Abstract
Objective
To translate and culturally adapt the facial disability index (FDI) into Korean following established guidelines, and to evaluate its reliability and validity in patients with facial palsy.
Methods
The translation process followed Beaton's cross-cultural adaptation methodology, involving forward translation, back translation, expert committee review, and prefinal testing. This prospective cross-sectional observational validation study included 100 adult patients with facial palsy recruited from a facial palsy center between May and July 2021. Reliability was assessed using Cronbach's α for internal consistency and intraclass correlation coefficient (ICC) for test–retest reliability (3-week interval). Construct validity was examined using Spearman's correlation between the Korean FDI and House–Brackmann (HB) grade, 36-Item Short Form Health Survey (SF-36), and Synkinesis Assessment Questionnaire.
Results
The Korean FDI demonstrated acceptable internal consistency (total Cronbach's α = 0.783; physical function = 0.810; social/well-being = 0.683). Test–retest reliability showed ICC values of 0.762 for physical function and 0.645 for social/well-being. The physical function subscale correlated moderately with HB grade (r = –0.461), whereas the social/well-being subscale correlated strongly with SF-36 mental component summary scores (r = 0.639). These findings were generally consistent with Turkish and Spanish validation studies, although ICC values were slightly lower, possibly due to the longer retest interval and inclusion of patients undergoing treatment.
Conclusions
The Korean version of the FDI is a reliable and valid patient-reported tool for assessing functional and psychosocial outcomes in patients with facial palsy. Its use may enhance patient-centered treatment planning, monitor treatment responses, and facilitate standardized data collection in clinical research. Future studies should examine its responsiveness in larger and more diverse patient populations and explore its application in clinical trials or real-world studies of integrative treatment strategies. Embedding the Korean FDI within multidisciplinary care frameworks may further strengthen evidence for patient-centered management of facial palsy.
Keywords
Introduction
The facial nerve plays an essential role in daily life, not only in fundamental functions such as mastication but also in speech and emotional expression. Peripheral facial nerve palsy is a lower motor neuron disorder of the facial nerve that can result from various causes, including infections, cholesteatoma, traumatic injury, malignancy, autoimmune diseases, or even pregnancy-related conditions. 1 Among these, acute idiopathic peripheral facial palsy, also known as Bell's palsy, has an incidence of approximately 20 per 100,000 persons per year and occurs most frequently between the ages of 15 and 40. 2 Standard treatments include corticosteroids and antiviral agents, 1 whereas therapeutic options such as acupuncture and physical therapy are also recommended. 3
Assessment of facial palsy severity is most commonly performed using the six-grade House–Brackmann (HB) scale. 4 Although this scale provides an objective measure of paralysis and recovery, it primarily reflects motor impairment and does not adequately capture the patient's subjective discomfort or the functional limitations they perceive in daily life. Similarly, general health-related quality of life instruments, which are often used in clinical research, are typically designed to evaluate gross motor function and activities of daily living. As such, they may fail to fully reflect the decline in quality of life specifically associated with facial dysfunction.
Facial palsy is a condition that has profound implications not only for objective recovery but also for patients’ subjective discomfort and overall quality of life. To accurately evaluate recovery, it is essential to use patient-reported instruments that capture the functional and psychosocial impact of facial dysfunction in detail. Several tools have been developed for this purpose, including the Derriford Appearance Scale (DAS59), 5 facial disability index (FDI), 6 and Synkinesis Assessment Questionnaire (SAQ). 7 Among these, the FDI, developed in 1996, 6 is particularly notable as it evaluates facial function across two domains—physical and social well-being—thereby assessing not only the physical consequences of facial nerve disorders but also their psychological and social impact. Because of these properties, the FDI has been widely used in clinical studies of facial palsy to assess treatment outcomes.8,9
Moreover, the FDI has been translated and culturally adapted into several languages, including Spanish and Italian,10–12 demonstrating its broad applicability. However, no culturally adapted and validated Korean version has been available to date. Therefore, in this study, we translated the FDI into Korean following Beaton et al.'s 13 cross-cultural adaptation guidelines and evaluated its reliability and validity for use in Korean patients with facial palsy.
Methods
Translation and expert committee
The overall translation process is illustrated in Supplemental Figure 1. Following internationally recommended guidelines, we established an expert committee for the translation of the instrument. The committee consisted of eight members: two forward translators, two backward translators, two medical professionals, one language expert, and one methodology expert.
