Abstract
Electromagnetic interference shielding clothing has been developed for people who is sensitive to electromagnetic radiation or workers working under extremely high electromagnetic radiation circumstances. The challenge was developing the fabric with good washability, durability, and air permeability. After three machine washing cycles, the electromagnetic interference shielding effectiveness can drop more than 99% for untreated copper-coated fabric. In this research, the chlorinated poly-para-xylylene (parylene C) encapsulating technology was used to protect the fiber's copper particles. The result shows that the treated sample's washing ability will significantly improve after processing 15 g parylene-C on the copper-coated fabrics. The electromagnetic interference shielding effectiveness can remain at 39.93 dB and 25 dB on average from 30 MHz to 3 GHz after 10 hand washing cycles and 10 machine washing cycles, respectively. The air permeability remains around 1043.6 mm/s for a 15 g parylene-C encapsulated sample. The chemical resistance property was also improved significantly after encapsulation of 15 g parylene-C. For the 15 g parylene-C encapsulated samples, there is less than 3% loss of electromagnetic interference shielding effectiveness after 8 h of immersing into pH = 2 and pH = 12 solution. The overall structure of the 15 g parylene-C encapsulated fabric remains intact after 6000 abrasion cycles. The study presents an effective method for fabricating highly durable, comfortable electromagnetic interference shielding fabric, guaranteeing reliability for technical clothing applications and showing great potential for further development.
Keywords
The debate about the hazard of high electromagnetic interference (EMI) pollution generated by the telecommunication of electronic devices to human health has been growing due to the fast development of modern electronic technology.1,2 As we know, people encounter electromagnetic radiation in everyday life which is not harmful to human health. 3 However, for special sensitive groups, such as patients who have undergone cardiac pacemaker implantation operations, workers who have long worked in environments with high electromagnetic radiation, and pregnant women living in environments with high electromagnetic radiation, 4 appropriate electromagnetic shielding measures need to be taken to shield extra electromagnetic radiation. 5 Especially for patients with implanted cardiac pacemakers, a higher electromagnetic radiation environment will affect the regular work of the pacemaker, which causes a lot of inconvenience and health risks for patients.6,7
Wearing the EMI shielding technical clothing to block the extra electromagnetic radiation, has been approved as one of the efficient methods for EMI shielding, which is capable of attenuating electromagnetic radiation propagation efficiently via reflection, absorption, and multi-reflection inside the material.8–10 Among them, conductive textiles fabricated by assembling metal particle coatings on fabrics through the mass-producible ways of electroless or electroplating are one of the most typical representatives. These metalized textiles are widely used as fabric for EMI shielding clothing.11–14 The carbon-based material also performs high EMI shielding effectiveness (SE) originating from highly conductive carbon fiber, particles, and carbon nanotube (CNT) coating, as well as excellent mechanical strength and flexibility.15–17 The nanostructured MXene/metal oxides integration for textile EMI shielding was also developed for EMI shielding application18–20 but still not widely used for protective clothing.
Regardless of the surface treatment method or mixing conductive fiber, one of the main problems of modified textiles for EMI shielding textiles was the washing durability and air permeability after the protection layer coating. 21 The majority of electromagnetic shielding clothing available on the market was not washable. 22 To solve this problem, a protection layer was necessary for better washing durability to keep the conductive layer stable after the abrasion process during the washing cycle. Wang et al. 23 reported that the silicon-coated MXene-decorated polyester textiles performed outstanding EMI shielding properties about 66 dB. The EMI shielding performance could maintain stability (>30 dB) over washing with the commercial detergent solution for different cycles. Xing et al. 9 developed the polyurethane film-covered carbon fabric with plated Ag particles that performed high conductivity and ultra-high SE (102.98 dB). However, the air permeability and washability of this material was not tested. Zou et al. 24 reported that the polyaniline (PAni) coated fabric with carbon nanotubes (CNTs) coating shows good SE around 23 dB with a high absorption ratio. After being washed in standard laundry detergent (OMO brand), the average SE of the coated fabric was 21.1 dB. Unfortunately, the air permeability of the fabric has not been tested as well.
From the result of our following research review,even if a certain degree of washing resistance was guaranteed, the air permeability of the treated fabric will be significantly reduced or even airtight. For example, as referred to in the study of Wang et al., 23 the air permeability of a silicon-coated sample was around 130 mm/s. The choice of coating technology and material of the protective layer significantly affects the fabric's breathability. Yang et al. 25 used the parylene deposition method to encapsulate the Cu/Ni coated polyamide fibrous membrane. The result showed superior electromechanical stability after 50 cycles of machine washing.
Parylene-C is the generic name for chlorinated poly-para-xylylene polymer. 26 The molecular structure is linear and polycrystalline in nature, which has been widely employed as an encapsulation material due to its superior moisture and chemical resistance properties, thermal and ulytraviolet (UV) stability. 27 Parylene coatings were effective at thin levels between 0.1–70 µm. This advantage makes it superior for employing smaller items or thickness-sensitive subjects. The specialized chemical vapor deposition (CVD) process ensures that parylene penetrates entirely into the structure and generates film to encapsulate the substrate surface. 28 These properties make parylene coatings ideal for several medical devices, electronics, transportation, defense, and aerospace applications. 29
Inspired by this study 25 , this research successfully developed ultrathin washable and air-permeable EMI shielding textiles with parylene-C encapsulation technology. In this article, to realize the combined function of washing ability and air permeability, a porous, stable, and anti-abrasion copper-coated polyester fabric with encapsulated parylene-C film (Py/Cu/PET) structure was elaborately designed and fabricated. The developed material shows high washing stability and maintains excellent air permeability. After the parylene encapsulation process, the sample's acid and alkali resistance properties improved significantly. It also demonstrated suitable anti-abrasion properties, guaranteeing reliable structure integrity even after 6000 abrasion cycles.
Regarding the enhanced mechanical property, the parylene-C encapsulated material should be able to be used as the technical clothing fabric for EMI shielding technical clothing. The excellent air permeability and flexible structure are suitable for integrating the fabric as a functional layer for EMI shielding clothing.
Experimental section
Materials
Greige fabric
The commercial name of the greige fabric was “MILIFE,” which was produced by JX Nippon ANCI, Japan. MILIFE is 100% polyethylene terephthalate (PET) nonwoven fabric which constructed from aligned warp PET monofilaments and PET monofilaments via thermal bonding method. The different orientated fiber (not regular net) was due to inaccuracies of industrial fabrication mainly. This structure and orientation of fibrous elements cannot be changed because of solid bonding spots. The basic information of the greige PET nonwoven fabric, which referred from the technical sheet of MILIFE is listed in Table 1. 30
Basic characteristics of polyethylene terephthalate (PET) nonwoven fabrics MILIFE

Metal coating process
The copper (Cu) particles loaded on the nonwoven polyester fabric was successfully realized via the electroless plating method from our previous research.31,32 The detailed steps and parameters are presented in the Table 2. This process includes three steps: surface treatment, activation, and deposition. Considering the requested size of sample and to ensure the better homogeneity of the material, in this study, the Cu-coated nonwoven fabric, commercial name “MEFTEX,” was purchased from Bochemie a.s, Czech Republic. To ensure the specific porosity of the fabrics, Meftex 10 was selected, which has the highest porosity compared to other company products. Meftex 10 was produced via a patent pended technological process based on subsequent chemical and continuous electroplating processes. The first step for greige PET fabric is activation. Surface activation was performed by using an activation solution (CATAPOSIT44, supplied by Rohm and Hass company, The Netherlands) at 45°C for 5 min, the fabrics were immersed in 10% hydrochloric acid. After activation, the treated fabric goes through the “roll to roll” Cu metal deposition process. The pre-activated PET fabric passes through a bath containing CuSO4. Similar to the electroless plating process, the Cu nanoparticles on the fiber surface were obtained by a reducing bath (based on borohydride). This production process created a continuous metal particle dense layer (Cu) on the fabric’s surface. The Meftex 10 is renamed Cu/PET for brevity in this article. The basic characteristics of Cu/PET are presented in Table 3. All chemicals used in this research were supplied from Sigma Aldrich, Czech Republic. All reagents were of analytical grade.
