Abstract
Objectives:
Pulmonary Embolism has been frequently reported in Acute Exacerbations of Chronic Obstructive Pulmonary Disease (AE-COPD). The study aimed to determine whether COPD patients who receive anticoagulant (AC) therapy have a reduced risk of hospitalization due to AE-COPD and death.
Methods:
This nationwide population-based study was based on data from the Danish Register of COPD (DrCOPD), which contains complete data on COPD outpatients between 1st January 2010 and 31st December 2018. National registers were used to obtain information regarding comorbidities and vital status. Propensity-score matching and Cox proportional hazards models were used to assess AE-COPD and death after one year.
Results:
The study cohort consisted of 58,067 patients with COPD. Of these, 5194 patients were on AC therapy. The population was matched 1:1 based on clinical confounders and AC therapy, resulting in two groups of 5180 patients. We found no association between AC therapy and AE-COPD or all-cause mortality in the propensity-score matched population (HR 1.03, 95% CI 0.96-1.10, p = 0.37). These findings were confirmed in a competing risk analysis. In the sensitivity analysis, we performed an adjusted analysis of the complete cohort and found a slightly increased risk of AE-COPD or death in patients treated with AC therapy. This study found a low incidence of pulmonary embolisms and deep venous thrombosis in both groups.
Conclusions:
AC therapy was not associated with the risk of hospitalization due to AE-COPD or all-cause mortality.
Keywords
Introduction
Chronic Obstructive Pulmonary Disease (COPD) is the third leading cause of death in the world. 1 Patients with COPD can experience recurrent acute exacerbations of COPD (AE-COPD), which are defined as clinical symptoms e.g. dyspnoea, increased sputum, and coughing combined with positive biomarkers e.g. increased C-reactive protein, eosinophilia, or neutrophilia. 2 The causes of AE-COPD are heterogeneous 3 and the condition is associated with impaired health status and a worsened prognosis.4,5 In addition to microbiological agents, pulmonary embolisms (PE) can be an important factor in the etiology of AE-COPD. According to a systematic review and meta-analysis of 7 studies and a total of 880 patients, PE was found in 16.1% of unexplained AE-COPD (95% confidence interval (CI), 8.3%−25.8%; p < .0001). The prevalence of deep venous thrombosis (DVT) in undescribed AE-COPD was 10.5% (95% CI, 4.3%–19.0%; p < .0001). 6 A recent study found a lower, but still substantial risk of 5.9% for PE during AE-COPD admission. 7
In general, patients with COPD and especially AE-COPD have been demonstrated to have a higher platelet activation. 8 A community study evaluating data from 14,654 participants between 1987 and 1989, found that COPD was associated with a greater risk of venous thromboembolism (VTE). 9 These findings were confirmed later in a cohort study, which found that COPD patients had a 1.6-fold higher risk of VTE compared to the general population. Additionally, VTE was a strong predictor of all-cause mortality, especially in patients with severe COPD. 10 In summary, patients with AE-COPD have an increased risk of VTE, but the possible beneficial effect of anticoagulant (AC) therapy to patients with COPD is unresolved. Therefore, the study aimed to evaluate whether pre-existing AC therapy may reduce the risk of hospitalization due to AE-COPD and death by all causes in COPD patients.
Methods
Study design
This is a national cohort study of COPD outpatients in Denmark.
Data sources
We used data from nationwide administrative registers. The following registers were used:
The Danish Register of Chronic Obstructive Pulmonary Disease (DrCOPD) contains individual and quality-assured data on age, gender, Forced Expiratory Volume after 1 s (FEV1), Body Mass Index (BMI), Medical Research Council Dyspnoea Scale (MRC), smoking status, hospitalization due to AE-COPD during the past year, prescriptions on inhaled corticosteroids, and all out-patient visits and hospital admissions due to AE-COPD since 1st January 2010. The Danish National Patient Register contains information on all Danish in-hospital and out-patient clinic contacts since 1977. We assessed data on patients’ comorbidities within 10 years prior to study entry: heart failure, ischaemic heart disease, atrial fibrillation, pulmonary embolism, deep venous thrombosis, hypertension, diabetes mellitus type 1, diabetes mellitus type 2, renal failure, asthma, cerebrovascular disease, and malignant disease. Furthermore, data on hospitalization due to AE-COPD within one year after study entry was obtained from the register. Definitions of the diagnoses are presented in Table 1. The Danish National Database of Reimbursed Prescriptions was used to assess data on AC therapy, acetylsalicylate, and beta-blocker treatment (ATC-codes in Table 2) during one year prior to study entry. This register contains information on all reimbursed drugs sold in community pharmacies and hospital-based outpatient pharmacies in Denmark since 2004. AC therapy was defined as at least one prescription during the one year prior to study entry. According to current Danish guidelines, patients with the following conditions indicate AC therapy: mechanical heart valve, moderate to severe mitral stenosis, severe renal failure, antiphospholipid syndrome, atrial fibrillation or flutter, venous thromboembolism, or intracavitary thrombosis.
