Abstract
Extranodal natural killer (NK)/T-cell lymphoma, nasal type (NNKTL) is a rare and highly aggressive non-Hodgkin lymphoma originating from NK or γδ T cells infected by Epstein-Barr virus (EBV). In the United States, NNKTL is usually noted in people of Asian or Hispanic descent. Natural killer/T-cell lymphoma, nasal type commonly involves the upper aerodigestive tract, including the nasopharynx, nasal cavity, Waldeyer’s ring, and oropharynx. Extensive local destruction and invasion has been noted, especially of the paranasal sinuses, hard palate, and central nervous system; involvement of the nasolacrimal duct with dacryocystitis is yet to be reported. We report a rare case of a Hispanic man with extranodal NNKTL masquerading as persistent dacryocystitis and necrotizing sinusitis unresponsive to antibiotics and surgical intervention. An extensive background of necrosis and inflammation was noted on pathology, and additional analysis with immunohistochemistry and in situ hybridization after repeat biopsy were necessary for accurate diagnosis.
Keywords
Introduction
Extranodal natural killer (NK)/T-cell lymphoma, nasal type (NNKTL) is a rare and highly aggressive non-Hodgkin lymphoma originating from NK or γδ T-cells infected by Epstein-Barr virus (EBV).1-3 It was initially known as “lethal midline granuloma” in the United States or “rhinitis gangrenosa progressiva” in Europe after it was first reported in 1897 by Dr P. McBride in a 28-year-old house painter as an aggressive and lethal disease that caused rapid destruction of the midline facial structures with worsening necrotic granuloma.1,3-6 Tse et al report the incidence of NNKTL in Southeast Asia and Central/South America is about 5.2% and 3%, respectively, while incidence in North American and European nations is about 0.3%. 2 In the United States, NNKTL is usually noted in people of Asian or Hispanic descent.1-3 Natural killer/T-cell lymphoma, nasal type commonly involves the upper aerodigestive tract, including the nasopharynx, nasal cavity, Waldeyer’s ring, and oropharynx.1,2,4 Extensive local destruction and invasion has been noted, especially of the paranasal sinuses, hard palate, and central nervous system.1-4,7 Invasion of the nasolacrimal duct with dacryocystitis is yet to be reported. Symptoms of NNKTL include nasal obstruction, bloody rhinorrhea, sore throat, necrotizing lesions of the hard palate or nose, hard palate perforation, hoarseness, edema of cheek or orbit, and systemic symptoms such as prolonged fever, night sweats, and weight loss.1-5 It has a 2:1 male predominance in adults between the ages of 40 and 50.3-5 Few pediatric cases have been reported. 1 We report a rare case of a Hispanic man with extranodal NK/T-cell lymphoma, nasal type, masquerading as persistent dacryocystitis, and necrotizing sinusitis unresponsive to antibiotics and surgical intervention.
Case Report
A 38-year-old Hispanic man presented to our hospital with a 3-week history of persistent left-sided dacryocystitis, preseptal cellulitis, ethmoid and maxillary sinusitis, with periorbital pain and epiphora. He had been hospitalized at an outside hospital and underwent incision and drainage of left dacryocystitis with preseptal abscess. He tested positive for SARS-CoV-2 infection but had no respiratory symptoms. He was discharged on amoxicillin and sulfamethoxazole–trimethoprim for methicillin–resistant Staphylococcus aureus, group A Streptococcus and Morganella morganii positive incision and drainage cultures. Clindamycin was later added to his regimen during an emergency department visit for worsening facial pain and edema. At the time he presented to our hospital, he noted worsening facial pain with new onset fever and chills for 2 days. He had a 30-lb weight loss within 3 months but denied night sweats, visual changes, epistaxis, headache, and rhinorrhea. His past medical history was significant for chronic sinusitis requiring frequent antibiotics treatment, especially in the preceding 4 months. His social, surgical, and family histories were unremarkable.
