Abstract
Objective
Inflammatory markers, such as the neutrophil-to-lymphocyte Ratio (NLR), systemic immune-inflammation index (SII), systemic inflammation response index (SIRI), and panimmune-inflammatory value (PIV), have previously been identified as prognostic factors in head and neck cancer (HNC). Notably, time-dependent receiver operating characteristic (ROC) curves have rarely been used to assess the prognostic role of inflammatory markers in head and neck cancer. This study aimed to evaluate the significance of the NLR, SII, SIRI, and PIV in this context.
Methods
This retrospective cohort study was conducted at Far Eastern Memorial Hospital, a tertiary medical center between July 2022 and January 2025, and 153 patients with HNC were enrolled in this study. Inflammatory markers were compared by tumor status, nodal involvement, and stage. Time-dependent ROC analysis was used to determine the area under the curve (AUC) and optimal cutoff values for overall survival (OS) and disease-specific survival (DSS) with Cox regression.
Results
The NLR, SII, SIRI, and PIV were significantly associated with tumor status and stage (p < 0.05). The optimal cutoff values were 2.9, 630, 1.6, and 400, with corresponding time-dependent AUCs of 0.73, 0.71, 0.64, and 0.62, with corresponding sensitivities of 81.7%, 81.2%, 55.9%, and 60.8%, and specificities of 64.1%, 54.9%, 68.5%, and 60.4%., respectively. Higher NLR, SII, SIRI, and PIV values were associated with poor OS and DSS.
Conclusions
The NLR, SII, SIRI, and PIV are associated with tumor status and poor survival outcomes (OS and DSS) and may provide additional stratification and risk-based management in patients with HNC.
Keywords
Introduction
Head and neck cancer (HNC) is one of the most common types of cancer worldwide. 1 The disease's clinical behavior is closely linked to the systemic inflammatory response. 2 Changes in components of the complete blood count, such as neutrophils, lymphocytes, monocytes, and platelets, are important indicators of immune system responses that are influenced by various factors and play critical roles in systemic inflammation, injury, and stress. Inflammation is a key element of the tumor microenvironment (TME) and contributes to tumorigenesis. 3 Because these hematological parameters, owing to their simplicity and accessibility, serve as valuable prognostic markers in cancer. Inflammatory markers, such as the neutrophil-to-lymphocyte ratio (NLR), 4 systemic immune-inflammation index (SII), and systemic inflammation response index (SIRI),5,6 have previously been identified as prognostic factors in HNC.
The pretreatment panimmune-inflammatory value (PIV) has also been reported as a promising biomarker for predicting survival in patients with nasopharyngeal carcinoma 7 and in those with locally advanced head and neck squamous cell carcinoma treated with chemoradiotherapy (CRT). 8 Recent studies exploring the prognostic role of PIV in patients with recurrent or metastatic head and neck cancer treated with immune checkpoint inhibitors indicate that an elevated PIV is associated with poor survival outcomes. 9
However, data on the PIV in the general HNC population are still scarce. Receiver operating characteristic (ROC) curves are commonly used to evaluate the predictive accuracy of continuous biomarkers, as they provide estimates of sensitivity and specificity for each potential cutoff point in the biomarker distribution for a binary outcome. Because survival data are time dependent, and analyses should account for this. 10 Accordingly, time-dependent ROC analysis has been proposed as an approach to evaluate marker performance over time. 11
Notably, prior studies have not utilized time-dependent analysis to assess the role of these inflammatory biomarkers in HNC. The study aimed to evaluate the prognostic significance of the NLR, SII, SIRI, and PIV in patients with HNC.
Materials and methods
This retrospective cohort study was conducted at a tertiary medical center and approved by the Institutional Review Board of Far Eastern Memorial Hospital (IRB No. 113206-E; approval date: September 12, 2024). The requirement for informed consent was waived by the Research Ethics Review Committee because of the retrospective nature of the study and the use of anonymized data.
The study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki (1975), as revised in 2024. To ensure confidentiality and anonymity, all patient data were fully de-identified prior to analysis. The reporting of this study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 12
The enrolled patients met the following criteria: (1) had histologically confirmed HNC; (2) had tumors located mostly in the oral cavity, oropharynx, hypopharynx, larynx, or nasopharynx; (3) were older than 20 years; and (4) had no previous history of HNC.
