Abstract
Pneumocephalus is usually induced by trauma, infections, tumors of the skull base, and surgical interventions. Spontaneous pneumocephalus occurs due to a defect in the temporal bone with no obvious cause. Few cases have been reported with spontaneous otogenic pneumocephalus. However, delayed postoperative pneumocephalus is rarely reported in the literature. Here, we present a case of otogenic pneumocephalus through Eustachian tube (ET) preceded by nose blowing 10 days after surgical treatment of meningoencephalocele of the right middle ear (ME) cleft and reconstruction of tegmen and dural defects. Pneumocephalus was provoked by decreased intracranial pressure (ICP) secondary to placement of lumbar drain, which caused direct communication between unsutured dural defect and the defective posterior wall of external auditory canal skin. A revision surgery of combined transmastoid/middle cranial fossa approach was performed for intracranial decompression followed by appropriate closure by suturing the dura, obliterating the ET and ME.
Keywords
Introduction
Pneumocephalus usually develops after trauma, neurosurgical procedures, surgery of anterior skull base, and rarely with otological lateral skull base surgeries. 1 It can be classified as acute <72 hours or delayed ≥72 hours. 2 On the other hand, spontaneous pneumocephalus is not related to any of the previous causes, and its incidence in the literature is rare and commonly associated with congenital skull base defect. 3 Common factors are associated with spontaneous pneumocephalus, which create a forceful air pressure changes and facilitate air entry into the skull such as flying, scuba diving, nose blowing, sneezing, coughing, and Valsalva maneuver. 3 We present a case of otogenic pneumocephalus which developed 10 days after surgical treatment of right temporal meningoencephalocele (MEC) and reconstruction of the tegmen and dural defects. This type of pneumocephalus was managed surgically by suturing the dura, obliterating the Eustachian tube (ET) during a revision of combined transmastoid/middle cranial fossa (MCF) approach. The patient showed improvement with no recurrence.
Case Presentation
A 58-year-old male patient, who had recurrent right chronic suppurative otitis media with a history of multiple right ear surgeries, ended with right tympanomastoid obliteration with blind sac closure 15 years ago. Patient presented with hearing loss in the right ear, no ear pain, discharge, tinnitus, or vertigo. There was no fever, headache, neck pain, photophobia, visual changes, neurological deficits, history of trauma, or meningitis. Examination showed normal left ear, and right ear blind sac with bulging cyst and no discharge, no tenderness, or redness. Rinne test was positive on left ear and negative on right, Weber test lateralized to the right ear, and bilateral normal facial nerve examination. Other ENT and neurological examinations were also unremarkable. Pure-tone audiometry indicated that left ear had normal hearing level, while right ear had moderate–severe conductive hearing loss with air–bone gap of 45 dB. Tympanometry showed type A on left ear but could not be done on right ear because of the blind sac. Computed tomography (CT) of the temporal bone demonstrated a defect in the right ear tegmen tympani and mastoideum, measured approximately 19 mm in diameter, with opacification filling the external auditory canal (EAC) and middle ear (ME) space (Figure 1A). A magnetic resonance imaging was obtained to examine the brain tissue for possible herniation, revealing an MEC herniating from tegmen defect extending anteriorly and filling the right ME cavity (Figure 1B). Diffusion-weighted imaging also pointed to cholesteatoma in ME medial to herniation (Figure 1C).
