Abstract
Esophageal melanosis is an uncommon condition characterized by melanin deposition in the esophageal mucosa. We report the case of a female in her late 40s with systemic lupus erythematosus and Sjögren's syndrome, in whom an incidental hyperpigmented lesion was identified in the mid-esophagus during endoscopy performed for iron-deficiency anemia. Histopathological examination confirmed melanocytosis without evidence of dysplasia or malignancy. Although its etiology remains unclear, esophageal melanosis has been linked to chronic mucosal inflammation or injury, which may be triggered by autoimmune alterations. Further studies are warranted to clarify this potential association.
Keywords
Introduction
Esophageal melanosis is a rare condition characterized by the presence of melanin pigmentation in the esophageal mucosa. This entity was first described in 1963 by de la Pava et al., 1 who documented the presence of melanoblasts in esophageal tissue samples. In a classic study from India, pigmented patches containing melanocytes were identified in 21 of 1000 consecutive routine esophagoscopies, corresponding to an incidence of 2.1%. 2
Histologically, it is characterized by melanocyte proliferation within the squamous epithelium and melanin deposition in the mucosa. Although its etiology and natural history remain poorly understood, it has classically been associated with esophageal injury processes, such as those seen in gastroesophageal reflux disease or chronic esophagitis. 3
Some authors have suggested a potential association between esophageal melanosis and the development of primary malignant neoplasms, including squamous cell carcinoma and primary esophageal melanoma, 4 highlighting the importance of accurate diagnosis and appropriate surveillance. Magnifying endoscopy may assist in differentiating melanosis from melanoma by revealing specific endoscopic features. 5
Case presentation
A female in her late 40s with a known history of systemic lupus erythematosus (SLE) and Sjögren's syndrome presented to the emergency department of Fundación Valle del Lili in November 2024 with a two-week history of left hemifacial edema, followed by the development of periorbital blistering lesions and left ocular ecchymosis.
On admission, the patient reported associated craniofacial and polyarticular pain. Physical examination revealed bilateral periorbital involvement; the left eye showed blistering lesions with mild erythema and warmth, predominantly at the lateral canthus. The right eye presented with milder periorbital edema, conjunctival injection, and linear hyperpigmented lesions, without impairment of visual acuity. No other relevant findings were identified. She was diagnosed with left preseptal cellulitis with involvement of the masticator and ipsilateral parotid spaces, originating from an odontogenic source due to chronic periodontal disease in teeth 28, 27, and 38. Management included dental extraction and empirical antimicrobial therapy with intravenous piperacillin–tazobactam (4.5 g every 6 h) with vancomycin (1 g every 12 h). After two days of treatment, the patient developed a cutaneous rash, leading to a modification of the regimen to linezolid 600 mg every 12 h. A total of 10 days of effective antimicrobial therapy was completed with satisfactory clinical improvement.
Upon admission, she had moderate anemia (hemoglobin: 10.4 g/dL), and during hospitalization her hemoglobin levels progressively declined to a nadir of 5.5 g/dL. Laboratory tests were consistent with iron-deficiency anemia, with normal vitamin B12 and folic acid levels (Table 1). As part of the anemia workup, an esophagogastroduodenoscopy was performed, which incidentally revealed a hyperpigmented area located 20 cm from the dental arch, involving 80% of the esophageal circumference and extending for 4 cm. Biopsies were obtained for histopathological evaluation (Figure 1).

Hyperpigmented esophageal lesion observed during esophagogastroduodenoscopy.
Baseline hematological and biochemical laboratory findings.
