Abstract
Ameloblastoma (AM) is a slow growing and aggressive benign tumor with an odontogenic epithelial origin arising from the mandible or maxilla. The odontogenic neoplasm invades local tissues asymptomatically and accounts for 1% of oral tumors and over 10% of odontogenic tumors. A 64-year-old man with a history of allergic fungal rhinosinusitis (AFRS) undergoing a revision image-guided endoscopic sinus surgery was found to have a fibrous mass suspicious of malignancy projecting inferolaterally and attached to the floor of the left maxillary sinus. Diagnostic biopsies were taken, and additional surgery was required to successfully resect the tumor via a transnasal endoscopic dissection. Multiple permanent pathology samples concluded the diagnosis of an AM. Endoscopic investigations led to the incidental discovery and ultimate complete endoscopic resection of the AM. The utilization of an endoscopic resection compared to the traditional maxillectomy with reconstruction results in significant less short and long-term morbidity for the patient.
Introduction
Ameloblastoma (AM) is an uncommon odontogenic neoplasm that originates from the epithelium of the dental lamina. 1 Ameloblastoma comes from the French word “amel” denoting enamel and the Greek word “blastos” referring to sprout or germ(inate). 3 The histologically benign tumor presents within the mandible (80%) or maxilla (20%) as an aggressive odontogenic tumor that slowly invades surrounding tissues in an asymptomatic and painless manner.2,3,4 In extraordinarily rare cases, AMs can arise from a primary locus within the sinonasal tract in contrast to their predominant gnathic origin. 5 Although it is a persistent lesion with a high recurrence rate to various conservative treatments, ameloblastic carcinomas are only present in 2% of the cases. 6
Ameloblastomas have a male predilection and tend to occur in the posterior aspect of the mandibular molar region. 6 According to the new 2017 World Health Organization (WHO) classification of AMs, they are further classified into clinical subtypes: conventional, peripheral, unicystic, and metastatic. 7 Conventional AM, historically known as multicystic AM, has various histopathological subtypes: follicular, plexiform, desmoplastic, and acanthomatous AMs. 7 Unicystic AMs can further be broken down into luminal, intraluminal, and mural. 7 Metastatic AM, despite being histologically benign, most commonly metastasize to the lungs.7,8
Ameloblastomas are almost always resected via an open (partial maxillectomy) approach with wide margins, leaving the patient with a large defect that is then reconstructed with free flaps. The surgery and recovery are fairly morbid for the patient in most cases. There have only been 2 reported cases in the literature of endoscopic resection of AM, one with a 4-year follow-up and one with an 11-month follow-up period.9,10 This report presents a patient whose diagnosis of AM was made as a secondary discovery to his initial underlying disease. The diagnostic and endoscopic surgical techniques that were utilized throughout the patients’ care are discussed alongside a brief review of the literature focusing on AMs.
Case Report
A 64-year-old asthmatic Caucasian male diagnosed with recalcitrant allergic fungal rhinosinusitis (AFRS) and no history of smoking presented to the Sinus Centre for revision image-guided endoscopic sinus surgery (ESS). During the procedure, a fibrous mass arising from the floor of the left maxillary sinus was discovered incidentally and biopsied. Postoperative histopathological findings concluded that the lesion contained cyst-like structures, lined by columnar epithelial cells and divided by spindle cell-type material of stromal appearance. The diagnosis of a maxillary AM was made and an additional surgery via an all endoscopic approach was scheduled for its removal.
Six weeks after the patient’s ESS, the patient had been continued on medical management for his AFRS, including topical budesonide via a Mucosal Atomization Device, saline rinses, betadine intranasal rinses, and long-term clarithromycin.
A referral was made to the cancer agency to have a positron emission tomography scan completed, and subsequently, a computed tomography (CT) scan was ordered (Figure 1). The AM appeared to be confined to the inferior aspect of the left maxillary sinus without bone invasion or metastasis. The mass was secured inferomedially to the dental root of the upper left third molar (tooth 2-7) and extended inferolaterally into the left cheek.
