Abstract
Background
Early changes in platelet count may reflect disease mechanisms affecting prognosis in critically ill patients, but their role in atrial fibrillation (AF) remains unclear.
Methods
This retrospective study analyzed intensive care unit (ICU) patients with AF from the Medical Information Mart for Intensive Care IV database. Platelet count within the first 36 h of ICU admission was assessed using latent growth mixture modeling to identify temporal trajectories. Associations between these trajectories and 28-day all-cause mortality were examined using Cox proportional hazards regression, Kaplan–Meier survival analysis, and subgroup analyses.
Results
Compared with the reference group, Class 2 showed a significantly higher risk of 28-day mortality in fully adjusted models (p = 0.013), whereas Class 3 showed no significant difference (p = 0.163). Kaplan–Meier analysis further confirmed reduced survival in Class 2 (log-rank p < 0.001). Stratified subgroup analysis indicated that the mortality association was more evident among female patients and those with a history of hypertension or heart failure.
Conclusion
In ICU patients with AF, early platelet count trajectories are independently associated with 28-day mortality. A downward trend in platelet levels signals a high-risk population, suggesting the potential utility of platelet dynamics in early prognostic assessment and risk stratification.
Introduction
Atrial fibrillation (AF) is the most prevalent form of sustained cardiac arrhythmia, marked by chaotic electrical impulses within the atria that result in irregular ventricular responses. Common triggers include hypertension, diabetes, systemic inflammation, and inherited genetic factors, which often act synergistically to initiate and perpetuate the arrhythmia.1–3 In recent years, both the prevalence and mortality of AF have been rising. Age is the most significant risk factor, with mortality rates among individuals over 65 nearly doubling in the past two decades. Rates are particularly higher in women, especially white women. 4
Platelets play a critical role in AF-related thromboembolic events. AF leads to blood stasis in the left atrial appendage, which is a major contributor to thrombus formation, as more than 90% of pathological thrombi originate there. This blood stasis constitutes part of Virchow's triad (blood stasis, endothelial injury, and hypercoagulability), which together form the pathological basis of the prothrombotic state in patients with AF. 5 One study found that serum levels of platelet glycoprotein Ib alpha (GP-Ibα) were downregulated in patients with recurrent AF. Since genetic variants of GP-Ibα are associated with stroke, this finding suggests that GP-Ibα may represent a potential link between AF recurrence and an increased risk of stroke. 6 In addition, inflammatory markers such as C-reactive protein can directly interact with platelet activation and aggregation. 5
Platelet count trajectory, a longitudinal biomarker that captures temporal changes in platelet levels rather than a single time-point measurement, has recently emerged as a promising indicator of sepsis, thrombopoietic function.7–9 To date, platelet trajectory analysis has been utilized across diverse clinical populations, including patients with sepsis, malignancies, and individuals undergoing major surgical procedures.10,11 Considering the well-recognized link between platelet activation, thrombus formation, and complications related to AF, it is reasonable to hypothesize that distinct platelet trajectory patterns may correlate with adverse clinical outcomes in this patient group.12,13 In these clinical contexts, specific platelet trajectory patterns—such as persistently low or declining counts—have been independently associated with adverse outcomes. Although previous studies have examined the association between single-point platelet counts and prognosis in patients with AF, these static measurements often fail to capture the dynamic physiological changes that reflect the evolving thrombo-inflammatory milieu in AF. As a result, single-time platelet counts may be insufficient to accurately predict clinical outcomes. In contrast, our study focuses on platelet count trajectories in patients with AF, aiming to explore whether distinct temporal patterns in platelet levels are linked to adverse outcomes.
This study investigates the prognostic value of platelet count trajectories in critically ill patients diagnosed with AF utilizing data from the Medical Information Mart for Intensive Care IV (MIMIC-IV) database. By delineating distinct subgroups based on dynamic platelet count trends and exploring their relationships with both short-term in-hospital outcomes and long-term prognoses, the study aims to enhance clinical risk stratification and shed light on the potential relevance of platelet trajectories as a predictive biomarker in AF management.
