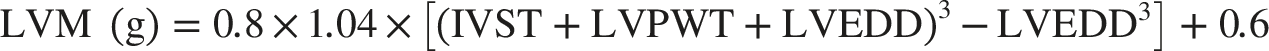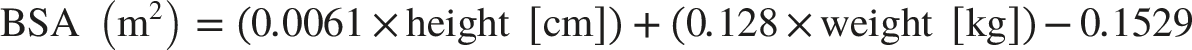Abstract
Objective
This study aimed to evaluate the associations of the Chinese Visceral Adiposity Index (CVAI), age, and clinic systolic blood pressure (SBP) with left ventricular hypertrophy (LVH) in postmenopausal women with primary hypertension.
Methods
We conducted a retrospective case-control study including 501 postmenopausal women hospitalized with primary hypertension between January and December 2023 at the Department of Hypertension at the Fifth Affiliated Hospital of Xinjiang Medical University. Participants were divided into an LVH group (cases, n=86) and a non-LVH group (controls, n=415) based on the left ventricular mass index (LVMI). Clinical data, biochemical parameters, echocardiographic results, and the CVAI were collected. Logistic regression and receiver operating characteristic (ROC) curve analyses were performed to determine the association between LVH and the outcome.
Results
Compared to the non-LVH group, the LVH group exhibited significantly higher age, clinical SBP, proportion of coronary artery disease, blood urea nitrogen (BUN) levels, CVAI, and cystatin C levels (all P<0.05). Left ventricular geometric patterns also differed significantly between the two groups (P < 0.001). Multivariate logistic regression analysis identified CVAI (odds ratio [OR] =1.025, 95% confidence interval [CI] = 1.013–1.038), age (OR=1.045, 95% CI = 1.013–1.079), and clinical SBP (OR=1.020, 95% CI = 1.007–1.033) as independent risk factors for LVH. Multinomial logistic regression showed that CVAI was associated with concentric (OR = 1.026, 95% CI = 1.008–1.044) and eccentric hypertrophy (OR = 1.026, 95% CI = 1.011–1.041). The receiver operating characteristic (ROC) curve showed an area under the curve (AUC) of 0.702 for CVAI alone, with a sensitivity of 83.7% and a specificity of 48%. The discriminative performance of CVAI was significantly better than that of the ventricular artery index (VAI; 0.551) and left anterior portal (LAP; 0.575). The combined discrimination using all three factors (CVAI, age, and clinical SBP) yielded an AUC of 0.751.
Conclusion
CVAI, age, and clinical SBP are independent risk factors for LVH in postmenopausal women with primary hypertension.
Keywords
1. Introduction
Hypertension is a significant health problem worldwide, affecting over 1.3 billion people and being a leading cause of death from heart disease. 1 Over 60% of adults over 65 have this condition, which is particularly problematic for older people. 2 When the heart experiences prolonged high pressure, it can lead to left ventricular hypertrophy (LVH), which is a serious issue. 3 This change in the structure of heart tissue increases the risk of heart failure, arrhythmias, and early death on its own. That is why it is so important to detect it early. 4
Postmenopausal women are at an increased risk of hypertension-induced target organ damage due to abrupt hormonal changes following ovarian cessation. 5 As estrogen levels decline, endothelial dysfunction worsens due to reduced nitric oxide availability. Simultaneously, vascular stiffness and visceral fat storage increase. 6 These physiological changes are interconnected and create a high-risk profile marked by systolic-dominant hypertension and metabolic imbalance. This dramatically increases the risk of LVH compared with men of the same age. 6 Current guidelines explicitly acknowledge this increased risk profile; however, echocardiographic screening remains inconsistently performed in resource-limited settings. 7
The Chinese Visceral Adiposity Index (CVAI) is a culturally suitable method for measuring visceral fat that avoids the issues associated with traditional anthropometric measurements. 8 CVAI offers a better way to assess the unhealthy metabolic activity of visceral adipose tissue than other indices, as it accounts for age, waist circumference, body mass index, triglycerides, and high-density lipoprotein cholesterol. Recent studies that confirmed this in several countries have shown that it is more effective at associating new cases of high blood pressure and heart problems across various Asian populations.9,10 This performance advantage demonstrates that CVAI can assess the pro-inflammatory adipokine environment, which directly influences pathways that alter heart muscle structure. 11
Despite these advances, there is limited evidence on the practical clinical application of CVAI. In particular, the extent to which CVAI is associated with structural cardiac changes in postmenopausal women with hypertension remains unclear. This population has a distinct pathophysiological profile, characterized by visceral fat–related insulin resistance and vascular ageing associated with estrogen deficiency. 12 Additionally, the extent to which predictive power can be gained by combining CVAI with known hemodynamic and chronological risk factors has not been measured. This is a significant issue with current risk stratification models. 13 Due to financial and infrastructure limitations, advanced cardiac imaging remains unavailable in real-world settings.