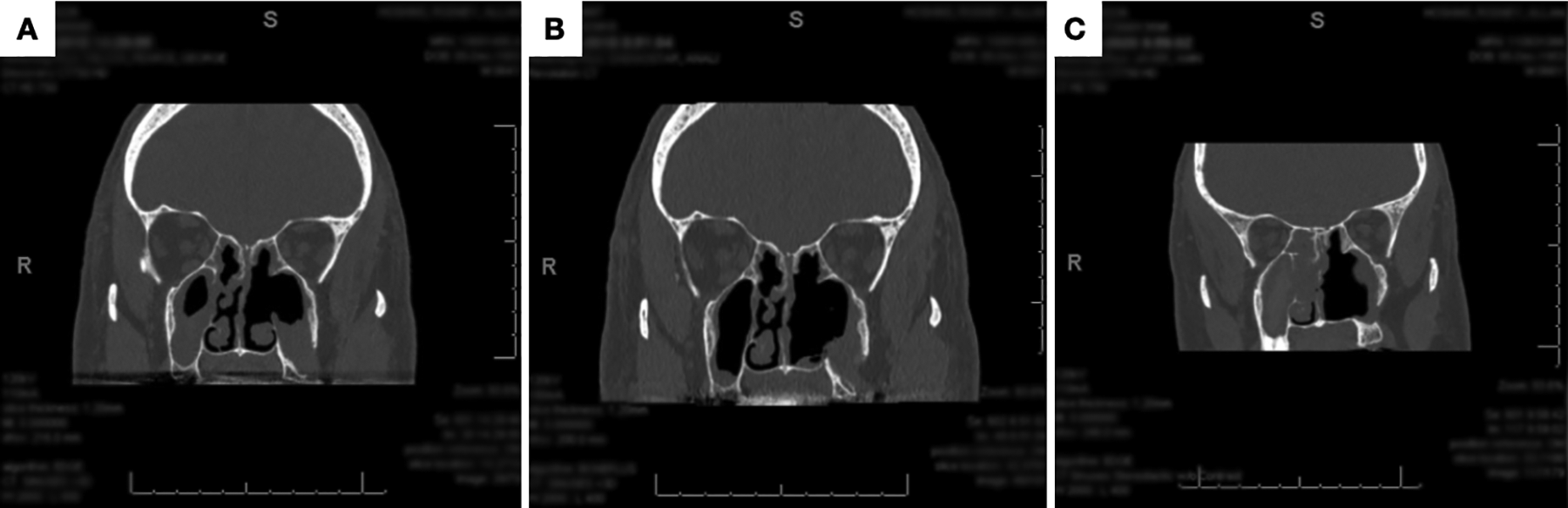
(A) A preoperative and (B) a 3-month postoperative coronal computed tomography (CT) of the growth shows the resection margins with minimal defect to surrounding structures. C, A 15-month follow-up postrecurrence surgery CT scan showing no signs of recurrence.
The patient was taken back to the operating room 3 months after the initial sinus surgery for endoscopic resection of the left maxillary sinus AM. An image-guided, transnasal endoscopic approach was utilized to perform an inferior turbinectomy and endoscopic medial maxillectomy. All recurrent nasal polyposis was debrided and allergic mucin removed, reinforcing the severity and symptoms of the patient’s recalcitrant AFRS. A left inferior turbinectomy and medial maxillectomy was then performed using angled instrumentation in order to maximize access to the site of the tumor within the inferior aspect of the maxillary sinus floor. The borders of the mass within the maxillary sinus floor were identifiable and were delineated with sharp cauterization approximately 5 mm from the gross edges of the tumor. The mucous membrane was then elevated from the maxilla leaving behind bare and dehiscent bone. Following the complete gross endoscopic removal of the mass lesion, a 4-mm 70° angle diamond burr was utilized to drill and polish the bone from the floor of the maxillary sinus extending into the root of the 2-7 molar. Monopolar cauterization was utilized at the tooth root to eradicate possible tumor remnants. Frozen and permanent histopathology samples were taken from the edges of the tumor dissection for margin inspection. Surgiflo hemostatic matrix (Johnson and Johnson Inc) was then applied to cover the area of the resection.