Materials and methods
Data source
The MIMIC-IV is a comprehensive and publicly accessible critical care database created through a collaboration between the Massachusetts Institute of Technology (MIT) and Beth Israel Deaconess Medical Center. Version 3.1 of MIMIC-IV contains detailed, de-identified clinical information from more than 70,000 intensive care unit (ICU) stays recorded between 2008 and 2022. 14 The dataset encompasses a wide range of variables, including patient demographics, vital signs, laboratory findings, administered medications, procedures, diagnostic classifications, and clinical outcomes. Due to its high temporal resolution and rich data granularity, MIMIC-IV serves as a valuable platform for conducting large-scale retrospective research in critically ill patient populations.
All data in MIMIC-IV are accessible to qualified researchers upon completion of data use training and adherence to access protocols designed to protect patient privacy and ensure ethical research conduct. In this study, one of the authors (Huan Zuo) completed the required training and obtained authorized access to the MIMIC-IV database. Zuo was responsible for data extraction and initial preprocessing, ensuring full compliance with all relevant data use agreements. The study was approved by the Institutional Review Board (IRB) of Beth Israel Deaconess Medical Centre under approval number 2001P-001699/14 and informed consent was obtained from all participants in the study.[Record ID: 65378168 (for HZ)].All study procedures were strictly conducted in accordance with the ethical principles set forth in the Declaration of Helsinki (revised 2024).
Inclusion and exclusion criteria
Patients diagnosed with AF were identified according to the standardized criteria of the International Classification of Diseases, Ninth and Tenth Revisions (ICD-9 and ICD-10). The study excluded patients who met any of the following criteria: (a) age less than 18 years or more than 100 years; (b) length of stay in the ICU shorter than 36 h; (c) missing platelet count measurements every 12 h after admission to the ICU; and (d) not a first-time admission to the ICU (Figure 1).

Flow-diagram illustrating patient inclusion in the study.
Data collection
Clinical information was retrieved from the MIMIC-IV database using PostgreSQL, with an emphasis on data recorded during the initial 24 h following ICU admission to accurately reflect baseline characteristics and reduce potential bias related to timing. Extracted variables encompassed demographic details (including age, sex, and race), vital signs (such as heart rate, systolic and diastolic blood pressure, mean arterial pressure, respiratory rate, body temperature, and oxygen saturation), and laboratory measurements (e.g. INR,PT, PTT, red cell distribution width, hemoglobin, hematocrit, blood urea nitrogen, electrolytes including potassium, sodium, calcium, bicarbonate, chloride, anion gap, and creatinine).
To assess illness severity, the Sequential Organ Failure Assessment (SOFA), Glasgow Coma Scale (GCS), and Charlson Comorbidity Index (CCI) were utilized. Comorbid conditions were identified through ICD codes and included acute kidney injury, renal disease, myocardial infarction, congestive heart failure, liver disease, cerebrovascular disease, peripheral vascular disease, chronic pulmonary conditions, rheumatic diseases, peptic ulcer disease, diabetes mellitus, hypertension, and hyperlipidemia. Therapeutic interventions within the first 24 h of ICU admission—such as administration of aspirin or warfarin, use of mechanical ventilation, and continuous renal replacement therapy (CRRT)–were also documented.
Outcome
The primary endpoint was all-cause mortality within 28 days during hospitalization. Follow-up commenced from the time of AF diagnosis and extended until either in-hospital death or discharge, depending on which occurred earlier.
Latent growth mixture modeling
Platelet counts were recorded at 12, 24, and 36 h following ICU admission. To uncover distinct patterns of platelet count changes within this early time frame, latent growth mixture modeling (LGMM) was employed. 15 The analysis focused specifically on patients with AF and was conducted using the “lcmm” package in R. LGMM enables the identification of hidden subpopulations within the cohort, each characterized by a unique trajectory defined by its own mean growth pattern over time. This approach operates under the assumption that the overall population is heterogeneous, consisting of several unobserved groups with distinct temporal profiles.
Selecting the optimal number of latent classes is a critical component of LGMM. To determine the most appropriate model, quadratic growth models incorporating between one and seven latent classes were evaluated. The final model was chosen based on a combination of statistical indicators and practical considerations: (1) lower values of the Akaike Information Criterion (AIC), Bayesian Information Criterion (BIC) were indicative of improved model fit; (2) greater log-likelihood and entropy values were favored, with entropy ≥ 0.7 regarded as acceptable for classification reliability; (3) each identified class was required to represent at least 1% of the overall study population; (4) the mean posterior probability for individuals within each class needed to meet or exceed 70% to ensure classification precision; and (5) model simplicity and clinical relevance were also taken into account during the selection process.
