Abstract
In elderly, hormones and oxidant-antioxidant interplay are suggested to mediate biochemical balance between adipose tissue to other tissues. Thus the study attempts to explore metabolic traits, plasma resistin, and oxidant-antioxidant markers in metabolic syndrome (MetS) in comparison to non-metabolic syndrome (non-MetS) elderly individuals. A total of 541 healthy elderly Caucasians, with no acute and/or chronic disorders were invited. After taking into account inclusion/exclusion criteria’s the MetS was defined as the presence of three out of five abnormal findings and allowed to divided groups into: non-metabolic syndrome, non-MetS (n = 25, median age 69.0 years), and newly diagnosed MetS (n = 29; median age 70.5 years) individuals. Glucose, plasma lipids, resistin (Res), thiobarbituric acid-reacting substances (TBARS), total antioxidant status (TAS), and Cu,Zn-superoxide dismutase (SOD-1) were measured. The MetS had higher resistin than non-MetS (p < 0.04). The linear correlation (all at p < 0.05) showed correlation for Res&triacylglycerols (R = 0.44), and for Res&diastolic blood pressure (R = −0.58) and for SOD-1&fasting glucose (R = −0.34) in MetS, while in the non-MetS group fasting glucose correlates with Res (R = 0.58) and with TAS (R = −0.43). The multiple regression analysis (alone and in combination) showed that independently from other factors resistin correlated positively with fasting glucose (β = 0.37; R = 0.58; R 2 = 0.23; p < 0.01) in all investigated elderly participants. In the MetS resistin correlated negatively with diastolic blood pressure (β = −0.68; R = 0.80; R 2 = 0.53; p = 0.0004) moreover in that group TAS correlated negatively with HDL-C (β = −0.71; R = 0.72; R 2 = 0.37; p = 0.01). While age correlated negatively with systolic blood pressure (β = −0.60; R = 0.62; R 2 = 0.14; p = 0.03) independently from other factors in the non-MetS group. Various metabolic factors contribute to maintain serum resistin and oxidant-antioxidant balance in the elderly people in the presence or absence of MetS. Resistin may serve as a predictor of MetS in the elderly, while strong antioxidant defense interactions in older individuals may indicate good health.
Aging is a process consist of clusters of increased prevalence of metabolic abnormalities and progressive decline in intrinsic physiological function. 1 Metabolic abnormalities include insulin-resistance, hyperglycemia, atherogenic dyslipidemia, and increased in visceral obesity.2–4 Together with age-related clinical conditions: hypertension and diabetes are elements of metabolic syndrome (MetS). Moreover, a number of additional characteristics have been suggested to be constituents of the MetS, containing: adiposity, oxidative stress, hepatic steatosis, inflammation, enhanced clotting factor activity, and endothelial dysfunction.5–8
Adiposity is associated with oxidative stress which is suggested that it may be a premature event in the pathology of MetS instead of the usual consequence or innocent bystander. 9 Furthermore, Holvoet et al. 10 noted that elderly patients with increased oxidative stress exhibit an accelerated risk of MetS. Yet, oxidative stress was associated with generally increased incidence of MetS 11 as well as its elements.12–14
Studies have demonstrated relationship between MetS and increased serum resistin. 15 Resistin, although is an adipokine, plays important roles in inflammation and may increase oxidative stress. 16 Moreover, similarly to leptin, another adipokine, may be potential biomarker for MetS and patient stratification. 17
Thus, the study attempts to explore metabolic traits, plasma resistin, and oxidant-antioxidant markers in MetS elderly individuals.
Materials and methods
The Bioethics Committees: of Medical University of Silesia in Katowice (statement number: KNW/0022/KB1/38/IV/16/17/18/19/20) and Poznan University of Medical Sciences in Poznan (statement number: 595/11) approved the study protocol; The study was performed in accordance with the Declaration of Helsinki of 1975 for Human Research revised in 2008. All participants signed consent, after they had been informed.
Inclusion criteria
A total of 541 individuals aged ≥60 years, healthy elderly Caucasians, with no clinically diagnosed acute and/or chronic disorders, drug-naïve, using neither special diet, nor supplements were invited to the study.