The patient fared well with complete resolution of the tumor mass with no signs of recurrence at his follow-up at 8 weeks (Figure 2) and 4 months after surgery. The patient did develop permanent left upper molar tooth (2-7 tooth) numbness, which was therefore extracted by his dentist. Unfortunately, 6 months after surgery, a small recurrence was noted, and the mass was again endoscopically excised with frozen and permanent negative histopathological margins. There are currently no signs of recurrence 15 months post recurrence surgery. To our knowledge, this is only the third strictly endoscopic excision of a maxillary AM without the need for external maxillary excision and reconstruction.
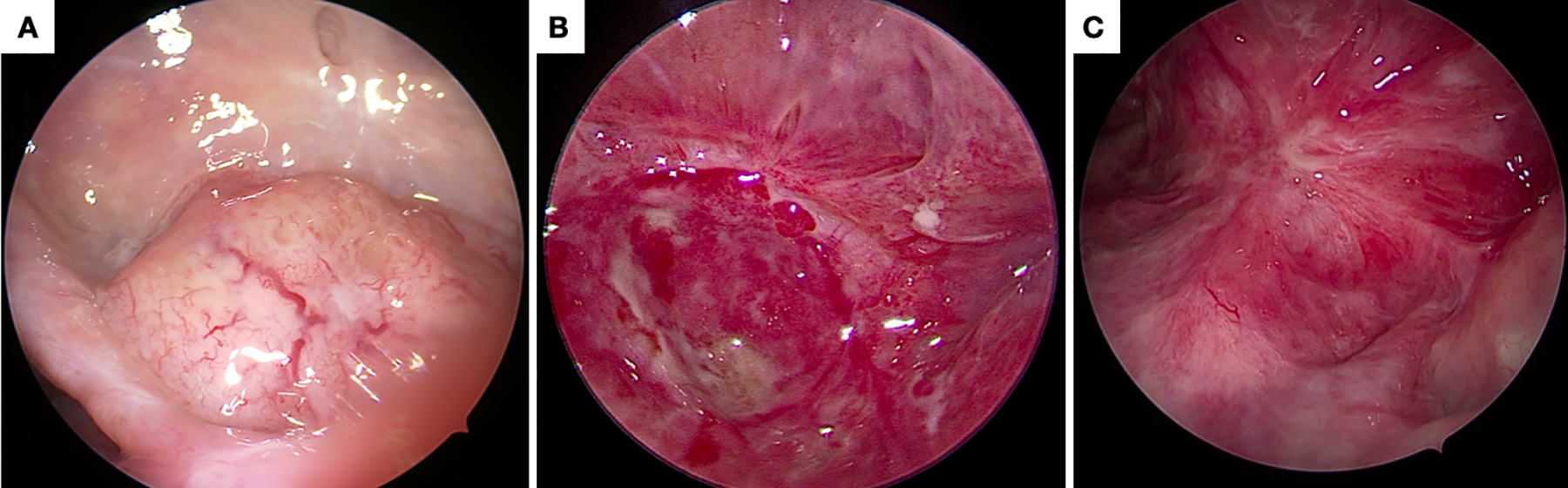
A, A nasal endoscopic image displaying an intraoperative view of the intact left maxillary ameloblastoma. B, Two-month postoperative endoscopic image of the healed maxillary sinus floor. C, Endoscopic image at 15 months post recurrence surgery showing healed sinus floor mucosa.