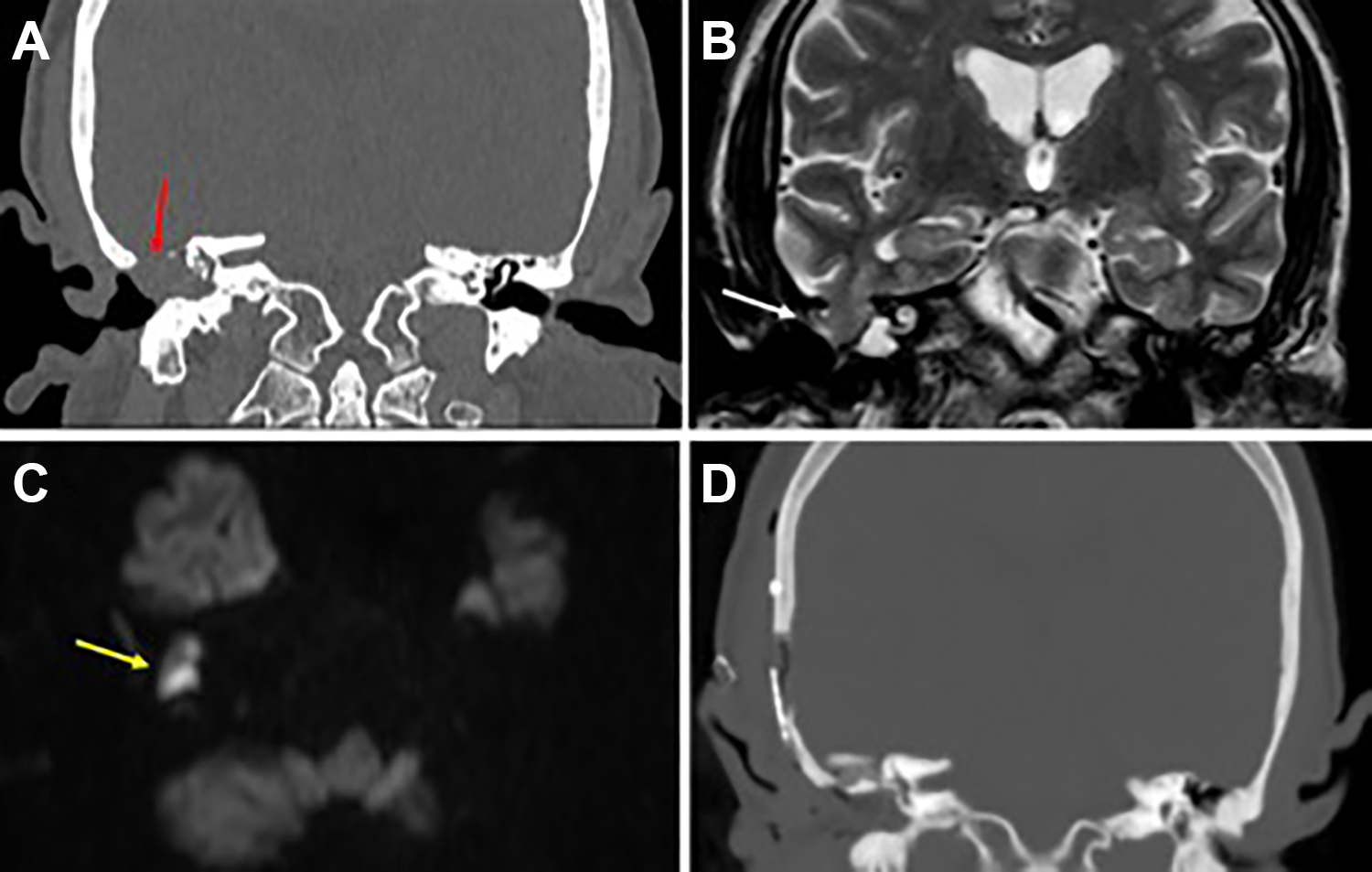
Preoperative computed tomography (CT) and magnetic resonance imaging (MRI) coronal views showing the tegmen defect (red arrow) and brain herniation (white arrow) in the right middle ear cavity (A and B). The MRI axial diffusion-weighted imaging view showing right hyperintense lesion (yellow arrow) which represents cholesteatoma (C). Postoperative CT coronal view showing removal of meningoencephalocele with defect repair and no postoperative pneumocephalus or other complications (D).
