Abstract
Objective
Congestive heart failure (CHF) is a chronic disease marked by impaired cardiac function, and excess visceral fat has been reported to be associated with CHF. While the age-adjusted visceral adiposity index (AVAI) is a novel metric for evaluating visceral fat, its association with CHF has not been well elucidated.
Methods
The association between AVAI and CHF prevalence was assessed using cross-sectional data from the 2013–2018 National Health and Nutrition Examination Survey (NHANES). Logistic regression was used to assess the association between AVAI and CHF prevalence, with sequential adjustment for covariates. Subgroup analyses stratified by demographic variables and lifestyle factors were done to examine the robustness of the results. Lastly, the discriminative ability of VAI and AVAI in distinguishing individuals with and without prevalent CHF was evaluated using receiver operating characteristic (ROC) curve analysis.
Results
A total of 6,695 participants were included in the analysis, comprising 6,470 without CHF and 225 with CHF. Logistic regression revealed a significant positive association between AVAI and CHF prevalence (OR = 1.75, 95% CI: 1.62–2.89), which remained robust after adjusting for confounders (adjusted OR = 1.57, 95% CI: 1.31–1.87). In subgroup studies, AVAI was consistently associated with the prevalence of CHF. ROC analysis indicated that AVAI demonstrated superior discriminative performance compared with VAI in distinguishing individuals with and without prevalent CHF.
Conclusions
This cross-sectional study identified a significant association between AVAI and prevalent CHF. AVAI may represent a clinically relevant marker associated with the prevalence of CHF and may help identify populations with a higher likelihood of prevalent CHF for further prospective investigation.
Keywords
1. Background
Dyspnea, edema, and exhaustion are common clinical symptoms of congestive heart failure (CHF), a chronic illness marked by compromised cardiac pumping performance that significantly lowers patients’ quality of life. 1 Epidemiological studies indicate that the global prevalence and mortality rates of CHF are steadily rising, particularly in older populations, where these rates remain persistently high.2,3 CHF has become one of the most common cardiovascular conditions among adults in the United States, according to data from the American Heart Association, and its social cost is steadily rising. 4 CHF adversely impacts patients’ physical health, leading to a substantial deterioration in quality of life, while also imposing a considerable cost strain on healthcare systems due to recurrent hospitalizations and emergency visits.5,6 Identifying factors associated with CHF, particularly through the development of effective markers for early identification, remains of important clinical and public health relevance.
The growing prevalence of obesity has coincided with an increasing burden of metabolic and cardiovascular diseases, representing a substantial global public health concern. 7 Obesity, especially excess visceral fat, is strongly linked to chronic conditions such as metabolic syndrome, type 2 diabetes, and cardiovascular disease. 8 The visceral adiposity index (VAI) was developed as a non-invasive indicator of visceral fat based on combined anthropometric and metabolic parameters. Several imaging modalities, including computed tomography (CT), magnetic resonance imaging (MRI), dual-energy X-ray absorptiometry (DXA), and ultrasound-based techniques, have been used to assess visceral adiposity.9–11 Although these methods provide relatively accurate measurements, their use in large-scale population studies is often limited by factors such as cost, accessibility, technical requirements, and, in the case of CT, radiation exposure. In this context, VAI provides a practical, cost-effective, and scalable surrogate for estimating visceral adiposity, particularly in large-scale population studies or resource-limited settings. VAI has been validated against imaging-derived measurements of visceral fat and has demonstrated strong correlations with visceral adiposity assessed by MRI. 12 Beyond its role as a proxy for fat distribution, VAI has also been consistently associated with adverse metabolic profiles and cardiovascular conditions, including insulin resistance, metabolic syndrome, and various cardiovascular diseases such as heart failure.13–15 A cross-sectional study showed that VAI was positively correlated with CHF. However, a critical limitation of VAI is its inability to adequately account for age, a crucial determinant in visceral fat distribution and metabolism. 16 Given that visceral fat distribution and metabolic function vary with age, the predictive efficacy of VAI may differ across age groups, thereby limiting its clinical applicability. 17 To overcome this limitation, researchers have introduced the age-adjusted visceral adiposity index (AVAI). By incorporating age as an adjustment factor, AVAI offers a more accurate estimation of visceral fat levels and their potential disease impacts across different age groups. AVAI provides a more refined evaluation of visceral fat–related metabolic burden than traditional VAI. Previous studies have demonstrated that age enhances the association of VAI with cardiovascular and all-cause mortality, and AVAI has also been associated with mortality. These findings support its potential role as a clinically relevant marker and its value in informing future prospective studies. 18 Although AVAI has emerged as a refined marker of visceral fat burden, its association with CHF has not been systematically examined. Considering the central role of age in fat redistribution and cardiometabolic dysfunction, it is hypothesized that AVAI may demonstrate superior discriminative ability for prevalent CHF compared to the traditional VAI. This study investigates the association between AVAI and CHF prevalence and compares its discriminative performance with that of VAI in a nationally representative cohort.