Patients were excluded if they did not receive curative treatment or if histopathological confirmation of squamous cell carcinoma (SCC) was lacking. We also excluded patients with distant metastasis (M1) or those with a secondary primary cancer.
A total of 153 patients were included in this study between July 2022 and January 2025. All eligible patients who met the inclusion and exclusion criteria were consecutively enrolled during the study period. The medical records of all patients with HNC treated were reviewed.
We recorded body weight and height, smoking habits, ECOG performance status, 13 and other basic patient characteristics. The clinical stage of the tumor was determined according to the AJCC 8th edition TNM classification at diagnosis. 14
Blood samples for hematologic and inflammatory marker analyses were collected before initiation of treatment during hospital admission to minimize variability due to treatment effects. The analyses included red blood cell (RBC) count, hemoglobin level, and multiple systemic inflammatory markers, such as the NLR (neutrophil count (103/μL)/lymphocyte count (103/μL)), SII (platelet count (103/μL)*neutrophil count (103/μL))/lymphocyte count (103/μL)), SIRI (neutrophil count (103/μL)* monocyte count (103/μL))/lymphocyte count (103/μL)) and PIV (neutrophil count (103/μL)* platelet count (103/μL)*monocyte count (103/μL)/lymphocyte count (103/μL)).
Body height and weight were gathered from medical records and used to calculate body mass index (BMI, kg/m2). The following cutoff values were used in the analysis: underweight BMI < 18.5 kg/m2, normal weight BMI 18.5–24 kg/m2, and overweight/obese BMI >24 kg/m2. The Eastern Cooperative Oncology Group (ECOG) Performance Status Scale describes a patient's level of functioning in terms of their ability to care for themselves, daily activity, and physical ability.
Statistical analysis
We used time-dependent ROC analysis to calculate the area under the ROC curve at 550 days (approximately 18.3 months) and determine the optimal cutoff values for continuous variables based on OS.10,15 OS was defined as the time from diagnosis to death from any cause or the last follow-up visit, whereas disease-specific survival (DSS) was defined as the time from diagnosis to death due to HNC. Survival outcomes for categorical data were compared using Kaplan–Meier plots and tested with the log-rank test. The potential effects of clinical variables on OS and DSS were examined via univariate and multivariate Cox regression model. The hazard ratios (HRs) and 95% confidence intervals (95% CIs) were subsequently calculated according to Cox regression models. 16
The performance of the multivariate Cox regression models is evaluated using Harrell's C-index. 17 The proportional hazards (PH) assumption is assessed using Schoenfeld residuals to ensure the validity of the Cox regression models. Comparisons of continuous variables between two independent groups were performed using unpaired Student's t-tests. All the statistical tests performed in this study were two-sided, and p values less than 0.05 were regarded as statistically significant. All statistical analyses were performed using STATA version 14 (StataCorp, College Station, TX, USA). Time-dependent AUC analysis was conducted using the stroccurve package. 15
Results
From July 2022 to Jan 2025, a total of 153 patients with HNC were enrolled in this study, with a predominant male representation (86.3%) compared with females (13.7%). The patients’ characteristics are displayed in Table 1. The median age of the participants was 57 years (IQR = 14), with a slight majority (56.2%) aged 60 years or younger. In terms of cancer location, 51.6% of cases involved the oral cavity, whereas the remaining 48.4% involved other head and neck regions.
Baseline clinicopathological characteristics of the patients.

Abbreviations: BMI: body mass index; ECOG: Eastern Cooperative Oncology Group performance status; NLR: Neutrophil-to-lymphocyte ratio = Neutrophil count (103/μL) ÷ Lymphocyte count (103/μL); SII: Systemic Immune Inflammation (Platelet count (103/μL) × Neutrophil count(103/μL)) ÷ Lymphocyte count(103/μL); SIRI: Systemic Inflammation Response Index = (Neutrophil count(103/μL) × Monocyte count (103/μL)) ÷ Lymphocyte count (103/μL); PIV: Panimmune-inflammation value = PIV = (Neutrophil count (103/μL) × Platelet count (103/μL) × Monocyte count (103/μL)) ÷ Lymphocyte count (103/μL).