Exclusion criteria
Participants who were previously diagnosed cardio-vascular diseases, diabetes, malignant tumors, inflammatory disease, liver cirrhosis, and impaired renal function (eGFR p < 60 mL/min/1.73 m2) were excluded from the present study. Moreover current smokers and alcohol consumers were excluded from the study. In addition, elderly individuals were also excluded if they used to use or were currently on any drug treatment for: hypertriglicerydemia, low HDL cholesterol, antihyperglycemic agents, and/or using antihypertensive drugs or drugs with antioxidant capacity (including over the counter (OTC) vitamins and supplements)
Clinical assessment
Following recruitment, elderly individuals undergo a complete clinical assessment.
Arterial blood pressure
Arterial blood pressure was measured following the recommendations of the European Society of Hypertension. 18 The mean of three BP measurements was calculated and used in all analyses.
Body composition
Body weight and height were measured to the nearest 0.1 kg and 0.1 cm, respectively with the participants in a standing position, wearing light clothing and without shoes, and body mass index (BMI) was calculated as weight in kilograms divided by the square of the height in meters (kg/m2). Waist circumference (WC) was measured as the smallest girth at the level of the navel using a non-elastic tape (to the nearest 0.1 cm).
Percentage of body fat
Percentage of body fat (FAT) was measured by bio-electrical impedance analysis (BIA) using BodyStat equipment (Bodystat 1500, Bodystat Limited, Douglas, British Isles).
Blood sampling and biochemical analysis
Ultimately, out of 541 seniors, 62 persons were entitled to oral glucose tolerance test (OGTT) according to WHO recommendation. 19 Patients diagnosed with type 2 diabetes were excluded (n = 8).
Finally 54 individuals were investigated blood samples and fasting blood samples were collected into vacutainer tubes, in the morning after at least 12 h of fasting.
Glucose and lipids measurements
Glucose and plasma lipids were measured using enzymatic methods (bioMerieux, Marcy I’Etoile, France), and the UV-160A Shimadzu spectrophotometer (Shimadzu Corporation, Kyoto, Japan).
The glucose concentrations was obtained from OGTT at 0 min (G0′) and after 2 h (G120′).
RANDOX Assayed Human Multi-Sera Level 1 (as normal) and RANDOX Assayed Human Multi-Sera Level 2 (as pathological) (Randox, Crumlin, United Kingdom) were used for monitoring the accuracy of the determinations of fasting plasma lipids: total cholesterol (T-C), high density lipoproteins cholesterol (HDL-C), and triacylglycerols (TAG). Low density lipoproteins (LDL) was calculated using Friedewald formula.
Criteria for metabolic syndrome
The metabolic syndrome (MetS) was based on the IDF (International Diabetes Federation) and AHA/NHLBI (American Heart Association/National Heart, Lung, and Blood Institute) joint scientific statement criteria. 20 The presence of three out of five abnormal findings constituted a diagnosis of the MetS and allowed to divide groups into: MetS and non-MetS. Finally the newly diagnosed abnormalities were taken into account:
Abdominal obesity with waist circumference (WC) ≥94 cm in men and WC ≥80 cm in women.
Elevated triglycerides (TAG): ≥150 mg/dL (1.7 mmol/L).
Reduced HDL cholesterol (HDL-C): <40 mg/dL (1.03 mmol/L) in men and <50 mg/dL (1.3 mmol/L) in women.
Elevated blood pressure (BP): systolic BP ≥130 mmHg or diastolic BP ≥85 mmHg.
Elevated fasting glucose ≥100 mg/dL (5.6 mmol/L).
Resistin
Circulating level of plasma resistin was determined using an enzyme-linked immunosorbent assay (ELISA) kit (R&D, Minneapolis, USA) with the between sample replicate coefficients of variations (CVs) established as an intra-assay CV% of 4.9% and inter-assay CV% of 9.6%. All samples were analyzed according to the manufacturer’s instructions.
Oxidative stress markers measurement
Concentration of plasma total antioxidant status (TAS), activity of erythrocyte cytoplasmatic superoxide dismutase Cu-,Zn-SOD (EC: 1.15.1.1) (SOD-1) were determined by method described previously by Dzięgielewska-Gęsiak et al. 21 and thiobarbituric acid-reacting substances plasma (TBARS) was determined by Okhawa method. 22
The intra- and inter-series of coefficients of variation (CV) were respectively for TAS 2.50% and 4.80%, TBARS 2.80% and 4.70%, and SOD-1 1.60% and 2.70%.