Of the two forward translators, one (T1) was familiar with the instrument and had clinical experience, whereas the other (T2) was not familiar with the instrument. This approach was intended to reflect both clinical and lay perspectives in the translation process. The two backward translators were native English speakers with no prior exposure to the original FDI. The medical professionals contributed their clinical expertise, including patients with facial palsy; the language expert ensured grammatical accuracy and cultural appropriateness of expressions; and the methodology expert supervised the process to ensure methodological rigor.
Each forward translator independently produced a Korean version of the FDI and submitted a written report. T1 and T2 then discussed their translations and reached a consensus version, which was subsequently provided to the two backward translators. Each backward translator independently retranslated the Korean version into English and prepared a written report documenting their lexical and syntactic choices. After completion of the back translations, the expert committee convened a face-to-face meeting to review all versions and finalize the prefinal version.
Approval for the translation of the instrument was obtained from the original author and the publisher. The finalized Korean version of the FDI, developed through the translation process, is presented in the Supplemental File.
Clinical validation study
To evaluate the validity of the Korean version of the FDI, this prospective, cross-sectional observational validation study was conducted at the Facial Palsy Center of Kyung Hee University Hospital in Gangdong between May and July 2021. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 14 The study protocol was reviewed and approved by the Institutional Review Board of Kyung Hee University Hospital in Gangdong, Republic of Korea (KHNMCOH 2021-04-007) on 10 May 2021. Written informed consent was obtained from all participants. All patient data were de-identified prior to analysis, and no information that could directly or indirectly identify individual participants was included in this study. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki of 1975, as revised in 2024.
Eligible participants were adults (≥19 years) diagnosed with facial palsy who continued to experience neuromuscular dysfunction and reported at least one discomfort in either the SAQ or FDI domains. Exclusion criteria included inability to read, understand, or complete Korean-language questionnaires and cognitive disorientation. In this prospective observational study, patients who met the predefined eligibility criteria were consecutively enrolled during the study period until the target sample size of 100 participants was reached.
Facial palsy severity was assessed using the HB grading system, 4 a six-grade physician-rated scale evaluating forehead, periorbital, and perioral muscles, where higher grades indicate more severe paralysis. To reduce measurement bias, all assessments were performed by a Korean medical doctor with more than 4 years of residency training and at least 5 years of clinical experience in facial palsy.
Patient-reported outcomes included the FDI, 6 which consists of physical function and social/well-being subscales, with lower scores indicating greater functional impairment. Patients completed the validated Korean version of this questionnaire, for which permission for translation and use was obtained from the original authors and publisher. The 36-Item Short Form Health Survey (SF-36), 15 a widely used quality-of-life instrument with 36 items across eight domains and summarized into physical component summary and mental component summary (MCS) scores, was administered using its validated Korean version. 16 The SF-36 was used in accordance with RAND Corporation guidelines, which permit its use without formal permission. 17 The SAQ7, where higher scores indicate more severe synkinesis, was also employed, using its previously validated Korean version, with permission obtained from the developer of the original English version. 18
Reliability
Internal consistency was evaluated using Cronbach's α for the overall questionnaire and for each subscale (physical function and social/well-being). A Cronbach's α > 0.7 was considered acceptable, and >0.8 was regarded as good reliability. 19
To assess test–retest reliability, patients who met the eligibility criteria and had persistent sequelae of facial palsy for more than 1 year, with no expected substantial change in symptoms within 1 month, were asked to complete the questionnaire twice at a 3-week interval. Test–retest reliability was assessed using the intraclass correlation coefficient (ICC), based on the Bland–Altman method. 20
Construct validity
Convergent validity was examined using Spearman's rank correlation coefficient. We hypothesized that lower scores on the FDI physical function subscale would be correlated with higher HB grades and higher SAQ scores, whereas higher scores on the FDI social/well-being subscale would be correlated with higher SF-36 MCS scores.
Additionally, correlations between the FDI and the eight domains of the SF-36 (Physical Functioning, Role-Physical, Bodily Pain, General Health, Vitality, Social Functioning, Role-Emotional, and Mental Health) were explored. Correlation strength was defined as absent (<0.1), weak (0.1–0.3), moderate (0.3–0.5), or strong (≥0.5). 21
Sample size
Recommendations for validation studies typically rely on subject-to-item ratios or absolute minimums. A systematic review of sample sizes in validation studies for patient-reported outcomes found that 90% of studies included ≥100 subjects, with a median subject-to-item ratio of 11. 22 Based on these recommendations, we adopted a subject-to-item ratio of ≥5, with an absolute minimum of 100 participants. 23 Given that the FDI consists of 10 items, we set a target sample size of 100 and did not account for dropouts.