With respect to tumor stage, a majority of patients presented with early-stage tumors (T1 & T2: 58.8%), whereas 39.9% presented with advanced T3 or T4 stage tumors. Similarly, 50.3% of the participants had nodal involvement (N1 to N3), whereas 35.3% were node negative. When considering the overall clinical stage, more than half of the patients (57.5%) were classified as stage III or IV, indicating a predominance of advanced disease at diagnosis. Treatment modalities varied, with the majority (68%) receiving a combination of surgery, chemotherapy and radiotherapy (CRT). Smaller proportions of patients underwent surgery with only chemotherapy (9.8%), radiotherapy (5.2%), or surgery alone (13.1%). Nutritional status, as indicated by BMI, revealed that 49% of patients had a BMI ≥24, suggesting that a substantial number were overweight or obese, whereas only 7.2% were underweight.
The performance status of most participants was favorable, with 96.7% scoring ECOG 0 or 1, indicating that they were fully active or restricted only in performing physically strenuous activity. Hematologic parameters revealed a mean platelet count of 244.7 × 103/μL and average neutrophil and lymphocyte counts of 5.0 × 103/μL and 1.9 × 103/μL, respectively. Inflammatory indices, which have prognostic implications, were elevated in some cases: the NLR, SII and SIRI averaged 3.3, 811.9, and 1.5, respectively, and the PIV averaged 387.5, with wide ranges indicating interpatient variability.
Comparisons of various inflammatory markers according to the tumor status, nodal status and stage of HNC are displayed in Table 2. The comparison between early (T1&2) and advanced (T3&4) tumor stages revealed significantly higher levels of systemic inflammatory markers in patients with more advanced disease. Specifically, the NLR was markedly greater in T3&4 patients with stage T3 and T4 tumors (4.26 ± 3.36) than in patients with stage T1 and T2 tumors (2.66 ± 1.59), with a p value of 0.0001. Similarly, the SII, SIRI, and PIV were significantly greater in patients with advanced-stage tumors: SII (1076.15 ± 978.96 vs. 626.62 ± 430.23, p = 0.0002), SIRI (2.28 ± 2.16 vs. 1.03 ± 0.69, p < 0.0001), and PIV (589.51 ± 633.65 vs. 247.90 ± 186.52, p < 0.0001). These results indicate a strong association between tumor progression and elevated systemic inflammatory responses.
Comparisons of various inflammatory markers according to the tumor status (A), nodal status (B) and stage (C) of head and neck cancer.

Abbreviations: NLR: neutrophil-to-lymphocyte ratio; SII: systemic immune-inflammation index; SIRI: systemic inflammation response index; PIV: panimmune-inflammation value.
When patients without lymph node involvement (N0) were compared with those with nodal metastasis (N positive), no statistically significant differences in systemic inflammatory markers were detected. The mean NLR was similar between groups (3.30 ± 0.38 for N0 vs. 3.40 ± 0.30 for N-positive; p = 0.8429), as were the SII (763.40 ± 96.5 vs. 869.27 ± 92.72; p = 0.4414), SIRI (1.48 ± 0.28 vs. 1.63 ± 0.15; p = 0.6076), and PIV (358.54 ± 75.65 vs. 426.32 ± 48.32; p = 0.4302). These findings suggest that, unlike tumor size or T stage, nodal involvement may not be strongly associated with the systemic inflammatory response in this cohort.
A comparison between patients with early-stage (I&II) tumors and those with advanced-stage (III&IV) tumors revealed significantly greater levels of systemic inflammation in those with more advanced disease. Specifically, the NLR was significantly greater in stage III&IV patients (4.05 ± 4.11) than in stage I&II patients (2.66 ± 1.51; p = 0.0102). Similarly, the SII was significantly greater in advanced-stage patients (1113.35 ± 1744.55 vs. 608.32 ± 363.65; p = 0.0231). While the SIRI showed a notable increase in advanced stages (2.50 ± 5.99 vs. 1.04 ± 0.72), the result approached but did not reach statistical significance (p = 0.0517). The PIV was also greater in stage III&IV patients (802.98 ± 2979.76 vs. 242.74 ± 180.41), although this difference was not statistically significant (p = 0.1325). These results suggest a trend toward heightened systemic inflammation in more advanced cancer stages, with statistically significant differences observed for the NLR and the SII.