2. Methods
2.1. Study design and population
This cross-sectional study was based on data from the 2013–2018 National Health and Nutrition Examination Survey (NHANES), a nationally representative survey designed to assess the health and nutritional status of the U.S. population. NHANES includes data from physical examinations, laboratory tests, questionnaire interviews, and medical conditions. All participants provided written informed consent, and the NHANES study protocol was approved by the NCHS Institutional Review Board. This study was conducted in accordance with the Declaration of Helsinki of 1975, as revised in 2024. Data from three consecutive NHANES cycles (2013–2014, 2015–2016, and 2017–2018) were included in this analysis. Participants were eligible if they were aged over 20 years, had available self-reported CHF diagnosis data, and had sufficient data for AVAI calculation. Individuals with missing critical variables were excluded to ensure the validity and reliability of the analyses. This study was reported in accordance with the STROBE guidelines. 19
2.2. Calculation of AVAI
In this investigation, AVAI functioned as the main exposure variable and was computed using a method derived from previous research.18,20 Laboratory data included triglycerides (TG) and high-density lipoprotein (HDL), whilst waist circumference (WC) and body mass index (BMI) were assessed at mobile screening sites. HDL and TG values were quantified in mmol/L, while WC was documented in centimeters. The AVAI calculation formula is presented as follows:
2.3. Definition of congestive heart failure
The diagnosis of CHF was established based on participants’ self-reported answers to the question: “Has a doctor ever informed you that you have congestive heart failure?”
Affirmative responses classified participants as CHF patients. This self-reported diagnostic method has been validated in multiple studies, demonstrating reliable accuracy and feasibility.21–23
2.4. Covariates
A variety of variables was used to account for any confounding factors that might affect the findings. Demographic factors included sex, race, poverty levels, and level of education. Poverty levels were classified into three categories based on the poverty index: <1, 1–3, and >3. 24 Lifestyle determinants including smoking, alcohol intake, and physical exercise. Participants who had consumed more than 12 alcoholic drinks in their lifetime were classified as drinkers, while those who had smoked at least 100 cigarettes were considered smokers. 25 Physical activity was assessed using self-reported questionnaires and quantified as metabolic equivalents (METs), with values <600 minutes/week classified as inactivity. Chronic diseases—including hypertension, hypercholesterolemia, chronic kidney disease (CKD), and diabetes—were identified via self-report. Relevant laboratory measures such as blood urea nitrogen (BUN), alanine aminotransferase (ALT), and aspartate aminotransferase (AST) were also included.
2.5. Statistical analysis
A cross-sectional investigation used data from the 2013–2018 NHANES cycles, with individuals chosen based on established inclusion criteria. All analyses in this study were conducted in accordance with the NHANES analysis guidelines, employing a complex sampling design for weighted analysis, incorporating sample weights (WTMEC2YR), stratification variables (SDMVSTRA), and master sampling units (SDMVPSU) to obtain nationally representative estimation results. Baseline characteristics were stratified by CHF status, with continuous variables reported as mean ± standard deviation and categorical variables as frequencies and percentages. Logistic regression was performed to assess the association between AVAI and CHF prevalence. To investigate the correlation between AVAI levels and the occurrence of CHF, AVAI was divided into four quartiles: Q1 (<-9.64), Q2 (-9.64 to -7.52), Q3 (-7.52 to -5.50), and Q4 (>-5.50). Restricted cubic spline (RCS) analysis was employed to explore the potential dose–response relationship between AVAI and CHF prevalence. Subgroup analyses were used to evaluate potential factors that may contribute to the association between AVAI and CHF in various demographics. All statistical analyses were performed using R software, with P-value < 0.05 considered statistically significant.
