collection, manuscript writing, imaging formatting, and presentation of manuscript. Hong-Tao Zhen: data review, manuscript appraisal, clinical analysis, and content analysis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (NSFC) grants 82201262 (Z.-Z.W.).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are not publicly available due to privacy reasons but are available from the corresponding author upon reasonable request.