Statistical analysis
All statistical analyses were conducted using R software. For continuous variables, data were presented as mean ± standard deviation when normally distributed, or as median and interquartile range for non-normally distributed data. Categorical variables were expressed as frequencies and percentages. Comparisons across groups were made using one-way ANOVA for normally distributed continuous data, the Kruskal–Wallis test for skewed distributions, and either the Chi-square test or Fisher's exact test for categorical variables, depending on data characteristics.
LGMM was employed to classify platelet count trajectories during the first 36 h after ICU admission. The optimal number of latent classes was determined based on model fit indices, including AIC, BIC, entropy, and the proportion of subjects in each class. For variables with less than 15% missing data, multiple imputation was performed using the random forest method under the assumption of missing at random. Variables with more than 15% missing data were excluded from multivariate analysis.
The relationship between identified platelet trajectory groups and in-hospital mortality among AF patients was examined using Cox proportional hazards regression. Three models were constructed: Model 1 was unadjusted; Model 2 adjusted for demographic factors (age, sex, and race); and Model 3 additionally included covariates found to be significant (p < 0.05) in univariate analyses. Pairwise comparisons were conducted by alternately using each trajectory class as the reference group.
Kaplan–Meier survival analysis was used to visualize 28-day survival across the different trajectory groups, with statistical comparisons performed via the log-rank test. To evaluate the consistency of associations, subgroup analyses were stratified by age, sex, and key comorbidities. A two-sided p-value < 0.05 was considered statistically significant. Results were reported as hazard ratios (HRs) with corresponding 95% confidence intervals (CIs).
Result
Latent class analysis of platelet dynamics in atrial fibrillation
Model fit indices for latent platelet trajectory models in AF patients are summarized in Table 1. The three-class model provided the best balance of fit, classification accuracy, and subgroup size, with lower AIC, lower BIC, and higher entropy compared to the two-class model. All three classes had adequate representation.
Model fit indices for latent class trajectory models based on platelet levels in patients with AF.
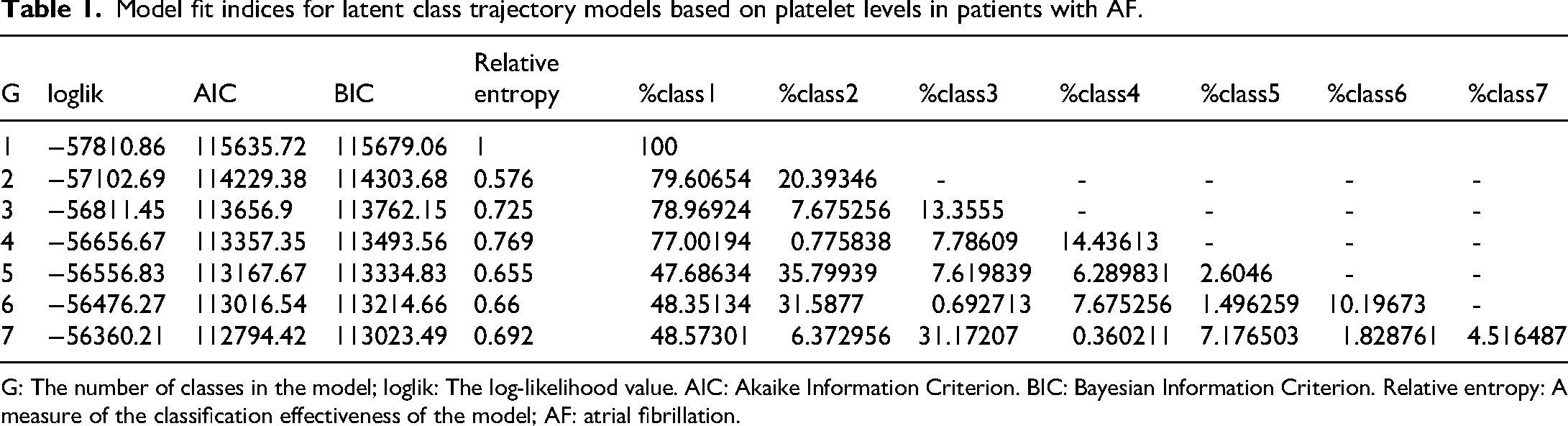
G: The number of classes in the model; loglik: The log-likelihood value. AIC: Akaike Information Criterion. BIC: Bayesian Information Criterion. Relative entropy: A measure of the classification effectiveness of the model; AF: atrial fibrillation.
