Abstract
Objective
The Agatston score quantifies coronary artery calcium (CAC) but shows limited specificity for obstructive coronary artery disease (CAD) with extensive calcification. The Naples Prognostic Score (NPS) integrates inflammatory and nutritional parameters. This study evaluated NPS performance alongside Agatston scoring.
Methods
This is a retrospective, single-center study that analyzed 426 stable angina patients undergoing coronary CT angiography and invasive angiography at İzmir City Hospital in 2024. Agatston and NPS scores were calculated from routine parameters. Obstructive CAD was defined as significant lesions. Performance was assessed using ROC analysis and multivariate regression.
Results
Among 426 patients (age 61.7 ± 9.5 years, 63.4% male), 277 (65.0%) had obstructive disease. Obstructive CAD patients showed lower HDL (P = .005), higher glucose (P = .008), and elevated HbA1c (P < .001). Agatston score showed moderate accuracy (area under the curve (AUC) 0.644), NPS limited performance (AUC 0.554). In Agatston ≥1000 subgroup (n = 43), NPS achieved AUC 0.680, combined model AUC 0.725. NPS was an independent predictor (OR 1.22, P = .006). Correlation was negligible (ρ=0.048).
Conclusions
Metabolic markers (HDL, glucose, HbA1c) are more discriminative than inflammatory markers for obstructive CAD. NPS provides complementary value in extensive calcification (Agatston ≥1000), capturing distinct pathophysiologic domains.
Keywords
Introduction
Coronary artery disease remains the leading cause of mortality worldwide. 1 While coronary artery calcium (CAC) scoring via the Agatston method strongly predicts cardiovascular events, its diagnostic accuracy for detecting obstructive stenosis shows heterogeneity, particularly in patients with extensive calcification where anatomic burden may obscure flow-limiting lesions.2–4
However, CAC scoring has recognized limitations in symptomatic patients.5,6 While calcium burden correlates with total atherosclerotic plaque, it does not directly quantify stenosis severity or plaque vulnerability. The COME-CCT Consortium meta-analysis demonstrated that the Agatston score had moderate diagnostic accuracy (68.8%) but was significantly inferior to CT angiography (81.1%, P < .001) for detecting obstructive CAD. 5 Notably, 17% of patients with zero CAC scores had obstructive CAD on invasive angiography.5,7 Patients with extensive calcification may have either stable plaques or critical stenoses, limiting diagnostic specificity.6,8
Chronic inflammation drives atherosclerotic progression. Recent evidence emphasizes integrating inflammatory biomarkers with traditional risk assessment to improve cardiovascular risk stratification.9,10
The Naples Prognostic Score (NPS) integrates inflammatory and nutritional parameters: serum albumin, total cholesterol, neutrophil-to-lymphocyte ratio (NLR), and lymphocyte-to-monocyte ratio (LMR). Originally developed for cancer prognosis, NPS has demonstrated cardiovascular prognostic value.11,12
NPS predicts adverse outcomes in heart failure, acute coronary syndromes, and post-intervention populations. Each component reflects distinct pathophysiology: albumin indicates systemic inflammation and nutritional status, lipids reflect metabolic function, while NLR and LMR capture immune-inflammatory balance. 13
Despite growing evidence for NPS in cardiovascular prognostication, critical knowledge gaps remain. First, the diagnostic utility of NPS for detecting obstructive CAD has not been systematically evaluated with comprehensive laboratory parameters. Second, whether NPS provides incremental diagnostic value beyond anatomic calcium quantification is unknown, particularly in different calcium burden strata. Third, the relationship between comprehensive cardiovascular risk markers and combined anatomic-inflammatory assessment has not been characterized. Fourth, the impact of concurrent cardiovascular medications on these diagnostic relationships remains unexplored.14–17
We hypothesized that inflammatory and nutritional parameters captured by NPS might complement calcium scoring, particularly in patients with extensive calcification, where anatomic assessment alone may have limited discriminative capacity. This study aimed to: (1) evaluate the diagnostic performance of Agatston and Naples scores for detecting obstructive CAD; (2) analyze the contribution of detailed laboratory parameters; (3) assess the impact of cardiovascular medications; (4) determine whether Agatston and Naples scores provide complementary or redundant information; and (5) explore whether NPS offers incremental diagnostic value in specific patient subgroups stratified by calcium burden.
Methods
Study design and population
This retrospective study was approved by İzmir City Hospital Ethics Committee (Approval Number: 2024/578, Date: November 2024) and was conducted in accordance with the Declaration of Helsinki (1975, revised 2024). Due to the retrospective nature of the study, the requirement for informed consent was waived by the ethics committee. Patient recruitment occurred between January 2024 and December 2024.
Inclusion criteria: (1) age >18 years, (2) symptoms of stable angina pectoris, (3) coronary CT angiography (coronary computed tomography angiography (CCTA)) with calcium scoring performed, (4) invasive coronary angiography completed within 90 days of CCTA, (5) availability of complete laboratory data for NPS calculation including serum albumin, total cholesterol, complete blood count with differential, and (6) availability of comprehensive metabolic panel.