Electroless copper deposit method for polyethylene terephthalate (PET) nonwoven fabrics

Basic characteristics of copper coated polyethylene terephthalate (PET) nonwoven fabrics MEFTEX

Preparation of Py/Cu/PET by parylene deposition system
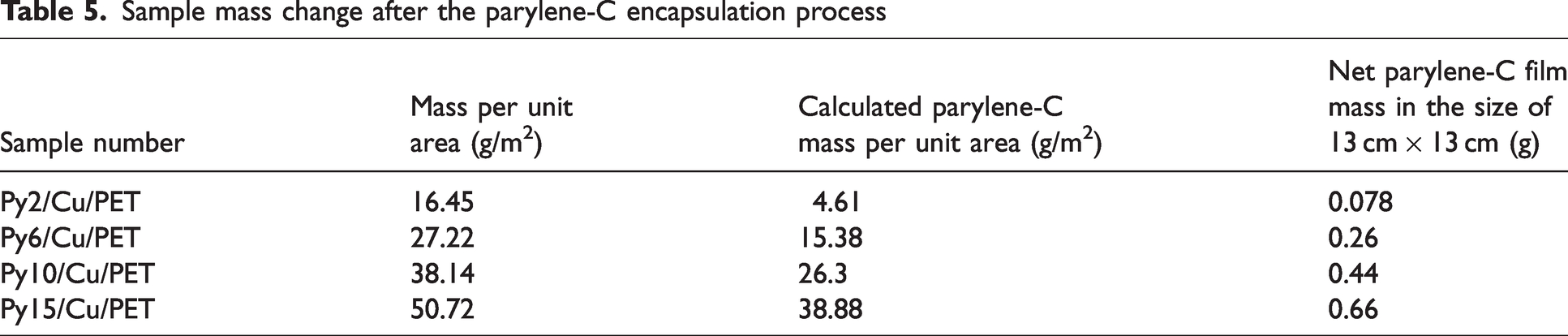
The parylene CVD processing was performed by the parylene deposition system (SCS PDS2010) located in CEITEC, Brno, Czech Republic. The basic properties of parylene-C are presented in Table 4.33–36 The Cu/PET sample was cut in the size of 13 cm × 13 cm, which matched the size of the sample holder in SCS PDS2010. After 24 h of conditioning in room circumstances, the Cu/PET was placed on a homemade supporting frame made from paper for a complete and uniform coating. Figure 1 presents the deposition process. The parylene deposition process was mainly performed in three steps: The first step was the vaporization of the dimer precursor at 150°C. The parylene-C dimer is the raw form of parylene-C. It is the solid particle form which can be inserted into the machine to be broken down through the deposition process. The addition of parylene-C dimer was controlled differently as 2 g, 6 g, 10 g, and 15 g for one deposition on the fabric size of 13 cm × 13 cm. The next step was pyrolysis of dimer into its monomeric form (para-xylene) at 680°C and the last step was the deposition of transparent parylene-C on target surfaces under a vacuum of 25 mTorr. According to the amount of parylene-C used in the encapsulation process, for brevity, the final parylene-C encapsulated sample was named Py2/Cu/PET, Py6/Cu/PET, Py10/Cu/PET, and Py15/Cu/PET. Due to the size of the Cu/PET and limited volume, adding 2 g parylene-C in the deposition machine does not mean that the sample will gain 2 g mass after the coating process. In the size of 13 cm ×13 cm sample, the mass is increased only 0.078 g after adding 2 g parylene-C in the deposition system. There is a similar effect for the 6 g, 10 g, 15 g samples. The increased mass of parylene-C is listed in Table 5.
Basic properties value of pure parylene-C film

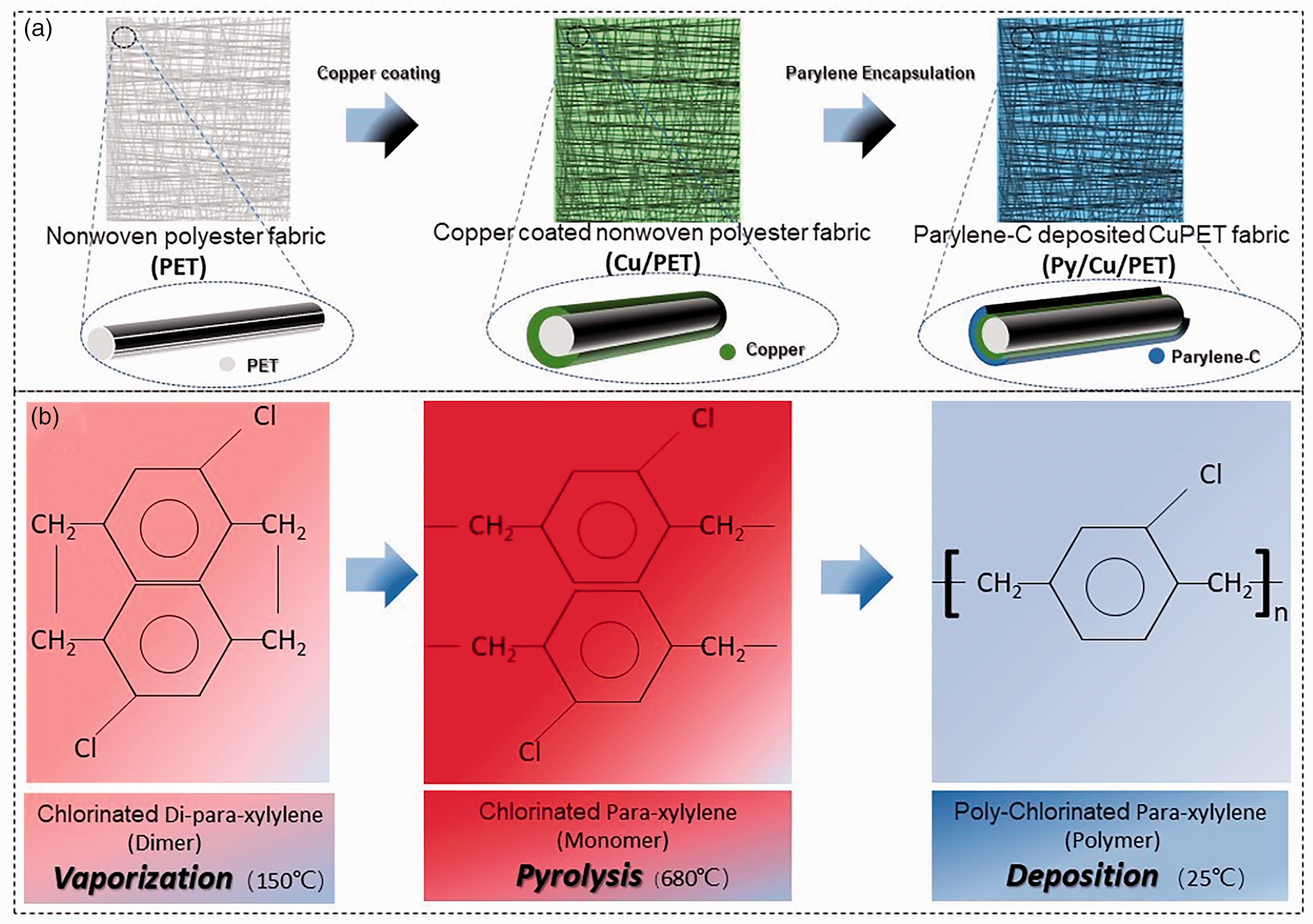
A schematic illustration of the fabrication process: (a) parylene-C (Py)/copper (Cu)/ polyethylene terephthalate (PET) preparation process and sample simulation photo. The insert fiber picture displays the feature of different material layer structures after parylene encapsulation and (b) The parylene deposition process on Cu/PET.