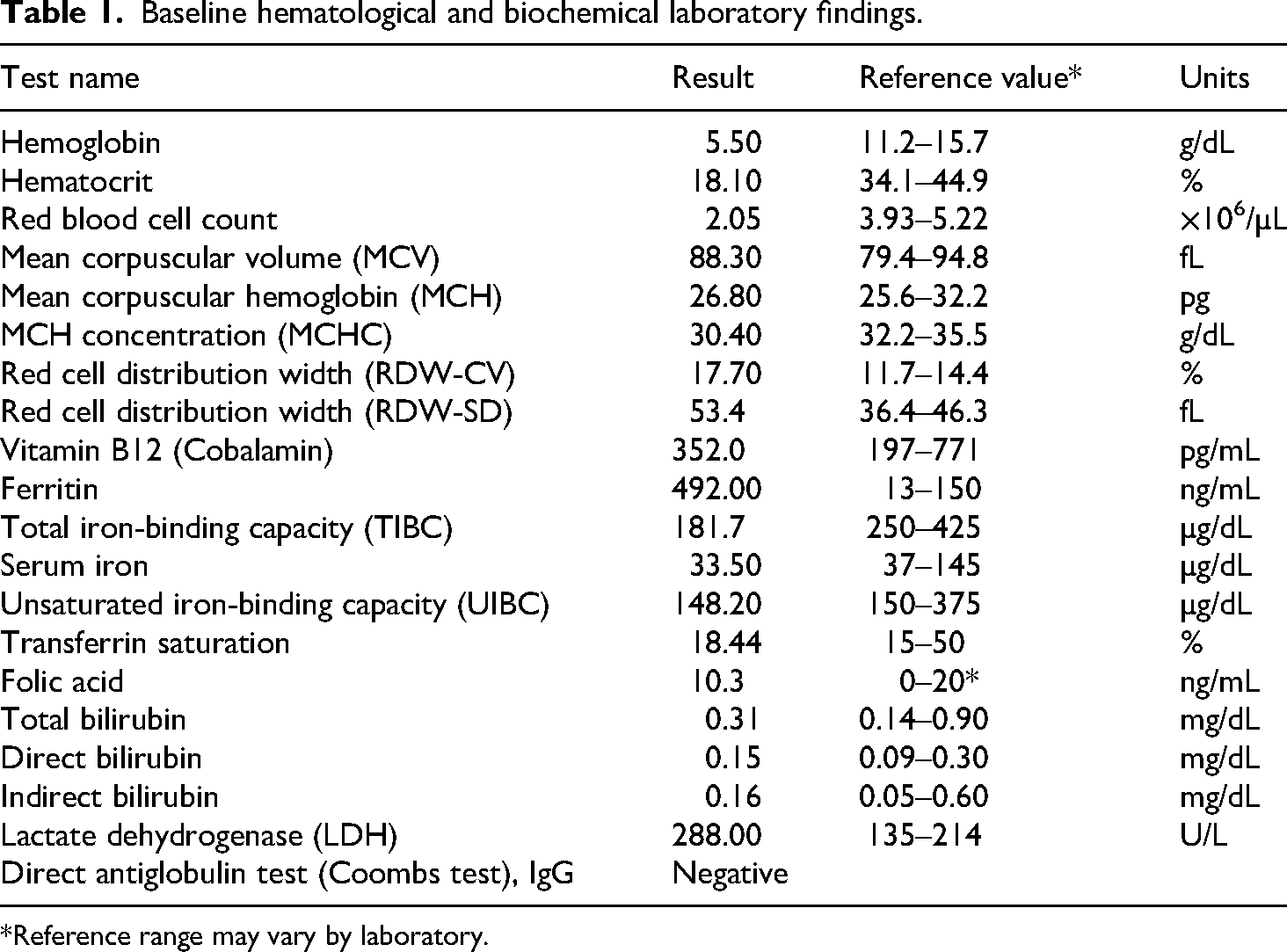
*Reference range may vary by laboratory.
A colonoscopy was also performed, and it was normal. The patient received treatment with erythropoietin (4000 IU administered weekly for four weeks) and intravenous iron sucrose (200 mg on days 1 and 5 of treatment). Additionally, bone marrow biopsy revealed erythroid hyperplasia with no evidence of malignancy.
Histological analysis of the esophageal biopsies showed basal layer hyperplasia, an increased number of dendritic melanocytes in the basal epithelium, and elevated melanin pigment in the mucosa. These melanocytes exhibited no dysplastic or atypical changes, and no features suggestive of malignancy were observed. Immunohistochemistry showed melanocyte immunoreactivity to SOX-10, Melan A, and HMB-45 (Figure 2), confirming the melanocytic nature of the pigmentation and findings consistent with esophageal melanocytosis.

Biopsy showing increased dendritic melanocytes and melanin pigment in the basal epithelial layer (A, hematoxylin and eosin, 4×, 10×, 40× magnifications). Melanocytes showing immunoreactivity for (B) SOX-10, (C) HMB-45, and (D) Melan A (immunohistochemistry, 4×, 10×, 40× magnifications).
When queried about her medical history, the patient denied alcohol or tobacco use but reported long-standing symptoms of dyspepsia, gastroesophageal reflux, and progressive dysphagia.