Exclusion criteria: (1) acute coronary syndrome at presentation, (2) previous coronary revascularization, (3) significant valvular heart disease, (4) known malignancy (active or within 5 years), (5) chronic inflammatory conditions, (6) acute infection within 2 weeks, (7) chronic kidney disease stage 4 or 5, (8) chronic liver disease (Child-Pugh class B or C), (9) incomplete laboratory data, or (10) incomplete imaging data.
Clinical data and laboratory assessments
Demographic data, cardiovascular risk factors, and medical history were obtained through electronic medical records review. Hypertension, diabetes mellitus, and hyperlipidemia were defined according to standard criteria or current medication use. Blood samples were obtained after 8–12 h of overnight fasting within 24 h of CCTA.
Complete blood count was analyzed using an automated hematology analyzer (Sysmex XN-1000). Biochemical analyses were performed using an automated clinical chemistry analyzer (Cobas 8000, Roche Diagnostics). Serum albumin was measured by the bromocresol green method. Lipid profile included total cholesterol, triglycerides, HDL, and LDL cholesterol (calculated using the Friedewald equation or measured directly if triglycerides ≥400 mg/dL). Fasting glucose was measured by the glucose oxidase method. HbA1c was measured by high-performance liquid chromatography. Electrolytes were measured by the ion-selective electrode method. High-sensitivity CRP was measured by immunoturbidimetric assay.
Naples prognostic score calculation
NPS was calculated according to the original definition by Galizia et al. 12 Points were assigned as follows: (1) serum albumin <40 g/L: 1 point; (2) total cholesterol <180 mg/dL: 1 point; (3) NLR >3.0: 1 point; and (4) LMR <4.44: 1 point. The total score ranges from 0 to 4, with higher scores indicating worse inflammatory-nutritional status.
Medication history
Systematic medication review was performed through electronic medical record review and structured patient interviews. Current use was defined as continuous medication use for at least 30 days prior to CCTA. Medications recorded included: ACE inhibitors or ARBs, beta-blockers, calcium channel blockers, oral antidiabetic agents, insulin therapy, and lipid-lowering therapy, including statins.
Coronary Ct angiography and calcium scoring
CCTA was performed using a 512-slice multidetector Ct scanner (Revolution Apex, Ge Healthcare) according to the guidelines of the Society of Cardiovascular Computed Tomography (SCCT). 18 Non-contrast ECG-gated coronary calcium scoring was performed using prospective ECG triggering. Calcium scores were calculated using dedicated GE post-processing software on the AW workstation (AW VolumeShare/CardIQ Suite, GE Healthcare) by experienced radiologists blinded to clinical outcomes. The Agatston method was applied, defining coronary calcification as regions ≥1 mm2 with attenuation values >130 Hounsfield units. Patients were categorized into coronary calcium score groups as follows: 0, 1–100, 101–400, 401–1000, and >1000. 2
Invasive coronary angiography
Invasive coronary angiography was performed via radial or femoral approach using standard techniques by experienced interventional cardiologists. Angiograms were reviewed by two experienced interventional cardiologists blinded to CCTA results, laboratory values, and NPS calculations. Obstructive CAD was defined as the presence of ≥50% diameter stenosis in the left main coronary artery or ≥70% diameter stenosis in any major epicardial vessel (left anterior descending, left circumflex, or right coronary artery) or their major branches (≥2.0 mm diameter), determined by visual estimation.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, Version 26.0 (IBM Corp., Armonk, NY, USA). Continuous variables were tested for normality using the Shapiro-Wilk test. Due to non-normal distributions of most laboratory parameters, continuous variables are presented as mean ± standard deviation and compared using the Mann-Whitney U test (two-group comparisons) or Kruskal-Wallis test (multiple-group comparisons). Categorical variables are presented as numbers (percentages) and compared using the chi-square test or Fisher exact test as appropriate (when expected cell counts <5).
Diagnostic performance was evaluated using receiver operating characteristic (ROC) curve analysis with calculation of area under the curve (AUC) and 95% confidence intervals using the DeLong method. Optimal cutoff values were determined using the Youden index (maximizing sensitivity + specificity − 1). Sensitivity, specificity, positive predictive value, and negative predictive value were calculated for clinically relevant cutoffs.
Multivariate logistic regression analysis was performed to identify independent predictors of obstructive CAD. Variables with P < .10 in univariate analysis were considered for inclusion in the multivariate model. A backward stepwise approach was used with P < .05 for retention in the final model. Results are presented as odds ratios (OR) with 95% confidence intervals. Model fit was assessed using the Hosmer-Lemeshow goodness-of-fit test. Multicollinearity was assessed using variance inflation factors (VIF), with VIF >5 indicating problematic multicollinearity.
