Abstract
Background
Previous studies suggest that obesity may exacerbate clinical manifestations of chronic obstructive pulmonary disease (COPD). Early detection of COPD could improve therapeutic outcomes. This study investigates the relationship between a novel metabolic biomarker, the Visceral Adiposity Index (VAI), and COPD risk. We hypothesized that higher VAI levels would be independently associated with an increased prevalence of COPD.
Methods
This study is a cross-sectional analysis of data from the National Health and Nutrition Examination Survey (NHANES) spanning the periods 1999–2018 and 2021–2023. Multivariable logistic regression was employed to assess the association between VAI and COPD, supplemented by subgroup stratification, smoothed curve fitting, and threshold effect analyses.
Results
Among 16,167 participants, 1397 were diagnosed with COPD. Multivariable logistic regression revealed a positive correlation between VAI levels and COPD prevalence, independent of confounding factors. Subgroup analyses indicated stronger associations in hypertensive patients and females. Smoothed curve fitting identified a threshold effect for VAI, with an inflection point at 2.55 (P = 0.002).
Conclusion
The findings of this study indicate a significant positive correlation between VAI levels and the prevalence of COPD. Maybe incorporating visceral adipose function assessment into the initial risk stratification system for high-risk populations with COPD holds certain clinical value.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a progressive pulmonary disorder characterized by irreversible airflow limitation, predominantly associated with tobacco exposure. 1 It frequently coexists with multiple comorbidities including metabolic syndrome, cardiovascular diseases, anxiety-depressive disorders, diabetes, and obesity.2,3
Recent investigations into the prevalence of metabolic syndrome among patients with COPD reveal that a substantial proportion exhibit metabolic disturbances characterized by central obesity and dyslipidemia. 4 Obesity, as a global health concern, represents one of the most prevalent comorbidities in COPD populations. 3 Patients with COPD with concurrent obesity often experience diverse pulmonary complications.5,6 Evidence indicates that obese patients with COPD manifest more severe respiratory symptoms, greater limitations in daily activities, reduced health-related quality of life, and increased healthcare resource utilization compared to their nonobese counterparts.7–9 While obesity is typically characterized by visceral fat accumulation, clinical practice predominantly relies on generic metrics such as body mass index (BMI), which merely reflect total adiposity without distinguishing subcutaneous from visceral fat distribution. The visceral adiposity index (VAI), designed to evaluate both visceral fat content and functional status,10,11 is derived through a computational model incorporating waist circumference (WC), BMI, triglycerides (TG), and high-density lipoprotein cholesterol (HDL-C). Compared to conventional indices like BMI and WC, VAI provides a more precise assessment of pathophysiological alterations, including fluctuations in adipocytokine levels and elevated plasma free fatty acid concentrations, thereby serving as an effective clinical tool for visceral adiposity evaluation.
The National Health and Nutrition Examination Survey (NHANES), a comprehensive health surveillance program administered by the Centers for Disease Control and Prevention (CDC), offers an exceptional research platform for investigating health-related associations such as the VAI–COPD relationship. With its large sample size, population heterogeneity, and high representativeness, NHANES provides robust epidemiological data. This study represents the first investigation utilizing the NHANES database to explore the potential association between VAI and COPD, aiming to evaluate VAI's viability as a predictive biomarker for COPD. We specifically hypothesized that a higher VAI, indicative of adverse visceral adiposity and metabolic dysfunction, would be positively associated with the prevalence of COPD.
Materials and methods
Study population
The reporting of this study conforms to STROBE guidelines. 12 This study utilized data from the NHANES, a biennial population-based survey funded by the US CDC since 1999. This study is a retrospective cross-sectional study. NHANES employs a multistage probability sampling design to recruit approximately 5000 individuals annually through household interviews, followed by physical examinations and biological sample collection in mobile examination centers. The survey encompasses 15 urban areas across the United States and includes diverse racial/ethnic groups such as African American, Asian, and Hispanic populations. 13 All patient information has been de-identified. Detailed information about the NHANES database is available at http://www.cdc.gov/nhanes.