The median follow-up duration was 550 days (approximately 18.3 months). Time-dependent ROC curves for OS at 550 days (approximately 18.3 months) were constructed to assess the predictive ability of systemic inflammatory markers. The optimal cutoff points were 2.9, 630, 1.6 and 400 for the NLR, SII, SIRI and PIV, respectively, for OS with time-dependent ROC analysis (Figure 1). The AUC values for NLR (A), SII (B), SIRI (C), and PIV (D) were 0.73, 0.71, 0.64, and 0.62, with corresponding sensitivities of 81.7%, 81.2%, 55.9%, and 60.8%, and specificities of 64.1%, 54.9%, 68.5%, and 60.4%. We used these cutoff points for further analysis.

Time-dependent ROC curves for overall survival (OS). The AUCs were 0.73, 0.71, 0.64, and 0.62 for NLR (a), SII (b), SIRI (c), and PIV (d), respectively. The corresponding sensitivities were 81.7%, 81.2%, 55.9%, and 60.8%, and specificities were 64.1%, 54.9%, 68.5%, and 60.4%. Cutoff values were determined using the nearest point to (0,1) on the ROC curve follow-up.
Kaplan-Meier plot survival analysis was conducted to assess the prognostic significance of systemic inflammatory markers in relation to overall survival, using established cutoff values for each index. For the NLR with a cutoff of 2.9, patients with a high NLR (≥2.9) demonstrated significantly poorer overall survival than those with a low NLR (<2.9), suggesting that an elevated NLR was associated with a worse prognosis (Figure 2(a)). Similarly, when analyzing the SII (cutoff: 630), higher SII values were linked to reduced overall survival (Figure 2(b)), indicating the potential of the SII as a strong prognostic marker reflecting immune and inflammatory status.

Overall survival analysis with a Kaplan-Meier plot for the NLR (cutoff 2.9) (a), SII (630) (b), SIRI (1.6) (c) and PIV (400) (d).
In contrast, for the SIRI (cutoff: 1.6), patients with elevated values tended toward having decreased survival, although the separation between survival curves may have been less pronounced (Figure 2(c)), possibly due to greater variability or the influence of other confounding factors. Finally, PIV analysis (cutoff: 400) revealed that patients with higher PIV values had worse overall survival than those with lower PIV values did (Figure 2(d)), highlighting the cumulative prognostic impact of multiple immune cell parameters. These Kaplan-Meier plots collectively underscore the prognostic utility of inflammation-based indices, particularly the NLR and the SII, which clearly distinguish survival outcomes among patients.
Kaplan–Meier survival analysis was performed to evaluate the prognostic significance of systemic inflammatory markers in relation to disease-specific survival, using the same cutoff values applied for overall survival. Patients with high NLR (cutoff: 2.9) had significantly lower disease-specific survival than those with low NLR (Figure 3(a)). Similarly, higher SII values (cutoff: 630) were associated with poorer disease-specific survival (Figure 3(b)). For the SIRI (cutoff: 1.6), elevated values were also significantly associated with shorter survival (Figure 3(c)). Patients with high PIV (cutoff: 400) showed significantly lower disease-specific survival compared with those with low PIV (Figure 3(d)).