His temperature was 98.5 F, blood pressure was 106/81 mm Hg, pulse rate was 108 bpm, respiratory rate was 16, and oxygen saturation on room air was 95%. On physical examination, there was left facial–periorbital erythema and edema along the left medial canthus which was tender to palpation, but no purulent discharge could be expressed. Induration was also noted along the left nasal sidewall. There was no crepitus and anterior rhinoscopy was unremarkable.
Initial laboratory examination revealed blood glucose 121 mg/dL, white blood cell count 9.75 × 10E9, absolute immature granulocyte 0.09 × 10E9, hemoglobin 13.9 g/dL, hematocrit 44.0%, and negative peripheral blood cultures. Otherwise, the coagulation panel and urinalysis were within normal limits. He was started on intravenous piperacillin–tazobactam and vancomycin. Computed tomography (CT) maxillofacial with contrast (Figure 1) was performed and showed findings consistent with left dacryocystitis with adjacent subperiosteal abscess formation lateral to the left nasal bone, significant absence of much of the left inferior turbinate, and acute on chronic left paranasal sinusitis. The working differential diagnosis included dacryocystitis, invasive fungal sinusitis, and necrotizing sinonasal infection with nasal abscess.
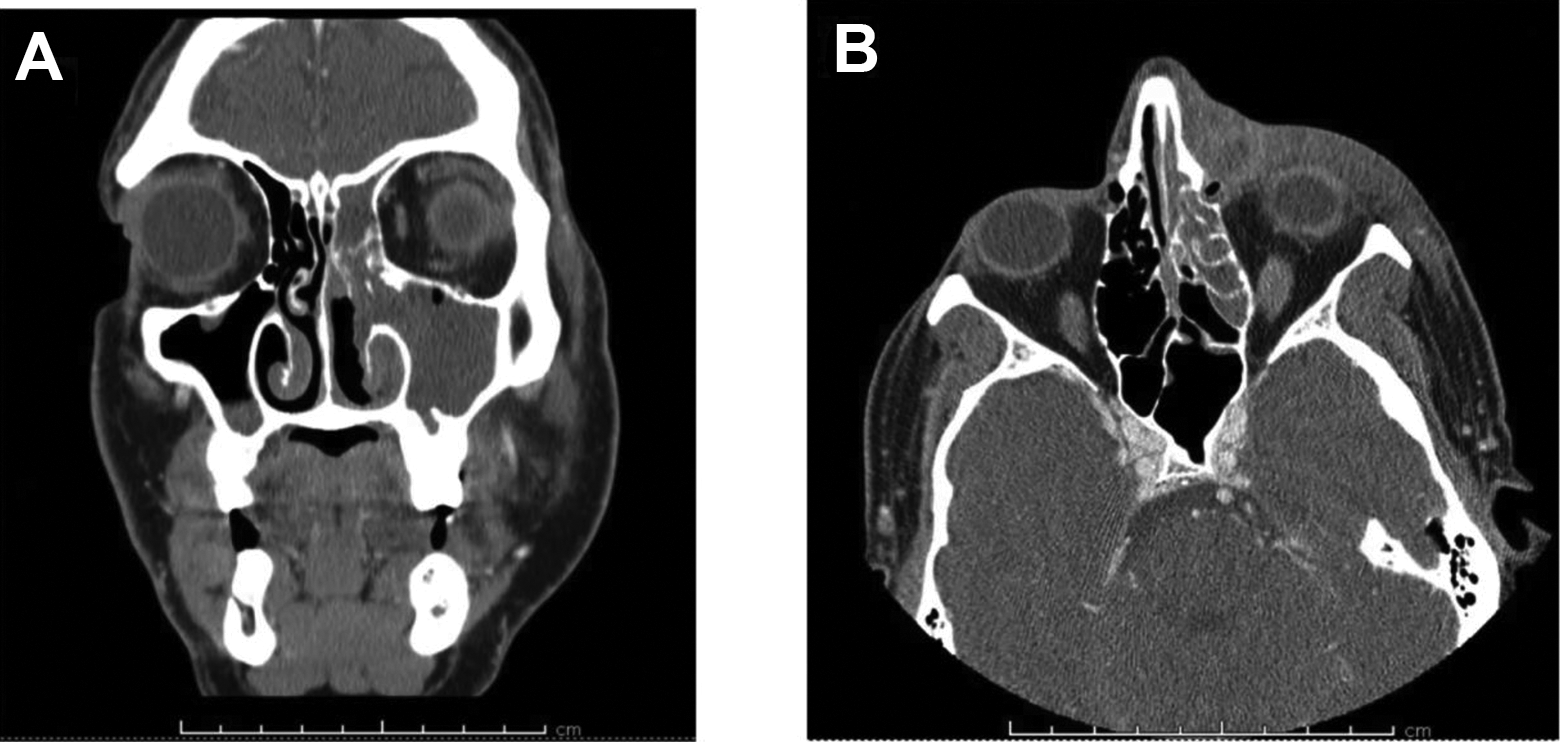
Computed tomography maxillofacial with contrast. A, Complete opacification of the left maxillary sinus, absence of the left inferior turbinate, and considerable opacification of the left ethmoid and frontal sinuses. B, Significant soft tissue swelling of the left side of the face. A low-attenuation area with peripheral enhancement around the inner canthus in the region of the lacrimal sac; consistent with left dacryocystitis. A low-attenuation area with peripheral enhancement lateral to the left nasal bone measuring approximately 1.5 × 1 × 1 cm, suggestive of subperiosteal abscess formation.