We conducted a retrospective case-control study to address these evidence gaps. This study had two main objectives. First, we aimed to determine whether CVAI is associated with echocardiographically confirmed LVH in postmenopausal women with essential hypertension, even after adjusting for all relevant confounding factors. Second, we sought to evaluate whether a model combining CVAI, chronological age, and clinic-measured systolic blood pressure is more effective at predicting outcomes than each parameter individually. We believed that visceral adiposity dysfunction would intensify the effects of hemodynamic load and biological aging on heart muscle remodelling in this already at-risk group.
2. Methods
2.1. Study design
This was a retrospective, single-center, unmatched case-control study. We identified postmenopausal women with primary hypertension from inpatient medical records and classified them into two groups: those with echocardiographically confirmed left ventricular hypertrophy (LVH; cases) and those without LVH (controls), all of whom were drawn from the same source population. No formal matching criteria were applied, and standard logistic regression was used to identify factors independently associated with LVH. The reporting of this observational study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 14
2.2. Ethical approval
This study was conducted in accordance with the ethical principles of the Declaration of Helsinki (1975; revised in 2024). Ethical approval was granted by the Ethics Committee of the Fifth Affiliated Hospital of Xinjiang Medical University (Approval No. XYDWFYLSK-2025-25; approved on 2 January 2025; see Supplemental File 1). Written informed consent was obtained from all patients.
2.3. Setting
This study was conducted at the Hypertension Department of the Fifth Affiliated Hospital of Xinjiang Medical University, a tertiary referral center in Urumqi, China. The electronic medical records of patients admitted between 1 January and 31 December 2023 were thoroughly reviewed. All clinical, laboratory, and echocardiographic data were obtained from routine inpatient assessments without additional follow-up visits. All patient data were fully de-identified prior to analysis.
2.4. Participants