Figure 2 displays the mean predicted platelet count trajectories over the observation period for different classes (Class 1, Class 2, and Class 3) of individuals. The three trajectories show distinct divergence, revealing heterogeneity in the dynamics of platelet changes within the population. Specifically, Class 1 (black line) shows platelet count remaining nearly constant with a slight decline throughout the period, indicating relatively stable platelet regulation in this group. Class 2 (red line) presents a “U-shaped” trend, with initially high platelet values rapidly declining to a nadir in the early phase, followed by a slight rebound in the later phase. In contrast, Class 3 (green line) shows an “inverted U-shaped” trajectory, where the platelet count slowly rises in the early phase and then gradually declines in the middle to late stages, demonstrating a moderate amplitude of dynamic change.

Class-specific predicted trajectories of serum platelet levels over time in patients with AF. Three distinct trajectory classes were identified within the first 36 h after ICU admission: Class 1: Persistently low (black line), characterized by consistently low platelet counts with a slight downward trend; Class 2: Decline–rebound (red line), starting from higher platelet levels with a rapid early decline followed by partial recovery; Class 3: Rise–fall (green line), showing an initial increase in platelet counts followed by a gradual decline. These trajectories highlight substantial heterogeneity in early platelet dynamics and may carry prognostic significance. AF; atrial fibrillation; ICU: intensive care unit.
Baseline characteristics and mortality-associated factors
Significant differences across glucose trajectory groups were found in baseline characteristics, including age (p = 0.034), sex (p < 0.001), and several vital signs (heart rate, blood pressure, respiratory rate, temperature, SpO₂; all p < 0.05). Key lab values—glucose, RDW, hemoglobin, hematocrit, sodium, chloride, anion gap, and bicarbonate—also differed significantly (all p < 0.01). SOFA score (p < 0.001) and CCI (p = 0.015) varied across groups as well.
The prevalence of complications such as AKI, myocardial infarction, liver disease, renal disease, peripheral vascular disease, and chronic pulmonary disease differed significantly (all p < 0.05), as did aspirin and warfarin use (both p < 0.001) (Table 2).
Clinical characteristics and outcomes by platelet trajectory in AF patients.

HR: hazard ratio, CI: confidence interval. SBP: systolic blood pressure; DBP: diastolic blood pressure; MBP: mean blood pressure; SpO₂: peripheral oxygen saturation; INR: international normalized ratio; PT: prothrombin time; PTT: partial thromboplastin time; RDW: red cell distribution width; BUN: blood urea nitrogen; SOFA: Sequential Organ Failure Assessment; GCS: Glasgow Coma Scale; AKI: acute kidney injury; CRRT: continuous renal replacement therapy; AF: atrial fibrillation.
Univariate Cox analysis showed that 28-day mortality was significantly associated with male sex (HR = 0.77, p < 0.001), AKI, liver disease, heart failure, cerebrovascular disease, CRRT use, and multiple clinical/lab variables including age, respiratory rate, SpO₂, coagulation indices, RDW, BUN, electrolytes, SOFA, GCS, CCI, and use of aspirin or warfarin (all p < 0.05) (Supplementary Table 1).
Association between platelet trajectories and 28-day mortality
In the unadjusted multivariate Cox regression model (Model 1), both Class 2 (p < 0.001) and Class 3 (p = 0.003) were significantly linked to higher 28-day mortality when compared with Class 1. These associations remained statistically significant in Model 2, which adjusted for age, sex, and race (Class 2: p < 0.001; Class 3: p = 0.009). In the fully adjusted Model 3, which incorporated demographic factors, comorbid conditions, therapeutic interventions, and key clinical and laboratory variables, Class 2 continued to show a significant association with increased risk of 28-day mortality (p = 0.013), while the association observed for Class 3 was attenuated and no longer reached statistical significance (p = 0.163) (Table 3).
Multivariate Cox regression analysis of platelet trajectories and 28-day mortality in AF.

HR: hazard ratio. CI: confidence interval. Model1: Crude. Model2: Adjust: gender, age, race. Model3: Adjust: gender, race, age, liver disease, congestive heart failure, hyperlipidemia, aspirin, cerebrovascular disease, warfarin, heart rate, resp rate, SpO2, INR, PT, PTT, RDW, BUN, potassium, CRRT, AKI, creatinine, chloride, anion gap, bicarbonate, SOFA, GCS, Charlson comorbidity index.