Discussion
Maxillary AMs are rarer than their mandibular counterparts and are presumed to be more aggressive with a 50% rate of recurrence within 5 years of initial resection. 7 This case presents a maxillary AM that expanded into the floor of the maxillary sinus. Ameloblastomas that develop in the sinonasal tract tend to develop around the age of 60 arising in continuation with the sinonasal mucosa and lack a connection to gnathic origin. 7 A single biopsy sample provided to the pathology department from the initial surgery contained an array of plexiform, follicular, and acanthomatous-type characteristics (Figure 3). According to Vickers and Gorlin criteria, there are certain histopathological variations that delineate AMs from other odontogenic neoplasms. These include hyperchromatism of basal cell nuclei, nuclear palisading with reverse polarization of basal cell nuclei, and vacuolization of basal cell cytoplasm. 11 Each of the determinants listed were visible within the tumor distinguishing the neoplasm from other possible differential diagnoses such as a dentigerous cyst or an odontogenic myxoma. 12
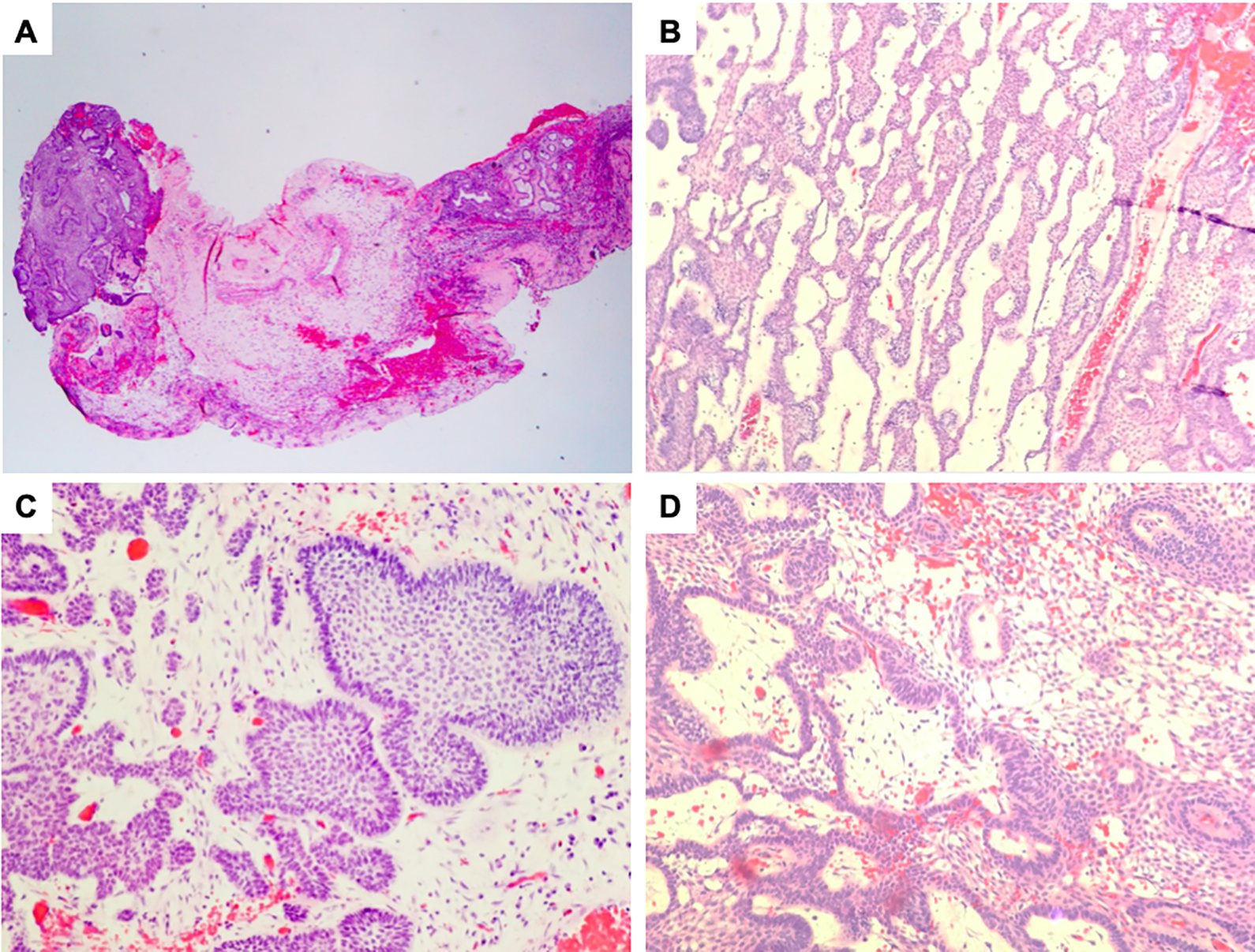
A, Ameloblastoma involving sinonasal mucosa. B, Anastomosing cords of epithelial cells form a plexiform arrangement. C, Peripheral palisading columnar cells with nuclear reverse polarization form a follicular arrangement. D, Edematous cysts mimic the stellate reticulum with palisading columnar cells exhibiting reverse polarization of the nuclei.