Statistical analysis
Statistical analysis was calculated by Statistica (version 13.3) for Windows. The Shapiro-Wilk test was used to check the normality of distributions of variables in the 54 elderly individuals (all elderly) and metabolic syndrome and without metabolic syndrome elderly groups. Because most of the data had a non-normal distribution, data was shown as medians, lower, and upper quartiles and a nonparametric Mann-Whitney U test was used to assess the differences between the MetS and non-MetS groups. To evaluate the strength of association between the variables The Spearman rank correlation test was used. The strength of the relationship between variables was classified based on the value of the correlation coefficient as a slight R < 0.0; weak 0.1 ≤ R < 0.3; average 0.3 ≤ R < 0.5; high 0.5 ≤ R < 0.7; very high 0.7 ≤ R < 0.9; almost full 0.9 ≤ R < 1. The multiple regression analysis was performed in the all elderly individuals, as well as in the MetS and non-MetS elderly groups. The waist, SBP, DBP, HDL-C, TAG, and G0′ were taken as independent variables and resistin, TBARS, TAS, and SOD-1 and other metabolic parameters were taken as dependent variables (LDL-C was not included as a derivative of analyzed variables). A p-value <0.05 or lower was considered statistically significant.
Results
The investigated elderly MetS and non-MetS groups differ by definition in WC (p = 0.001), HDL-C (p < 0.05), BP (p = 0.003), and fasting glycemia (p < 0.0001) but TAG did not differ statistically significant (p > 0.05). Figure 1 shows the percentage distribution of metabolic syndrome traits in the elderly MetS and non-MetS groups.

The percentage distribution of metabolic syndrome traits in the elderly MetS and non-MetS group.
Table 1 summarize the clinical and biochemical data of the studied elderly groups. We noted no gender differences in MetS vs. non-MetS groups, except higher FAT in MetS females (p = 0.0003). Therefore, males and females were investigated in both groups together. Resistin concentration was higher in MetS vs. non-MetS (p < 0.04) group.
Clinical and biochemical characteristic of all investigated elderly individuals and with (MetS) or without (non-MetS) metabolic syndrome.

BMI: body mass index; DBP: diastolic blood pressure; FAT: fat tissue; G0′: fasting glucose concentration; G120′: glucose concentration after 120 min of oral glucose tolerance test; HDL-C: high density lipoproteins cholesterol; HGB: hemoglobin; LDL-C: low density lipoproteins cholesterol; MetS: metabolic syndrome group; non-MetS: individuals without metabolic syndrome; SBP: systolic blood pressure; SOD-1: Cu,Zn-superoxide dismutase; TAS: total antioxidant status; T-C: total cholesterol; TAG: triglicerydes; TBARS: thiobarbituric acid-reacting substances; n: number.
p is calculated comparing MetS with non-MetS.
The strength of association between the variables regarding metabolic syndrome traits and resistin and oxidant-antioxidant markers was performed in all elderly participants, those with MetS and non-MetS (Tables 2–4 respectively).
The linear correlation analysis regarding resistin, oxidant-antioxidant markers, and continuous variables in all elderly participants in relation to metabolic syndrome traits.

BMI: body mass index; DBP: diastolic blood pressure; FAT: fat tissue; G0′: fasting glucose; G120′: glucose after oral glucose tolerance test; HDL-C: high density lipoproteins cholesterol; LDL-C: low density lipoproteins cholesterol; SBP: systolic blood pressure; SOD-1: Cu,Zn-superoxide dismutase; TAG: triacylglycerols; TAS: total antioxidant status; TBARS: thiobarbituric acid-reacting substances; T-C: total cholesterol.
The correlations are calculated at p < 0.05, significant R are bold.
The linear correlation analysis regarding resistin, oxidant-antioxidant markers, and continuous variables in elderly participants with MetS in relation to metabolic syndrome traits.