Patient was operated using a combined transmastoid/MCF approach for resection of the MEC and reconstruction of the base of skull defect, assisted with a neurosurgical team. The surgical team used a reverse question mark incision from the mastoid bone to posterior–superior direction. The temporalis muscle was reflected along its pedicle, and craniotomy performed. The defect was found at tegmen tympani and mastoideum, which measured nearly 1 cm × 1.9 cm in greatest dimension. Then canal wall-up mastoidectomy was performed, revealing a sclerotic mastoid. The herniated dura and brain tissue which occupied the attic, antrum, part of EAC, and extended deeply in the ME were resected. Cholesteatoma was identified and removed from mesotympanum, hypotympanum, and protympanum. Ossicles were absent except for stapes, and the facial nerve was preserved. The tegmen was reconstructed in an underlay fashion with multiple layers, from up to down as follows: fibrin sealant patch (Tachosil) to cover the dura, a periosteal tissue, and bone graft. Then the attic was closed to support the previous reconstruction by a chip of bone and cartilage under it without obliteration of ET. A mix of bone dust and Tachosil was used to obliterate the mastoid, while cartilage graft was used for tympanic membrane. The surgical team planned for second-stage surgery for recurrent cholesteatoma and hearing reconstruction. Immediate minimal cerebrospinal fluid (CSF) leak was managed by lumbar drain, which was placed at a level of 15 cm H2O. In the second postoperative day, CT scan of the temporal bone showed no complications (Figure 1D). During hospitalization, no further CSF leak was noticed and the lumbar drain was minimal with an average of 90 mL/d for 4 days and was removed. Upon discharge, patient showed normal neurological and otological evaluation and patient was instructed to avoid straining, nose blowing, and Valsalva.