3. Results
3.1. Baseline characteristics of participants
The participant selection procedure is illustrated in Figure 1, which comprises a total of 6,695 individuals, 225 CHF participants and 6,470 non-CHF participants. Baseline characteristics are summarized in Table 1. Compared to non-CHF participants, those with CHF were generally older, more likely to smoke, and less physically active. Patients with CHF had significantly higher AVAI values compared to non-CHF patients, indicating a potential association between AVAI levels and the prevalence of CHF. Flow diagram displaying selection process for including participants. AVAI: age-adjusted visceral adiposity index Baseline characteristics of the study population. Mean (SD) for continuous variables, % for categorical variables. ALT: alanine aminotransferase; AST: aspartate aminotransferase; AVAI: age-adjusted visceral adiposity index; BMI: body mass index; BUN: Blood Urea Nitrogen; CHD: coronary heart disease; CHF: congestive heart failure; CKD: chronic kidney disease; HDL: high-density lipoprotein; PIR: poverty index ratio; SCR: Serum Creatinine; TG: Triglyceride; VAI: visceral adiposity index; WC: waist circumference.

3.2. Association between AVAI and prevalence of Congestive heart failure
Logistic regression analysis of the association between AVAI and CHF prevalence.

AVAI: age-adjusted visceral adiposity index; CI: confidence interval; CHF: congestive heart failure; OR: odds ratio; Q: quartiles.
Model 1: no covariates adjusted; Model 2: adjusted for sex and race; Model 3: adjusted for sex, race, PIR, educational level, smoke, drinking, activity status, diabetes, hypertension, CKD, hypercholesterolemia, BUN, ALT, AST.

RCS curve illustrating the dose-response relationship between AVAI levels and CHF prevalence. Adjusted for sex, race, PIR, educational level, smoke, drinking, activity status, diabetes, hypertension, CKD, hypercholesterolemia, BUN, ALT, AST. AVAI: age-adjusted visceral adiposity index; CI: confidence interval; CHF: congestive heart failure; OR: odds ratio.
3.3. Subgroup analysis
Subgroup analysis between AVAI and CHF.

3.4. ROC analysis
Figure 3 presents the ROC curves and corresponding AUC values for the discrimination of prevalent CHF. AVAI demonstrated a higher AUC (0.8070) compared with VAI (0.5726), indicating superior discriminative ability. To further ensure a fair comparison, we additionally evaluated a VAI model incorporating age (VAI + age), which showed an improved AUC of 0.7143; however, AVAI still exhibited better performance. These findings suggest that AVAI is more strongly associated with prevalent CHF and may better distinguish individuals with and without CHF in this cross-sectional setting. Given the established association between visceral adiposity and cardiometabolic disorders, AVAI may serve as a simple and non-invasive indicator associated with prevalent CHF in clinical practice. ROC curve assessing the diagnostic performance of VAI, VAI+Age and AVAI for predicting CHF prevalence.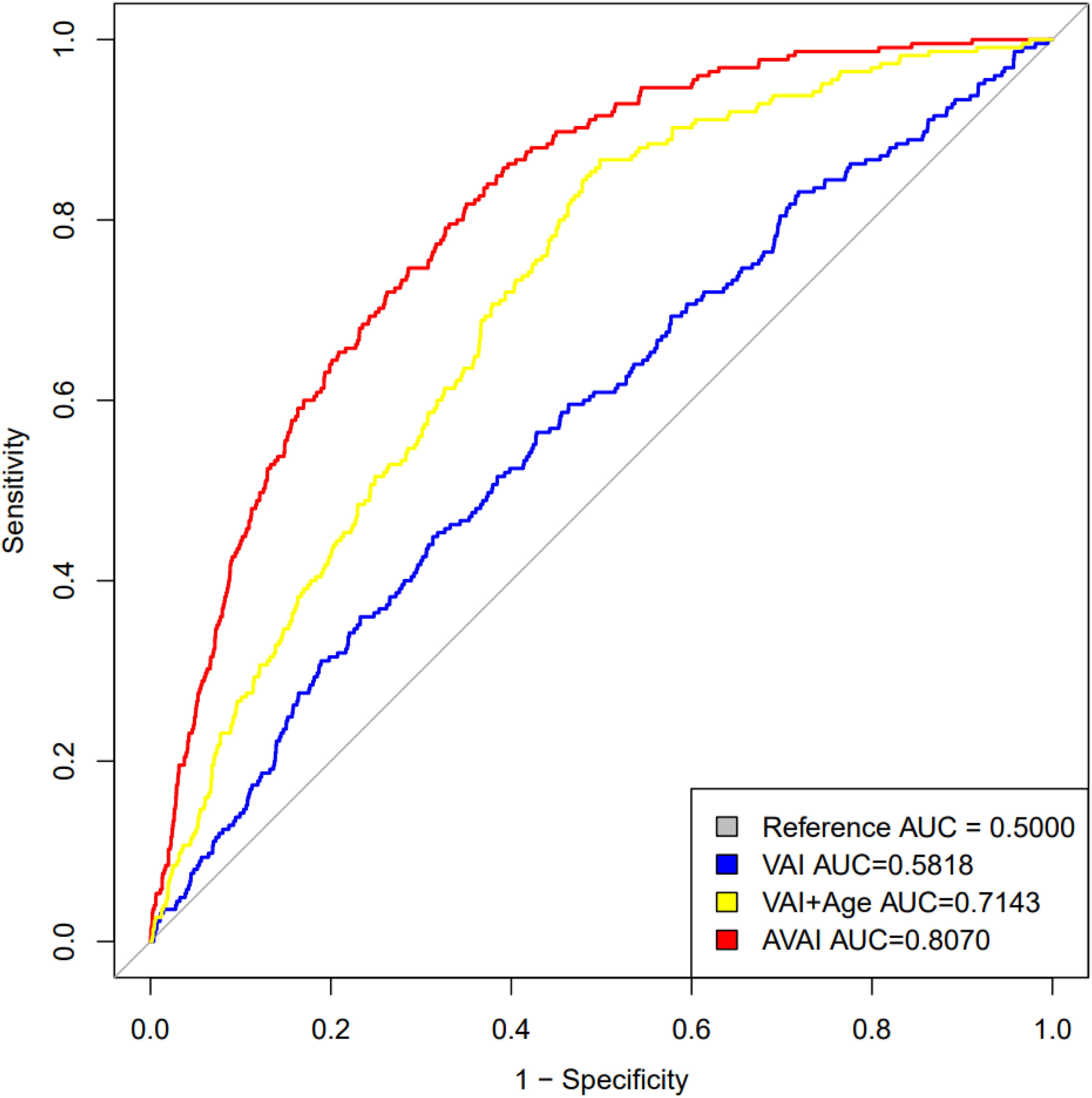
4. Discussion
This study, based on NHANES 2013–2018 data, examined the association between AVAI and CHF. The results demonstrated a significant association between AVAI and the prevalence of CHF, and this relationship remained consistent across multiple subgroups. ROC curve analysis indicated that AVAI showed better discriminative ability than traditional VAI for distinguishing individuals with and without prevalent CHF. These findings highlight the potential of AVAI as a reliable surrogate for visceral adiposity, a key factor in metabolic and cardiovascular disorders. Given the high burden of CHF and its clinical implications, AVAI may serve as a marker associated with adverse cardiometabolic profiles and prevalent CHF and serve as a basis for future prospective and mechanistic studies.