Eligible participants were postmenopausal women aged 18 or older with a confirmed diagnosis of primary hypertension, defined according to the Chinese Guidelines for Prevention and Treatment of Hypertension (2018 Revision) as systolic blood pressure (SBP) of 140 mmHg or higher and/or diastolic blood pressure (DBP) of 90 mmHg or higher measured on three separate visits. 7 Menopausal status was classified according to the Stages of Reproductive Ageing Workshop (STRAW+10) criteria, defined as ≥12 months since the last menstrual period. 15 Patients diagnosed with LVH based on left ventricular mass index (LVMI) thresholds (LVMI ≥ 95 g/m2 for women) constituted the case group, while those without LVH served as controls. 13
A total of 2,321 men and women with essential hypertension were recruited from the institutional registry (Project 2321) at the Department of Hypertension, The Fifth Affiliated Hospital of Xinjiang Medical University, Urumqi, Xinjiang, China. As this study used retrospective data, it was not registered in a clinical trial registry. We applied predefined exclusion criteria, including menopause < 12 months (no menstrual period for 12 months), white-coat or secondary hypertension (excluding secondary causes, such as pheochromocytoma), recent estrogen or glucocorticoid therapy (within the last 6 months), pregnancy, malignancy or significant cardiac disease (severe coronary artery disease, heart failure, etc.), thyroid/hepatic/renal dysfunction (e.g., eGFR < 30 mL/min/1.73m2), uric acid-lowering drugs, smoking or alcohol abuse (defined as smoking in the last 6 months or excessive alcohol consumption), recent surgery (within the past 3 months), and acute cardiovascular events (myocardial infarction, stroke, or acute heart failure in the last 6 months). These criteria led to the exclusion of 188 women, leaving 501 postmenopausal women with primary hypertension (mean age 62.0 ± 9.1 years) for analysis. This was an unmatched case-control study; therefore, no formal matching criteria (such as age or other variables) were used to select controls. Participants were stratified by left ventricular mass index (LVMI ≥95 g/m2 for women and LVMI≥ 115 g/m2 for womenLVMI ≥115 g/m2 for men,≥ 95 g/m2 for men) into LVH (n = 86) and non-LVH (n = 415) groups. The inclusion-exclusion process is shown in Figure 1. Participant flow diagram for the case–control study of left ventricular hypertrophy in postmenopausal women.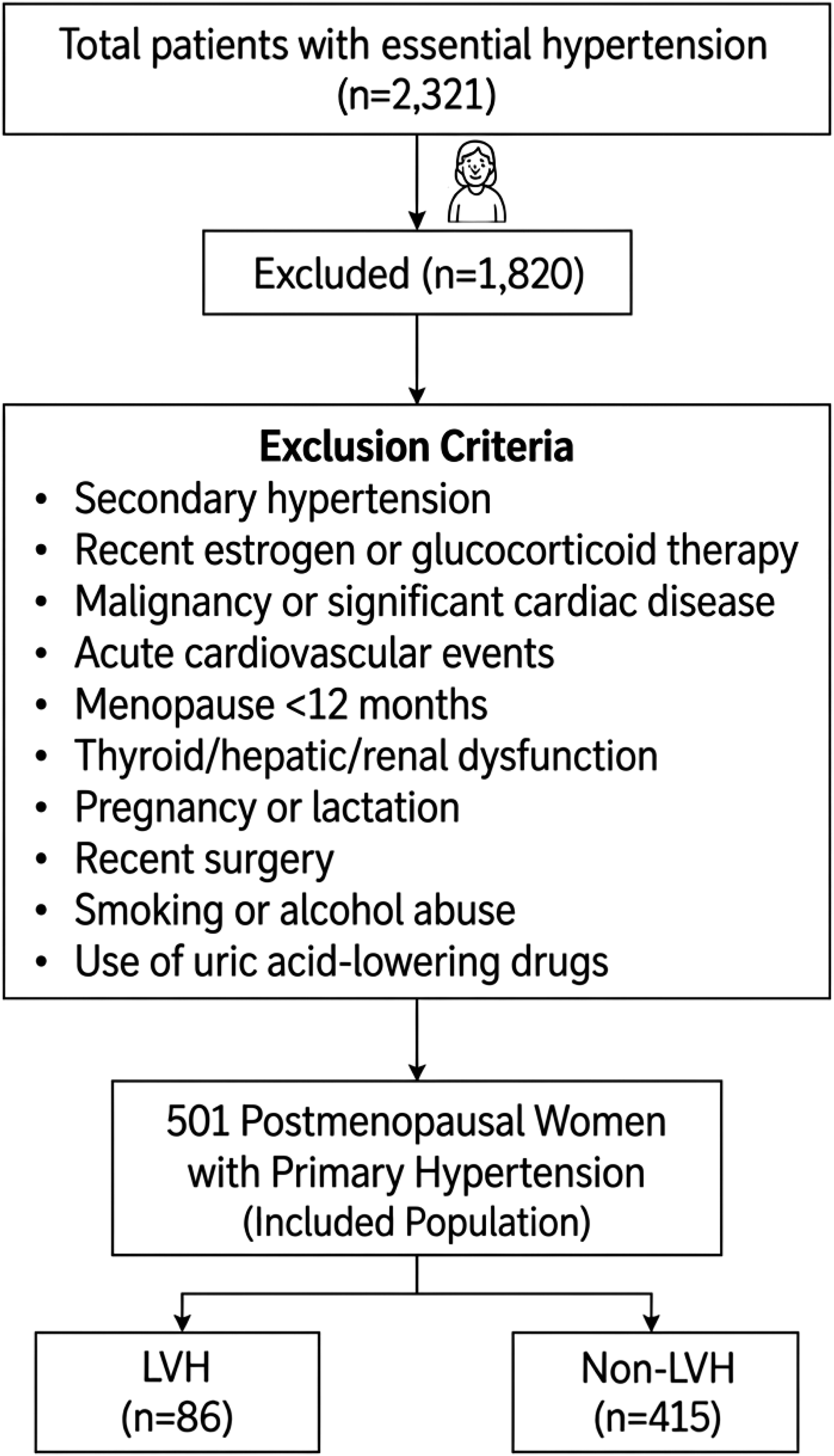
2.5. Variables
The primary outcome was LVH, defined as LVMI ≥95 g/m2 in women according to the American Society of Echocardiography guidelines.