Sample mass change after the parylene-C encapsulation process
Characterization and evaluation
Morphology and physical characterization
Scanning electron microscope (SEM, including Bruker EDS, USA, and LYRA3 TESCAN, Czech Republic), energy dispersive X-ray spectroscopy (EDS), and the Rigaku D/Max 2500 X-ray diffractometer (XRD) with Cu Kα radiation at a scanning speed of 4 min−1 were employed to study the morphology and microstructure of the sample. Infrared spectra were measured with Fourier transform infrared spectrometer (FTIR) type Tensor 27 (Bruker). The wavelength was between 4000 cm−1 and 650 cm−1. Samples were tested in powder form in attenuated total reflection (ATR) mode with diamond crystal. The number of scans was 50, and the resolution was 4 cm−1. Fabric thickness was measured under pressure of 1 kPa using a thickness gauge (mm), as per standards ASTM-D-572937 (nonwoven samples). The thickness of coated parylene-C film can be easily calculated by the thickness of the treated sample (Py/Cu/PET) minus the untreated sample (Cu/PET). The air permeability impedance was tested by FX 3300 (TEXTEST Instruments), which was used at 200 Pa pressure drop to conduct the air permeability test according to the ISO 9237 standard. 38 The porosity calculation was processed via the software IMAGE J. The ultra-thin structure was one characteristic of the Cu/PET and Py/Cu/PET samples. From the image analysis, the nonwoven porosity can be expressed by the optical porosity, which was calculated percentage value from the aperture area from the observation part divided by the area of the observation part.
EMI shielding test
The EMI SE test was operated by the coaxial transmission line method according to the standard ASTM 4935-10.
39
According to this standard, it presumes a plane wave's impact on a shielding material at frequencies from 30 MHz to 3 GHz. The measuring equipment was constructed with a coaxial specimen holder (supplied by Electro-Metrics, Inc., EM-2107A). The input and output signals were connected to a vector network analyzer from Rohde and Schwarz ZNC3. The power ratio from output and input signal can be used for calculating the SE (forward transmission coefficient S21) without and with shielding material. The calculation equation is as equation (1):
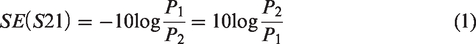
The electromagnetic wave reflection coefficient SER (S11) interpreted that the electromagnetic wave signal from the transmitting antenna was reflected by the shielding material and received by port 1. The ratio of the receiving reflected electromagnetic wave power P3 and P1 calculates the input reflection coefficient by equation (2):

According to the transmission line theory, the total SE can be calculated by the following equation (3):
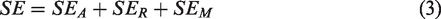
In equation (3), SE is shielding effectiveness, SER is direct reflection loss, SEA is absorption loss, and SEM is multiple reflection loss. The factor SEM can be ignored when the SEA > 6 dB. It is usually only significant when the shielding material is thin and at low frequencies (i.e. below approximately 20 kHz). 40
Washing test
Machine washing tests of Cu/PET and Py/Cu/PET were carried out with a washing machine (Miele Professional Maintenance System, Germany) according to the standard ISO-6330 with 20 ± 1 g ISO standard detergents for each cycle. 41 Considering the sample character and requirement of ISO-6330, washing procedure No.3G was selected. Samples were set into a protection bag. The washing cycle involved washing, rinsing, spin-drying, and room-temperature drying. The detailed washing procedure is listed in Table 6.
Washing cycle detailed procedure

The hand washing cycle used the same detergents from the machine cycle. To simulate the natural hand wash cycle, the whole cycle includes 2 min hand washing process and a 2 min rinsing process. As requested in the standard, the ballast cotton fabric is 100% cotton plain woven fabric, the piece size is 92 cm × 92 cm and fabric mass is 180 g/m2. The fabric was supplied from ZET, s.r.o, Czech Republic.
Abrasion test
Abrasive resistance is an essential factor in judging the abrasion durability of the parylene encapsulated sample, which the Martindale Abrasion Tester measured according to ASTM D4966-98. 42 Top to 10,000 abrasion cycle was applied for treated and untreated samples.
Chemical resistance test
The Cu-coated polyester is sensitive under an extreme pH environment. The Cu particle could be oxidated, and fiber corrosion can happen. The following method referred to the standard EN ISO 6529-2001: Protective clothing. Protection against chemicals. Determination of resistance of protective clothing materials to permeation by liquids and gases and GB 24539-2021 Protective clothing- Protective clothing against liquid acids and alkali. The acid and alkali resistance properties of Py/Cu/PET were tested for 8 h in NaOH (pH = 12) and H2SO4 (pH = 2) solution at ambient temperature (T = 23°C, RH = 49%). The sample was submersed into the pH-adjusted solution. After each 2 h, sample was cleaned with distilled water until pH = 7. After drying the sample, the EMI shielding test was performed to evaluate the loss of EMI shielding effectiveness.
Softness
The softness of the sample was evaluated by bending rigidity method regarding the standard CSN 800858, the samples were tested by using TH-5 equipment. The samples were made into rectangle shapes of 2.5 cm × 5 cm. The 2.5 cm side was clamped to the sensor jaws, then the device applied an external force to the sample. The final output of the device was the value of the bending force. Regarding the softness, with the lower bending force present the better the softness of the tested sample. The average value of five times of testing was calculated.
Results and discussion
Morphologies
The geometrical characterization of the parylene-C encapsulated sample Py/Cu/PET is listed in Table 7.
Geometrical characterization of copper (Cu)/polyethylene terephthalate (PET) and parylene-C (Py)/Cu/PET
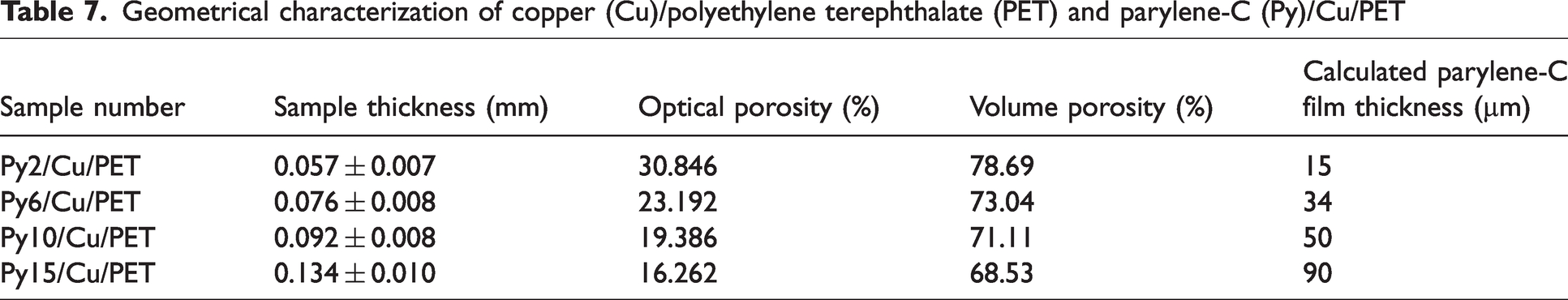
Compared with the operating mass of parylene-C, the thickness of parylene-film performs the positive linear correlation as shown in Figure 2(h). Py15/Cu/PET contains the heaviest coating material compared to other samples in the Py/Cu/PET. However, the Py15/Cu/PET thickness remains around 0.13 mm, which shows good potential for protective clothing or other applications for EMI shielding. The volume porosity (Vp%) was calculated according to the fiber volume ratio (Vfi%) when ignoring the mass of air in the sample, the calculation equation is presented as equation (4). The optical and volume porosity show the same trend that both porosities reduced with the increased amount of parylene-C.
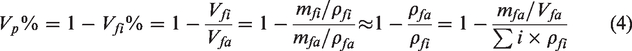
Morphology and physical characterization of copper (Cu)/ polyethylene terephthalate (PET) and parylene-C (Py)/Cu/PET: (a) scanning electron microscope (SEM) scanned picture of Cu/PET; (b) SEM scanned picture of Cu/PET focus on the copper particle on the fiber surface; (c) SEM scanned picture of Py15/Cu/PET; (d) SEM scanned picture of Py15/Cu/PET focus on the parylene film coated on the fiber surface; (e) EDS mapping of element Cu on the surface of Cu/PET; (f) EDS mapping of element Cu and element Cl on the surface of Py/Cu/PET; (g) EDS mapping of element Cu and Cl on the fiber of Py15/Cu/PET; (h) coated parylene-C thickness change with the increased amount of processed parylene-C; (i) XRD patterns of Cu/PET and Py10/Cu/PET and (j) attenuated total reflection (ATR)-Fourier transform infrared spectrometer (FTIR) spectra of sample Py15/Cu/PET.