After 10 days from surgery, patient presented with severe headache and new left lower limb weakness without any other neurological manifestations, preceded by a history of forceful nose blowing. Examination showed normal conscious, oriented patient, and vitally stable. Lower limb examination showed power of 4/5 bilateral and 5/5 for the upper limbs bilateral with normal sensation, and no pronator drift. The remainder of the neurological examination was unremarkable. Facial nerve examination was normal and right ear examination showed hard blood clot with no active ear discharge and good healing cartilage graft at tympanic membrane. Surgical wound area healed well with no dehiscence, fistula, or signs of inflammation. New CT imaging was obtained, showing a newly developed right-sided frontotemporal pneumocephalus and causing mass effect pressure (Figure 2A-C).
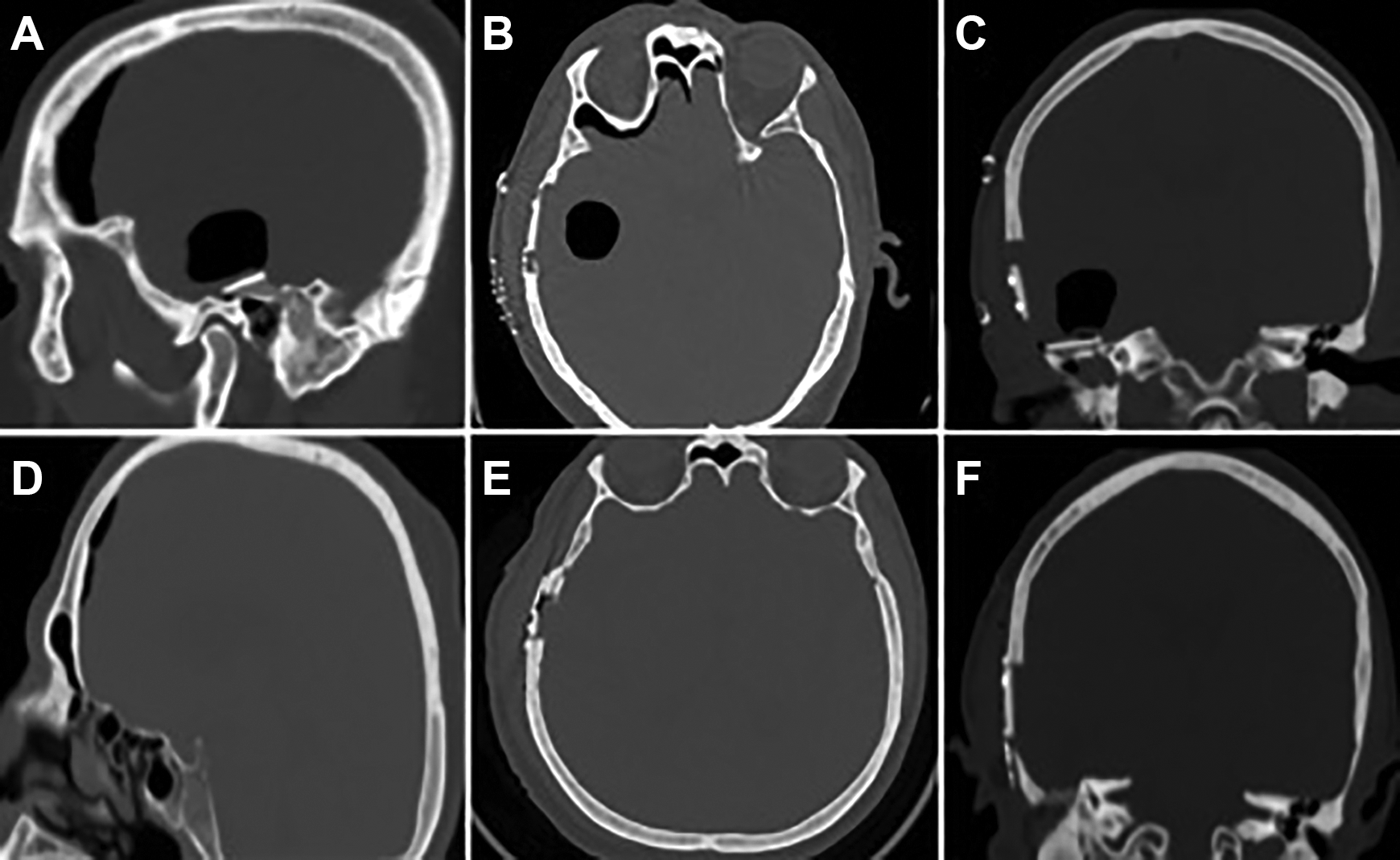
Computed tomography scan of the right side pneumocephalus after 10 days from surgery: sagittal, axial, and coronal views (A, B, C). Postoperative revision of tegmen repair with Eustachian tube obliteration showing improvement in pneumocephalus (D, E, F).
After admission, patient had one episode of seizure, received medications, and was controlled. Pneumocephalus was managed surgically by revision combined transmastoid/MCF approach for intracranial decompression, defect closure, and ET obliteration. The previous reconstructed tegmen defect was readily identified and the bone graft was removed. The previous periosteal graft was sutured to the dura for establishing better closure of dura, then Tachosil was applied and then muscle flap was used to cover the defect followed by glue and reinforced by small bone to seal the defect. The ET was obliterated with periosteum, fat tissue, and fascia. The ME and mastoid was filled with abdominal fat. Intraoperative decision was made to avoid blind sac closure of EAC since the patient is having fair residual hearing and a patent EAC will provide good monitoring for future recurrence of cholesteatoma. A follow-up CT temporal bone showed subsided pneumocephalus (Figure 2D, E, and F). After 2 months, there was unremarkable radiological, neurological, or otological findings.