13
The primary exposure of interest was the Chinese visceral adiposity index (CVAI), calculated using the following validated formula:
Other association variables included age, clinic SBP, and coronary artery disease (documented clinically or through angiography). Potential confounders encompassed blood urea nitrogen (BUN), fasting glucose, total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), and cystatin C. Age and SBP were evaluated as potential effect modifiers because of established interactions with LVH and cardiovascular risk factors.
2.6. Data sources and measurement
All clinical and demographic data, including age, height, weight, waist circumference, and history of coronary artery disease, were extracted from the electronic medical records. Clinic blood pressure was consistently measured by a single trained hypertension nurse using an Omron electronic sphygmomanometer (HEM-7117). After at least 5 minutes of seated rest, three consecutive measurements taken at 5-minute intervals (with the arm at heart level) were averaged for analysis. 7 Blood pressure was measured on the upper arm after the participant had been seated and rested for 5 min. At the initial visit, blood pressure was measured on both arms, and the higher reading was used for subsequent measurements. The arm was positioned at heart level during measurements. Two consecutive measurements were taken 2 min apart, and the average of the two readings was used. If the difference between the two readings for systolic or diastolic pressure exceeded 10 mmHg (1 mmHg = 0.133 kPa), a third measurement was taken, and the average of the three readings was used.
Body surface area (BSA) was calculated as:
Finally, LVMI was computed as follows:
2.7. Bias mitigation
Selection bias was minimized by including all consecutive eligible inpatients admitted during the study period (January to December 2023). Measurement bias was reduced by having all blood pressure measurements performed by the same trained nurse and by blinding the echocardiographers to the CVAI values. Data abstraction was independently conducted by two researchers, and discrepancies were resolved by consensus.
2.8. Study size
No formal a priori sample size calculation was conducted. Instead, all eligible postmenopausal women with hypertension who were admitted during the specified 12-month period were included. A post-hoc power analysis showed approximately 82% statistical power to detect an odds ratio of 1.025 per CVAI unit at α = 0.05, assuming an observed LVH prevalence of 17%.
2.9. Quantitative variables
Continuous associated factors (CVAI, age, SBP, BUN, and laboratory measures) were analyzed as continuous variables to maintain statistical power. CVAI was also divided into two groups based on the case group distribution for descriptive and ROC analyses. Age and SBP were split at clinically relevant thresholds to enable subgroup analysis.
2.10. Statistical methods
Normally distributed continuous data are presented as mean ± standard deviation (SD) and compared using independent-samples t-tests, whereas skewed variables are reported as median (interquartile range) and analyzed using the Mann–Whitney U test. Categorical variables were compared using the χ2 test. Logistic regression was initially performed with univariate analyses; factors with P<0.05 were included in a multivariate logistic regression model to estimate adjusted odds ratios (ORs) and 95% confidence intervals (CIs). Multiplicative interaction terms (CVAI × age, CVAI × SBP) were tested; however, no significant interactions were observed (P>0.10). Multinomial logistic regression was used to examine the association between CVAI and left ventricular geometric patterns. Missing data were minimal (<2%), mostly for high-density lipoprotein cholesterol (HDL-C; n=9 missing), and were handled using a complete-case analysis without imputation. Sensitivity analysis excluding extreme CVAI values (top and bottom 5%) confirmed the robustness of the results (adjusted OR remained at 1.025 [95% CI: 1.012–1.037]). Receiver operating characteristic (ROC) curves were plotted to evaluate the diagnostic value of CVAI, age, clinic systolic blood pressure, and their combination, VAI, and LAP for LVH, and the area under the curve (AUC) was compared pairwise using the DeLong test. All analyses were performed using SPSS version 29.0, with a two-tailed P<0.05 considered statistically significant. Internal validation of the optimal CVAI cutoff was performed using bootstrap resampling (1,000 iterations) to estimate the 95% confidence interval (CI) for the cutoff value.