The composited fiber density can be calculated via the sum of density from different component of the fiber times mass ratio (i).
The SEM image of the Cu/PET shows good porosity (Figure 2(a)). It can be observed that the Cu particles distributed evenly on the surface of the fiber (Figure 2(b)). During the parylene encapsulating process, the gaseous monomers of parylene-C were evenly distributed into the whole chamber under vacuum circumstance. Unlike traditional coating technology that blocks the pores of the fabric, the monomers of parylene-C were able to penetrate the apertures of the Cu/PET and deposit on the fiber surface evenly under ubiquitous gas-phase deposition at the molecular level. As a result, the apertures will not be blocked, which can maintain the good air permeability of the sample (Figure 2(c)). On the surface of the fiber, the parylene-C forms a dense film in 3D structure rather than two simple layers covering only on the top and bottom of the fabric surfaces. Such a hierarchical structure enables the highly pervasive protection for the Cu particles interconnector by parylene-C (Figure 2(d)).
Depending on the Cl element in the molecular structure of parylene-C, the Cl element can be the identity of the existing parylene-C film. By using EDS analysis, the untreated Cu/PET demonstrated that the Cu particles were evenly distributed on the surface of the sample (Figure 2(e)). After the deposition process, the EDS map shows the Cl element evenly spread on the treated sample Py15/Cu/PET (Figure 2(f)). On the focus of fiber level, the element Cl concentrated on the surface of the fiber. The Cu element was evenly distributed on the fiber surface. This EDS map illustrates that after the encapsulating process, the parylene-C film coved the Cu particles on the Cu/PET surface perfectly (Figure 2(g)).
The X-ray diffraction analysis (XRD) spectrum of Py/Cu/PET shows no apparent shift compared to untreated Cu/PET (Figure 2(i)). Therefore, the crystal morphology of Cu existing on the fibers did not change due to the chemical vapor deposition of parylene-C, which also shows that the presence of parylene-C does not affect the crystal morphology of Cu on the surface of the fabric, thus ensuring other functionalities of the sample.
The specific region of the IR spectra for parylene-C has been well identified in the literature.26,43,44 Figure 2(j) presents the FTIR spectrum of Py15/Cu/PET. All the bands shown in the spectrum match the notable band of parylene-C. The specific band occurs at 877 cm−1, and 1051 cm−1 of Cl bounded aromatic ring, which is the identified group of parylene-C. the bands at 1557 cm−1, and 1607 cm−1 present aromatic C-C bonding. The bands at 1452 cm−1 of C-H bonding and band at 827 cm−1 of two adjacent C-H bending on the benzene ring also match the typical bands of parylene-C. The persistence of the main spectrum bands and limited change of the above band shows the integrity of the parylene-C encapsulation process. This high cover ratio led to optimized enhancement for the mechanical properties of Py/Cu/PET.
Regarding the softness of the treated sample, the bending force of 2.5 cm × 5 cm sample was presented in the Table 8.
Basic characteristics of polyethylene terephthalate (PET) nonwoven fabrics MILIFE

From the average value of the bending force, the softness of the sample was reduced with increasing the encapsulated mass of parylene-C. After encapsulation, the fiber diameter and fabric thickness were increased significantly. Considering the character of parylene-C, the softness of treated sample will be influenced. However, thanks for the relatively thin structure of Cu/PET, Py15/Cu/PET still performs elegant softness even with the highest encapsulated mass of parylene-C, the Py15/Cu/PET fabric can fold and deformed easily which is still suitable for technical clothing application.
EMI shielding property change due to the machine washing cycle and hand washing cycle
Previous research has studied the EMI shielding property of Cu-coated non-woven polyester fabric. 12 Generally, the following factor influences the SE of the shielding material; (a) electrical conductivity, (b) porosity of the conductive layer and (c) conductive layer thickness. The average SE of Cu/PET in the frequency band from 30 MHz to 3 GHz was 45 dB. The parylene-C was a non-conductive material, and the electrical conductivity of Py/Cu/PET was extremely low after encapsulation. However, this process did not change the conductivity of the Cu particle on the fiber's surface; there was still one conductive “Cu layer” between PET and Parylene-C, which can still perform a good EMI shielding effect. After integrating 15 g parylene-C in the deposition process, the thickness of the sample was only increased 90 µm. In this case, the increased non-conductive layer thickness has no significant impact on the SE.
Regarding the porosity change of the conductive layer, the deposited parylene-C blocked part of the fabric porosity. However, the porosity of the conductive layer remains the same because there was no change in the Cu particle distribution of the fabric. For this reason, the SE of Cu/PET and Py/Cu/PET will not perform significantly differently, as illustrated in Figure 3(a). Especially at 3 GHz, the SE of all samples is between 42.9–46.5 dB. Py6/Cu/PET performs the relatively lowest SE in this group, and Py10/Cu/PET performs the highest SE compared with other samples. The uneven distribution of the Cu particles of Cu/PET could cause this result. The heterogeneous structure of Cu-coated non-woven PET performs unstable SE, which varies in the range of 5 dB. After the deposition of the parylene-C, the heterogeneous structure has not fundamentally changed. In this case, Py/Cu/PET samples also perform similar SE performance with Cu/PET; the SE value varied in the same range of 5 dB. The peak of the SE cure was obviously shifted after the deposition of parylene-C; this was caused by the porosity change. Regarding the frequency selective surface (FSS) theory, the transmitted electromagnetic wave was influenced by the shape of the apertures in the shielding material. 45 From Figure 2(a) and 2(c), the shape and distribution of the pores of the fabric were significantly reduced, which could influence the transmitted electromagnetic wave. According to the classification of electromagnetic SE values on textiles for general use, 46 all samples were evaluated in the “AAA” (good) category for Class I professional use.