28-Day survival by platelet trajectory
Figure 3 presents Kaplan–Meier survival curves for 28-day mortality across the three platelet trajectory classes. Patients in Class 1 (red line) demonstrated the highest probability of survival, followed by Class 3 (blue line), whereas Class 2 (green line) consistently showed the lowest survival throughout the 28-day follow-up. Separation between the survival curves emerged early and progressively widened over time. The differences in 28-day cumulative survival among the three groups were statistically significant, as assessed by the log-rank test (p < 0.001).

Kaplan–Meier curves of 28-day overall survival across platelet trajectory classes. Kaplan–Meier analysis showed significant differences in survival across the three trajectory classes (log-rank test p < 0.001). Patients in Class 2 (decline–rebound) exhibited the lowest survival probability early during the ICU stay, with the gap widening over time, whereas Class 1 (persistently low) and Class 3 (rise–fall) demonstrated relatively higher survival probabilities. The number at risk table is provided below the curves to enhance interpretability of the survival analysis. HR: hazard ratio.CI: confidence interval; ICU: intensive care unit.
Subgroup analysis of platelet trajectories and 28-day mortality
In the subgroup analysis of this study, we evaluated the impact of clinical characteristics such as age, gender, congestive heart failure, and hypertension on the primary outcome (Figure 4). A significant interaction was observed only for gender (p for interaction = 0.047), with females showing a lower risk than males (HR = 0.833, 95% CI: 0.710–0.977). No significant interactions were found for age (p = 0.689), CHF (p = 0.771), or hypertension (p = 0.985), suggesting consistent effects across these subgroups.

Subgroup analysis of the association between platelet trajectories and 28-day mortality in AF patients. AF: atrial fibrillation.
Discussion
This study is the first to apply a LGMM to analyze early platelet count trajectories in ICU patients with AF, identifying three clinically meaningful trajectory classes and demonstrating an independent association between these classes and 28-day mortality. As shown in Figure 2, the three trajectories exhibited distinct temporal dynamics, revealing substantial heterogeneity in platelet regulation patterns within the population. Specifically, Class 1 was characterized by relatively stable platelet count throughout the observation period, with only a slight decline. Class 2 (red line) followed a typical “U-shaped” trajectory. Notably, compared with Class 1, patients in Class 2 had a significantly higher risk of mortality, and this association persisted even after comprehensive adjustment for demographic characteristics, comorbidities, treatment measures, and key laboratory indicators. The Kaplan–Meier survival curves further demonstrated that Class 2 patients exhibited the lowest survival probability from the early ICU period, with the difference widening over time. Class 3 (green line) displayed an “inverted U-shaped” trajectory, with platelet count gradually increasing in the early phase, which may represent a relatively favorable adaptive recovery process. In addition, subgroup analysis showed a significant interaction only with sex: female patients had a lower risk of mortality compared with males, suggesting the possibility of sex-related biological mechanisms or differences in treatment.
Although platelets are anucleate cell fragments primarily recognized for their role in hemostasis and thrombosis, emerging evidence indicates that their functions extend far beyond traditional coagulation processes. In response to vascular injury, platelets promptly adhere to exposed subendothelial structures such as collagen and von Willebrand factor. This adhesion triggers platelet activation, leading to aggregation and the release of bioactive substances stored in α-granules and dense granules, thereby initiating and propagating the coagulation cascade. 16 In addition to their role in coagulation, platelets serve as important modulators of immune and inflammatory responses. They express a diverse array of surface receptors that enable them to detect pathogenic signals, engage in cross-talk with various immune cells, and secrete chemokines that facilitate the recruitment of leukocytes to sites of tissue injury or inflammation. 17 In addition, increasing evidence suggests that platelets play a role in central nervous system (CNS) disorders, such as epilepsy. Their infiltration into neural tissues and involvement in inflammatory and oxidative stress responses may contribute to the progression of neurological damage.18–20 Platelet dysfunction is linked to a range of serious complications. In CNS disorders such as Alzheimer's, Parkinson's, and multiple sclerosis, abnormal platelet activation may contribute to disease progression. In elderly patients, platelet hyperreactivity—often driven by oxidative stress—increases the risk of thrombotic events like stroke, myocardial infarction, and deep vein thrombosis.21–23 Thus, platelets are multifunctional effector cells with coagulative, immunomodulatory, and inflammation-related functions. These features make them promising therapeutic targets for a wide range of diseases.