We integrated data from NHANES cycles spanning 1999–2018 and 2021–2023, initially comprising 113,249 participants. After applying stringent inclusion and exclusion criteria, 16,167 US adults were retained for analysis. Specifically, 49,898 individuals were excluded due to missing COPD data, 466 for age < 20 years, 44,452 for incomplete VAI measurements, and 2266 for missing key covariates (Figure 1). In fact, following the screening process, the final study population was limited to subjects from 2003 to 2016.

Flow chart of patient screening. COPD: Chronic obstructive pulmonary disease; NHANES: National Health and Nutrition Examination Survey; VAI: visceral adiposity index.
The NHANES study protocol was approved by the Institutional Review Board (IRB) of the National Center for Health Statistics, and all participants provided written informed consent. 14 This secondary analysis was conducted in accordance with the ethical principles of the Declaration of Helsinki, as revised in 2024.
Assessment of COPD
Participants with COPD was defined as those with FEV1 (forced expiratory volume in one second)/FVC (forced vital capacity) < 0.7 15 after inhaling bronchodilators or those who answered “yes” to any of the following questions in their self-reports 16 : “Have doctors diagnosed you with chronic bronchitis?” “Have doctors diagnosed you with emphysema?” and “Have doctors or other health professionals diagnosed you with chronic obstructive pulmonary disease?”. 17
Assessment of LAP
The VAI was calculated by gender using the equation previously established by Amato et al., 18 as follows, where TG and HDL-C are in mmol/L, WC is in cm, and BMI is in kg/m².
Male:
Measurement of covariates
Based on previous studies,10,19–21 this research incorporated potential confounding factors that might influence the relationship between VAI and COPD in the final analysis. The examined covariates comprised two major dimensions: demographic variables and health behavioral variables. The demographic variables included: age, sex, race/ethnicity (categorized as Mexican American, non-Hispanic Black, non-Hispanic White, and Other races), poverty–income ratio (PIR), marital status (classified as married/cohabiting or living alone), and educational attainment (stratified into three levels: below high school, high school, and above high school). Health behavioral variables encompassed: baseline disease status (including diabetes and hypertension), along with smoking and alcohol consumption indicators. Disease status data were obtained through participants’ self-reported diagnoses confirmed by licensed physicians or other healthcare professionals. Smoking status was defined as “having smoked ≥100 cigarettes in one's lifetime,” while alcohol consumption was determined using the criterion of “having consumed alcoholic beverages ≥12 times in the past year.”
Statistical analysis
The VAI was categorized into four quartile groups (Q1–Q4) ranging from lowest to highest. Categorical variables were presented as proportions, while continuous variables were expressed as mean ± standard error (SE). Continuous variables were described using mean ± standard deviation (SD), and categorical variables were reported as frequencies or percentages. Linear regression was employed to calculate P-values for continuous variables, while chi-square tests were used for between-group comparisons of categorical variables. To assess the association between COPD and VAI, multivariable logistic regression models were utilized to compute odds ratios (ORs) with 95% confidence intervals (CIs). Three models were constructed: model 1 (unadjusted), model 2 (adjusted for age, race, and sex), and model 3 (fully adjusted model incorporating age, sex, race, PIR, marital status, education level, diabetes, hypertension, smoking status, and alcohol consumption).
Subgroup analyses were conducted to investigate potential effect modifiers through stratification by age, sex, race, PIR, marital status, education level, diabetes, hypertension, smoking status, and alcohol consumption. A generalized additive model (GAM) with smooth curve fitting was further applied to explore potential nonlinear relationships between VAI and COPD. Statistical significance was set at P < 0.05. All analyses were performed using R (version 4.2.0) and EmpowerStats (version 4.2). To mitigate the influence of outliers, extreme VAI values below the 1st percentile or above the 99th percentile were excluded from multivariable logistic regression, subgroup analyses, threshold effect analyses, and smooth curve fitting.
Results and analysis
Baseline characteristics of study participants
A total of 16,167 participants were included in this study. Table 1 summarizes the baseline characteristics of the study population. Participants were divided into four quartiles based on the mean VAI. The results demonstrated that compared to the lowest quartile, the highest VAI quartile exhibited a significantly higher proportion of males and a progressively increasing prevalence of COPD (Q1: 6.77%; Q2: 7.81%; Q3: 9.51%; Q4: 10.45%, P < 0.001). Additionally, significant differences were observed across VAI quartiles in age, sex, race, PIR, marital status, education level, diabetes, hypertension, alcohol consumption, and smoking status (all P < 0.001). Individuals in higher VAI quartiles were more likely to be male, older, non-Hispanic White or Mexican American, and to have a history of smoking, alcohol use, married/cohabiting status, lower income, lower education levels, as well as higher prevalence rates of hypertension and diabetes.