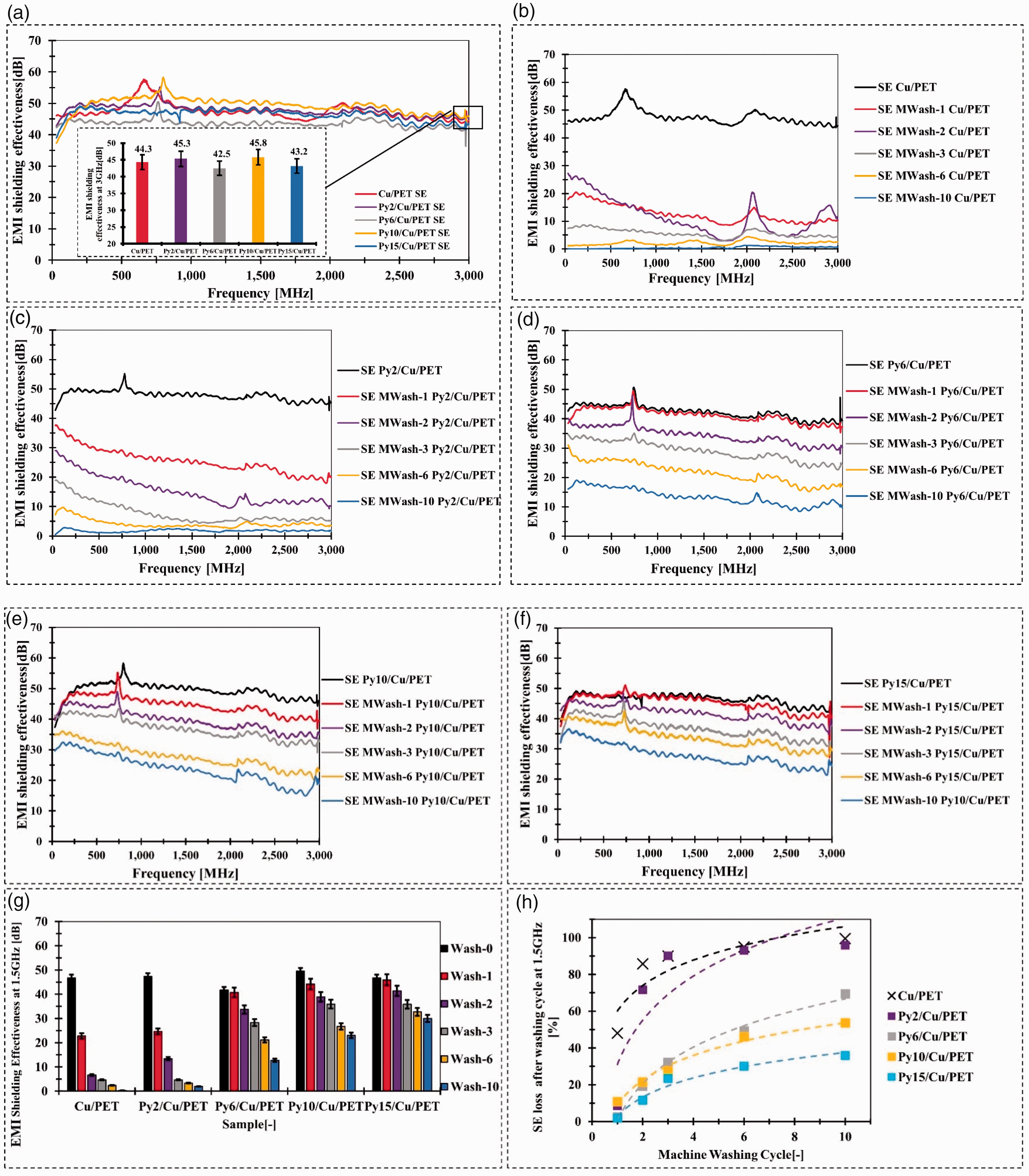
Electromagnetic interference (EMI) shielding after machine washing (MWash) cycle: (a) EMI shielding effectiveness (SE) of copper (Cu)/ polyethylene terephthalate (PET) and parylene-C (Py)/Cu/PET; (b) SE of Cu/PET with 10 machine washing cycle; (c) SE of Py2/Cu/PET with 10 machine washing cycles; (d) SE of Py6/Cu/PET with 10 machine washing cycles; (e) SE of Py10/Cu/PET with 10 machine washing cycles; (f) SE of Py15/Cu/PET with 10 machine washing cycles; (g) SE at 1.5 GHz compared with Cu/PET and Py/Cu/PET after 10 machine washing cycles and (h) SE loss at 1.5 GHz for Cu/PET and Py/Cu/PET after 10 machine washing cycles.
The SE after 10 machine washing cycles of untreated sample Cu/PET is presented in Figure 3(b). After two washing cycles, the SE at 1.5 GHz drops from 47.5 dB to 6.83 dB. There was nearly no EMI shielding property after 10 times of machine washing (SE = 0.35 dB at 1.5 GHz). As a result, the SE obtained at 1.5 GHz frequency was used for evaluation in this article because it was close to the frequency used by many working devices (e.g. cell phones, GPS, and Wi-Fi routers). 13
The parylene-C encapsulated fabric performs enhanced machine-washing ability with the increased amount implemented in the deposition process. Using 2 g parylene-C for deposition was not enough for 10 machine washing cycles (Figure 3(c)). With the increasing used amount of the parylene-C, the EMI shielding property was gradually improved from 12.76 dB to 26.6 dB via integrating increased parylene-C from 6 g to 15 g (Figure 3(d)–(f)). The best machine-washing ability regarding EMI shielding performance compared to other samples was Py15/Cu/PET. The average SE between frequency band from 30 MHz to 3 GHz remains 27.5 dB after 10 times machine washing cycle, which can be classified as Class II − general use, Grade 4 “very good”. The SE loss at 1.5 GHz after the machine-washing cycle can be observed in Figure 3(g). For Cu/PET, the SE loss at 1.5 GHz reaches 99.5%. The improvement was significant with processing 15 g parylene-C, and the SE loss at 1.5 GHz was 35.9% (Figure 3(h)).
The results of SE after hand washing cycles are presented in Figure 4. For Cu/PET after the hand washing cycle, the SE dropped significantly (Figure 4(a)). The SE dropped around 10 dB after each hand washing cycle in the first three washing round. After 10 hand washing cycles, the SE at 1.5 GHz was 1.3 dB, which lost 97.19% of EMI shielding property.
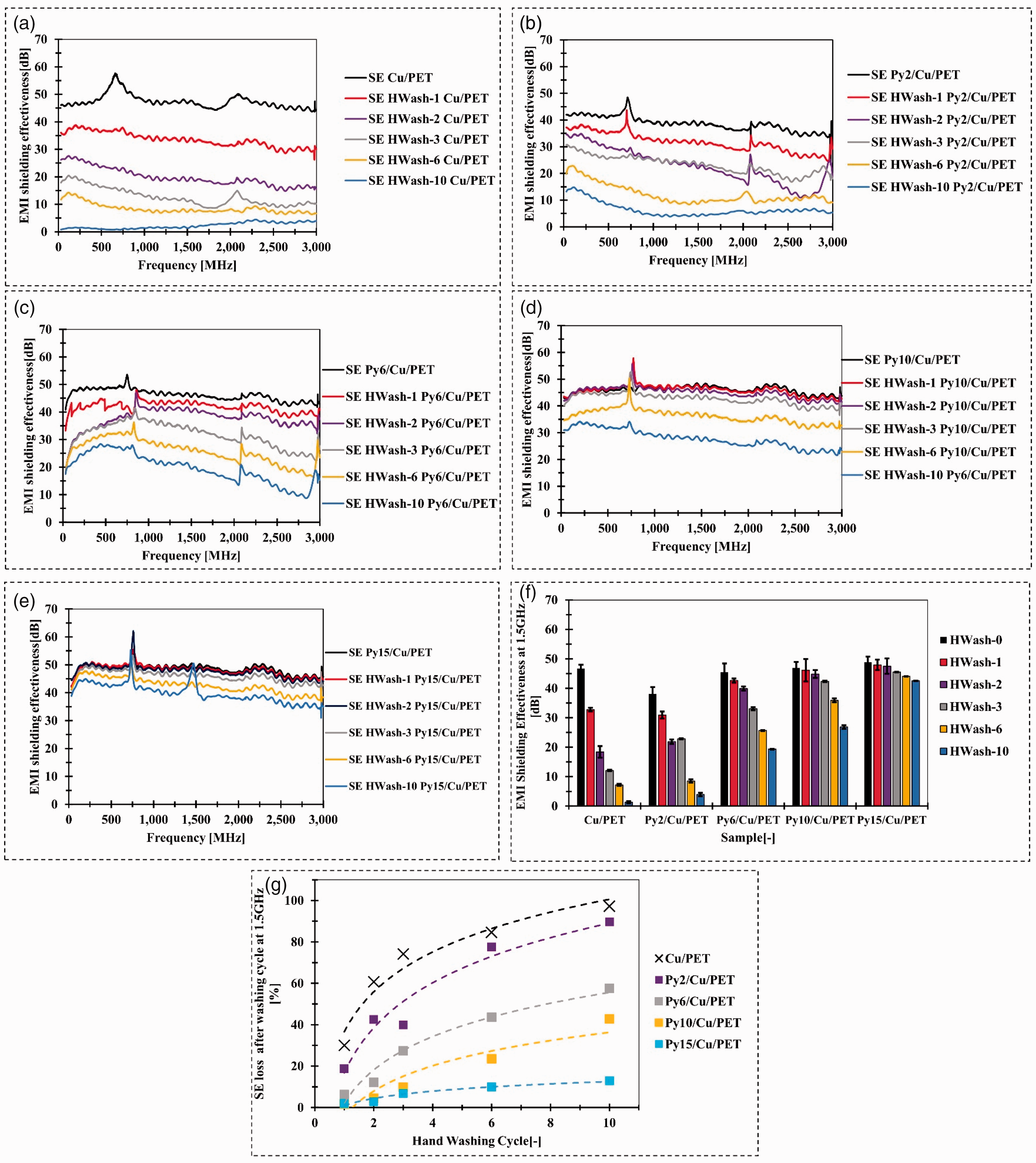
Electromagnetic interference (EMI) shielding effectiveness (SE) after hand washing (HWash) cycle: (a) SE of copper (Cu)/ polyethylene terephthalate (PET) with 10 hand washing cycles; (b) SE of parylene-C (Py)2/Cu/PET with 10 hand washing cycles; (c) SE of Py6/Cu/PET with 10 hand washing cycles; (d) SE of Py10/Cu/PET with 10 hand washing cycle; (e) SE of Py15/Cu/PET with 10 hand washing cycles; (f) SE at 1.5 GHz compared with Cu/PET and Py/Cu/PET after 10 hand washing cycles and (g) SE loss at 1.5GHz for Cu/PET and Py/Cu/PET after 10 hand washing cycles.