Operative nasal endoscopy was performed with biopsy and debridement of necrotizing sinonasal tissue, left maxillary antrostomy, left total ethmoidectomy, and left frontal sinusotomy. We noted extensive inflammation, crusting and purulence in the left nasal cavity, significant absence of bone and mucosa of the left inferior turbinate, and severely inflamed middle turbinates with friable tissues but no frank necrosis or black eschar. Incision and drainage of the left nasal sidewall induration was performed, but no purulence was noted.
A frozen section of the sinonasal tissue was suggestive of necrotizing infection without fungal elements. Final pathology showed widespread inflammation, necrosis, fibrinopurulent exudate, and granulation tissue with no evidence of vascular invasion or fungal infection.
Subsequent laboratory examination showed preexisting undiagnosed diabetes (hemoglobin A1C 7.3%), worsening anemia (hemoglobin 10.4 g/dL and hematocrit 31.8%), new-onset acute kidney injury (serum creatinine 2.77 mg/dL), worsening hyperglycemia (blood glucose 220 mg/dL), SARS-CoV-2 RNA negative, and elevated phosphorus, lactate dehydrogenase (LDH), and D-Dimer of 4.9 mg/dL, 310 IU/L, and 1.32 ug/mL, respectively.
The patient’s facial erythema and edema initially improved, but he developed progressive swelling of the left nare. Computed tomography maxillofacial was repeated without contrast due to ongoing acute kidney injury, and it demonstrated unresolved and more extensive acute on chronic paranasal sinusitis and increased left nare, nasolabial, and infraorbital soft tissue swelling. Repeat nasal endoscopy with further left sinonasal debridement and biopsy of the left nare and left lateral nasal wall was performed. Final pathology revealed extranodal NK/T cell lymphoma, nasal type (NNKTL) along with tumor necrosis with necrobiotic debris (Figure 2A-B).