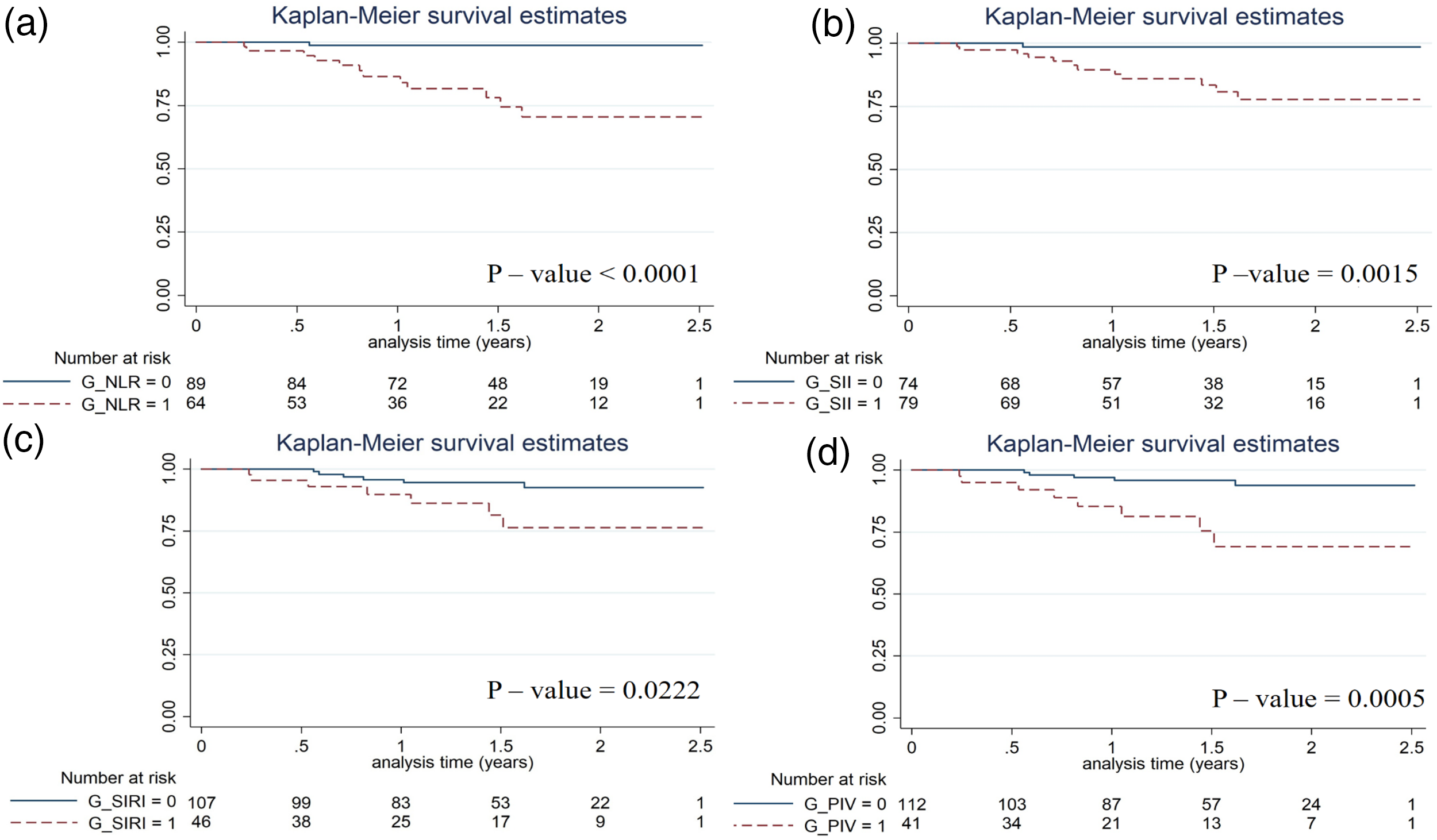
Disease-specific survival analysis with a Kaplan-Meier plot for the NLR (cutoff 2.9) (a), SII (630) (b), SIRI (1.6) (c) and PIV (400) (d).
The Cox regression results are displayed in Table 3. Univariate Cox regression analysis for OS and DSS revealed several significant prognostic factors among the clinical and inflammatory variables. Patients with advanced-stage tumors (T3&4) had significantly worse outcomes than those with early-stage disease (T1&2), with hazard ratio (HR) of 4.51 (p = 0.006) for OS and 4.99 (p = 0.006) for DSS. A low BMI (<18.5 kg/m2) was also a negative prognostic factor associated with a significantly increased risk of mortality for both OS (HR = 4.07, p = 0.047) and DSS (HR = 4.74, p = 0.033).
Univariable Cox regression analysis of potential factors associated with OS and DSS.