Also, from a molecular viewpoint, maxillary AMs have been shown to exhibit the gene mutation Smoothened (SMO) at a high incidence. 13 AMs occurring in the maxilla harbored mutations of the sonic hedgehog (SHH) pathway, which then activated SMO gene mutations. 13 The SHH pathway mutations are instrumental in the formation of the initial growth of the maxillary AMs. SHH pathway inhibitors have been shown to be beneficial in other cancers and may prove to be beneficial for nonsurgical treatment of maxillary AMs. Agents such as cyclopamine, which antagonizes SHH signaling, and KAAD-cyclopamine, which inhibit GLI proteins that are responsible for mediating SHH signaling, may be potential chemotherapeutic contenders. 14
The primary modality used to treat AMs is surgery. Conventional variants of AM are the most common and infiltrate aggressively into surrounding tissues. Radical surgical resection with a margin of at least 1 to 2 cm is ideal to ensure all tumor remnants are removed.15,16 A partial or a complete maxillectomy has been shown to be extremely effective in preventing recurrence when compared to conservative treatment, such as curettage, which has a recurrence rate of 60% to 80%.7,15,17 A maxillectomy can leave large maxillary defects and scaring that can subsequently lead to various morbidities. 17 Morbidities such as velopharyngeal incompetence and muscle scarring can lead to decreased facial movement and trismus. 17 Although chemotherapy and radiation therapy may be beneficial for a variety of tumors, they are used primarily as a palliative treatment or for when surgery is not an option in the case of AM. 15
Endoscopic sinus surgery can revolutionize the management of select AMs. It has proven to have very good outcomes for various other sinonasal tumors such as inverting papillomas and skull base tumors. 18 Endoscopic techniques spare facial scarring and minimizes the occurrences of maxillary defects in comparison to a partial or total maxillectomy. It is an invaluable tool that can be used to aid localization and access of a tumor using a miniature lens with a magnified operative field of view.
Although ESS is very practical, it does have its limitations, such as accessing the maxillary sinus. 19 Access through the middle nasal meatus is very complex. As a result, a transnasal endoscopic medial maxillectomy can be performed facilitating access to the maxillary sinuses and beyond. 19
In this case, the patient underwent an endoscopic transnasal resection of an AM that had grown into the floor of the left maxillary sinus. The AM was felt to be small and accessible enough to be amenable for an endoscopic approach. Also, during counseling, the patient was given a choice between an open approach partial maxillectomy versus an endoscopic medial maxillectomy approach.
The mass was confined within the maxillary sinus superior to the maxillary third molar (2-7 tooth) and had not infiltrated into either the oral cavity or intracranially. Therefore, the mucosa overlying the implicated tooth root was removed and the tumor mass widely drilled off. In an attempt to preserve the functionality of the patients’ teeth, the tooth root was polished using a diamond tip burr, with the tooth itself being left intact initially. The tissue within the maxillary sinus healed in the immediate postoperative period. However, between the second and fourth month postoperative visit, the patient had developed tooth paresthesia of the implicated dental region. Terminal branches of the posterior superior alveolar nerve can often be damaged during a maxillary floor drill out.20,21 The patient’s implicated 2-7 tooth was therefore extracted by his dentist. At the patient’s 6-month follow-up visit, a small recurrence was noted on endoscopic examination which was promptly resected with negative margins. At 15 months post his recurrence (final) surgery, the patient has not had any further symptoms or signs of recurrence both endoscopically or on CT imaging studies. As per the WHO’s description of AM prognosis, we strongly recommend q3 monthly endoscopic evaluation and q6 monthly follow-up CT imaging studies during the first 2 years, followed by yearly CT imaging studies for the following 3 to 5 years to continually monitor for any future recurrences. Beyond this, we further recommend q6 to 12-month lifelong endoscopic follow-up. 6
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