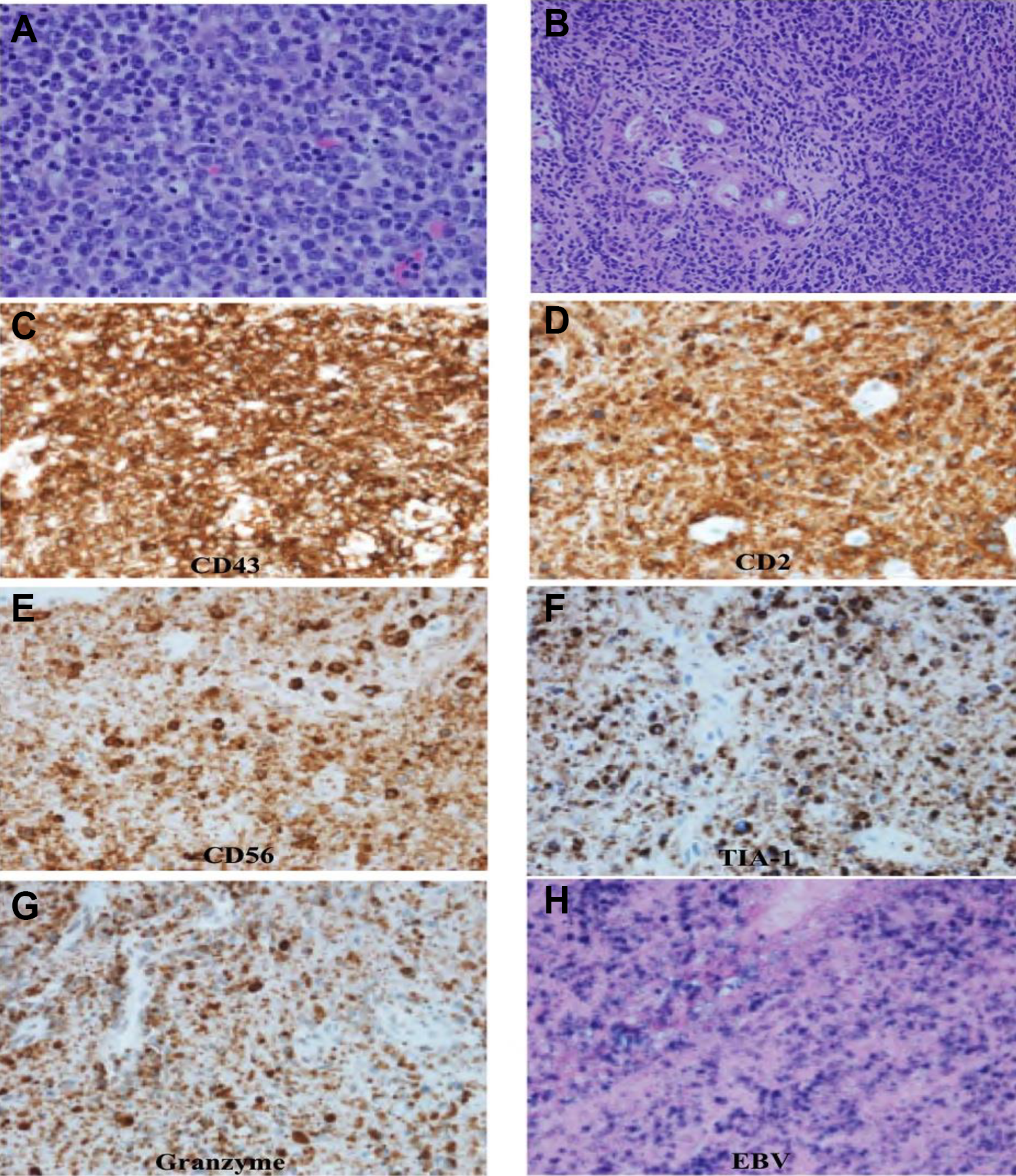
A, Multiple atypical cells with enlarged, pleomorphic nuclei with vesicular chromatin and variably prominent nucleoli. Multiple mitotic figures and apoptotic bodies are visible. B, Normal respiratory mucosa entrapped by atypical lymphoid cells. C-G, Immunohistochemistry—The atypical cells are positive for CD43, CD2, CD56, T-cell intracellular antigen-1 (TIA-1), and granzyme, respectively; consistent with the diagnosis of natural killer (NK)/T cell lymphoma. H, In situ hybridization—The atypical cells are positive for Epstein-Barr virus (EBV); consistent with the diagnosis of NK/T cell lymphoma. Source: Rachel Gordezky, MD; Department of Pathology, Howard University College of Medicine, Washington, District of Columbia.