This study demonstrated a significant association between AVAI and prevalent CHF, providing new insight into the relationship between visceral adiposity and CHF. Prior research has demonstrated a significant association between visceral fat and cardiovascular diseases, particularly in the presence of metabolic abnormalities such as metabolic syndrome, diabetes, and hypertension, with excessive visceral fat accumulation being linked to these conditions.26–28 Compared with traditional VAI, AVAI introduced in this study demonstrates improved discriminative ability for prevalent CHF. This enhancement may be related to the important role of age in determining visceral fat distribution and metabolic activity. Aging is associated with a progressive redistribution of fat from subcutaneous to visceral compartments, along with increased adipose tissue inflammation, insulin resistance, and metabolic dysfunction, which have been associated with CHF.29–31 However, traditional VAI does not account for age-related metabolic changes, which may lead to underestimation in older adults. By incorporating age as an adjustment factor, AVAI may better capture age-related differences in visceral fat burden and cardiometabolic status across different age groups. This refinement strengthens the pathophysiological relevance of the index and supports the relevance of AVAI as a more tailored marker associated with prevalent CHF. There is a significant association between AVAI and CHF, and this finding is consistent with existing literature. Previous studies suggest that visceral fat accumulation is strongly associated with insulin resistance, inflammation, and metabolic dysregulation, all of which contribute to cardiovascular disease development and progression.32–34 Visceral fat has been associated with insulin resistance and inflammation and is considered to play an important role in cardiovascular pathology. In this study, AVAI was significantly associated with the prevalence of CHF across all subgroups, with no significant interaction effects observed. This consistency suggests that the observed association remains stable across different populations. The absence of interaction further supports the robustness of this association. ROC analysis indicated that AVAI demonstrated better discriminative ability than VAI for distinguishing individuals with and without prevalent CHF in this cross-sectional analysis. The higher AUC of AVAI may have potential clinical relevance, as it suggests improved cross-sectional discrimination of prevalent CHF compared with VAI; however, the practical impact of this difference remains to be established in prospective studies. Therefore, AVAI may serve as a valuable complementary marker associated with cardiometabolic status and prevalent CHF. While its prognostic or predictive utility cannot be determined from this cross-sectional study, its non-invasive and easily accessible nature supports its potential application in clinical research. Rather than functioning as a standalone diagnostic or prognostic tool, AVAI may provide additional cross-sectional information when used alongside conventional biomarkers, imaging modalities, and clinical indices. Prospective studies are warranted to clarify its temporal relationship with CHF and to determine whether AVAI has prognostic relevance in longitudinal settings.