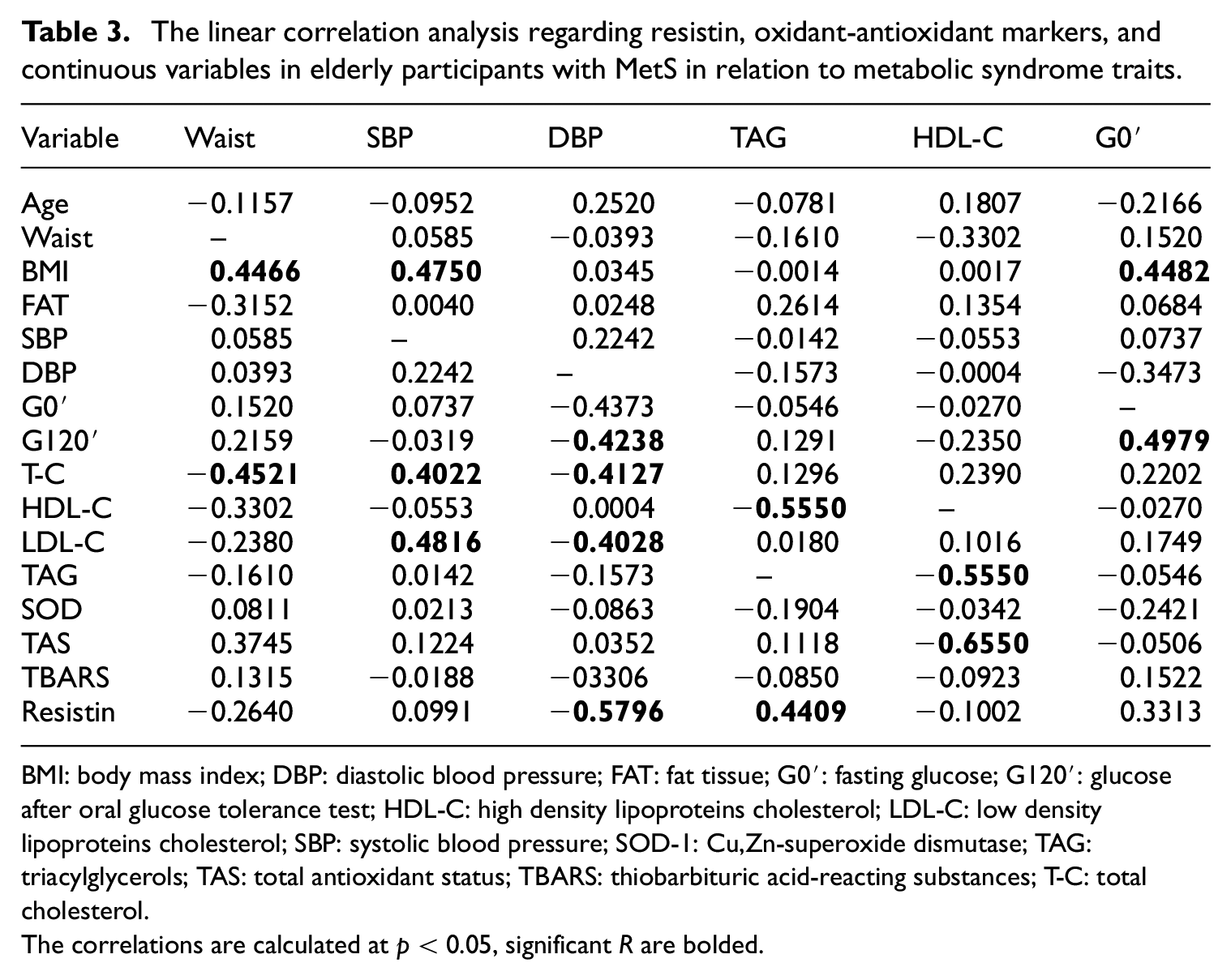
BMI: body mass index; DBP: diastolic blood pressure; FAT: fat tissue; G0′: fasting glucose; G120′: glucose after oral glucose tolerance test; HDL-C: high density lipoproteins cholesterol; LDL-C: low density lipoproteins cholesterol; SBP: systolic blood pressure; SOD-1: Cu,Zn-superoxide dismutase; TAG: triacylglycerols; TAS: total antioxidant status; TBARS: thiobarbituric acid-reacting substances; T-C: total cholesterol.