Fluctuations in platelet counts are closely linked to mortality in critically ill patients. Puertas et al. found that in post-cardiac surgery and ICU patients, non-survivors often had persistently low or slowly recovering platelet counts, while a rapid rebound correlated with better survival. Mortality rose notably when platelet counts stayed below 100 × 10⁹/L. Dynamic platelet changes were as predictive of short-term mortality as traditional severity scores. These findings highlight the value of tracking platelet counts trends in critical care to identify high-risk patients early and guide timely intervention.24,25 The present study further confirmed by trajectory analysis that the short-term risk of death was significantly higher in patients with progressively decreasing platelet counts.
In this study, the three platelet counts trajectory classes identified through LGMM revealed distinct clinical implications. Patients in Class 1 exhibited persistently low but stable platelet levels (approximately 120–130 K/uL) throughout the early ICU stay, with minimal fluctuations. This pattern suggests a relatively stable hematologic state without signs of acute deterioration and was associated with the lowest 28-day mortality, possibly reflecting a “protective” or at least non-aggressive disease course. In contrast, Class 2 displayed a pronounced U-shaped trajectory, with platelet counts rapidly declining from around 300 to below 200 shortly after admission, followed by a modest rebound. This pattern may indicate acute consumption or destruction due to conditions such as inflammation, disseminated intravascular coagulation (DIC), or significant bleeding, and was independently associated with the highest mortality, highlighting its value in identifying high-risk patients. Early recognition of this trajectory could facilitate timely intervention, including intensified monitoring and tailored treatment strategies. Class 3 patients began with the highest platelet counts (>300 K/uL), followed by a gradual decline to approximately 250 K/uL. This relatively mild fluctuation may reflect a compensatory response to systemic inflammation, gradually stabilizing with treatment. Mortality in this group was intermediate, suggesting a moderate risk profile. Overall, these dynamic platelet trajectories underscore the biological heterogeneity among critically ill AF patients.
A gradual decrease in platelet count often indicates underlying serious diseases and is significantly associated with increased mortality risk. In patients with multiple myeloma undergoing CAR-T therapy, persistent thrombocytopenia is linked to impaired immune reconstitution and markedly reduced overall survival. 26 In atherosclerosis, elevated platelet activity and increased mean platelet volume are closely associated with higher risks of myocardial infarction and stroke. 27 Among patients with CKD, especially those with coexisting peripheral artery disease, heightened platelet reactivity raises the risk of cardiovascular death by 2–3 times and accounts for up to 26% of excess myocardial infarction or mortality risk. 28 Therefore, persistent thrombocytopenia serves as a critical warning indicator of several high-risk conditions.
Thrombocytopenia can arise from reduced production, increased destruction or clearance, abnormal sequestration, and dilution. In sepsis, inflammatory cytokines (e.g. TNF-α, IL-6) cause endothelial damage and DIC, leading to excessive platelet consumption, while reduced thrombopoietin (TPO) impairs platelet production. 29 In chronic liver disease, it is often due to hypersplenism and decreased hepatic TPO synthesis. 30 Extracorporeal circulation or ECMO can increase shear stress, causing loss of platelet surface receptors like GPIbα and rapid clearance. 31 Large-volume fluid resuscitation may also dilute platelets. 29 These mechanisms frequently coexist and require comprehensive evaluation for accurate diagnosis and management. In addition, the progressive decline in platelet count may be caused by a variety of pathologic mechanisms, including bone marrow dysfunction (e.g. myelodysplastic syndromes, acute leukemia), immune-mediated destruction (e.g. ITP, HIT), abnormal retention due to hypersplenism, and pregnancy-related factors (e.g. preeclampsia, HELLP syndrome). The etiology of the disease needs to be determined by a combination of detailed history, laboratory tests, and clinical manifestations.32–35
This study found that in female patients, the platelet count trajectory (Class 2) was significantly associated with an increased risk of 28-day mortality, whereas no such association was observed in males, suggesting a potential sex interaction. This difference may be closely related to sex-specific electrophysiological and physiological characteristics in AF. As reviewed by Smith et al., females exhibit shorter P-wave duration, smaller atrial size, and distinct ion channel expression even under normal sinus rhythm. Additionally, they are more prone to spontaneous calcium release and transient inward current (Iti), indicating a higher susceptibility to calcium handling dysregulation and triggered activity under stress conditions. 36 These characteristics may be further exacerbated during the course of AF, rendering the female atria more sensitive to inflammation, coagulation, and electrical remodeling. As a result, the impact of platelet count on disease progression may be amplified in females. Furthermore, as noted by Gillis et al., although female patients with AF tend to develop the condition at a later age, they face higher risks of stroke, heart failure, and mortality. Additionally, they respond less favorably to antiarrhythmic drugs such as amiodarone and experience a higher incidence of adverse effects. 37 These differences in treatment and management may also indirectly affect females’ tolerance to platelet fluctuations under critical conditions, thereby increasing their risk of mortality. As a result, dynamic changes in platelet count may more sensitively reflect the underlying electrophysiological and inflammatory vulnerability in females, making it a stronger prognostic marker in this population.