3. Results
3.1. Participant characteristics
From January 2023 to December 2023, we screened 2,321 consecutive patients with essential hypertension. Among them, 689 were postmenopausal women who met the initial inclusion criteria. After applying the exclusion criteria (detailed in Figure 1), 188 women were excluded, leaving 501 postmenopausal women with primary hypertension (mean age 62.0 ± 9.1 years) for analysis. Based on echocardiographically measured left ventricular mass index (LVMI ≥95 g/m2 for women and ≥115 g/m2 for men), 86 participants (17.2%) were classified into the LVH group and 415 (82.8%) into the non-LVH group, yielding an approximate 1:5 case-to-control ratio.
Comparison of general clinical characteristics between NLVH and LVH groups.

Abbreviations: NLVH, non-left ventricular hypertrophy; LVH, left ventricular hypertrophy; SBP, systolic blood pressure; DBP, diastolic blood pressure; BMI, body mass index; BUN, blood urea nitrogen; SCr, serum creatinine; UA, uric acid; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; CVAI, Chinese Visceral Adiposity Index.
No significant differences were observed between groups for clinic diastolic blood pressure (DBP), body mass index (BMI), serum creatinine (SCr), uric acid (UA), fasting glucose, total cholesterol (TC), triglycerides (TG), HDL-C, low-density lipoprotein cholesterol (LDL-C), or serum potassium and sodium (all P > 0.05). These findings suggest that LVH in this cohort is associated with older age, SBP, CVAI, and renal biomarkers, consistent with established cardiovascular risk profiles.
3.2. Correlation between CVAI and clinical variables
Spearman correlation coefficients between CVAI and clinical variables.

aLVMI, left ventricular mass index.
In contrast, CVAI demonstrated significant negative correlations with clinic diastolic blood pressure (DBP; r = –0.201, P < 0.001), high-density lipoprotein cholesterol (HDL-C; r = –0.313, P < 0.001), and serum calcium (Ca; r = –0.112, P = 0.012). Notably, CVAI did not show significant correlations with clinic systolic blood pressure (SBP), serum creatinine (SCr), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), or serum sodium (Na) (all P > 0.05). CVAI was positively correlated with age, body mass index (BMI), blood urea nitrogen (BUN), uric acid, fasting glucose, triglycerides, cystatin C, LVMI, and potassium, suggesting its role in reflecting adiposity-related metabolic dysregulation and subclinical cardiovascular remodelling in postmenopausal hypertensive women. However, it showed no significant correlation with SBP, SCr, TC, LDL-C, or sodium, suggesting that its potential as a surrogate marker may be more closely linked to metabolic and cardiovascular changes than to traditional risk factors.
3.3. Univariate analysis of LVH risk factors
Univariate logistic regression analysis of factors associated with LVH.

*P < 0.05 indicates statistical significance. Age, clinic SBP, CAD, BUN, and CVAI were significant variables.
The presence of coronary artery disease also significantly increased the odds of LVH (OR = 2.251; 95% CI: 1.326–3.821; P = 0.003). Elevated blood urea nitrogen (BUN) emerged as another critical factor (OR = 1.228 per mmol/L; 95% CI: 1.054–1.430; P = 0.008). Most notably, the Chinese Visceral Adiposity Index (CVAI) showed a strong association with LVH (OR = 1.034 per unit; 95% CI: 1.023–1.046; P < 0.001), emphasising its potential as a marker of cardiovascular remodelling in this population.
In contrast, no significant links were found between clinic diastolic blood pressure (DBP), body mass index (BMI), serum creatinine (SCr), uric acid (UA), fasting glucose, total cholesterol (TC), triglycerides, HDL-C, LDL-C, cystatin C, or electrolyte levels (serum potassium, sodium, and calcium).
3.4. Multivariate logistic regression analysis
Collinearity diagnostics.

Multivariate logistic regression analysis of factors associated with LVH.