The correlations are calculated at p < 0.05, significant R are bolded.
The linear correlation analysis regarding resistin, oxidant-antioxidant markers, and continuous variables in elderly participants without MetS in relation to metabolic syndrome traits.
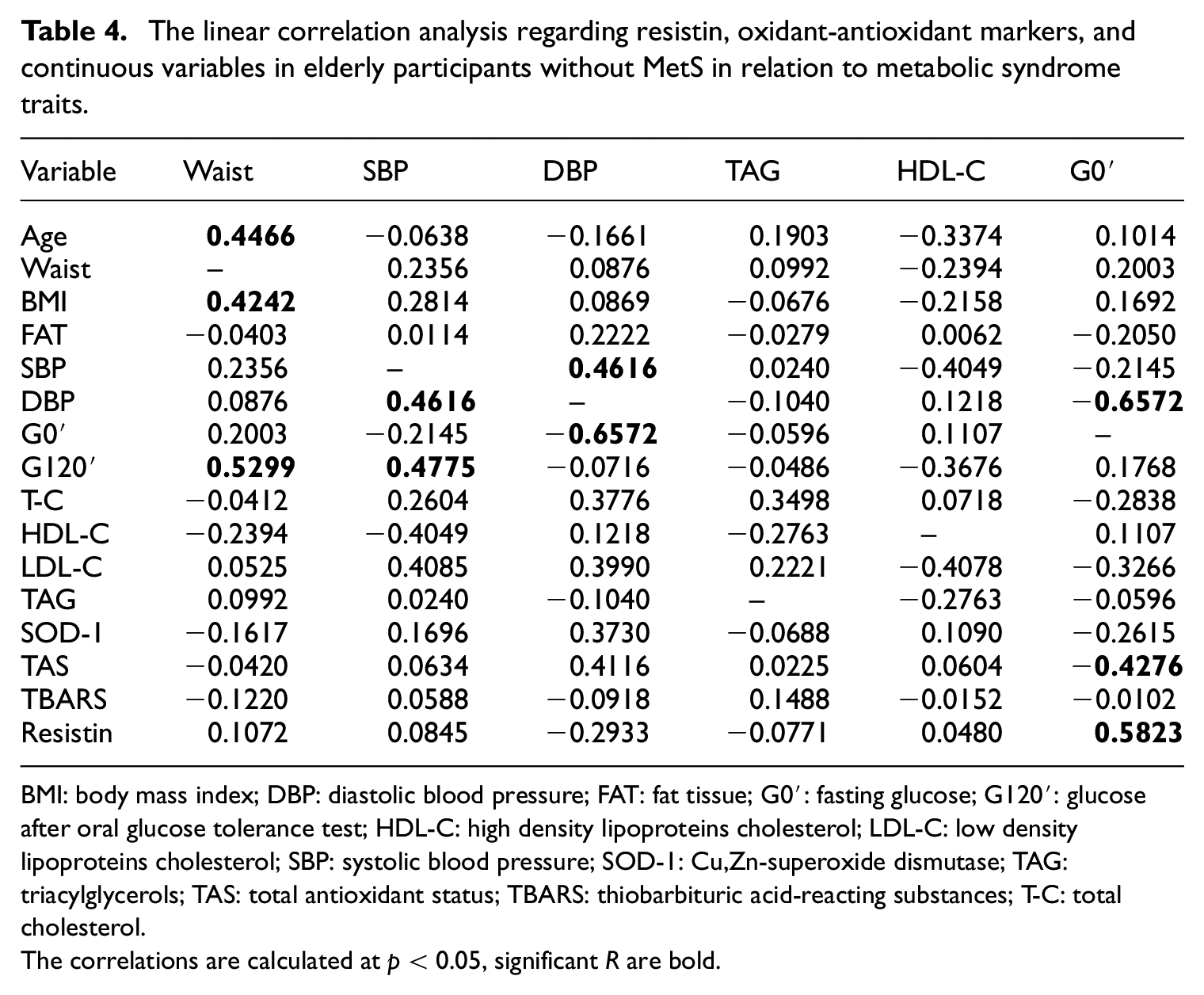
BMI: body mass index; DBP: diastolic blood pressure; FAT: fat tissue; G0′: fasting glucose; G120′: glucose after oral glucose tolerance test; HDL-C: high density lipoproteins cholesterol; LDL-C: low density lipoproteins cholesterol; SBP: systolic blood pressure; SOD-1: Cu,Zn-superoxide dismutase; TAG: triacylglycerols; TAS: total antioxidant status; TBARS: thiobarbituric acid-reacting substances; T-C: total cholesterol.