In this study, LGMM was used to model the early platelet count trajectories of ICU patients with AF, identifying three distinct patterns within the first 36 h after admission. It is noteworthy that one trajectory, characterized by an initial decline followed by a slight rebound (Class 2), was significantly associated with a higher 28-day in-hospital mortality rate. This finding underscores the important prognostic value of dynamic indicators in outcome assessment, surpassing traditional single time-point measurements. The application of LGMM in this clinical context is a key innovation of this study, as it helps to capture temporal heterogeneity in critically ill patients. These results suggest that early platelet dynamics may reflect underlying pathophysiological processes closely related to prognosis. However, several limitations of this study should be considered. First, due to the observational design, causal relationships cannot be inferred, and residual confounding factors may still exist despite multivariate adjustments. Second, platelet data were limited to three early time points (12, 24, and 36 h), potentially missing later or non-linear trends. Third, the lack of an external validation cohort limits the generalizability of the findings to other populations or medical settings. Fourth, excluding patients with missing data may have introduced selection bias, especially survivor bias, where patients who died early may not have had sufficient laboratory tests, thus underrepresenting severe cases in the sample and affecting the generalizability to more severe cases. In conclusion, early platelet count trajectories are associated with short-term outcomes in AF patients and may be valuable for future risk stratification. Prospective studies with longer observation periods, the inclusion of external validation cohorts, and measures to handle missing data are warranted to further validate and expand on these findings.
Conclusion
In this retrospective cohort study of ICU patients with AF, we identified three distinct early platelet count trajectories patterns using LGMM. Among them, a U-shaped trajectory (Class 2) was independently associated with the highest 28-day in-hospital mortality. These findings suggest that early dynamic changes in platelet count hold important prognostic value and may reflect underlying pathophysiological processes. Incorporating trajectory-based monitoring into clinical practice could improve early risk stratification and support individualized management strategies.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251399568 - Supplemental material for Latent trajectory modeling of platelet counts and mortality risk in intensive care unit patients with atrial fibrillation
Supplemental material, sj-docx-1-sci-10.1177_00368504251399568 for Latent trajectory modeling of platelet counts and mortality risk in intensive care unit patients with atrial fibrillation by Huan Zuo, Xin Zuo and Zhuohui Tan in Science Progress
Footnotes
Acknowledgments
Not applicable
Ethics approval and consent to participate
The study was approved by the Institutional Review Board (IRB) of Beth Israel Deaconess Medical Centre under approval number 2001P-001699/14 and informed consent was obtained from all participants in the study.
The study strictly adhered to the following international norms: Declaration of Helsinki (DH) on ethical principles for medical research; The US Health Insurance Privacy and Accountability Act (HIPAA) regarding patient data privacy; MIMIC database use agreement
Consent to participate declaration
Every human participant should provide their consent.
Author contributions
Study concept and design by TZ and HZ; acquisition, analysis, or interpretation of data by all authors; drafting of the manuscript by HZ; critical revision of the manuscript by HZ and XZ; statistical analyses by HZ; supervision of research by TZ. All authors contributed to the article and approved the submitted version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest
The authors declare that this research was conducted without any commercial or financial relationships that could be perceived as conflicts of interest.
Supplemental material
Supplemental material for this article is available online.
Clinical trial number
Not applicable.
Declaration of generative AI in scientific writing
It is hereby declared that the content presented in this manuscript has been developed without the assistance of generative artificial intelligence tools for the writing, analysis, or interpretation of the data.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