The Spearman correlation coefficient was calculated to assess the relationship between Agatston and Naples scores, as both demonstrated non-normal distributions. Correlation strength was interpreted as: <0.3 weak, 0.3–0.7 moderate, >0.7 strong. Subgroup analyses were performed stratifying patients by Agatston score categories (0, 1–100, 101–400, 401–1000, >1000). Diagnostic performance metrics (AUC, sensitivity, specificity) were calculated within each calcium burden stratum. All statistical tests were two-sided, and statistical significance was defined as P < .05. No adjustment for multiple comparisons was made for exploratory subgroup analyses, which should be considered hypothesis-generating.
Sensitivity analysis: modified Naples score
To address the potential confounding effect of lipid-lowering therapy on the cholesterol component, we performed sensitivity analysis using a Modified Naples Score. The Modified NPS was calculated using only three components (serum albumin, NLR, and LMR), excluding the total cholesterol component, with a possible score range of 0–9 points. The diagnostic performance of the Modified NPS was compared to the standard 4-component NPS using bootstrap methodology (1000 iterations) to calculate 95% confidence intervals and statistical significance.
This study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting observational studies. 19
Results
Study population and baseline characteristics
A total of 426 consecutive patients met all inclusion criteria and were included in the final analysis. Mean age was 61.7 ± 9.5 years (range: 32–84 years), and 63% were male. The median time between CCTA and invasive coronary angiography was 14 days (interquartile range: 7–28 days).
Obstructive CAD was identified in 277 patients (65.0%) on invasive coronary angiography. The distribution of lesions by vessel territory was: LAD 125 patients (45%), RCA 89 patients (32%), LCx 78 patients (28%), and LMCA 22 patients (8%). Multi-vessel disease was present in 105 patients (38% of those with obstructive CAD).
Baseline demographic and clinical characteristics stratified by the presence of obstructive CAD are presented in Table 1. Groups were well-matched for age (61.5 ± 9.4 vs 61.9 ± 9.6 years, P = .688) and sex distribution (65.7% vs 59.1% male, P = .136). Traditional cardiovascular risk factors showed similar prevalence between groups. Hypertension showed borderline significance (64.3% vs 60.4%, P = .040).
Baseline demographic and clinical characteristics.

Interobserver Agreement. Calcium scoring was performed by two independent observers (board-certified cardiologists with >5 years CCTA experience). For quality control, a random subset of 50 cases (11.7%) underwent duplicate analysis. Interobserver agreement for Agatston score categories was excellent (weighted kappa 0.89, 95% CI 0.83–0.95). For total Agatston scores, the intraclass correlation coefficient was 0.94 (95% CI 0.89–0.97), with a mean absolute difference of 32±47 Agatston units. Disagreements were resolved by consensus reading with a third experienced reader.
Overfitting Prevention. To minimize overfitting risk given our sample size, we employed several protective strategies. The multivariable model included only variables with biological plausibility and univariate significance (P<.10). We limited the number of predictors relative to outcome events (approximately 1 predictor per 10 outcome events) and used stepwise backward elimination with stringent retention criteria (P<.05). Model performance was assessed using bootstrap internal validation procedures where applicable. For subgroup analyses with limited sample sizes (particularly Agatston ≥1000, n=43), we acknowledge these as hypothesis-generating and interpret findings with appropriate caution regarding potential overfitting.
Data are presented as mean±SD or n (%). CAD, coronary artery disease.
Laboratory parameters and metabolic markers
Laboratory parameters stratified by CAD status are shown in Table 2. Standard cardiovascular risk markers, including lipids and glucose, showed expected patterns.
Laboratory parameters stratified by coronary artery disease status.

Data are presented as mean ± SD. NLR, neutrophil-to-lymphocyte ratio; LMR, lymphocyte-to-monocyte ratio; LDL, low-density lipoprotein; HDL, high-density lipoprotein; CRP, C-reactive protein.
Liver function tests were similar between groups.05) and ALT (P > .05), showing no significant differences. Renal function parameters were similarly matched, with mean creatinine (P > .05) and estimated GFR (P > .05) demonstrating no significant associations. Serum albumin, a component of the Naples score, was comparable (P > .05).
In contrast, several metabolic markers demonstrated robust associations with obstructive CAD. Most notably, HDL cholesterol was significantly lower in patients with obstructive disease (P > .05), representing an 8.2% reduction. This difference persisted across all calcium burden categories and remained significant in multivariate analysis. Total cholesterol (P > .05), LDL cholesterol (P > .05), and triglycerides (P > .05) showed no significant differences, suggesting that the HDL-obstructive CAD relationship is independent of other lipid parameters.
Glucose metabolism parameters demonstrated particularly strong associations. Fasting glucose was significantly elevated in obstructive disease (P > .05), representing a 9.5% increase. More strikingly, HbA1c reflecting long-term glycemic control was substantially higher in the obstructive group (P > .05), with 11.7% relative difference. This association remained significant even among patients without diagnosed diabetes mellitus, suggesting that subclinical dysglycemia may contribute to atherosclerotic disease progression.