Abbreviations: BMI: body mass index; ECOG: Eastern Cooperative Oncology Group performance status; NLR: neutrophil–lymphocyte ratio; SII: systemic immune–inflammation index; SIRI: systemic inflammation response index; PIV: panimmune–inflammation value.
Among the systemic inflammatory markers in univariate Cox regression analysis, an elevated NLR (≥2.9) was strongly associated with poor prognosis, with an HR of 7.33 (95% CI: 2.06–26.03, p = 0.002) for OS and an even more pronounced HR of 21.74 (95% CI: 2.82–167.42, p = 0.003) for DSS. Similarly, a high SII (≥630) was significantly associated with poor outcomes (HR = 7.01, 95% CI: 1.58–31.10 for OS, p = 0.010; HR = 12.84, 95% CI: 1.67–98.81 for DSS, p = 0.014). The SIRI (≥1.6) approached significance for OS (HR = 2.51, 95% CI: 0.91–6.93, p = 0.076) and was significant for DSS (HR = 3.33, 95% CI: 1.12–9.92, p = 0.031). Finally, a PIV (≥400) was significantly associated with both OS (HR = 4.17, 95% CI: 1.51–11.54, p = 0.006) and DSS (HR = 5.77, 95% CI: 1.88–17.72, p = 0.002), indicating its potential prognostic value. These results suggest that tumor stage, nutritional status, and elevated inflammatory markers—particularly the NLR, SII, and PIV—are important predictors of survival in this population of patients with HNC.
To further confirm the independent prognostic value of these inflammatory markers, we perform multivariate Cox regression analyses. These models are adjusted for age, BMI, treatment modality, and T stage to account for potential confounding factors.
For OS, the HRs are 5.27 (95% CI: 1.34–20.74) for NLR, 5.10 (95% CI: 1.04–24.94) for SII, 1.61 (95% CI: 0.54–4.82) for SIRI, and 2.37 (95% CI: 0.80–7.05) for PIV.
For DSS, the HRs are 15.27 (95% CI: 1.82–128.0) for NLR, 8.95 (95% CI: 1.06–75.84) for SII, 2.08 (95% CI: 0.63–6.84) for SIRI, and 3.42 (95% CI: 1.02–11.47) for PIV. In addition, Harrell's C-index is calculated (Table 4).
Multivariate Cox regression analysis and Harrell’s C-index for OS and DSS models.

Abbreviations: PH assumption: proportional hazards assumption.
Multivariable analysis adjusted for age, BMI, treatments, and pathologic T stage.
The results show that elevated NLR (≥2.9), SII (≥630), SIRI (≥1.6), and PIV (≥400) demonstrate strong predictive performance, with the C-index values of 0.81, 0.79, 0.78, and 0.78 for OS and 0.85, 0.83, 0.82, and 0.83 for DSS, respectively.
Discussion
The present study demonstrated that several systemic inflammatory markers—including NLR, SII, SIRI, and PIV—were significantly associated with tumor status and survival in patients with HNC. Time-dependent ROC analysis further showed that NLR and SII possessed comparatively stronger discriminatory ability among these indices. As expected, advanced tumor stage (T3–4) was associated with poorer OS and DSS, consistent with established evidence that larger and more invasive tumors confer a worse prognosis. 18 Patients with more advanced disease exhibited higher levels of inflammatory markers, further supporting the link between tumor burden and systemic immune dysregulation. 5 19–21
Inflammation-based biomarkers have emerged as particularly potent predictors of survival. Elevated NLR (≥2.9), SII (≥630) and PIV (≥400) were significantly associated with worse outcomes for both OS and DSS, highlighting their potential utility as accessible, cost-effective markers for risk stratification. These indices reflect a heightened proinflammatory and immunosuppressive state, which may promote tumor progression and resistance to therapy.
To further evaluate the prognostic performance of these inflammatory markers, time-dependent ROC analysis was applied, as recommended in previous methodological work. 10 This analysis showed that NLR and SII had relatively better discrimination for 550-day survival compared with SIRI and PIV, and the resulting cutoffs were used in subsequent outcome analyses.
