Abstract
Objectives
Diabetes mellitus (DM) is a common comorbidity in intensive care unit (ICU) patients. The Braden skin score (BSS) has increasingly been recognized as an indicator of patient frailty. However, the association between the BSS and clinical outcomes in critically ill diabetic patients remains unclear. This study aimed to investigate the relationship between the BSS and clinical outcomes in diabetic patients in ICU settings.
Methods
A retrospective cohort of diabetic patients with measured BSS was identified from the Medical Information Mart for Intensive Care IV (MIMIC-IV) database. The primary outcomes included mortality at 30, 60, and 90 days. The association between BSS and survival outcomes was evaluated using time-dependent Cox proportional hazards model. Further validation was conducted through Kaplan–Meier survival analysis, restricted cubic spline fitting, and subgroup analyses.
Results
A total of 20,590 patients were included in this study, those with higher BSS had a 14% decreased risk of 30-day mortality (HR: 0.86, 95% CI: 0.84–0.87, p < .01). Based on cutoff values of 13, patients were categorized into two risk groups. After adjusting for covariates, time-dependent Cox proportional hazards model indicated that the high-risk group exhibited a significantly increased 30-day all-cause mortality compared with the low-risk groups (HR: 1.76, 95% CI: 1.62–1.91, p < .01). Kaplan–Meier curves consistently demonstrated poorer survival across all time points in the high-risk group. Subgroup analysis further indicated that the association between BSS and mortality was particularly pronounced in patients with cerebrovascular disease.
Conclusions
A low BSS was independently linked to higher mortality among critically ill patients with diabetes, especially in those with concomitant cerebrovascular diseases. These results support the potential utility of BSS as a prognostic indicator in this population. Further validation through larger prospective studies remains necessary.
Introduction
Diabetes mellitus (DM) is a chronic metabolic disorder characterized hyperglycemia resulting from either inadequate insulin secretion by the pancreas or impaired utilization of insulin by the body.1,2 As a major worldwide public health issue, DM continues to impose growing demands on medical systems and societies across the globe. In 2021, approximately 529 million individuals were affected by diabetes. This figure is expected to rise sharply to around 1.31 billion by 2050, with associated global health expenditures projected to exceed 1054 billion dollars by 2045. 3 Furthermore, patients with DM hospitalized have a higher risk of intensive care unit (ICU) admission, at the same time, DM is the second most common comorbidity in patients admitted to the ICU.4,5 For patients in the ICU, the presence of DM is a significant risk factor that negatively impacts their clinical outcome.6,7 Short-term all-cause mortality is a key indicator for ICU quality management and clinical decision-making. The mortality risk among ICU patients with diabetes is complex and multifactorial. Although traditional scores, such as sequential organ failure assessment (SOFA), are effective, they can be complex to use and may fail to capture the overall frailty status.
The Braden skin score (BSS), originally developed by Barbara Braden and Nancy Bergstrom in 1987, remains a foundational instrument for assessing patient susceptibility to pressure injury. This evaluative tool incorporates six distinct domains: sensory perception, moisture, activity, mobility, nutrition, and friction and shear. 8 Recognized for its high predictive sensitivity in both identifying the progression and informing prevention strategies for pressure injuries, the BSS has been extensively integrated into routine clinical care. Beyond its original purpose, a reduced BSS provides an integrative measure of physiological reserve and frailty in critically ill patients by encapsulating deficits in mobility, sensory perception, and nutritional status. This composite assessment is independently associated with increased short term mortality, linking it to core pathophysiological mechanisms in critical illness such as systemic inflammatory response, immunosuppression, and hypercatabolism. In diabetic patients, who are particularly vulnerable due to inherent microcirculatory impairment, peripheral neuropathy, and delayed wound healing, skin integrity may serve as a sensitive window into systemic health. Previous studies have demonstrated the prognostic utility of BSS in specific ICU populations. Zhu et al. found BSS to be a significant predictor of 30-day all-cause mortality in elderly heart failure patients admitted to the ICU. 9 Similarly, Tang et al. reported that BSS effectively identifies high mortality risk in ischemic stroke patients in ICU settings. 10 However, few studies have specifically examined this association in diabetic patients admitted to the ICU. This study hypothesizes that in this high risk population, early BSS assessment may serve as a simple yet comprehensive clinical tool to effectively identify patients at exceptionally high risk of short term mortality. Utilizing data extracted from the Medical Information Mart for Intensive Care IV (MIMIC-IV), this study seeks to evaluate the prognostic value of BSS in diabetic ICU patients, thereby addressing an important gap in the existing literature.
Methods
Data source and study population
The retrospective cohort study extracted data from the MIMIC-IV database v3.0, which contains deidentified electronic health records of patients admitted to the Beth Israel Deaconess Medical Center from 2008 to 2019. After training and assessment, we were granted access rights to extract relevant variables (record ID: 62192466). As this study was a retrospective analysis based on existing anonymised data, the patient information has been deidentified, and therefore the need for ethical consent has been waived. This study was conducted in accordance with the principles of the Declaration of Helsinki, as revised in 2024. The reporting of this study follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 11
Eligible patients were those with a diagnosis of DM who were admitted to the ICU for the first time. Patients who met the following criteria were excluded from the study: having ICU stays shorter than 24 h, being under 18 years of age, lacking BSS and mortality information. For analysis, patients were divided into two groups based on BSS, The patients selection process is shown in Figure 1.