11
Betablocker treatment was defined as at least two prescriptions during the one year prior to study entry. The Cause of Death register contains data on the cause, date, location of death, and post-mortem data on Danes since 1970. We assessed the date of death of the patients included in this study.
ICD10 classification of diseases.

ATC codes.

Population
We included all patients with COPD above 40 years of age, who were registered with an out-patient-clinic visit from the 1st January 2010 to 31st December 2018 in DrCOPD. Study entry was defined as the date of the first out-patient-clinic visit in DrCOPD. Patient baseline characteristics were assessed at study entry. Due to a strong association between malignant neoplasm, mortality, and hospitalizations, we excluded all patients with malignant neoplasms (except other skin cancers than malignant melanoma) (Table 1) within five years before study entry. Patients diagnosed with malignant neoplasms more than five years before study entry remained in the study cohort since we expected their mortality and hospitalizations rates to be similar to the background population. The flowchart of inclusion is presented in Figure 1.

Flowchart of inclusion of the study cohort.
Outcome measures
The primary outcome was a combined endpoint of hospitalization due to AE-COPD or all-cause mortality. Secondary outcomes were all-cause mortality, AE-COPD admission, and the number of PE and DVT. All endpoints were evaluated after one year from baseline. Patients were censored at the first event.
Ethics
Danish Data Protection Agency approved this study (journal no. VD-2018-264, with I-Suite no. 6504). Informed consent and ethical approval from the patients are not required for registry-based studies according to current Danish legislation. The data from the different registries have been compiled by using encrypted unique personal identification numbers. 12
Statistical methods
In case of missing BMI, FEV1, or MRC at any visit, the last observation was carried forward. If values were missing from all visits, the patients received the median value.
A subpopulation was formed using a greedy-match propensity score. 13 Patients in pre-existing AC therapy were matched in a 1:1 ratio with patients without AC therapy. The patients were matched on characteristics at study entry; age, gender (male/female), BMI, smoking status (current/not-current smokers), FEV1, inhaled corticosteroids (yes/no), previous DVT (yes/no), previous PE (yes/no), heart failure (yes/no), ischemic heart disease (yes/no) and Metoprolol prescription (yes/no). AC therapy for patients with atrial fibrillation is decided upon the CHA2DC2-VASc score, which is based on the patient's characteristics such as age, gender, and cardiovascular comorbidities. 14 Thus we were not able to match the population completely since patients with atrial fibrillation on AC therapy are not comparable to patients with atrial fibrillation without AC therapy. Additionally, we calculated a modified Charlson Comorbidity Index (CCI) score based on patients’ comorbidities; Apoplexia cerebri, dementia, peptic ulcer, diabetes mellitus, renal failure, liver failure (mild or severe), peripheral vascular disease, hemiplegia, and connective tissue diseases. Regarding AIDS/HIV, which typically are included in the CCI, we decided not to include the diagnoses because they are not considered to decrease life expectancy if treated.
The primary endpoint was evaluated in a univariable analysis on the propensity-score matched subpopulation. We tested whether there is an interaction between AC therapy and 1) smoking, 2) atrial fibrillation, or 3) congestive heart failure towards the primary outcome. We report no interaction between AC therapy and smoking, nor between AC therapy and atrial fibrillation. We found a positive interaction in the heart failure interaction analysis, p = 0.03. Therefore the primary analysis was stratified for congestive heart failure. Furthermore, a sensitivity analysis was performed to evaluate the endpoint in a multivariable analysis, including the whole cohort. A competing risk model was performed to evaluate the secondary endpoint of death and AE-COPD after 1 year. The number of DVT and PE was evaluated by Fischer's Exact Test
Cox proportional hazard regression models are presented as hazard ratios (HRs) with 95% CI. Statistical analyses were performed using SAS statistical software 9.4. Plots were customized by the NewSurv macro. 15
Results
Between 1st January 2010 and 31st December 2018, 58,067 individual COPD outpatients were identified in DrCOPD, of these 43 patients emigrated within the first year from baseline and therefore only followed until the date of emigration. In the cohort, we identified 5194 patients who claimed at least one prescription for AC therapy one year prior to study entry. The majority of the patients’ (79.5%) first anticoagulant prescription was vitamin-K-antagonist, and fewer patients (20.5%) claimed non-vitamin K antagonist oral anticoagulants as their first AC therapy prescription. The baseline characteristics of the study population are presented in Table 3. The groups differed in every variable except malignant diagnoses and treatment with inhalation medication prior to matching. After propensity-scores matching the two groups were fairly comparable except for the number of patients with atrial fibrillation (70.7% on AC therapy vs. 29.3% not receiving AC therapy). The propensity-score matched population included a total of 10,360 outpatients, and the patient characteristics are presented in Table 4.