Statistical analysis
All data are presented as mean ± standard deviation or n (%), unless otherwise specified. Statistical significance was set at p < 0.05. To minimize bias, all analyses were conducted by an independent statistician. Demographic characteristics were compared using independent (unpaired) t-tests or chi-square tests, as appropriate. Quantitative variables were handled as continuous data in the correlation analyses. Because this was a single-time-point survey study, no imputation was performed for missing data. Instead, efforts were made to minimize missing responses by assisting participants in completing all items during data collection.
Results
Patient characteristics
The clinical characteristics of the 100 enrolled patients are presented in Supplemental Table 1. Of the total sample, 45% were male and 55% were female. Most patients (92%) were diagnosed with Bell's palsy. Sixty-four percent had disease duration longer than 1 year, whereas 36% had a duration between 3 months and 1 year.
Among the 31 patients who participated in the test–retest reliability study, clinical characteristics were generally comparable to the overall sample. However, the proportion of female patients and those with disease duration >1 year was slightly higher.
Reliability
The overall Cronbach's α for the Korean version of the FDI was 0.783 (95% confidence interval: 0.714–0.841), indicating acceptable internal consistency. The Cronbach's α was 0.810 for the physical function subscale and 0.683 for the social/well-being subscale (Table 1).
Facial disability index score at baseline and internal consistency.
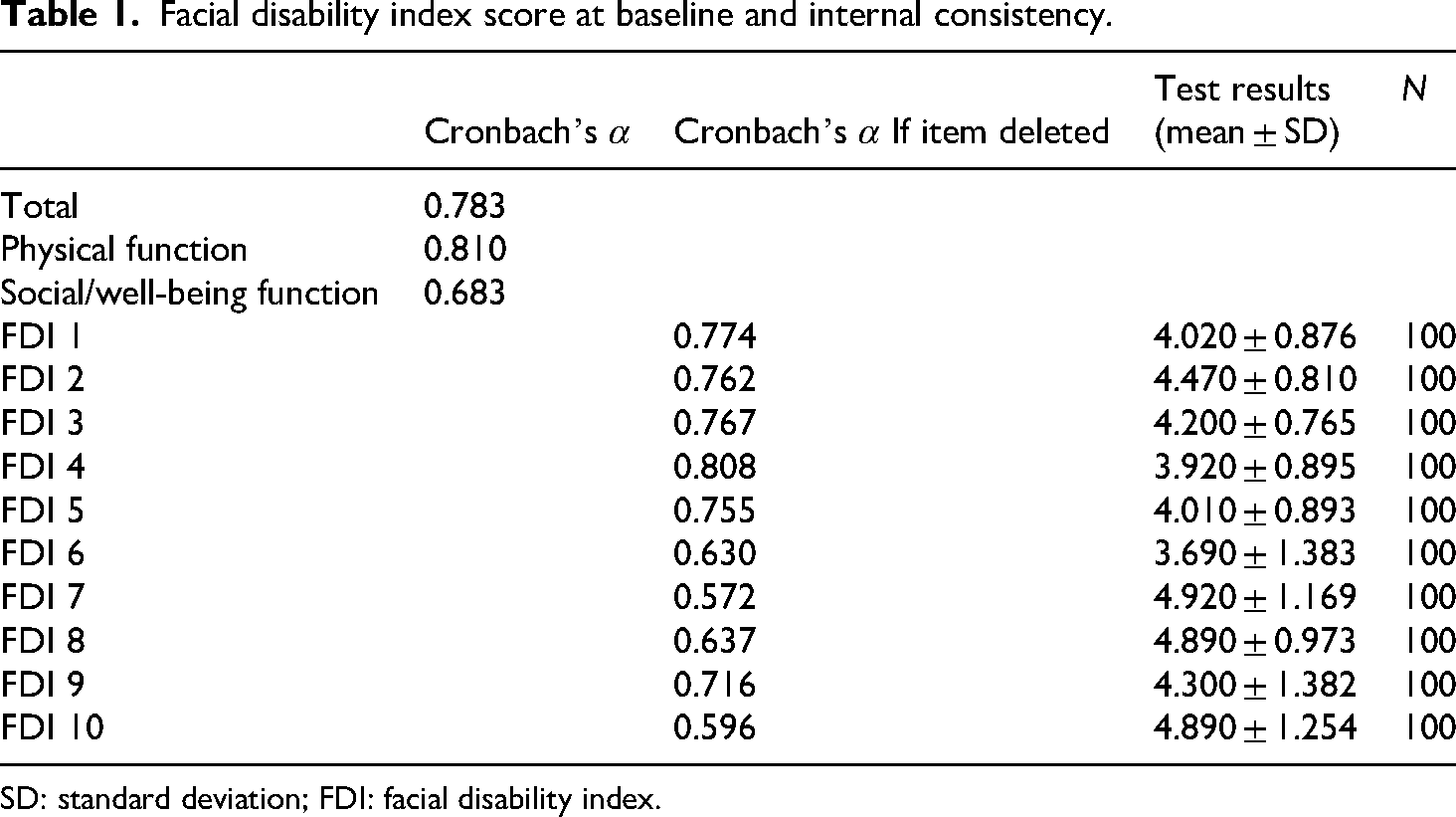
SD: standard deviation; FDI: facial disability index.