The correlations are calculated at p < 0.05, significant R are bold.
Moreover we checked correlation regarding resistin and TBARS, TAS, and SOD-1 in all investigated MetS and non-MetS elderly individuals.
In all elderly participants, in addition to the significant correlations (at p < 0.05) indicated in the Table 2, resistin correlated with G120′ (R = 0.3796). Regarding oxidant-antioxidant markers TBARS correlated with age (R = −0.3573; p < 0.05), and percentage of body fat (R = −0.5474; p < 0.05); SOD-1 correlated with LDL-C (R = 0.3368; p < 0.05) and with TAS (R = 0.5445; p < 0.05).
In the MetS elderly group, in addition to the significant correlations indicated in the Table 3, resistin correlated with G120′ (R = 0.4196; p < 0.05), T-C (R = 0.4589; p = 0.05). For the oxidant-antioxidant markers in the MetS elderly group we found correlation for TBARS and age (R = −0.5339; p < 0.05) and TBARS and percentage of body fat (R = −0.4794; p < 0.05).
In the case of healthy non-MetS elderly participants linear correlations (at p < 0.05) were observed for resistin and G0′ (R = 0.5823). In addition to the significant correlations indicated in the Table 4 the correlation was found for TBARS and percentage of body fat (R = −0.5855; p < 0.05) and TBARS and BMI (R = −0.6224; p < 0.05). SOD-1 correlated with T-C (R = 0.5284; p < 0.05), with LDL-C (R = 0.5784; p < 0.05) and with TAS (R = 0.8814; p < 0.05), moreover TAS correlated with T-C (R = 0.5216; p < 0.05) and with LDL-C (0.5479; p < 0.05.
The multiple regression analysis was performed to evaluate whether the emerged univariate correlations were independent of potential confounding factors (alone and in combination).
The multiple regression analysis revealed that in all investigated elderly participants resistin correlated with G0′ (β = 0.37; R = 0.58; R 2 = 0.23; p < 0.01) independently from other factors. In the group of MetS elderly individuals resistin correlated with DBP (β = −0.68; R = 0.80; R 2 = 0.53; p = 0.0004) and TAS correlated with HDL-C (β = −0.71; R = 0.72; R 2 = 0.37; p = 0.01) independently from other factors. And in the non-MetS elderly group, age correlated with SBP (β = −0.60; R = 0.62; R 2 = 0.14; p = 0.03) independently from other factors.
Discussion
The metabolic syndrome brings the most dangerous risk factors for cardiovascular disease. It is estimated that it affects about a quarter of the world’s adult population. People with the syndrome are five times more likely to develop type 2 diabetes (if it is not yet diagnosed), three-fold higher risk to have an acute coronary episode or infarction and two times more likely to die in comparison with those individuals without metabolic syndrome. 23
The results of the presented work are a voice in the discussion on the characteristic of older adults with and without metabolic syndrome in whom there appears no overt disease. In the terms of metabolic and oxidative stress complications we explored metabolic traits among elderly population without other clinical complication in the terms of the plasma resistin concentration and oxidant-antioxidant markers.
Resistin is an adipocyte-and monocyte-derived cytokine which modulates glucose and lipid levels, insulin function and metabolism homeostasis and may contribute to the inflammation and vascular dysfunction. 24 Moreover it has been proposed that adipocytokine, such as resistin, may be possible association between obesity and other metabolic syndrome constituents. 25 Additionally it has been proposed that resistin might put together metabolic components, proinflammatory, and prothrombic state and thus play a part in the development of type 2 diabetes mellitus (T2DM), metabolic syndrome, and cardio-vascular diseases (CVD). 26 Moreover Chen et al. 27 reported that raised resistin concentration significantly reduced an endothelial nitric oxide synthase expression and nitric oxide production through oxidative stress in cultured human coronary artery endothelial cells. Its serum concentration is increased in diabetes and cardio-vascular disease and other inflammation-related diseases. 28 In our elderly individuals we found that those with diagnosed metabolic syndrome had elevated plasma resistin.