Baseline characteristics of the total study population.
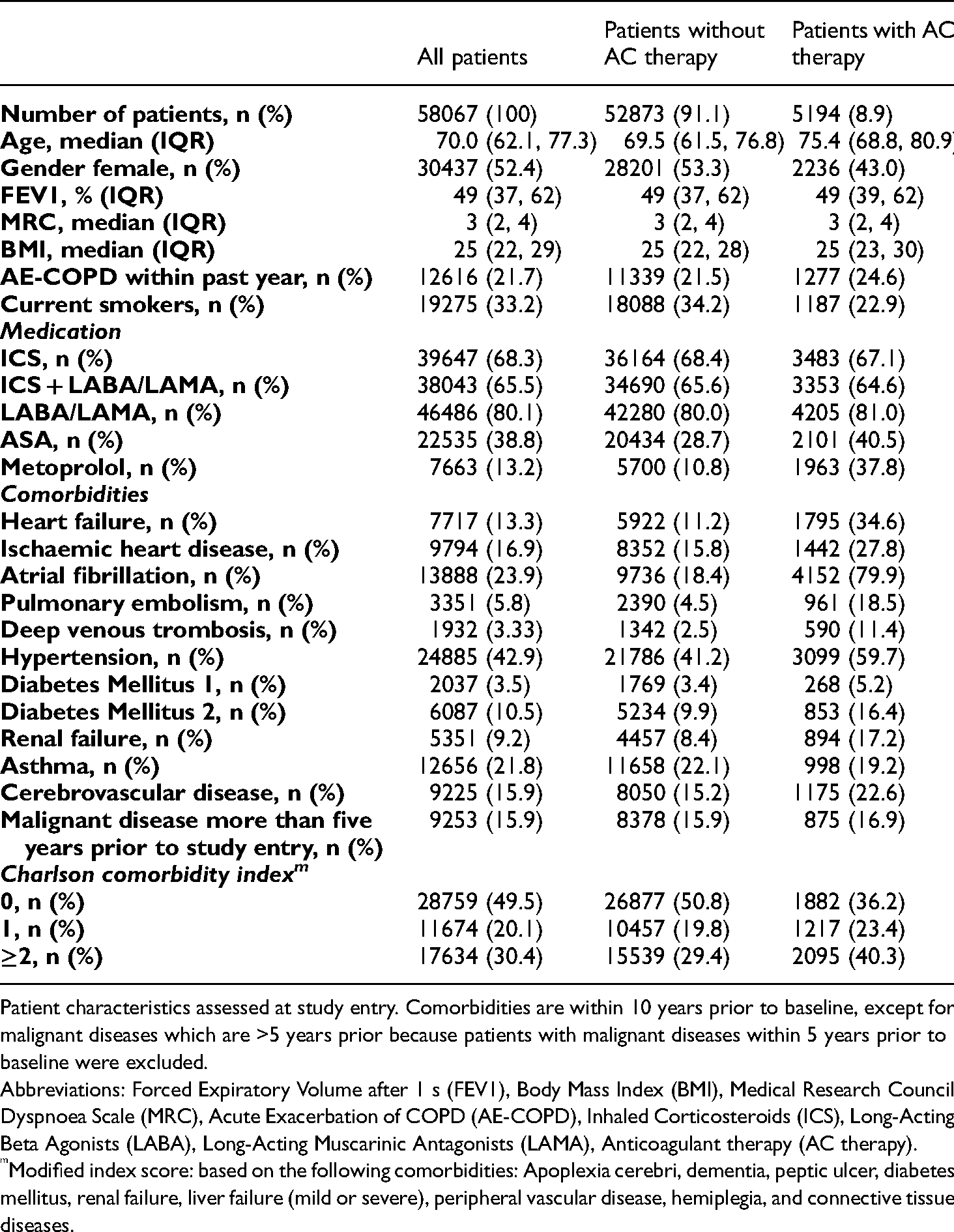
Patient characteristics assessed at study entry. Comorbidities are within 10 years prior to baseline, except for malignant diseases which are >5 years prior because patients with malignant diseases within 5 years prior to baseline were excluded.