Baseline characteristics of participants.

Mean ± SD for continuous variables: the P-value was calculated by the linear regression model; (%) for categorical variables: the P-value was calculated by the chi-square test. COPD: chronic obstructive pulmonary disease; PIR: poverty–income ratio; Q: quartile; VAI: visceral adiposity index.
Association between VAI and COPD
To investigate the relationship between VAI and COPD, three multivariable logistic regression models were constructed (Table 2). In the unadjusted model 1, logistic regression revealed a significant positive association between VAI and COPD. After adjusting for age, race, and sex in model 2, the association remained statistically significant (OR = 1.0239, 95% CI: 1.0122–1.0358, P = 0.000059). However, in the fully adjusted model 3 (further controlled for age, sex, race, PIR, education level, marital status, alcohol consumption, smoking status, hypertension, and diabetes), the association was substantially attenuated (OR = 1.0009, 95% CI: 0.9951–1.0220, P = 0.216553). Sensitivity analysis using VAI quartiles in model 3 showed that compared to the lowest quartile, the highest VAI quartile was associated with a 23.44% increased risk of COPD (OR = 1.2344, 95% CI: 1.0287–1.4812, P = 0.023527).
Multivariable logistic regression models for the association between VAI and COPD in adults.

CI: confidence interval; COPD: Chronic obstructive pulmonary disease; OR: odds ratio; VAI: Visceral adiposity index.
Based on model 3, a GAM with smooth curve fitting was applied to further explore the nonlinear relationship between VAI and COPD (Figure 2). Threshold effect analysis identified an inflection point at VAI = 2.55 (P = 0.002). Below this threshold (VAI < 2.55), each 1-unit increase in VAI was significantly associated with a 16.24% elevated COPD risk (OR = 1.1624, 95% CI: 1.0609–1.2737, P = 0.0013). Above the threshold (VAI ≥ 2.55), the association diminished (OR = 0.9993, 95% CI: 0.9832–1.0158, P = 0.9358), indicating a saturation effect where the impact of VAI on COPD risk stabilized (Table 3).
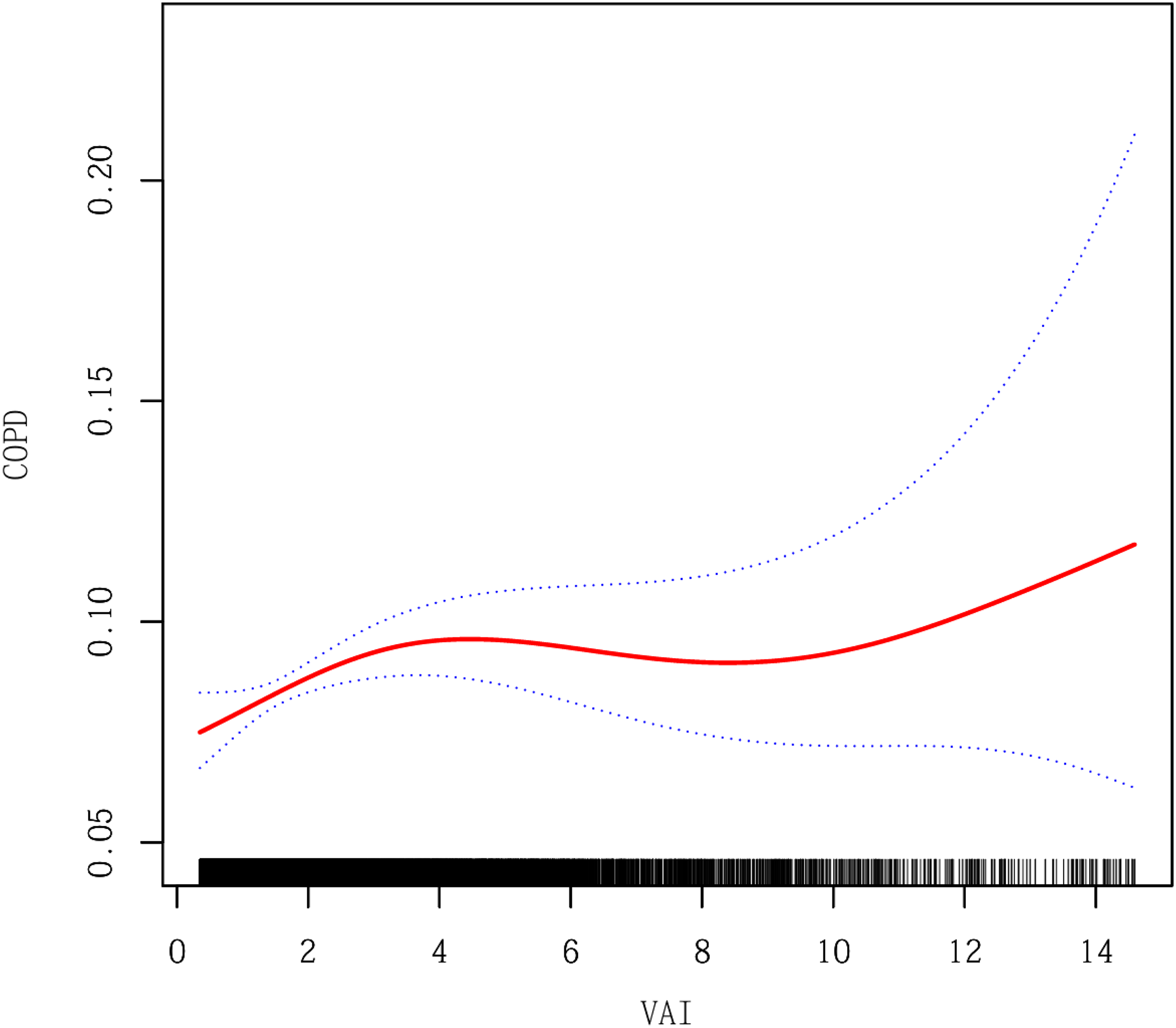
The nonlinear associations between the VAI and COPD. Notes: The solid red line represents the smooth curve fit between variables. Blue bands represent the 95% confidence interval from the fit. COPD: Chronic obstructive pulmonary disease; VAI: visceral adiposity index.
Threshold effect analysis of VAI on COPD using a two-piecewise logistic regression model in adults.