Serum potassium was lower in obstructive CAD patients (4.2 ± 0.4 vs 4.4 ± 0.4 mmol/L, P = .004), potentially reflecting dietary patterns or medication effects.
Cardiovascular medication use
Current medication use showed remarkably similar distribution between obstructive and non-obstructive groups (Table 3). ACE inhibitors or ARBs were used by 59.2% of obstructive and 59.1% of non-obstructive patients (P = .337). Beta-blockers were used by 40.1% versus 43.6% (P = .671), calcium channel blockers by 37.2% versus 40.3% (P = .463), and lipid-lowering therapy by 34.3% versus 35.6% (P = 1.000). The uniform distribution strengthens the interpretation of laboratory findings and ensures observed differences are not confounded by differential medication use.
Cardiovascular medication use.
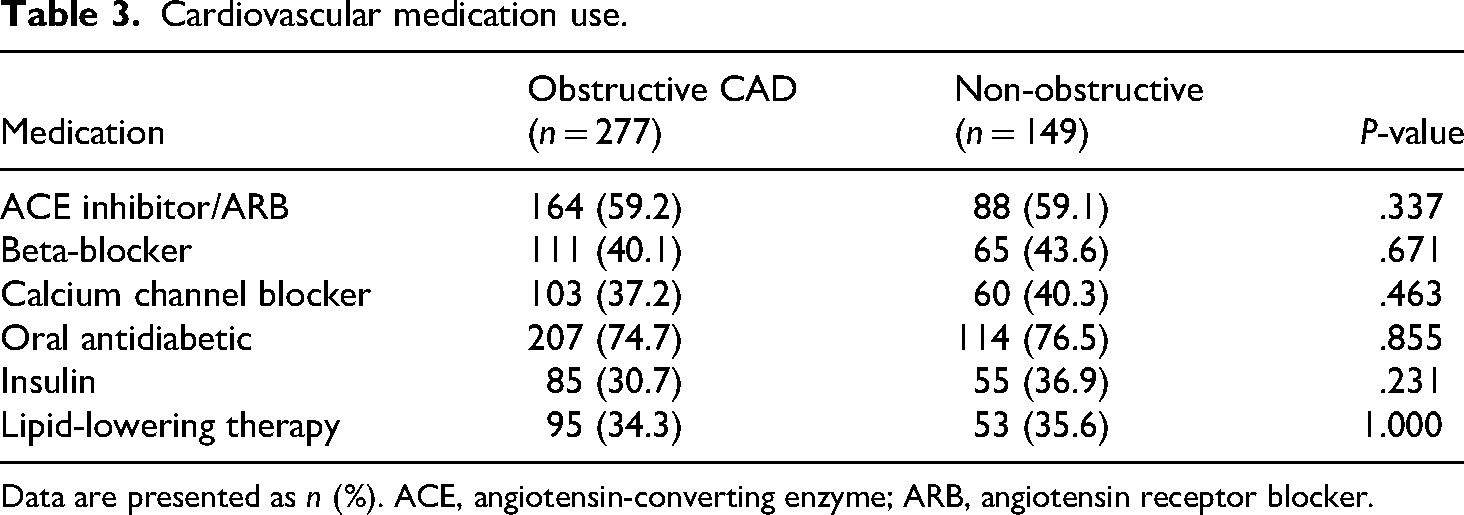
Data are presented as n (%). ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker.
Agatston and Naples scores
Mean Agatston and Naples scores were both significantly higher in patients with obstructive CAD (Table 4). The Agatston score difference was substantial, reflecting the expected association between calcium burden and obstructive disease. However, considerable overlap existed. Naples scores showed similar patterns with significant between-group differences but substantial overlap. The prevalence of obstructive CAD increased progressively with Naples score category: 48.6% (score 0), 62.1% (score 1), 70.2% (score 2), 78.7% (score 3), and 81.8% (score 4).
Coronary calcium and Naples scores.

Data are presented as mean±SD.
Diagnostic performance by calcium burden categories
Table 5 presents diagnostic performance stratified by Agatston score categories, revealing important heterogeneity. In the overall cohort, the combined model (Agatston + Naples) achieved AUC 0.643, essentially identical to Agatston alone (AUC 0.644), while Naples alone showed limited discrimination (AUC 0.554).
Diagnostic performance by calcium burden categories.

Most notably, in patients with very extensive coronary calcification (Agatston ≥1000, n = 43, 81.4% with obstructive CAD), the Naples score demonstrated markedly improved discrimination (AUC 0.680) compared to its overall performance. The combined model achieved AUC 0.725, representing a 13.7% improvement over Agatston alone. This improvement was the largest observed across all calcium burden strata.