Abbreviations: Forced Expiratory Volume after 1 s (FEV1), Body Mass Index (BMI), Medical Research Council Dyspnoea Scale (MRC), Acute Exacerbation of COPD (AE-COPD), Inhaled Corticosteroids (ICS), Long-Acting Beta Agonists (LABA), Long-Acting Muscarinic Antagonists (LAMA), Anticoagulant therapy (AC therapy).
Modified index score: based on the following comorbidities: Apoplexia cerebri, dementia, peptic ulcer, diabetes mellitus, renal failure, liver failure (mild or severe), peripheral vascular disease, hemiplegia, and connective tissue diseases.
Baseline characteristics of the propensity-score matched study population.

Patient characteristics assessed at study entry. Comorbidities are within 10 years prior to baseline, except for malignant diseases which are >5 years prior because patients with malignant diseases within 5 years prior to baseline were excluded.
Abbreviations: Forced Expiratory Volume after 1 s (FEV1), Body Mass Index (BMI), Medical Research Council Dyspnoea Scale (MRC), Acute Exacerbation of COPD (AE-COPD), Inhaled Corticosteroids (ICS), Long-Acting Beta Agonists (LABA), Long-Acting Muscarinic Antagonists (LAMA), Anticoagulant therapy (AC therapy).
Modified index score: based on the following comorbidities: apoplexy, dementia, peptic ulcer, diabetes mellitus, renal failure, liver failure (mild or severe), peripheral vascular disease, hemiplegia, and connective tissue diseases.
Outcomes
The number of deaths and AE-COPD hospitalizations in the total cohort and the propensity-matched population after one year of follow-up is presented in Table 5.
Mortality and AE-COPD hospitalization in the study cohort.

Abbreviations: Anticoagulant (AC), Acute Exacerbations of COPD (AE-COPD).
We found no association between the risk of AE-COPD hospitalization or all-cause mortality and AC therapy in the propensity-score matched population in the primary analysis, which was adjusted for CCI score [modified] (HR 1.03, 95% CI 0.96-1.10, p = 0.3555) Figure 2. When stratifying for congestive heart failure, the risk was also unaltered for patients with heart failure on AC therapy (HR 0.90, 95% CI 0.81-1.00, p = 0.0514). For patients without congestive heart failure on AC therapy, we found an increased risk of AE-COPD or all-cause mortality after 365 days (HR 1.15, 95% CI 1.05-1.26, p = 0.0023).
A competing risk analysis of the propensity-score matched population applying all-cause mortality as a competing risk to AE-COPD hospitalization showed no difference in risk of AE-COPD between the two groups (HR 0.96, 95% CI 0.87–1.05, p = 0.3520), and also no difference in mortality after 365 days (HR 1.02, 95% CI 0.91–1.16, p = 0.70).
In the propensity-matched population, the number of patients registered with PE within 365 days in the group without AC therapy was 147 patients (2,8%) and 105 patients (2.0%) in the group with AC therapy (p = 0.01). The number of patients registered with DVT within 365 days in the group without AC therapy was 65 patients (1.3%) and 60 patients (1.2%) in the group with AC therapy (p = 0.72). Regarding simultaneous PE and DVT, 12 patients without AC therapy and 6 patients with AC therapy were registered with both diagnoses simultaneously.

Time (days) to hospitalization due to AE-COPD or all-cause mortality in patients with or without AC therapy in the propensity-score matched population.
Sensitivity analyses
Multivariable analysis of the total population of 58,067 patients adjusted for age, gender, smoking status, FEV1, BMI, use of inhaled corticosteroids, heart failure, ischaemic heart disease, DVT, and PE showed an increased risk of hospitalization due to AE-COPD or all-cause mortality within 365 days in the group of patients receiving AC therapy compared to the patients without AC therapy (HR 1.10, 95% CI 1.04–1.16, p = 0.0005).