For test–retest reliability, the ICC was 0.762 for the physical function subscale and 0.645 for the social/well-being subscale (Table 2).
Individual test–retest reliability for the sub-scores and items of the facial disability index (n = 31).
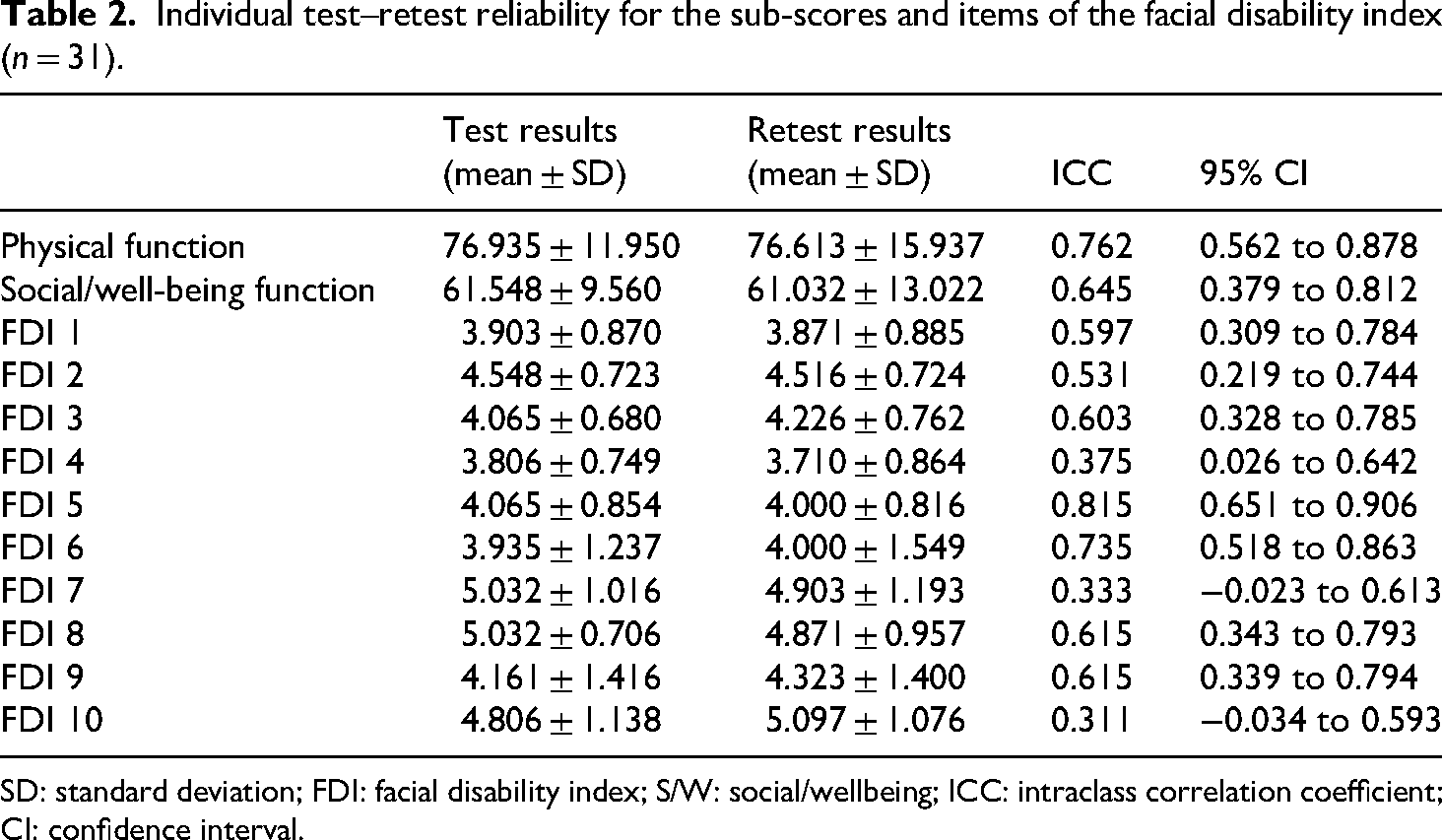
SD: standard deviation; FDI: facial disability index; S/W: social/wellbeing; ICC: intraclass correlation coefficient; CI: confidence interval.