Multinomial logistic regression showed that in Model 2, CVAI was associated with concentric hypertrophy (odds ratio [OR] = 1.026; 95% confidence interval [CI] = 1.008–1.044; P = 0.004) and eccentric hypertrophy (OR = 1.026; 95% CI = 1.011–1.041; P < 0.001). In contrast, coronary artery disease and BUN, although significant in univariate analyses, did not remain significant after adjustment for other variables (P = 0.294 and P = 0.101, respectively), suggesting that their links to LVH may be confounded by age, blood pressure, or visceral adiposity (Table 5).
Multinomial logistic regression analysis of the association between CVAI and left ventricular geometric patterns.

Model 1: crude model (unadjusted for confounders).
Model 2: adjusted for age, systolic blood pressure, coronary artery disease, and BUN.
3.5. Interaction and sensitivity analyses
To examine potential effect modification, interaction terms between the CVAI and two biologically plausible modifiers, age and clinic SBP, were created and included in a multivariate logistic regression model. Neither the CVAI × age interaction (P = 0.325) nor the CVAI × SBP interaction (P = 0.412) was statistically significant, indicating that the association between visceral adiposity and LVH does not vary significantly across age groups or systolic pressure levels. These results suggest a consistent and independent contribution of CVAI to LVH risk, regardless of age or hemodynamic factors.
As part of the sensitivity analyses, the adjusted regression model was re-estimated after excluding participants in the extreme percentiles of the CVAI distribution (i.e.,the top and bottom 5%). This was done to evaluate the stability of the association and to minimize the impact of potential outliers. The strength and direction of the association remained unchanged, with CVAI remaining statistically significant (adjusted OR = 1.024; 95% CI: 1.011–1.037; P < 0.001), thereby confirming the robustness of the primary findings. Overall, these analyses support the independent associative role of CVAI and strengthen the study’s internal validity.
3.6. Association analysis of CVAI, age, and clinic SBP with LVH
ROC curve analysis for discriminating left ventricular hypertrophy.

Note. aP < 0.05 vs. CVAI; bP < 0.05 vs. LAP; cP < 0.05 vs. VAI; dP < 0.05 vs. age; and eP < 0.05 vs. clinic systolic blood pressure.
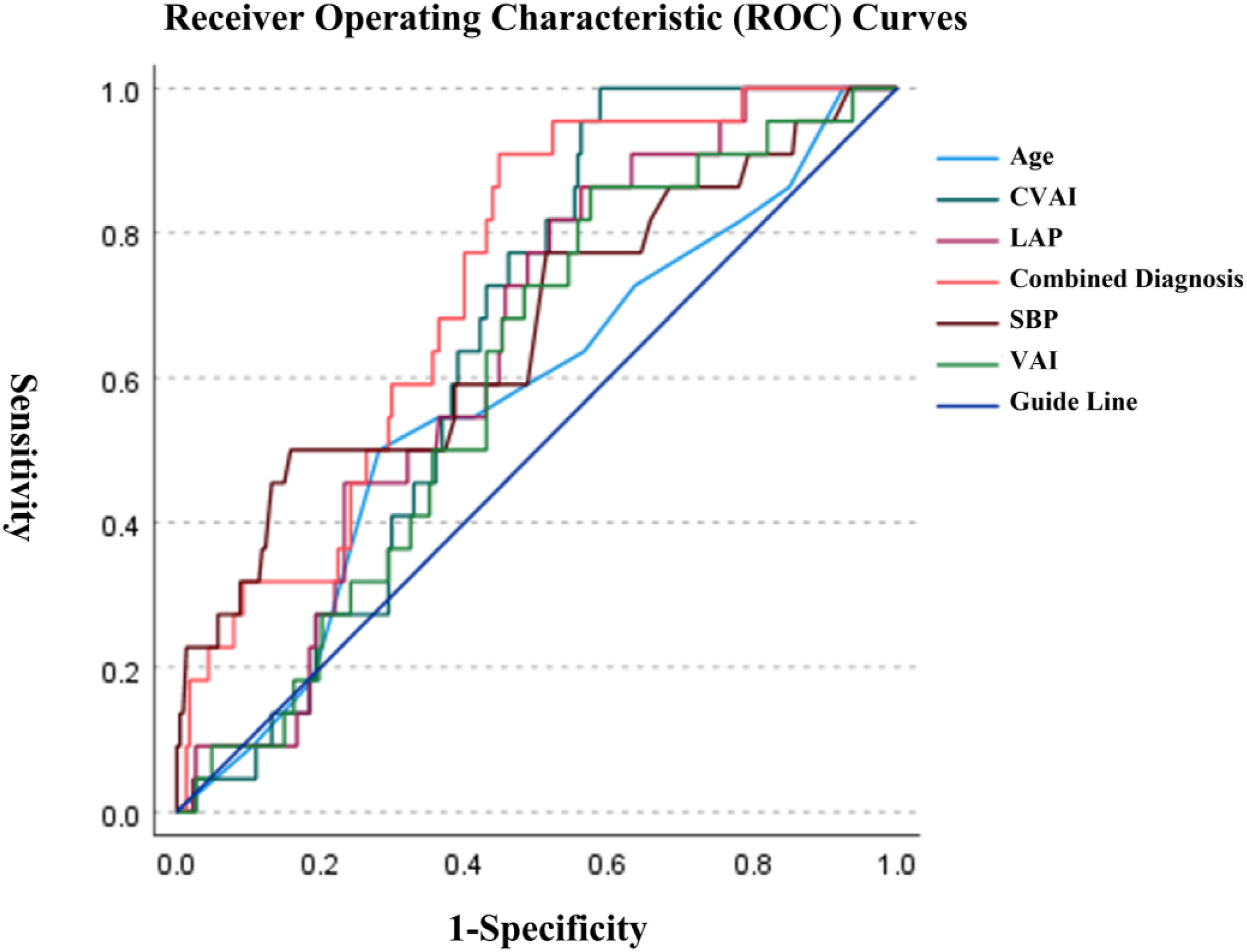