Very high calcium burden subgroup analysis
Given the notable performance of the Naples score in the Agatston ≥1000 subgroup, we performed detailed characterization (Table 6). This subgroup comprised 43 patients (10.1% of the total cohort) with a mean age of 65.2 ± 8.1 years, 74.4% male, and a high prevalence of traditional risk factors.
Very high calcium burden subgroup (Agatston ≥1000, n = 43).

AUC: area under the curve; CI: confidence interval. Sensitivity and specificity calculated for NPS ≥2 cutoff. The combined model represents logistic regression including both Agatston and Naples scores.
Comparison of standard and modified Naples prognostic score.

Alb: serum albumin; Chol: total cholesterol; NLR: neutrophil-to-lymphocyte ratio; LMR: lymphocyte-to-monocyte ratio; AUC: area under the curve; CI: confidence interval.
Sensitivity and specificity calculated at NPS cutoff ≥2.
P-value for AUC comparison using the bootstrap method.
Spearman correlation analysis between Agatston and Naples scores revealed a negligible correlation coefficient of ρ=0.048 (P = .324), confirming that these scores capture fundamentally orthogonal pathophysiologic information. This lack of correlation indicates Naples score provides information distinct from and additive to anatomic plaque burden.
In this subgroup, the Agatston score alone achieved AUC 0.637 (95% CI 0.437–0.815). Naples score demonstrated notably improved performance with AUC 0.680 (95% CI 0.523–0.837). Using a cutoff of NPS ≥2, sensitivity was 54.3% and specificity was 87.5%. The combined model achieved the highest diagnostic accuracy with AUC 0.725 (95% CI 0.541–0.878), representing 13.7% improvement over Agatston alone.
Multivariate analysis
Multivariate logistic regression identified Naples score as an independent predictor of obstructive CAD (OR 1.22, 95% CI 1.06–1.40, P = .006). Other significant independent predictors included Agatston score (OR 1.001, 95% CI 1.000–1.002, P = .048), HDL cholesterol (OR 0.97, 95% CI 0.95–0.99, P = .012), and HbA1c (OR 1.28, 95% CI 1.08–1.52, P = .004). CRP did not retain significance (P = .52). The comprehensive multivariate model achieved AUC 0.684, representing 6.2% improvement over the Agatston score alone.
Correlation analysis
Sensitivity analysis: modified Naples score performance
Given that 35% of our cohort received lipid-lowering therapy and total cholesterol showed no discrimination between obstructive and non-obstructive groups (P = .762), we performed sensitivity analysis using a Modified Naples Score, excluding the cholesterol component. The Modified NPS (calculated from albumin, NLR, and LMR only) demonstrated virtually identical diagnostic performance to the standard NPS (AUC 0.527, 95% CI 0.474–0.580 versus AUC 0.554, 95% CI 0.501–0.609; difference 0.027, P = .214). Among the 426 patients, 182 (42.7%) had total cholesterol <180 mg/dL, with equal distribution between obstructive and non-obstructive groups (42.6% vs 43.0%, P = 1.000). These findings confirm that the cholesterol component contributes no discriminative value in this treated cardiovascular population (Table 7).
The “cholesterol paradox” in cardiovascular NPS application
A critical finding of our sensitivity analysis relates to the cholesterol component of the Naples Score. Originally derived in oncology populations, the NPS assigns one point to patients with total cholesterol <180 mg/dL, reflecting malnutrition, cachexia, and reduced hepatic biosynthetic capacity, all markers of poor prognosis in cancer patients. However, this scoring paradigm creates a paradox in cardiovascular populations receiving guideline-directed medical therapy. In our cohort, 35% of patients were on lipid-lowering therapy, and statin-treated patients achieving excellent cholesterol control would paradoxically receive a “worse” score than untreated hyperlipidemic patients.
Our data strongly support this concern. Total cholesterol showed no significant difference between obstructive and non-obstructive groups (194.6 ± 56.0 vs 192.5 ± 61.5 mg/dL, P = .762), and the cholesterol component demonstrated no association with obstructive CAD (P = 1.000). Most compellingly, our sensitivity analysis demonstrated that the Modified NPS (excluding cholesterol) performed identically to the standard NPS (AUC 0.527 vs 0.554, P = .955), confirming that the cholesterol component contributes zero discriminative value in this setting.
This finding has important implications for NPS application in cardiology. While the composite score remains useful due to its metabolic (albumin, glucose) and inflammatory (NLR, LMR) components, the cholesterol component requires reconsideration in treated cardiovascular populations. We recommend that future cardiovascular studies consider using a Modified Naples Score (three components) or develop cardiovascular-specific scoring cutoffs that account for lipid-lowering therapy. This “cholesterol paradox” illustrates the challenge of translating prognostic scores across clinical contexts and emphasizes the need for population-specific validation.