Discussion
This is the first study to investigate if pre-existing AC therapy could be related to improved prognosis in a large cohort of patients with COPD. We found, that AC therapy in this population of severe COPD outpatients did not reduce the risk of hospitalization due to AE-COPD or all-cause mortality within 365 days in the primary analysis of the propensity-score matched subpopulation. A secondary analysis was performed to estimate the separate outcomes of hospitalization due to AE-COPD after one year using a competing risk model, in which all-cause mortality was accounted for as a competing risk. The signal remained unchanged. In the sensitivity analysis, an adjusted analysis of the total cohort, we found a slightly increased risk of hospitalization due to AE-COPD or all-cause mortality in patients with AC therapy. Stratified for congestive heart failure, the risk associated with AC therapy for all-cause mortality or AE-COPD was unaltered for patients with heart failure but slightly increased for patients without heart failure. Further, we found no difference in the frequency of venous thromboembolisms within one year from baseline.
In a small study of patients with atrial fibrillation, a subgroup analysis of patients with concomitant COPD indicated that treatment with oral AC was associated with a decreased risk of death. 16 Explanations for the discrepancy between these results and the present study results include reverse bias by indication (patients with atrial fibrillation not receiving AC therapy, will most often not be following guidelines) and type I error in the other study, and further that we chose not to stratify for atrial fibrillation to avoid reverse bias by indication. Atrial fibrillation is very common among patients with COPD, and in our study, we found a higher prevalence than described in the literature 17 or the same, 18 which is not unreasonable when considering the severity of COPD in our population. Atrial fibrillation is an independent predictor of death in patients with COPD.19–22 Thus, our neutral results may be explained by a “neutralizing effect” i.e. benefits from AC therapy and attributable risk to atrial fibrillation, since nearly all patients in AC therapy had atrial fibrillation.
Among patients with heart failure, we did not observe any association between AC therapy and AE-COPD or all-cause mortality. These findings are consistent with earlier studies evaluating the effect of AC therapy on mortality in patients with heart failure.23,24 The small increased risk of AE-COPD or all-cause mortality among patients without heart failure in the present study could probably be due to underlying factors.
VTE during COPD exacerbation is considered to be associated with a worse prognosis. 25 Unfortunately, both stable COPD and AE-COPD are associated with a hypercoagulable state.26,27 Mechanisms to explain the association between COPD and change in coagulability primarily include systemic chronic inflammation (increases of C-reactive protein, fibrinogen, interleukins 6 and 8, and tumor necrosis factor α), and secondarily hypoxia-related platelet activation and oxidative stress. 28 Circulating active coagulation factors (e.g. Factor XI and tissue factor) have also been found in patients with COPD. 29 In our study, VTE was very infrequently present as an event in the national health register, and the frequency of DVT 30 and PE7,31–34 in our study is much lower than other findings, which indicates that the register may not be sensitive to this outcome. Perhaps due to a clinical underdiagnosing, since PE can occur in COPD patients with acute respiratory symptoms even without clinical suspicion. 7
This nationwide study had a large sample size since DrCOPD contains data on all COPD outpatient visits between 1st January 2010 and 31st December 2018, therefore chance findings are unlikely. We partly overcame information bias by using the Danish national registers to obtain data on patient characteristics (e.g. age, FEV1, smoking, and BMI), comorbidities, and outcomes. The registers are known to have a high degree of completeness and are regularly validated, and thus lead-time bias is unlikely. Second, we had a complete follow-up, since all admissions and deaths are entered into the Danish national registers in real-time. Despite these strengths, some limitations deserve careful consideration: There may be some compliance bias, thus we defined AC therapy as at least one prescription during the one year prior to study entry, but we are not able to know if the patients adhered to the therapy. Therefore we might underestimate the possible beneficial effect of AC therapy. Furthermore, there may be some diagnostic bias as the control and case group are not completely identical. Lastly, due to the observational study design, it is impossible to account for unknown confounders fully.
In conclusion, AC therapy did not appear to be beneficial or, in a clinically meaningful way, harmful to patients with severe COPD. However, even with the large sample size and complete follow-up, the registered incidence of VTE was low and all events may not have been registered. Our findings need confirmation in other settings and other geographical regions.
Contribution
AK, JE, and JSJ contributed to the conception and design of the study, data collection and analysis, data interpretation, and writing the manuscript. PS, RS, and TSI contributed to the conception and design of the study, data interpretation, and revising the manuscript. KB contributed to the data collection and revising the manuscript. All authors have approved the final manuscript and agreed to be accountable for all aspects of the work.
Footnotes
Acknowledgments
We thank Barbara Bonnesen Bertelsen, MD, cand.scient.biol for graphical support.
Data availability
Data are not publicly available but can be obtained through appropriate approval. According to Danish legislation regarding sharing of population data, source data collected for this study cannot be made available for others. However, Danish citizens who have a legitimate reason can apply for access to the data via the Danish National Health Authority.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