Discussion
Temporal bone MEC consists of herniated brain tissue and meninges, with or without CSF leak, through bony defect in the skull, which can be divided into acquired or congenital which lead to spontaneous type.4,5 There are rarely reported conditions with spontaneous type with an incidence of 8.6%. 5 The spontaneous type can be categorized into congenital or idiopathic. 5 Congenital cases usually present during childhood which result from defects in otic capsule, while idiopathic cases are present during adulthood which result from defect in tegmen tympani or mastoideum. 5 An elevation in baseline intracranial pressure (ICP), focal inflammation, or aging process can lead to rupture of dura, which attributed to forming MEC. 6 Acquired otogenic MEC is common, caused by erosion of tegmen tympani or mastoideum secondary to ear infections, cholesteatoma, tumors, trauma, and surgeries such as mastoidectomy, which is the most common individual cause. 5 A study conducted by Jackson et al reported a series of 35 cases with MEC, 88.6% of them attributed to otologic infections or surgery. 7 Otogenic MEC may remain asymptomatic for years, until it develops hearing loss, CSF leak, headache, intracranial infections, or facial nerve weakness. 8
The treatment of otogenic MEC is surgical, and different approaches can be used depending on many factors such as the size of the defect and its location, hearing condition, ME status, and associated comorbid conditions. 4 For patients with small defects in tegmen tympani, mastoideum, or posterior fossa, transmastoid approach can be used. 4 The MCF approach is typically used in large defects (>2 cm2), multiple tegmen defects, or lesion involving anterior epitympanum with normal hearing. 4 If the ossicular chain is involved by the lesion, or ME infection/cholesteatoma present, a combined transmastoid/MCF is optimal, which provides meticulous eradication of mastoid and ME pathology, accurate dural closure, and adequate support of intracranial contents. 9 The success rate of MCF or combined transmastoid/MCF approach has been evaluated with an average of 80%. 10
Otogenic pneumocephalus usually occurs due to a defect in the temporal bone that leads to development of fistula permitting communication of ME with the intracranial cavity, which is related to trauma, ear infections, ear surgeries, or congenital defects. 11 In literature, the most common presenting symptom is headache followed by otorrhea, meningeal signs, aphasia, vestibular symptoms, paralysis, seizures, and coma.1,12 Our patient presented with headache and new left lower limb weakness with no other otological or neurological complaints. Generally, for pneumocephalus to occur, persistent negative ICP and/or positive pressure should preclude. Negative ICP occurs after CSF loss from pathological or intentional drainage, which will be replaced by sucked air through dural weak points. This mechanism is known as “inverted soda bottle.”2,3 The second mechanism is called “ ball valve,” which is induced by a defect in the temporal bone with a positive pressure gradient by factors such as nose blowing, straining, or Valsalva that eventually cause air entry.2,3 In the present case, the mechanism of pneumocephalus can be explained as follows: an intracranial hypotension was induced by previous placement of lumbar drain. Then, an existing defect in the skin of posterior wall of EAC and a nonsutured dural defect from previous surgery of MEC caused a direct communication among the EAC, subcutaneous tissue, and the subarachnoid space, which were associated with a history of nose blowing (Figure 2C).
Management of pneumocephalus depends on the underlying cause and severity of condition. Tension pneumocephalus is considered life-threatening and requires emergent surgical evacuation, while nontension or asymptomatic cases can be managed conservatively.2,13 Surgical management is preferred over conservative management to equalize the pressure and seal the defect using a combination of autologous, muscle, fascia, bone grafts, with artificial fibrin patches or glue.14,15 In addition to the previous steps, ET obliteration was mentioned in a few reports, which has been used to eliminate the route of air entry and reduce the recurrence.11,15
In this case, the development of pneumocephalus could be avoided with complete separation of the subarachnoid space from the subcutaneous tissue by placing a barrier by suturing the dura with temporalis fascia and avoiding lumber drain insertion as possible in the early days after surgery except in heavy CSF leak. Also, according to the patient history, the onset of symptoms related to pneumocephalus, started shortly after an attempt of forceful nose blowing which pointed out to a possible route of air entry through a patent ET in addition to a potential route through a defect in the EAC as mentioned above. Therefore, to address all probable sources of air entry into the ME and pressure changes, ET obliteration is reasonably considered as an additional attempt of prevention of such scenario.
Conclusion
Otogenic pneumocephalus is rarely reported in the literature. For its occurrence, it requires both factors to be present: a defect in the temporal bone and a pressure gradient among this defect. It should be suspected when patients present with nonspecific neurological manifestations and otological symptoms. It is usually managed surgically to equalize the pressure, repair the dura, and seal the defect, finally obliterating the ET.
Footnotes
Authors’ Note
We declare that this manuscript is original, have never been published, or under the consideration for publication elsewhere (in part or in whole). We also declare that the corresponding author and all of the coauthors have actively participated in this manuscript. No Arabic abstract and Arabic article for this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