Discussion
This comprehensive multi-parameter study provides novel insights into the diagnostic evaluation of suspected obstructive coronary artery disease through integrated assessment of anatomic calcium burden and systemic metabolic-inflammatory status. Our principal findings demonstrate that: (1) metabolic dysfunction markers (HDL, glucose, HbA1c) show stronger associations with obstructive CAD than traditional inflammatory markers; (2) Naples score demonstrates specific diagnostic value in patients with very extensive calcification (Agatston ≥1000); (3) these relationships persist independent of cardiovascular medication use; and (4) the negligible correlation between scores confirms they capture complementary pathophysiologic domains.
Metabolic markers versus inflammatory markers
A striking finding is the divergence between metabolic and traditional inflammatory markers in their association with obstructive CAD. While CRP showed no significant difference between groups (P = .589), metabolic markers demonstrated robust associations: HDL cholesterol (P = .005), fasting glucose (P = .008), and HbA1c (P < .001). This challenges traditional focus on inflammatory biomarkers alone and supports emerging concepts of metabolic-inflammatory crosstalk in atherosclerosis.3,4 This pattern likely reflects the stable plaque phenotype characteristic of chronic angina, where metabolic dysfunction outweighs acute inflammatory processes in determining stenosis severity.
HDL cholesterol's protective role extends beyond reverse cholesterol transport to include anti-inflammatory and endothelial protective functions. The 8.2% lower HDL in obstructive CAD reflects both quantitative deficiency and qualitative dysfunction. HDL particles lose protective properties through oxidation and glycation in inflammatory states. 20 HDL's ability to promote cholesterol efflux from arterial macrophages determines its atheroprotective capacity, with efflux capacity more strongly predicting events than HDL levels alone. 21
Glucose metabolism parameters reflect insulin resistance and chronic hyperglycemia, driving atherosclerosis through oxidative stress, AGE formation, and endothelial dysfunction. 22 The 11.7% higher HbA1c in obstructive disease suggests subclinical dysglycemia contributes to atherosclerotic progression, supporting treatment of glycemia as a continuous risk factor. 22
CRP's lack of association is notable. While CRP predicts future events in primary prevention, it may not detect prevalent obstructive disease in symptomatic patients. 10 CRP elevations in stable CAD are often modest, and hepatic CRP production may not capture localized vascular inflammation. 9 Composite scores integrating multiple parameters may capture cardiovascular risk more effectively than single markers.
These findings reflect our stable angina population characteristics.
Naples score performance across calcium burden Spectrum
The subgroup-specific performance of the Naples score represents our study's most clinically actionable finding. In the overall cohort, Naples score demonstrated limited diagnostic utility (AUC 0.554). However, in patients with Agatston ≥1000, Naples score achieved substantially improved discrimination (AUC 0.680), and the combined model reached AUC 0.725 (13.7% improvement over Agatston alone).
Inflammatory-nutritional status becomes diagnostically relevant when anatomic plaque burden is extensive but heterogeneous in significance. In a very high calcium burden, 81.4% had obstructive disease, yet 18.6% did not. The Naples score helped identify patients whose extensive calcium represents stable plaques without critical stenosis.
Coronary calcium heterogeneity matters: calcium density, distribution patterns, and plaque characteristics vary considerably. MESA demonstrated that calcium density inversely correlates with events—denser calcification indicates stable plaque. 4 Naples score may identify patients whose extensive calcium represents ongoing inflammation (higher scores) versus stable calcification (lower scores).
The high specificity (87.5%) of the NPS ≥2 cutoff in the Agatston ≥1000 subgroup is clinically valuable. Among patients with alarming calcium burden (≥1000) but NPS <2, 95% actually had obstructive disease. This suggests the Naples score helps refine risk assessment in a population where calcium scoring alone provides limited discrimination, potentially improving triage decisions for invasive evaluation.
Multiparameter integration and complementary information
The multivariate model achieved AUC 0.684 (6.2% improvement over Agatston alone), exemplifying precision medicine through integration of readily available data without additional cost. Naples score's independent predictive value (OR 1.22, P = .006) demonstrates that the composite score captures information beyond individual components.
Atherosclerotic disease is multifactorial. Naples score integrates four parameters representing distinct processes: albumin (inflammation, nutrition), cholesterol (metabolic reserve, though requiring careful interpretation due to “the cholesterol paradox” where guideline-directed lipid-lowering therapy may confound this component in cardiovascular populations), NLR (innate immunity), and LMR (adaptive immunity). The score's independent predictive value suggests synergistic information beyond individual components.
The negligible correlation (ρ=0.048) confirms complementary value. Agatston quantifies cumulative anatomic burden over decades; Naples captures current inflammatory-nutritional status. This orthogonality, measuring different risk dimensions, provides true complementarity rather than redundant information.
Clinical implementation
Our findings support a tiered diagnostic approach: standard risk assessment for most patients, with the Naples score potentially adding value in high-calcium cases. However, prospective validation is required before clinical implementation.