Construct validity
The FDI physical function subscale showed a moderate negative correlation with the HB grade (Spearman's ρ = –0.461), whereas the FDI social/well-being subscale demonstrated a strong positive correlation with the SF-36 MCS score (ρ = 0.639) (Table 3).
Correlation between HB and SF-36 with FDI.

FDI: facial disability index; HB: House–Brackmann; SAQ: synkinesis assessment questionnaire; MCS: mental component summary; PCS: physical component summary; SF-36: 36-Item Short Form Health Survey.
*p < 0.05; **p < 0.01; r = Spearman correlation coefficient.
Discussion
In this study, we translated and culturally adapted the FDI into Korean and evaluated its reliability and validity in patients with facial palsy, predominantly those with an idiopathic etiology. The Korean version of the FDI demonstrated good internal consistency across both the physical function and social/well-being subscales (Cronbach's α) and acceptable test–retest reliability (ICC).
Our findings are comparable with previous validation studies in other languages. For example, the Turkish version reported Cronbach's α values of 0.82 (physical) and 0.63 (social/well-being), which are consistent with the trends observed in our study. 12 This discrepancy in internal consistency between the two subscales may be attributed to the heterogeneous nature of the items. While the physical function subscale focuses on direct and observable functional impairments, the social/well-being subscale encompasses more multifaceted psychosocial experiences. Specifically, Item 9, which assesses sleep quality, is sensitive to a broad range of external determinants—such as general psychological stress or lifestyle habits—that extend beyond the direct impact of facial dysfunction. Such inherent variability in psychosocial responses likely contributes to the lower Cronbach's α in the social domain compared to the more homogenous physical function items. Correlation patterns also differed slightly: while the Turkish version demonstrated a strong negative correlation between physical function and the HB grade (ρ = –0.837) and a weak positive correlation between social/well-being and SF-36 (ρ = 0.292), our study found a moderate negative correlation between physical function and HB grade (ρ = –0.461) and a strong positive correlation between social/well-being and SF-36 MCS (ρ = 0.639). These differences may be attributable to variations in patient characteristics and sample composition. Similarly, the Spanish version reported Cronbach's α values ranging from 0.77 to 0.82, which also align with our results. 10
The ICC values in our study were slightly lower than those reported in the Turkish version, 12 which may be explained by differences in the test–retest interval. While a 1- to 2-week period is commonly used, 24 we selected a 3-week interval to reduce recall bias and avoid artificially inflated reliability estimates. All participants included in the test–retest analysis had chronic, stable facial palsy with no expected substantial clinical change during the retest period. Although the longer interval may yield more conservative ICC values than shorter intervals, it provides a more realistic assessment of measurement stability under typical clinical conditions.
This study has several limitations. First, the assessment of construct validity was constrained by the limited availability of validated Korean-language instruments suitable for evaluating facial function and patient-reported experiences. A Korean version of the DAS59 25 exists; however, this instrument is primarily intended for patients with facial trauma, burns, or marked disfigurement and was therefore not appropriate for the study population. Other internationally recognized patient-reported outcome measures, such as the Facial Clinimetric Evaluation (FaCE) scale, 26 have demonstrated good psychometric properties but have not yet been translated or validated in Korean. Consequently, construct validity was examined using clinician-rated facial nerve grading systems rather than patient-reported instruments. Although both the HB grade and the Sunnybrook Facial Grading System 27 are clinician-based tools that may not fully capture patient-reported experiences, the HB grade was selected to enhance comparability with previous international validation studies of the FDI,10–12 as several non-English versions have employed the HB grading system as a reference measure. Second, most participants (92%) had idiopathic facial palsy, and patients with trauma, tumor, or other secondary causes were underrepresented, limiting generalizability. Third, the sample size was relatively small, which may restrict the robustness of subgroup analyses.