The possible biological pathways between AVAI and CHF may be investigated via the various functions of visceral fat. Visceral fat operates as an active endocrine organ, releasing pro-inflammatory agents, which provoke systemic chronic low-grade inflammation and detrimentally affect heart structure and function.35–38 Excess visceral fat is closely linked to insulin resistance, which increases systemic metabolic stress and impairs cardiac metabolism, thereby contributing to metabolic dysfunction and worsening heart failure.36,39–41 Increased visceral fat also promotes cardiac remodeling, exacerbates myocardial fibrosis, and induces structural changes in the heart, thereby advancing the progression of CHF.42,43 Studies have also shown that visceral fat alters sympathetic nervous system activity, increasing cardiac workload and subsequently impairing heart function. 44 Additionally, metabolic byproducts of visceral fat exacerbate oxidative stress and myocardial cell damage, contributing to the onset of heart failure.41,45,46 These findings suggest that visceral fat may be associated with CHF through multiple pathways, including inflammatory responses, metabolic disturbances, cardiac remodeling, and neural regulation. However, although existing evidence indicates a close association between visceral adiposity and CHF, the underlying mechanisms remain unclear. Further studies are needed to better elucidate the mechanisms linking AVAI and CHF.
5. Study strengths and limitations
The principal strength of this study lies in the use of NHANES, a nationally representative large-scale dataset, in conjunction with AVAI to examine its association with congestive heart failure (CHF). By introducing AVAI as a novel marker, this study incorporates age-related factors, providing a more refined characterization of visceral fat distribution across age groups and its potential clinical relevance compared with traditional VAI. Nevertheless, several limitations should be acknowledged. The cross-sectional design precludes causal inference. Although a significant association between AVAI and CHF was observed, its role requires further validation in longitudinal studies. Secondly, the self-reported diagnosis of CHF by participants may be subject to recall bias or inaccuracies. Since the NHANES dataset relies on self-reported questionnaire data and is designed for large-scale epidemiological surveys using non-invasive methods, it lacks professional diagnostic imaging data. Moreover, while the NHANES dataset includes a broad and varied population, the lack of long-term individual follow-up data may restrict the generalizability of the results. Subsequent research should, thus, include long-term follow-up data to better clarify the possible processes and prediction capabilities of AVAI in cardiovascular illnesses. Additionally, the NHANES dataset does not distinguish between HFpEF (Heart Failure with preserved Ejection Fraction) and HFrEF(Heart Failure with reduced Ejection Fraction), precluding evaluation of subtype-specific associations. Given the plausible mechanistic relevance of visceral adiposity in HFpEF, future studies incorporating echocardiographic data are warranted.
6. Conclusion
This cross-sectional study identified a significant association between AVAI and prevalent CHF. AVAI may represent a clinically relevant marker associated with the prevalence of CHF and may help identify populations with a higher likelihood of prevalent CHF for further prospective investigation.
Footnotes
Acknowledgements
The National Health and Nutrition Examination Survey was made accessible to us by the CDC’s National Center for Health Statistics.
Ethical considerations
The National Center for Health Statistics Research Ethics Review Board looked over the NHANES study plan and gave its approval. Each subject also signed a form saying they understood what the study was about. This study did not directly test on humans or involve any other treatments. Secondary analyses were based on a de-identified, publicly available dataset, and this type of research did not need any extra ethical approval.
Author contributions
DX conceived the study and planned the study. GL and XL analyzed the data and wrote the paper. SJ, DG and QZ performed the literature search. XW and TL made the critical revision of the paper. All authors have contributed significantly to the manuscript to be published.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.