Implementation is straightforward: automated Naples calculation in electronic health records using routine laboratory tests. No additional testing, equipment, or training required. Cost is negligible since the labs are already obtained.
Our findings highlight the heterogeneous diagnostic challenge across the calcium burden spectrum, where different biomarkers may offer complementary value at opposite ends of the Agatston scale. In patients with zero CAC (n = 22, 59.1% with obstructive CAD in our cohort), anatomic calcium scoring provides limited prognostic discrimination, and emerging evidence suggests that inflammatory and metabolic biomarkers may offer incremental risk stratification. Recent Asian population studies have demonstrated that biomarkers, including high-sensitivity CRP, metabolic indices, and comprehensive inflammatory scores, can identify subclinical atherosclerosis progression even in zero CAC cohorts. Chen et al. systematically reviewed evidence showing that zero CAC does not guarantee absence of obstructive disease, particularly in Asian populations, and that biomarker-enhanced stratification may improve risk assessment in this subgroup. 23 Machine learning approaches incorporating laboratory parameters have shown promise for predicting CAC progression in initially zero-calcium patients.
Conversely, in patients with extensive calcification (Agatston ≥1000), anatomic burden alone loses specificity, and our data suggest that Naples score components—particularly the inflammatory-nutritional axis may help discriminate obstructive from non-obstructive disease within this high-calcium subgroup (AUC 0.680 vs Agatston alone 0.637). This bidirectional utility biomarkers enhancing zero CAC risk stratification at one extreme and heavy CAC discrimination at the other suggests that integrated anatomic-biomarker approaches may be most valuable at the spectrum's extremes rather than its middle ranges. Shen et al. demonstrated that even in patients with initial zero CAC, metabolic and inflammatory parameters predict subsequent calcium progression, suggesting these pathways remain active throughout atherosclerosis evolution. 23 Future research should specifically evaluate whether the Naples score or other composite indices can improve event prediction across the full calcium burden continuum, from zero to very high scores, potentially using machine learning frameworks similar to those demonstrated by Wu et al. for CAC progression modeling. 25
Gender-specific considerations merit acknowledgment in interpreting Naples score performance. Our cohort comprised 63.4% male patients (270/426), baseline Naples scores differed significantly between sexes (male: 1.36 ± 0.90 vs female: 0.97 ± 0.82, P < .001), with males demonstrating higher inflammatory-nutritional burden despite similar ages. This gender difference aligns with established sex-specific patterns in inflammatory biomarkers and metabolic syndrome prevalence, where premenopausal hormonal protection may confer lower inflammatory profiles in females. Despite this baseline difference, the Naples score demonstrated similar discriminative performance for predicting obstructive CAD in both sexes (male AUC 0.519, female AUC 0.524), suggesting the score's diagnostic utility is preserved across genders even though absolute score distributions differ. Males showed higher obstructive CAD prevalence (54.1% vs 43.6%) and greater calcium burden (mean Agatston 457 ± 662 vs 338 ± 476). Males comprised 72.1% (31/43), limiting the power for sex-stratified analysis in this high-calcium population. These findings suggest that while Naples score thresholds may require sex-specific interpretation due to baseline differences, the score's relative discriminative capacity appears maintained across genders. However, our study was not powered for definitive gender-stratified analysis, particularly within calcium burden subgroups. Given that inflammatory biomarker profiles, lipid metabolism, and metabolic syndrome manifestations exhibit well-established sexual dimorphism, future studies should specifically evaluate whether Naples score component weightings or diagnostic thresholds require sex-specific calibration for optimal cardiovascular risk prediction. Such an investigation would align with broader efforts toward precision medicine and sex-specific risk assessment in cardiovascular disease.
The mechanistic basis for the Naples score's enhanced discriminative capacity in patients with extensive coronary calcification (Agatston ≥1000) warrants detailed consideration. In this high-calcium subgroup, anatomic plaque burden is universally severe (mean Agatston 1885 ± 945), yet clinical significance varies substantially—our data showed 81.4% had obstructive disease while 18.6% did not. The Naples score may capture pathophysiologic dimensions orthogonal to calcium burden that distinguish stable, heavily calcified plaques from those with flow-limiting stenosis.
Several potential mechanisms merit exploration. First, the inflammatory components (NLR, LMR) may reflect ongoing plaque activity versus quiescent calcification. Neutrophil and monocyte activation drive both plaque progression and positive remodeling through matrix metalloproteinase release and inflammatory cytokine production. In extensive calcification, elevated NLR might indicate continued inflammatory activation promoting stenosis development, while preserved LMR might suggest relative plaque stability. The observation that metabolic markers (HbA1c, glucose, HDL) showed stronger associations with obstructive CAD than acute inflammatory markers (CRP) in our cohort suggests chronic metabolic-inflammatory dysregulation rather than acute inflammation characterizes vulnerable plaques in stable angina with heavy calcification.