Similar to the machine washing cycle results, the Py/Cu/PET samples perform enhanced hand washing with the increased amount of parylene-C implemented in the deposition process. The 2 g parylene-C for deposited samples performs better washability compared to Cu/PET but still loses 89.65% EMI shielding property at 1.5 GHz after 10 hand washing cycles (Figure 4(b)). After increasing the encapsulated amount of the parylene-C, the SE loss at 1.5 GHz was gradually reduced from 57.5% to 12.9% via integrating increased parylene-C from 6 g to 15 g (Figure 4(c)–(e)). The best hand-washing ability regarding EMI shielding performance compared to other samples was Py15/Cu/PET. The average SE between the frequency band from 30 MHz to 3 GHz remains 39.93 dB after 10 times hand washing cycles, which dropped around 17.66% compared to the unwashed sample (Figure 4(e)). The SE loss level for parylene-C encapsulated sample of the hand washing cycle was less than machine washing cycle. This process was gentle compared with the machine washing cycle due to the less friction. Depending on the results of SE via a different method of washing cycle, the parylene-C encapsulated fabric was more suitable for the hand washing process than the machine washing cycle.
The machine-washed samples were used to discuss why SE dropped after the washing cycle. Since the machine-washing cycle was more intense than the hand-washing cycle, the morphology change after machine washing should be more evident than the hand-washing. The SEM image presents the Cu particle loss after machine washing of Cu/PET (Figure 5(a) and (b)), which could be the main reason that the SE of Cu/PET lost more than 99% after 10 machine washing cycles. Additionally, the fiber surface was damaged during the washing process, and several cracks on the fiber surface can be observed (Figure 5(b)). This damage can also be observed for sample MWash-10 Py2/Cu/PET and MWash-10 Py6/Cu/PET to varying degrees (Figure 5(d) and (f)). Generally, the parylene-C encapsulated fiber remains the structural integrity with the increased use amount of parylene-C to 15 g (Figure 5(c) and (e)). There was no significant damage on the fiber surface of sample MWash-10 Py15/Cu/PET that the Cu particle was covered by the parylene-C film (Figure 5(h)). This is the reason that MWash-10 Py15/Cu/PET remains a suitable EMI shielding property after the machine-washing cycle.

Scanning electron microscope (SEM) image presents the fiber surface morphology change before and after 10 machine washing cycles: (a) copper (Cu)/ polyethylene terephthalate (PET) before wash; (b) Cu/PET after 10 machine washing cycles; (c) parylene-C (Py)2/Cu/PET before wash; (d) Py2/Cu/PET after 10 machine washing cycles; (e) Py6/Cu/PET before wash; (f) Py6/Cu/PET after 10 machine washing cycles; (g) Py15/Cu/PET before wash and (h) Py15/Cu/PET after 10 machine washing cycles.
Air permeability
Figure 6(a) presents the air permeability of Cu/PET and Py/Cu/PET before and after the washing cycle. As referred to in the geometrical structure of Py/Cu/PET, due to the CVD method, the parylene-C film covered the fiber surface without blocking all the fabric apertures, which enabled the fabric to maintain good air permeability. The parylene-C film increased the fiber diameter, leading to the pore size decrease (Figure 2(a) and (c)). Such changes affect the air permeability of the fabric to some extent. With the increase of parylene-C to 15 g, the air permeability was reduced from 3930 mm/s (Cu/PET) to 1043.6 mm/s (Py15/Cu/PET). The reduction of the fabric porosity causes this change. The increasing deposited parylene-C made the fiber diameter thicker, which caused the synchronized drop in fabric porosity and air permeability (Figure 6(b)).
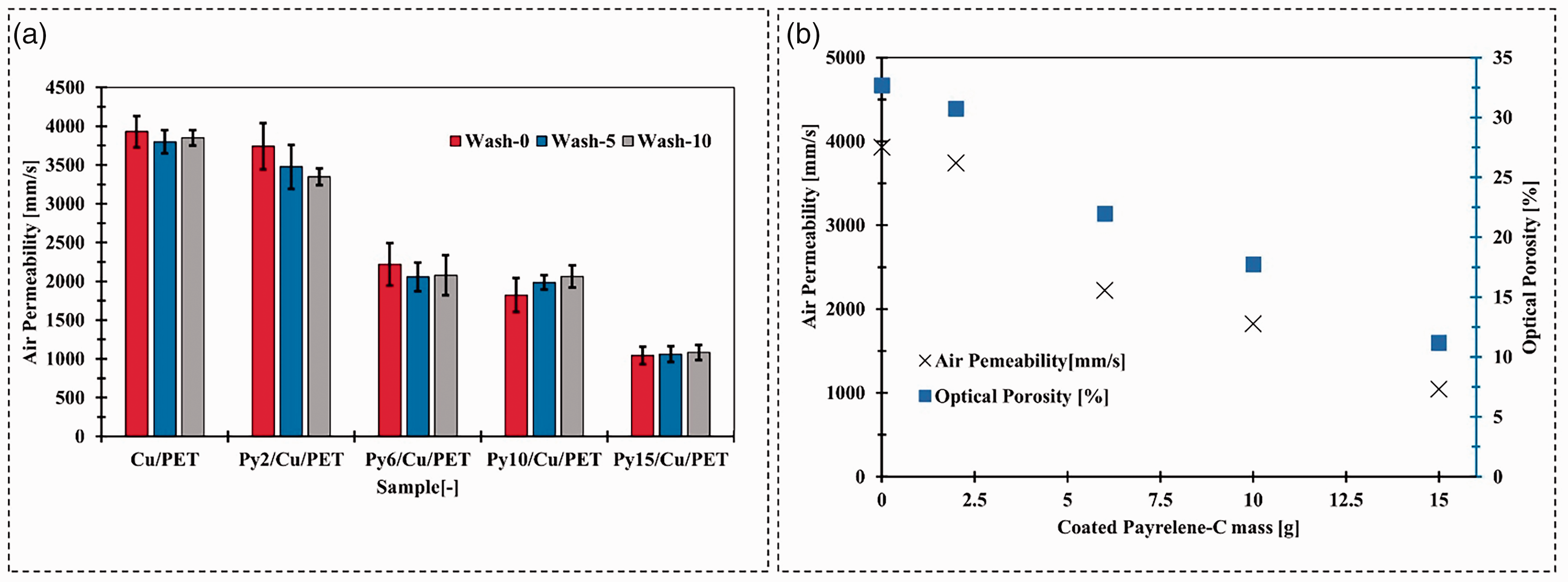
(a) Comparing air permeability for copper (Cu)/ polyethylene terephthalate (PET) and parylene-C (Py)/Cu/PET before and after the washing cycle; (b) the relationship between air permeability and optical porosity with different coated parylene-C.
No matter whether it was for Cu/PET or Py/Cu/PET, the air permeability change caused by the washing process was not significant. During the washing process, the fiber surface was damaged by friction with other textile fabric, which also caused the loss of Cu particles. But the fiber size and fabric structure may not have an extraordinary change, so for air permeability, there was no mathematically significant difference for the sample that experienced the machine-washing cycle compared to the original sample.