Notes: Age, gender, race, PIR, marital status, education level, diabetes, hypertension, smoking status, and alcohol consumption were adjusted. CI: confidence interval; COPD: Chronic obstructive pulmonary disease; OR: odds ratio; PIR: poverty–income ratio; VAI: visceral adiposity index.
Subgroup analysis
Subgroup analyses stratified by age, sex, race, marital status, PIR, education level, hypertension, diabetes, smoking, and alcohol consumption were conducted to validate the robustness of the VAI–COPD association and identify heterogeneity (Table 4). The positive association remained consistent across most subgroups. Notably, two key interactions were observed: Hypertension status: A significant interaction was detected, with VAI positively associated with COPD in nonhypertensive individuals (OR = 1.0257, 95% CI: 1.0075–1.0442, P = 0.0054) but not in hypertensive individuals. Sex: A sex-specific interaction was identified, showing a positive association between VAI and COPD in females (OR = 1.0237, 95% CI: 1.0034–1.0444, P = 0.0220) but not in males.
Stratified analysis of the correlation between VAI and COPD in adults.

Notes: Age, gender, race, PIR, marital status, education level, diabetes, hypertension, smoking status, and alcohol consumption were adjusted. The strata variable was not included in the model when stratifying by itself.
CI: confidence interval; COPD: chronic obstructive pulmonary disease; OR: odds ratio; PIR: poverty–income ratio; VAI: visceral adiposity index.
Discussion
This cross-sectional study analyzed data from 16,167 participants in the US NHANES to investigate the association between the VAI and the risk of COPD. Results revealed a positive correlation between VAI levels and COPD risk in both unadjusted and partially adjusted models. Quartile stratification analysis demonstrated a more pronounced COPD risk disparity in the highest quartile group (Q4) compared to the reference group (Q1), potentially attributable to visceral fat accumulation, metabolic dysregulation, and systemic inflammation prevalent in high-VAI populations—pathophysiological processes implicated in COPD pathogenesis. Additionally, insulin resistance, dyslipidemia, and elevated oxidative stress in these individuals may exacerbate airway inflammation and lung function decline. Although full adjustment for confounders attenuated the association, it remained statistically significant in the extreme VAI quartile (Q4), suggesting clinical utility of VAI in COPD risk stratification.