Second, the nutritional-metabolic components (albumin, cholesterol) may indicate systemic vulnerability rather than local plaque characteristics. Low albumin in the context of extensive calcification might reflect chronic inflammation-driven hypoalbuminemia and endothelial dysfunction, both promoting stenosis development through impaired vascular repair mechanisms. Third, the composite nature of the Naples score, integrating inflammation, nutrition, and metabolism, may capture the multifactorial nature of atherosclerotic progression better than any single biomarker in patients whose disease has advanced to extensive calcification.
The Asian population evidence base provides additional mechanistic insights. Studies examining CAC progression in Asian populations with initially zero calcium have demonstrated that inflammatory and metabolic dysregulation precede anatomic calcification, suggesting these biomarkers capture early atherogenic processes. Shen et al. showed that even in zero CAC patients, metabolic parameters predict subsequent calcium accumulation, implying continuous inflammatory-metabolic axis activation throughout atherogenesis.23,24 Chen et al.'s meta-analysis demonstrated that zero CAC does not preclude obstructive disease, and that biomarker-enhanced stratification improves risk prediction. 23 If these same inflammatory-metabolic axes continue to drive plaque vulnerability after extensive calcification has developed, potentially through ongoing plaque remodeling and stenosis progression Naples score would logically maintain prognostic value even in very high calcium burdens. This hypothesis requires prospective validation examining whether Naples score changes predict events or stenosis progression within heavy CAC populations, ideally with serial imaging and inflammatory biomarker assessment to directly test whether score dynamics reflect plaque evolution.
Study limitations
Limitations include a single-center retrospective design, a symptomatic population limiting generalizability, cross-sectional analysis precluding outcome assessment, and a small sample size in the high-calcium subgroup (n = 43, including eight controls), and medication dosage variability not captured in data collection (dose-dependent effects of statins, ACE inhibitors, and beta-blockers on biomarkers cannot be fully assessed). These findings require prospective validation in larger, diverse cohorts before clinical implementation.
Future directions
Prospective validation studies are needed to confirm the Naples score's diagnostic and prognostic value across diverse populations and calcium burden ranges.
Third, assessment of whether Naples-based risk stratification affects downstream management decisions and clinical outcomes would establish clinical utility. A randomized trial comparing standard calcium score-guided versus integrated Agatston-Naples score-guided management could evaluate whether the integrated approach improves outcomes. Fourth, investigation of whether the Naples score changes with targeted interventions and whether such changes predict outcomes would clarify therapeutic implications.
Conclusions
This comprehensive multi-parameter analysis demonstrates that integrated assessment combining anatomic calcium quantification with systemic metabolic-inflammatory evaluation enhances diagnostic accuracy for obstructive coronary artery disease beyond calcium scoring alone. While the Agatston score remains the primary anatomic marker, metabolic dysfunction markers, particularly HDL cholesterol, fasting glucose, and HbA1c, show stronger associations with obstructive disease than traditional inflammatory markers such as CRP.
The Naples Prognostic Score demonstrates specific diagnostic value in patients with very extensive coronary calcification (Agatston ≥1000), where anatomic assessment alone has limited discriminative capacity. In this subgroup, the combined Agatston-Naples model achieved 13.7% improvement in diagnostic performance. The negligible correlation between Agatston and Naples scores confirms they capture fundamentally orthogonal pathophysiologic information: anatomic plaque burden versus current systemic metabolic-inflammatory status.
These diagnostic relationships persist independent of current cardiovascular medication use, supporting real-world clinical applicability. From a clinical implementation perspective, calculation of the Naples score from routine laboratory data may enhance risk stratification, specifically in patients with extensive coronary calcification, potentially improving triage decisions for invasive evaluation. The score requires no additional testing or cost beyond routine laboratory panels, facilitating implementation.
This integrated diagnostic approach aligns with the 2025 ACC Scientific Statement emphasizing inflammation independent role in atherosclerosis. By combining anatomic assessment with biological markers reflecting metabolic-inflammatory status, we move toward more comprehensive, multi-dimensional cardiovascular risk assessment. These hypothesis-generating findings require prospective validation in larger, multicenter cohorts to evaluate reproducibility, establish optimal diagnostic cutoffs, and determine effects on patient outcomes.
Footnotes
Acknowledgments
The authors would like to thank the staff of the Cardiology and Radiology departments at İzmir City Hospital for their support during the data collection process.
Ethical approval
This study was approved by the İzmir City Hospital Non-Interventional Clinical Research Ethics Committee (Approval Number: 2024/578, Date: November 19, 2024). The study was conducted in accordance with the Declaration of Helsinki.
Patient consent
Due to the retrospective nature of this study, the requirement for informed consent was waived by the İzmir City Hospital Ethics Committee.
Author contributions
Murat Küçükukur: conceptualization, methodology, data curation, formal analysis, writing—original draft, writing—review and editing, supervision, and project administration. Ece İrem Zaman: investigation, data collection, validation, and writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request, subject to ethical approval and data protection regulations.