Receiver operating characteristic (ROC) curves for discriminating left ventricular hypertrophy (LVH) using the CVAI, age, and baseline systolic blood pressure. The receiver operating characteristic (ROC) curves are shown for the Chinese visceral adiposity index (CVAI), age, clinic systolic blood pressure (SBP), lipid accumulation product (LAP), visceral adiposity index (VAI), and the combined model (CVAI + age + clinic SBP) in postmenopausal women with primary hypertension. In the overall cohort, CVAI showed moderate discrimination for LVH (area under the curve [AUC] 0.702, 95% confidence interval [CI] 0.647–0.756), and the combined model showed improved discrimination (AUC 0.751, 95% CI 0.697–0.805). Pairwise AUC comparisons were performed using the DeLong test (Table 6). Subgroup ROC analyses stratified by body mass index (BMI) and age are presented in Table 6.
4. Discussion
This retrospective case–control study shows that CVAI, age, and clinic systolic blood pressure are each associated with left ventricular hypertrophy (LVH) in postmenopausal women with primary hypertension, and that a combined model incorporating these variables improves discrimination for LVH.5,16
More than half of Chinese adults are overweight or obese, 8 and there is growing evidence that visceral fat is more dangerous for the heart than general obesity. Therefore, it is crucial to find reliable methods to assess visceral fat dysfunction. CVAI showed a stronger association and discriminative performance for LVH than BMI, WC, and other anthropometric measures, likely because it incorporates lipid parameters (triglycerides and HDL-C) and age, providing a more comprehensive assessment of visceral adiposity. 9
Current research supports the physiological plausibility of our findings. Visceral adipose tissue releases pro-inflammatory cytokines (IL-6, TNF-α) and adipokines that directly activate myocardial fibroblasts and promote collagen deposition through pathways that depend on angiotensin II.17–21 This process is essential for women who have undergone menopause because a lack of estrogen worsens endothelial dysfunction and arterial stiffening. These changes increase systolic hypertension and ventricular afterload.17,22,23 The increased risk of heart remodelling associated with both older age and high systolic blood pressure demonstrates that metabolic, chronological, and hemodynamic insults all contribute to this process.24–27 Although no significant difference in serum creatinine levels was observed, BUN and cystatin C were significantly higher in the LVH group, suggesting that these renal biomarkers may be independently associated with LVH and cardiovascular risk.