Nevertheless, our study has important strengths. It represents the first cultural adaptation and validation of the FDI in Korean patients with facial palsy, providing a standardized patient-reported outcome measure that complements existing clinician-rated tools such as the HB grade. By capturing subjective perceptions of both functional and psychosocial impact, the Korean FDI enables more patient-centered assessment in both clinical practice and research settings. As a standardized instrument, it also enhances comparability across studies and may contribute to improving the quality and consistency of facial palsy research in Korea.
Future research should examine the performance of the Korean FDI in longitudinal and interventional studies, including clinical trials and real-world studies of integrative treatment strategies. From an integrative medicine perspective, comprehensive patient management requires not only symptom-focused treatment but also coordinated multidisciplinary collaboration across different medical fields. 28 Incorporating validated Patient Reported Outcome Measures such as the FDI into multidisciplinary frameworks may support more holistic, patient-centered care and strengthen the evidence base for facial palsy management. Additionally, validation of other internationally recognized facial-specific instruments, such as the FaCE scale, would provide complementary perspectives and further enhance the comparability of Korean facial palsy research.
Conclusion
In conclusion, our findings suggest that the Korean version of the FDI is a reliable and valid patient-reported instrument for assessing functional and psychosocial status in patients with facial palsy. The study demonstrated acceptable internal consistency and stable test–retest reliability in this clinical sample. As a standardized measure that complements clinician-rated tools, the Korean FDI may support more patient-centered assessment in both clinical practice and research settings. However, because this was a cross-sectional observational study with a limited sample size, the generalizability of these findings may be restricted. Future longitudinal studies in larger and more diverse populations are needed to further evaluate the instrument's responsiveness and clinical utility. Overall, the Korean FDI may contribute to a more standardized assessment of facial palsy outcomes in Korea.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261428981 - Supplemental material for Cross-cultural adaptation and validation of the Korean version of the facial disability index in patients with facial palsy
Supplemental material, sj-docx-1-sci-10.1177_00368504261428981 for Cross-cultural adaptation and validation of the Korean version of the facial disability index in patients with facial palsy by Sang-Soo Nam, Jee Young Lee, Bonhyuk Goo, Jung-Hyun Kim, Seung-Min Lee, Mi-Riong Kim, In-Hyuk Ha and Yoon Jae Lee in Science Progress
Supplemental Material
sj-pdf-2-sci-10.1177_00368504261428981 - Supplemental material for Cross-cultural adaptation and validation of the Korean version of the facial disability index in patients with facial palsy
Supplemental material, sj-pdf-2-sci-10.1177_00368504261428981 for Cross-cultural adaptation and validation of the Korean version of the facial disability index in patients with facial palsy by Sang-Soo Nam, Jee Young Lee, Bonhyuk Goo, Jung-Hyun Kim, Seung-Min Lee, Mi-Riong Kim, In-Hyuk Ha and Yoon Jae Lee in Science Progress
Supplemental Material
sj-docx-3-sci-10.1177_00368504261428981 - Supplemental material for Cross-cultural adaptation and validation of the Korean version of the facial disability index in patients with facial palsy
Supplemental material, sj-docx-3-sci-10.1177_00368504261428981 for Cross-cultural adaptation and validation of the Korean version of the facial disability index in patients with facial palsy by Sang-Soo Nam, Jee Young Lee, Bonhyuk Goo, Jung-Hyun Kim, Seung-Min Lee, Mi-Riong Kim, In-Hyuk Ha and Yoon Jae Lee in Science Progress
Footnotes
Ackowledgments
Not applicable.
Author contributions
Conceptualization: Sang-Soo Nam. Methodology: Jee Yoon Lee, In-Hyuk Ha, and Yoon Jae Lee. Investigation: Jee Young Lee, Bonhyuk Goo, Jung-Hyun Kim, Seung-Min Lee, and Mi-Riong Kim. Data Curation: Jee Young Lee, Bonhyuk Goo, and Jung-Hyun Kim. Translation and Cultural Adaptation: Seung-Min Lee and Mi-Riong Kim. Formal Analysis: Yoon Jae Lee. Writing—original draft: Yoon Jae Lee. Writing—review & editing: In-Hyuk Ha and Sang-Soo Nam. Supervision: Sang-Soo Nam. Project Administration: In-Hyuk Ha. All authors have read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: RS-2021-KH109727).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request, subject to institutional and ethical regulations.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