Smooth curve fitting and threshold effect analysis identified a significant inflection point at VAI = 2.55 (P = 0.0013) in the fully adjusted model. Below this threshold, VAI and COPD risk exhibited a positive correlation, whereas the association diminished above this critical value (P = 0.9358). This nonlinear relationship implies VAI may serve as a potential biomarker for early COPD intervention. Subgroup analyses further revealed significant effect modification by sex and hypertension status, underscoring the need for sex-specific clinical management strategies and blood pressure control in COPD care.
Emerging evidence establishes obesity not merely as a COPD exacerbator but as an independent risk factor for disease progression.8,9,22,23 Abdominal obesity restricts lung expansion and increases respiratory workload, while adipose tissue dysfunction-driven chronic low-grade inflammation accelerates COPD pathogenesis. 24 Notably, a dose–response relationship between obesity severity and COPD risk has been observed in nonsmokers, highlighting the necessity for enhanced COPD screening in older obese populations (>50 years). 9 The “obesity paradox” in BMI-COPD associations warrants further exploration—while obesity may worsen dyspnea, survival advantages in mild-to-moderate obesity likely stem from interactions among smoking history, comorbidities, and lifestyle factors. 25 Adipokines (e.g. anti-inflammatory adiponectin) critically modulate pulmonary inflammation and function, 8 emphasizing the need for comprehensive evaluation of BMI, fat distribution patterns, and medication history in personalized management.
Currently, the direct molecular mechanisms linking the VAI to COPD have not been fully elucidated. However, existing evidence suggests that the abnormal body fat distribution and metabolic dysregulation reflected by VAI may collectively impair pulmonary function through both inflammatory pathways and mechanical constraints. Calculated from anthropometric measures such as WC and BMI, along with lipid parameters (triglycerides and HDL-C), VAI provides a comprehensive indicator of overall fat distribution and abdominal fat accumulation.11,26 Therefore, the association between VAI and COPD involves not only metabolic inflammatory mechanisms but also alterations in respiratory mechanics resulting from abnormal fat distribution. Visceral adipose tissue functions as a highly active endocrine organ, releasing pro-inflammatory cytokines—including leptin, tumor necrosis factor-alpha (TNF-α), and interleukin-6 (IL-6)—while suppressing anti-inflammatory mediators like adiponectin. This establishes a state of chronic low-grade inflammation. In COPD, this systemic inflammatory milieu may synergize with localized pulmonary inflammation, exacerbating airway remodeling, parenchymal destruction, and the accelerated decline of lung function. This state leads to systemic chronic low-grade inflammation. Such systemic inflammation may synergize with the inherent chronic pulmonary inflammation, exacerbating airway remodeling, lung tissue destruction, and accelerated decline in lung function, thereby promoting the progression of COPD.27–30 Second, the metabolic dysfunction represented by VAI (i.e. insulin resistance and dyslipidemia) also exerts synergistic detrimental effects on the pulmonary microenvironment. Insulin resistance itself has been shown to be independently associated with decreased lung function, potentially through its pro-inflammatory effects or by influencing airway smooth muscle and vascular function. 31 In addition to systemic inflammation, mechanical factors are also crucial. Excessive visceral fat accumulation restricts diaphragmatic descent and thoracic expansion, thereby impairing lung ventilation and increasing the work of breathing.32,33 This mechanical disadvantage may lead to more severe air trapping and dyspnea symptoms in individuals with pre-existing airflow limitation. Therefore, the association between VAI and COPD involves not only metabolic inflammatory mechanisms but also alterations in respiratory mechanics resulting from abnormal fat distribution.