Our findings highlight the potential role of the CVAI in identifying postmenopausal women with hypertension at a higher risk of LVH. Echocardiography and advanced cardiac care are much less accessible in rural Chinese primary care facilities due to uneven resource distribution and economic disparities. 28 The high sensitivity (83.7%) of the CVAI in our study suggests that it could be further investigated as a simple screening tool to identify individuals who may benefit from prioritization for echocardiography in resource-limited settings.28,29 However, this application requires confirmation in prospective studies before it can be recommended for clinical practice.29,30
We propose an exploratory, hypothesis-generating, tiered risk stratification framework based on receiver operating characteristic (ROC)-derived cut-offs from this dataset; however, these thresholds and any related management implications require rigorous external validation and health-economic evaluation before clinical implementation. There are a few limitations to consider. First, our study was conducted at only one center (the Xinjiang region); therefore, the results may not apply to populations living along the coast or in southern China. Second, unmeasured confounders, especially sodium intake and physical activity, may independently influence LVH risk. Third, the study did not account for variations in antihypertensive regimens, although different drug classes affect cardiac remodelling in various ways. Detailed data on specific antihypertensive medication regimens (including drug class, dosage, and duration) were incomplete in our retrospective dataset, representing a significant source of unmeasured confounding. The lack of this detailed information prevented us from conducting reliable stratified analyses or adjusting for the potential impact of different antihypertensive treatments on LV remodelling. Fourth, this study was cross-sectional; therefore, no causal relationships can be drawn between the CVAI, age, systolic blood pressure, and LVH. Fifth, LVH was defined solely based on LVMI, without incorporating right ventricular wall thickness (RWT) measurements to differentiate between concentric and eccentric hypertrophy. Future studies should include RWT to refine LVH classification. Research should also validate findings in multiethnic populations, examine CVAI trajectories during LVH progression, and test interventions to reduce visceral adiposity (such as GLP-1 agonists) for LVH regression. Additionally, adipokine profiling in cardiac tissue could clarify the link between obesity, hypertension, and cardiac remodelling.
This study proposes a framework that integrates the Chinese visceral adiposity index (CVAI), age, and systolic blood pressure to evaluate their cross-sectional association with left ventricular hypertrophy (LVH) in postmenopausal women with primary hypertension. The CVAI has demonstrated stronger associations with cardiovascular outcomes than traditional indices, such as body mass index (BMI) and waist circumference, particularly in the Chinese population. Unlike previous studies that examined individual factors, our analysis considers these variables together to assess their combined discriminative performance. This hypothesis-generating approach suggests that these three easily available variables could be further investigated in prospective cohort studies to determine their utility for identifying individuals at a higher risk of LVH in routine clinical settings, particularly in resource-limited environments.
5. Conclusions
In postmenopausal women with primary hypertension, the CVAI, age, and clinic SBP were independently associated with left ventricular hypertrophy. Combining these three factors demonstrated improved discriminative performance compared with individual variables alone. This triad represents a low-cost, practical approach that may help identify individuals who warrant prioritization for echocardiography in resource-limited settings. However, as this was a cross-sectional, single-center study, these findings should be considered hypothesis-generating. Prospective multicenter studies are needed to validate these associations and establish the clinical utility of this approach for improving cardiovascular risk assessment and the early detection of LVH.
Supplemental Material
Supplemental Material - Association of CVAI, age, and clinic systolic blood pressure with left ventricular hypertrophy in postmenopausal women with primary hypertension: A single-centre retrospective case–control study
Supplemental Material for Association of CVAI, age, and clinic systolic blood pressure with left ventricular hypertrophy in postmenopausal women with primary hypertension: A single-centre retrospective case–control study by Xiaoyong Hu, Djandan Tadum Arthur Vithran, Qiuyu Zhang, Nuhanguli Malasadi, Adili Abudula, Hongjian Li in Science Progress.
Footnotes
Acknowledgments
The authors would like to thank the participants for their valuable contributions to this study.
ORCID iDs
Ethical considerations
Ethical approval was granted by the Ethics Committee of the Fifth Affiliated Hospital of Xinjiang Medical University (Approval No. XYDWFYLSK-2025-25).
Consent to participate
Written informed consent was obtained from all participants.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 82260089) and the High-level Leading Talent Project for Medical and Health Care Professionals of the Xinjiang Uygur Autonomous Region–Tian Shan Talent Elite Program (No. TSYC202301A057).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors declare that they have no conflict of interest.
Data Availability Statement
The datasets generated and analyzed in the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.