Concerning the air permeability of other developed machine-washable and wearable electromagnetic shielding textiles, Py15/Cu/PET was in the leading position (e.g. the air permeability of Cao et al. developed conductive carbon nanotubes (CNT) and screen-printed fabric is 88.2 m/s, 47 Du et al. developed the Cu-coated fabric via TA/APTE technology, the air permeability of this sample above 30 m/s, 48 Yang et al. developed the paper–based triboelectric nanogenerator (P-TENG) with good flexibility and conductivity, the air permeability of treated sample is 35.7 m/s). 49 Typically, the protection layer blocks nearly all the fabric's pores, significantly reducing air permeability. Regarding Py15/Cu/PET, the measured value was 1043.6 mm/s, which was higher than traditional clothing used fabric(e.g. air-permeability of 295.2 g/m2 warp knitted fleece PET fabric was 669.22 mm/s, for 201 g/m2 warp knitted underwear fabric the air permeability value was 900 mm/s). 50 In this case, the Py15/Cu/PET could be used as the functional layer integrated into the sandwich structure for technical clothing applications without a massive influence on air permeability.
Acid and alkali resistance property
After encapsulation, the Cu-coated PET fabrics, which was sensitive to different extreme pH environments before the treatment, showed good acid and alkali resistance. The results are presented in Figure 7.
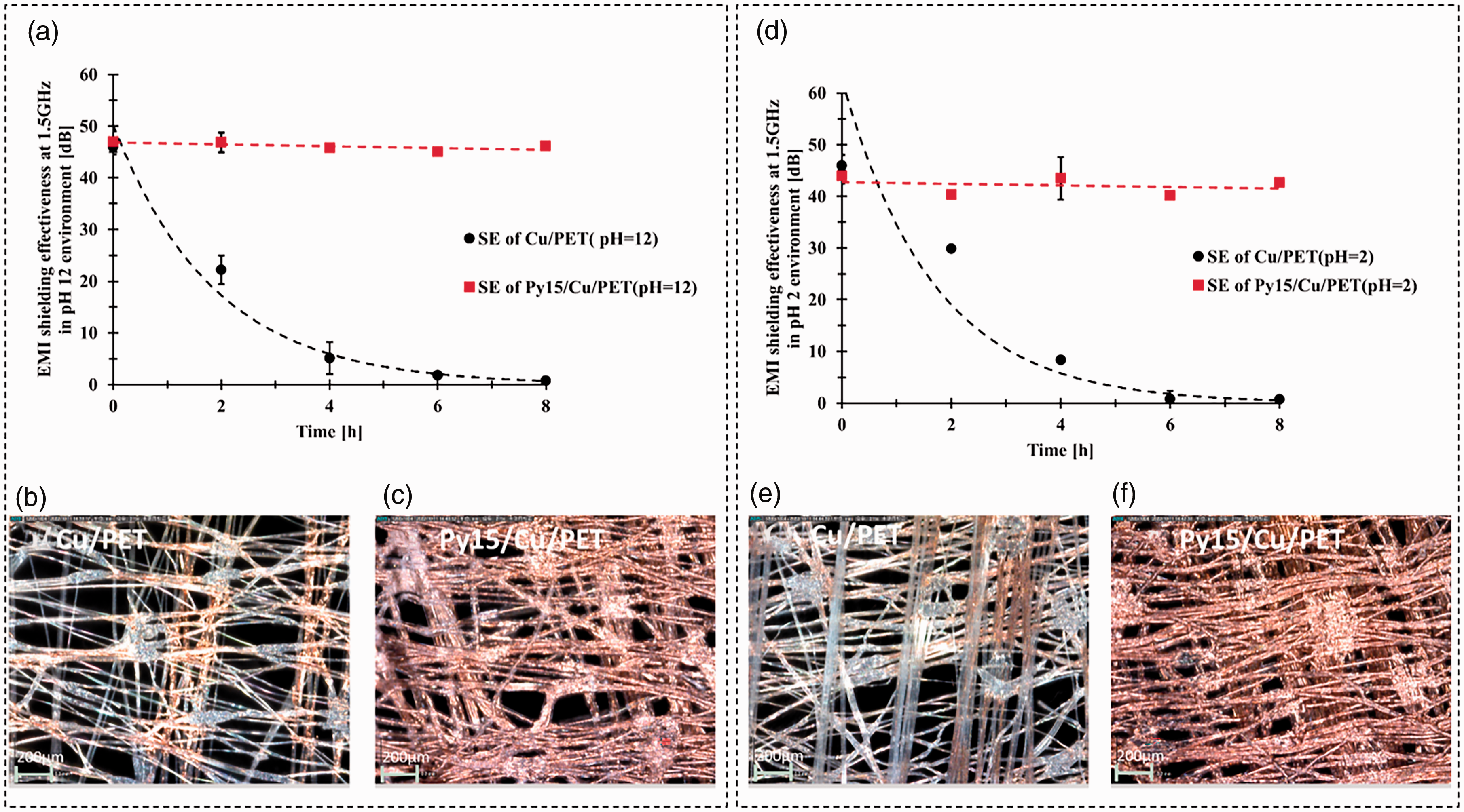
(a) Shielding effectiveness (SE) at 1.5 GHz of copper (Cu)/ polyethylene terephthalate (PET) and parylene-C (Py)15/Cu/PET in pH = 12 environment; (b) surface morphology change of Cu/PET after 8 h immersion in pH = 12 NaOH solution; (c) surface morphology change of Py15/Cu/PET after 8 h immersion in pH = 12 NaOH solution; (d) SE at 1.5 GHz of Cu/PET and Py15/Cu/PET in pH = 2 environment; (e) surface morphology change of Cu/PET after 8 h immersion in pH = 2 H2SO4 solution and (f) surface morphology change of Py15/Cu/PET after 8 h immersion in pH = 2 H2SO4 solution.
The SE of Cu/PET decreased significantly after immersing the samples into NaOH and H2SO4 solution. After 8 h, the average SE at 1.5 GHz dropped to 0.76 dB (pH = 12) and 0.75 dB (pH = 2), which performs nearly no EMI shielding property. On the contrary, the Py15/Cu/PET shows excellent acid and alkali resistance properties. The SE at 1.5 GHz was always above 40 dB for both situations; compared with the original sample, the SE at 1.5 GHz decrease was only 1.9% (pH = 12) and 3% (pH = 2) (Figure 7(a) and (d)).
There are ester bonds in the macromolecules of polyester, which can be hydrolyzed. Alkali has a catalytic effect on the hydrolysis of ester bonds. The degree of hydrolysis varies with the alkali type, concentration, temperature, and time. 51 In this case, the untreated sample immersed in NaOH will cause part of the polyester to hydrolyze. The fiber finesse was reduced after 8 h immersed in the NaOH solution compared with the encapsulated fabric (Figure 7(b) and (c)).
The sulfuric acid can oxidize and react with Cu particles on the fiber surface, reducing its electromagnetic shielding performance (Figure 7(e) and (f)). In the moisture environment, the coated Cu particles could react with oxygen which causes the loss of EMI shielding property. Additionally, the untreated sample was immersed under the pH = 2 and pH = 12 circumstances; the Cu particles will react with SO42− and OH− to form different valence Cu ions. Based on the above reasons, it may be possible to explain why untreated samples rapidly lose their electromagnetic shielding properties under acid and alkali conditions. According to the previously referenced research, 26 in the molecular structure of parylene C, the benzene ring and the methylene group are very stable. The molecule contains no hydrophilic or lipophilic groups except the -Cl functional group. In this case, the parylene-C has superior moisture and chemical resistance properties, which could prevent oxidation and corrosion of the Cu-coated fabric from acid and alkali. This structure makes parylene-C encapsulated fabrics more stable under high acid and alkali corrosion.
Abrasion resistance
The parylene deposition process gave the new material high abrasion resistance property, which was very important for EMI shielding technical clothing and other wearable applications. Figure 8(a) shows up to 0.07% mass loss for Cu/PET after abrasion of 1000 cycles. The surface of Cu/PET has been wholly worn in the abraded parts, as shown in Figure 8(b). It was observed clearly that a large area of fiber had been stripped and broken. The original thickness of the sample Cu/PET was relatively low, and the porosity was higher than other textile samples in this case. Unsurprisingly, the abrasion durability was poor for sample Cu/PET.