The initial pathology was then reviewed and was revised to note the presence of NNKTL. The neoplastic infiltrate displayed a wide range of morphologic features ranging from small cells with irregular serpentine nuclei to large pleomorphic cells with vesicular nuclei and abundant pale or clear cytoplasm. The neoplastic cells showed angiocentricity and angiodestruction with associated zonal necrosis. They were also associated with polymorphous infiltrates of plasma cells and small mature lymphocytes, mimicking an inflammatory process. Immunohistochemistry showed that the neoplastic cells exhibited the classic immunotype of extranodal NK/T cell lymphoma with strong diffuse expression of CD56, CD2, CD43, CD20, granzyme B, and T-cell intracellular antigen-1 (TIA-1), while surface CD3 was negative (Figure 2C-G). In situ hybridization revealed diffuse presence of EBV infection (Figure 2H). A bone marrow biopsy was performed, but no metastasis to the bone marrow was noted. Chemoradiotherapy was commenced after completion of staging with positron emission tomography CT (PET/CT).
Discussion
Extranodal NK/T-cell lymphoma is considered an EBV-associated malignancy with the discovery of EBV DNA, EBV-determined nuclear antigen (EBNA1), and EBV oncogenic proteins in lymphoma cells in the late 20th century. 1 The nasal type makes up 80% of extranodal NK/T-cell lymphoma, while the nonnasal type is 20%.2,7 Natural killer/T-cell lymphoma, nasal type presents as rapidly progressive ulceration and necrotic granuloma in the nasal cavity, the Waldeyer’s ring, and the upper aerodigestive tract.1-5,7 Natural killer/T-cell lymphoma, nasal type can invade the paranasal sinus, hard plate, central nervous system, orbits, and facial skin.1-4,7 To our knowledge, we report the first case of NNKTL specifically invading and obstructing the nasolacrimal duct to present as persistent dacryocystitis and paranasal sinusitis unresponsive to antibiotics and surgical intervention. Accurate diagnosis was only possible through additional pathological analysis with immunohistochemistry and in situ hybridization after repeat biopsy. Nonnasal type extranodal NK/T-cell lymphoma involves the digestive tract, skin, testis, salivary glands, spleen, muscle, and uterus.2-5 Though rare, aggressive NK-cell leukemia/lymphoma is possible when there is dissemination to the liver, blood, spleen, lymph nodes, and bone marrow.2,4 In our case, our patient had chronic and worsening anemia necessitating a bone marrow biopsy, but no metastasis was noted.