At the one-month outpatient follow-up, the patient showed marked improvement of the periorbital inflammatory lesions and a reduction in the associated craniofacial and joint pain, with no other relevant findings on physical examination. Follow-up laboratory tests demonstrated an increase in hemoglobin levels (10.7 g/dL). No endoscopic follow-up examinations have been performed to date. The reporting of this study conforms to CARE guidelines. 6
Discussion
We report a case of esophageal melanosis in a patient with significant comorbidities, including SLE and Sjögren's syndrome. While esophageal melanosis is generally considered a rare and benign condition, there is ongoing discussion about its potential role as a premalignant lesion, particularly in the development of primary esophageal cancer. 7
Its recognition is clinically relevant due to the need to differentiate it from other causes of esophageal pigmentation. Among benign entities, esophageal pseudomelanosis may mimic it endoscopically; however, it is distinguished histologically by the absence of true melanocytes and the presence of non-melanic pigments, such as hemosiderin or lipofuscin.2–4 Similarly, acute esophageal necrosis (“black esophagus”) may present with intensely dark mucosa, although it is typically associated with a critical clinical setting and is characterized by extensive tissue necrosis on histopathological examination.1,3,8
The most relevant differential diagnosis is with neoplastic melanocytic lesions, particularly primary malignant melanoma of the esophagus, an extremely rare but aggressive neoplasm that demonstrates invasion of the lamina propria, cytonuclear atypia, and high mitotic activity.7,9–11 Other less frequent entities that should be considered include benign melanocytic nevi, esophageal metastases from cutaneous melanoma, drug-induced pigmentation (such as that associated with minocycline), hemosiderin deposition secondary to chronic bleeding, and, rarely, esophageal squamous cell carcinoma with secondary pigmentation.5,8,12,13 In this context, clinic and endoscopic correlation, with detailed histological analysis and the use of immunohistochemistry (SOX-10, HMB-45, and Melan A), is essential to establish an accurate diagnosis and to exclude malignant pathology.3,4,7
There are currently no robust epidemiological data that allow accurate quantification of the risk of malignant transformation in esophageal melanosis, largely due to its very low prevalence and the limited number of reported cases. Nevertheless, available observational evidence suggests a potential association between esophageal melanosis and neoplastic conditions of the upper aerodigestive tract. In a cohort study of Japanese alcoholic men undergoing endoscopic screening, esophageal melanosis was significantly associated with iodine-unstained lesions, which are considered precursors of squamous neoplasia (odds ratio [OR] 2.81), as well as with esophageal squamous cell carcinoma (OR 6.54), compared with individuals without melanosis. 5 In addition, although exceedingly rare, isolated case reports have documented the progression of a benign melanotic esophageal lesion to primary malignant melanoma of the esophagus, with malignant transformation demonstrated through serial endoscopic follow-up.9,10
The reported male-to-female ratio is approximately 3:1, with a mean age of onset between 60 and 65 years. 11 Most cases involve the mid to lower third of the esophagus, as observed in this case. Although the exact pathogenesis remains unclear, two main theories have been proposed: aberrant migration of neural crest cells during embryogenesis and the differentiation of epithelial stem cells into melanoblasts in response to tissue injury. 12
A case series by Sharma et al. 2 demonstrated an association between esophageal melanosis and histological features of esophageal injury, such as basal cell proliferation, intercellular edema, and esophagitis in seven of 21 cases. Vincent Comraj et al. 14 also described this association. These histological features may support the hypothesis that basal stem cells differentiate into melanocytes as part of the initial response to mucosal injury.
Although no direct link between esophageal melanosis and autoimmune diseases such as SLE and Sjögren's syndrome has been established in the literature, these conditions are known to cause functional esophageal abnormalities, including dysmotility, dysphagia, and gastroesophageal reflux,15–17 in this context, chronic gastroesophageal reflux leads to mucosal injury and an inflammatory response characterized by increased pro-inflammatory cytokines and structural remodeling in the esophageal epithelium 18 and recent research has shown that impaired esophageal motility in patients with gastroesophageal reflux symptoms is correlated with increased and prolonged acid exposure and mucosal injury. 19 While the etiology of esophageal melanosis and other hyperpigmented esophageal lesions remains uncertain, some authors proposed that chronic mucosal lesions may contribute to reactive melanocytic proliferation and pigment deposition in the esophageal mucosa3,8,13; however, robust evidence to establish such causal associations between esophageal melanosis and autoimmune diseases is currently lacking in the literature.
Conclusion
Esophageal melanosis is an uncommon entity that has been highly associated with anemic processes in the literature, as observed in the present case. Additionally, several studies have suggested a potential association with malignancy. In suspected cases, a thorough diagnostic approach, including histopathological confirmation, is essential to rule out malignancy. Moreover, close follow-up of confirmed cases is warranted to allow early detection of histological changes suggestive of malignant transformation. As a limitation, this patient did not undergo endoscopic follow-up.
Although a direct association with autoimmune disorders has not been reported, the esophageal involvement observed in these diseases may represent a risk factor for the development of melanosis. Future studies should aim to clarify this potential association.
Footnotes
Acknowledgements
ChatGPT was used exclusively for language refinement during the translation of the manuscript and was not involved in the research design, data analysis, or interpretation.
Ethical considerations
This case report was reviewed and approved by the Institutional Biomedical Research Ethics Committee of Fundación Valle del Lili, Cali, Colombia (Approval No. 460; Act No. 17/2025), on 28 August 2025. The study was conducted in accordance with national and international regulations, including the Declaration of Helsinki.
Consent to participate
Written informed consent to participate was obtained from the patient. The study was reviewed and approved by the Institutional Biomedical Research Ethics Committee.
Consent for publication
Written informed consent for publication of clinical details and images was obtained from the patient.
Author contribution
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this case report are available from the corresponding author upon reasonable request by the journal.