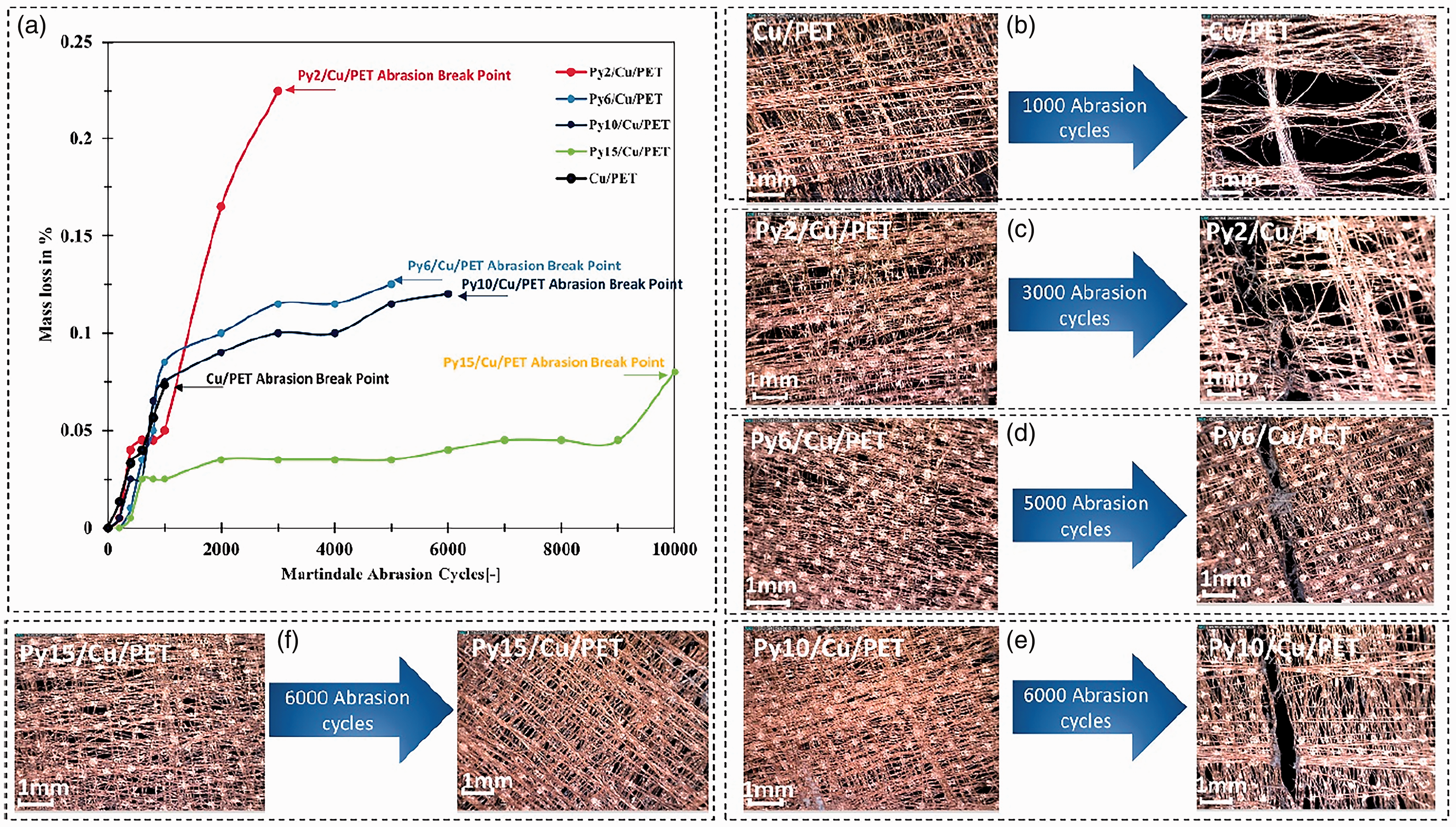
Abrasion test result: (a) mass loss after specific abrasion cycles; (b) structure change of copper (Cu)/ polyethylene terephthalate (PET) after 1000 abrasion cycles; (c) structure change of parylene-C (Py)2/Cu/PET after 3000 abrasion cycles; (d) structure change of Py6/Cu/PET after 5000 abrasion cycles; (e) structure change of Py10/Cu/PET after 6000 abrasion cycles and (f) structure change of Py15/Cu/PET after 6000 abrasion cycles.
Py2/Cu/PET was worn out due to the weak protection of too little parylene-C film to cover the Cu-coated fabric (Figure 8(c)), and after 3000 abrasion cycles the material was worn.
The abrasion durability of Py6/Cu/PET was improved. The breakpoint enables 5000 abrasion cycles (Figure 8(d)). The same trend for Py10/Cu/PET is that the break happened after 6000 abrasion cycles (Figure 8(e)). The Py15/Cu/PET exhibits supreme abrasive endurability in contrast with unencapsulated and less encapsulated samples. There was only a tiny amount of mass loss ratio (0.045%), and the surface almost kept intact after 6000 cycles (Figure 8(f)). After 10,000 abrasion cycles, there was a small crack that happened on the edge of the sample, which was caused by the edge of sharp sample holder but not the abrasion parts. In this case, more abrasion cycles can be realized if they take more protection between the edge of the sample and the sample holder. However, from another aspect, the parylene-C encapsulated sample shows better abrasion durability against the textile friction movement compared to sharp metal scratch damage.
The excellent abrasion resistance was mainly due to the stable bond between the parylene-C film and the fibers. Parylene-C is one of the polymers which has good mechanical stability and can be applied on the semi-conductor surface; due to the protection of parylene-C film, the friction interface was changed from Cu-particle coated PET fiber with textile to parylene-C film with textile. The change of the friction interface reduces the abrasion damage of the textile.
Conclusions
In this article, to solve the problem of poor washing ability of Cu-coated EMI shielding fabrics and maintain good air-permeability, parylene-C encapsulation via CVD technology was applied for processing the greige sample. The morphology analysis result identified the encapsulated parylene-C film via SEM image and EDS mapping. The operating mass of parylene-C performs the positive linear correlation with the thickness of the sample. The parylene-C film keeps part of the Cu particle encapsulated on the fiber surface during the washing process. After integrating 15 g parylene-C, the average SE between the frequency band from 30 MHz to 3 GHz remained at 27.5 dB and 39.93 dB after 10 times machine and hand washing cycles, respectively. This result was significantly improved compared to the untreated sample, which has more than 90% loss of EMI shielding property after 10 washing cycles. The encapsulated parylene-C film did not block all the pores of the fabrics, which enabled the sample to maintain good air permeability of 1043.6 mm/s after 15 g parylene-C processing. The superior chemical resistance has been approved, and there is less than a 3% loss of EMI shielding property after immersing the samples into pH = 2 and pH = 12 for 8 h. Due to the excellent anti-abrasion property of parylene-C, the encapsulated sample shows improved abrasion durability so that the fabric structure was intact until the 10,000 abrasion cycle with 15 g parylene-C processed in the deposition system.
Unlike the traditional coating anti-machine-washing treatment method that blocks all the fabric pores, the parylene CVD method not only improves the washing performance of the fabric but also ensures a certain degree of air permeability. Additionally, the fabric's chemical resistance properties and anti-abrasion properties are also improved accordingly. Based on the above excellent properties, especially for people who need to protect themselves from excessive electromagnetic wave radiation, such as patients with cardiac pacemaker implantation, the parylene-C encapsulated Cu-coated fabrics as electromagnetic shielding materials have great potential for further development and utilization.
Footnotes
Acknowledgments
The author(s) are grateful for the support from the student research project 2022 No. SGS-2022-6012 supported by the Czech Ministry of Education and Faculty of Textile, Technical University of Liberec. They also acknowledge CzechNanoLab Research Infrastructure supported by MEYS CR (LM2018110).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Youth and Sports of the Czech Republic and the European Union – European Structural and Investment Funds in the Frames of Operational Program Research, Development and Education – project Hybrid Materials for Hierarchical Structures (HyHi, Reg. No. CZ.02.1.01/0.0/0.0/16_019/0000843).