Prompt and effective treatment of NNKTL patients requires early diagnosis through thorough biopsy analysis. While 69% to 100% of patients often present within its early stages, prognosis is worse in advanced stages.1,2,4 Diagnosis of NNKTL requires detection of EBV infection through in situ hybridization for EBV-encoded early RNA.1,2,4,5 Natural killer/T-cell lymphoma, nasal type can be excluded if EBV infection is absent. 2 Diagnosis also requires detection of the presence of either CD56 or cytotoxic molecules such as granzyme B, TIA-1, and perforin. 2 If neither of these is present, then the diagnosis is EBV-positive peripheral T-cell lymphoma, not otherwise specified.1,2 Quantification of circulating EBV-DNAs (BamHI-W DNA and LMP1 DNA) and EBV-micro RNAs (miR-BART2-5p, miR-BART7-3p, miR-BART13-3p, and miR-BART1-5p) in sera is required for NNKTL diagnosis, prognostication, and as a clinical progression or recurrence marker.1,3,4 High pretreatment levels of both EBV DNAs or miR-BART2-5p are associated with an aggressive clinical course and poor prognosis. Natural killer/T-cell lymphoma, nasal type is also associated with elevated levels of LDH, as noted in our patient, and soluble IL-2 receptor in serum. 1
On immunohistochemistry, the cells express CD2+, surface CD3−, cytoplasmic CD3ε+, CD45+, CD56+, perforin, granzyme B, T-cell restricted intracellular antigen 1(TIA1), Fas ligand, and intercellular adhesion molecule-1.1-3,5,7 Histologically, NNKTL neoplastic infiltrates display angiocentricity and angiodestruction with zonal necrosis.1,2,7 Harabuchi et al report that the lymphoma cells show diffuse infiltration and appear as “pleomorphic large or small cells with mitosis” and “various inflammatory cells such as macrophages, granulocytes and plasma cells in a necrotic background.” 1 The surrounding inflammation and necrosis often makes diagnosis difficult, as was the case in our patient. 1 Diagnosis is often delayed and multiple biopsies may be required. In its early stages, NNKTL can easily be confused for a necrotizing sinonasal infection as was the case in our patient whose neoplasm was disguised as dacryocystitis and sinusitis. 8 It is imperative that when managing a patient with necrotizing or resistant sinonasal infection that we have a broad differential diagnosis that also includes plasmacytoid dendritic cell neoplasms, invasive fungal infections, Wegener’s granulomatosis, NK-cell lymphomatoid gastropathy, chronic lymphoproliferative disorder of NK-cells, and nasal type extranodal NK/T-cell lymphoma (NNKTL).2,3,8 While there is no standard staging system for NNKTL, F18 fluorodeoxyglucose PET/CT helps with staging using adopted systems like the T-staging system or the Ann Arbor system.2,5,7
Chemotherapy, combined with radiotherapy either concurrently or sequentially, is the main treatment strategy for NNKTL.1,3,4 Conventional anthracycline-containing chemotherapy regimens have been replaced by non-anthracycline-containing regimens due to P-glycoprotein-associated resistance.3-5 In early clinical stages, the 5-year survival rate is 40% to 50%.1,3 After diagnosis, there is a continued risk of relapse of up to 10 years. 3 Current treatment for stage I/II disease combines radiotherapy with chemotherapy regimens such as DeVIC (dexamethasone, etoposide, ifosfamide, and carboplatin) or MPVIC-P (methotrexate, peplomycin, etoposide, ifosfamide, carboplatin, and prednisolone).1-4 For advanced stage (III/IV) disease, the SMILE (steroid, methotrexate, ifosfamide, L-asparaginase, and etoposide) chemotherapy regimen combined with radiotherapy can be used.1-4 Hematopoietic stem cell transplantation may also be considered especially for relapsed/refractory cases or advanced-stage in high risk patients.1-5
Extranodal NNKTL is a potential cause of necrotizing sinusitis and may masquerade as dacryocystitis and sinusitis unresponsive to antibiotics and surgical intervention. Given the background of necrosis and inflammation noted on pathology, additional analysis with immunohistochemistry and in situ hybridization after repeat biopsy were critical for accurate diagnosis.
Supplemental Material
Supplemental Material, sj-jpeg-1-ear-10.1177_0145561320987643 - An Unusual Case of Extranodal Natural Killer/T-Cell Lymphoma, Nasal Type Masquerading as Dacryocystitis and Sinusitis
Supplemental Material, sj-jpeg-1-ear-10.1177_0145561320987643 for An Unusual Case of Extranodal Natural Killer/T-Cell Lymphoma, Nasal Type Masquerading as Dacryocystitis and Sinusitis by Anthony N. Eze and Adedoyin O. Kalejaiye in Ear, Nose & Throat Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