Through subgroup analyses, this study revealed a significant positive association between VAI and COPD risk in nonhypertensive populations, whereas no such association was observed in hypertensive individuals. This divergence may be attributed to the more complex pathophysiological characteristics inherent to hypertension patients, which could modulate VAI levels. Hypertension is not only linked to metabolic abnormalities but also induces cardiovascular dysfunction and systemic hemodynamic changes, 34 mechanisms that may influence the pathogenesis of COPD. Furthermore, the elevated systemic inflammation commonly observed in hypertensive populations, coupled with COPD's inflammatory nature, may attenuate VAI's independent predictive capacity for COPD risk in this subgroup. Conversely, in nonhypertensive populations, elevated VAI levels—as an indicator of visceral fat accumulation—may more directly reflect adipose metabolic dysregulation and its detrimental pulmonary effects, thereby accentuating the COPD association.
As the first exploratory investigation examining the VAI–COPD prevalence relationship, this study demonstrates methodological rigor through multiple strengths. The application of the large-sample NHANES database significantly enhanced the external validity of the findings. Comprehensive adjustments for confounders, combined with subgroup analyses, threshold effect evaluations, and smooth curve fitting, further reinforced result robustness. The clinical practicality of VAI measurement underscores its potential as a COPD screening tool, particularly valuable for early risk identification in resource-constrained settings. These attributes collectively highlight the study's innovative contributions and translational relevance. However, several study limitations warrant consideration: First, our COPD definition incorporated not only spirometric criteria but also self-reported physician diagnoses of emphysema or chronic bronchitis, which may introduce misclassification and recall bias—an inherent limitation of utilizing the NHANES database for the present study. Second, the cross-sectional design precludes causal inference and lacks COPD severity stratification, leaving its impact uncharacterized. Third, exclusive reliance on US population data may restrict generalizability to other ethnic groups. Finally, incomplete smoking exposure data constrained exploration of its potential confounding effects. Prudent interpretation of results should account for these limitations. Future research should prioritize the inclusion of more diverse population samples, particularly from socioeconomically disadvantaged regions and ethnic minority groups, to enhance the generalizability of findings. Future longitudinal studies with prospective designs are warranted to provide more definitive evidence. Such studies should prioritize the use of standardized spirometry protocols and systematically collect comprehensive clinical records to verify COPD diagnoses, thereby minimizing potential information bias. Longitudinal studies employing objective diagnostic modalities such as pulmonary function tests and advanced imaging examinations will provide stronger evidence for establishing causal relationships while better controlling for confounding variables. Future research could focus on exploring the correlation between VAI and persistent lung function indicators such as FEV1. This will help clarify the dose–response relationship between visceral adipose tissue volume and the severity of airflow limitation. Mechanistic investigations should focus on elucidating the precise biological pathways linking the VAI to COPD, with animal models and in vitro cellular systems playing pivotal roles in this exploration.
Conclusion
This study confirms a positive correlation between VAI and COPD prevalence. Based on this finding, incorporating VAI into COPD risk stratification for high-risk populations during clinical evaluation may enable more precise early identification and prevention through the quantification of visceral adipose function.
Footnotes
List of abbreviations
Acknowledgements
The authors acknowledge the National Center for Health Statistics (NCHS) at the Centers for Disease Control and Prevention (CDC) for their essential contributions to study design implementation, data collection protocols, systematic data curation, and creation of publicly accessible databases within the NHANES research program.
Ethics approval and consent to participate
This study used data from the National Health and Nutrition Examination Survey (NHANES), which are publicly available and openly licensed for unrestricted reuse. All data utilized in the study are fully anonymized and do not contain any personally identifiable information. The use of NHANES data is limited to statistical analysis and reporting purposes. Any attempt to identify individuals is prohibited and would violate the confidentiality protections provided under federal law. Therefore, no additional permission was required to access or use the data in this research. For more information, please visit the official NHANES website: ![]() .
.
Authors’ contribution
Qiang Xiao and Shaofeng Zhang conceptualized and designed the study. Jia Jiang, Yaling Zeng, and Jia Li extracted and organized the clinical data. Yidan Lin, Xiping Wu, Jiayi Liu, and Shaofeng Zhang contributed to statistical analysis, literature review, and drafted the manuscript. Xinyu Song, Qiang Xiao, and Kai Wang interpreted the data. Zekui Fang and Xinyu Song contributed to addressing reviewer comments, revising the manuscript format, and editing the Discussion section. All authors gave final approval for the version to be published.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Natural Science Foundation of Guangdong Province, the Demonstration Project of Technological Innovation of Hubei Province (grant number 2024A1515012935, 2022BCE031).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.