Nutritional status, reflected by low BMI (<18.5 kg/m2), was another independent adverse prognostic factor. Malnutrition is known to impair immune function, reduce treatment tolerance, and negatively affect clinical outcomes. 22
Interestingly, nodal status was not significantly associated with systemic inflammatory markers or survival, indicating that while nodal involvement is important, it may not be as closely linked to systemic inflammation as primary tumor burden is. This observation is consistent with a recent study by Cho et al., who reported no such correlation in patients with surgically treated oral cavity squamous cell carcinoma. 23
Limitations
Several study limitations must be acknowledged. First, although the overall sample size was reasonably sufficient, the study was conducted at a single institution, and the number of cases for each head and neck subsite (oral cavity, oropharynx, hypopharynx, larynx, and nasopharynx) was relatively limited, preventing further subgroup analysis. Second, patients with salivary gland carcinoma were not included in this study, because salivary gland cancers differ biologically from SCC. Systemic inflammatory markers have also been investigated in salivary gland malignancies. Previous studies have demonstrated that PLR values are higher in malignant parotid tumors than in benign lesions. 24 These observations suggest that inflammation-based indices might have prognostic relevance across a broader spectrum of head and neck neoplasms. Future studies could incorporate salivary gland cancers to validate these associations. Third, the retrospective nature of the study inherently carries risks of selection bias, incomplete data, and unmeasured confounding variables. Fourth, inflammatory markers were assessed at a single time point prior to treatment, and dynamic changes over time were not captured. Fifth, the optimal cut-off values for NLR (2.9), SII (630), SIRI (1.6), and PIV (400), determined using time-dependent ROC analysis within this cohort, have not been externally validated, which may limit their reproducibility and clinical applicability. Finally, the rate of HPV positivity in our cohort was low, which is consistent with national data showing that HPV-positive oropharyngeal cancers account for approximately 30% of cases, with even lower HPV prevalence observed in other head and neck subsites, 25 indicating that the findings may be less applicable to HPV-positive populations. Future prospective, multicenter studies with larger cohorts and longitudinal biomarker evaluations are warranted to validate and expand upon these findings.
In summary, the time-dependent ROC analysis demonstrated that NLR and SII provided comparatively superior discriminatory ability relative to the other inflammatory markers. When interpreted alongside clinical factors, these indices may further enhance the assessment of survival risk. Overall, integrating inflammatory markers with established prognostic factors may offer a more comprehensive approach to risk stratification in patients with HNC.
Conclusions
Inflammatory markers (NLR, SII, SIRI, and PIV) appear to be associated with tumor status and are related to poorer overall and disease-specific survival in patients with head and neck cancer. With further validation, these indices may be incorporated into prognostic assessment and considered for broader use in future clinical and research settings.
Footnotes
Acknowledgements
We gratefully acknowledge the support of our colleagues from the Head and Neck Multidisciplinary and Surveillance Team at Far Eastern Memorial Hospital, Taiwan.
Ethical statement
The study protocol was reviewed and approved by the Institutional Review Board of Far Eastern Memorial Hospital (IRB: 113206-E; approval date: September 12, 2024). Given the retrospective nature of the study and the use of anonymized data, the requirement for informed consent was waived by the IRB.
Author contributions
Ping-Yi Lin, Yu-Hsin Lin, and Ming-Hsun Wen contributed equally to this work. Ping-Yi Lin was responsible for study design, data collection, analysis, and manuscript drafting. Yu-Hsin Lin contributed to data acquisition, statistical analysis, and manuscript revision. Ming-Hsun Wen assisted with data interpretation and literature review. Ping-Chia Cheng, Po-Hsuan Wu, Wu-Chia Lo, and Chih-Ming Chang provided clinical data and critical revision of the manuscript. Li-Jen Liao supervised the study, validated the results, and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Far Eastern Memorial Hospital Research Program (FEMH-2024-C-023, PI20190002 and FEMH-2025-C-068).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in this published article. Further information is available from the corresponding author upon reasonable request.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this manuscript, the author(s) used ChatGPT (developed by OpenAI) to assist with language editing and improvement of readability. The tool was not used in any part of the research methods, data analysis, or result generation. All text generated by the AI tool was carefully reviewed, verified, and revised by the author(s), who take full responsibility for the integrity, accuracy, and originality of the final manuscript.