The investigated elderly population were divided according the metabolic syndrome defined by joint scientific statement criteria. 20 At this stage of the assessment of the metabolic balance in the studied population, we can discuss the term “metabolically healthy obese,” proposed in the literature for people with excessive body weight, without hypertension, dyslipidemia, and hyperglycemia. 29 The high positive correlation of fasting glucose with resistin was observed in the investigated non-MetS elderly participants, whereas in those elderly with diagnosed MetS average positive correlation was found for resistin and lipids traits. While, Thoan et al. 30 found weak negative correlation for resistin and fasting glucose and resistin and triglycerides in overweight and type 2 diabetes patients.
In the animal model, it was found that resistin positively correlate with triglycerides in overfed mice. 31 This findings coincide with our human research and shows that increase in triglycerides might explain an increase in plasma resistin in elderly MetS patients. Moreover, in elderly both with and without metabolic syndrome participants we found negative association for percentage of body fat and oxidant marker – TBARS (average and high, respectively). Furthermore, Hernaez et al. 32 found that both advanced age and hyperglycemia are related with increased lipid oxidation. However, in our non-MetS elderly individuals there were additionally very high association between antioxidant markers (intra- and extracellular). It corroborate with the previous study when we found high association of TAS and SOD-1 in elderly population with high HDL-C. 33
The metabolic syndrome still raises a lot of controversy. Some scientists even question it’s real existence, claiming that the traits that make up it should be considered separately. 34 Furthermore, the resistin levels were a vital influence factor in the correlation with insulin resistance in diabetes and obese individuals in the presence of hyperresistinemia only. 35 Our finding from multiple regression analysis revealed independent positive correlation between resistin and DBP in MetS elderly, what suggests that DBP in 53% accompanies resistin in metabolic syndrome development. Furthermore in the MetS elderly group the changes in HDL-C in 37% carry through TAS variability in that group. Besides, Cardoso et al. 36 showed that resistin is included as the putative hallmarks of aging, due to its mediator role on adipose tissue activity, insulin-resistance, inflammation, mitochondrial dysfunction, and apoptosis. Moreover we suggested previously that raised oxidative stress may be consequence of old age while an insulin action identifies those at high risk for cardio-vascular problems, trough congruent balance of oxidant-antioxidant systems. 37 In that context now we imply serum resistin as a predictor of metabolic syndrome in elderly individuals free of other complications.
Limitation of the study
Whether or not resistin is linked with the metabolic syndrome in well-functioning elderly persons we confirm that there are some limitations of this study. The main criticism should be to definition of metabolic syndrome which is a mere aggregation of metabolic abnormalities. In that context, there is a risk that the effects observed may have been underestimated if a greater proportion of those who were diagnosed metabolic syndrome would have higher triglycerides. Furthermore we investigated only elderly with impaired fasting glucose, excluding those with diabetes.
Conclusions
An increased incidence and clustering of metabolic traits has been observed in elderly individuals. Various metabolic factors contribute to maintaining serum resistin and oxidant-antioxidant balance in the elderly people in the presence or absence of MetS. In this context, resistin may serve as a predictor of MetS in the elderly, while strong antioxidant defense interactions in older individuals may indicate good health. Discovering the pathophysiological roles of resistin in in the elderly metabolic syndrome patients may result in new pharmacotherapeutic approaches.
Footnotes
Author contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript and APC were supported by the Medical University of Silesia [grant number: KNW-1–072/K/9/K].
Ethics approval
Ethical approval for this study was obtained from: The Bioethics Committees: of Medical University of Silesia in Katowice (statement number: KNW/0022/KB1/38/IV/16/17/18/19/20) and Poznan University of Medical Sciences in Poznan (statement number: 595/11).
Informed consent
Written informed consent was obtained from all subjects before the study.