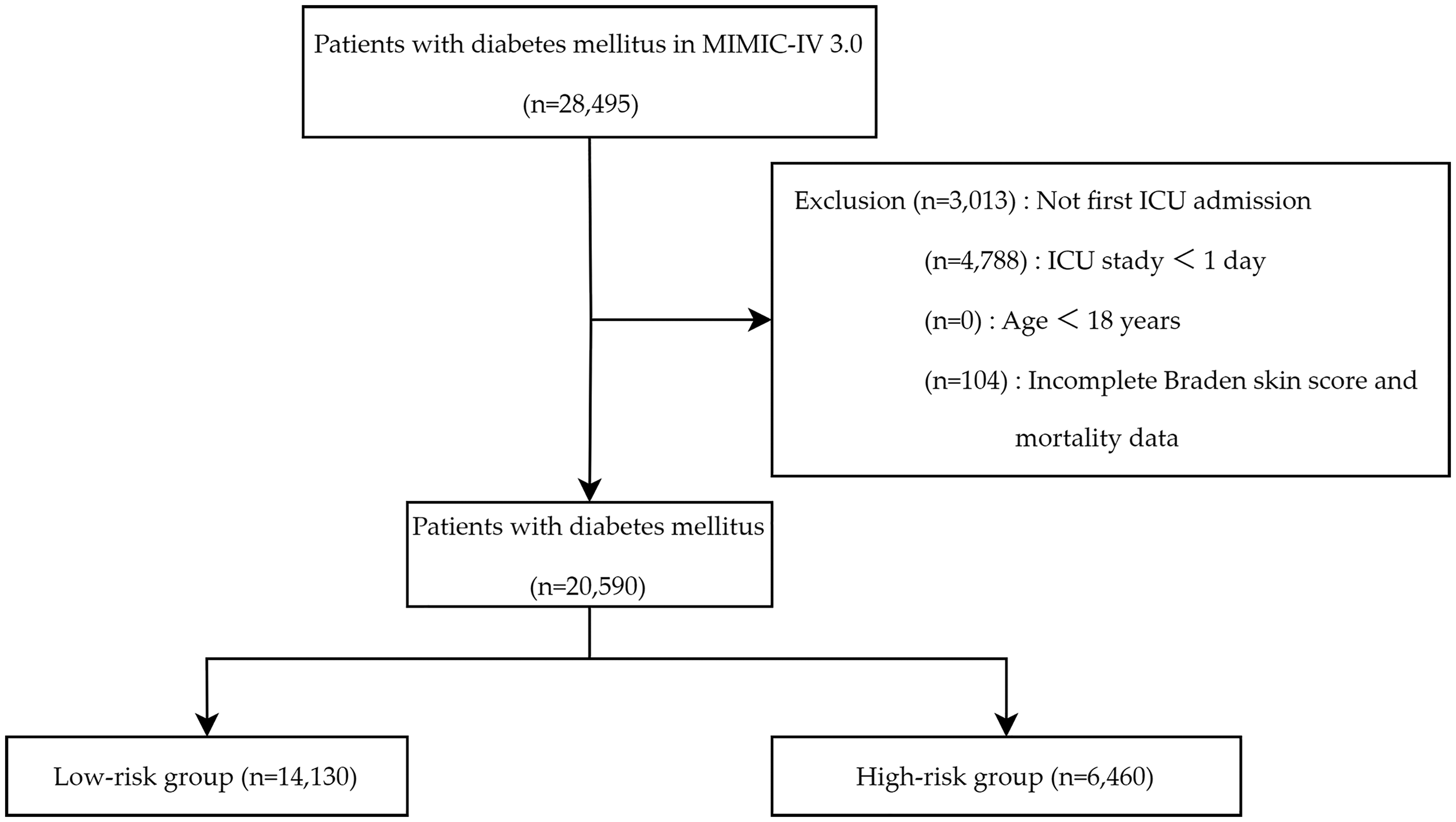
The flowchart of this study.
Data collection
Health data were extracted by structured query language (SQL) from the database, including: demographics, comorbidities, severity of illness score, vital signs, laboratory test results, treatments and medications, and outcomes. Data were collected during the first 24 h following ICU admission. In instances where laboratory tests were performed multiple times, the most abnormal value was selected. Comorbid conditions were identified and classified according to ICD-9 and ICD-10 coding systems. To reduce the impact of potential bias, the study excluded variables with a missing rate of more than 20%. For data with a missing rate of less than 20%, we used the ‘mice’ package of the R software and applied the random forest method of multiple imputation.
The study used the BSS on admission to the ICU as the exposure factor. This is a scale specifically designed to assess a patient's risk of skin pressure injury. It combines six key risk components: Sensory, Moisture, Activity, Mobility, Nutrition and Friction/Shear. 8 The cumulative index ranged from 6 to 23, with a lower score indicating a higher propensity to develop pressure injuries. Participants were categorized based on BSS assessed on the first day of ICU admission. The optimal cutoff for risk stratification was determined by adopting the receiver operating characteristic (ROC) curve analysis with Youden's index maximization for 30-day mortality in the cohort. The optimal cutoff was BSS=13. Accordingly, patients with a BSS <13 were classified as the high-risk group, and those with a BSS≥13 were classified as the low-risk group. The outcomes in this study were 30-day, 60-day, and 90-day all-cause mortality.
Statistical analysis
Statistical analyses were performed using R software (version 4.4.2). Based on BSS values, patients were classified into two risk categories, using the low-risk group as the reference for comparative analyses. The baseline characteristics of patients between two groups were compared. Normally distributed continuous variables were expressed as mean with standard error (SE) and compared using unpaired Student's t-test. Non-normally distributed variables were summarized as median (25th percentile, 75th percentile) and were compared using the Mann‒Whitney U test. Categorical variables were reported as frequencies (n) and percentages (%), with comparisons made via Chi-square tests. To evaluate the association between the BSS and mortality at 30, 60, and 90 days, a multivariable Cox proportional hazards model was initially constructed. Following model specification, the proportional hazards (PH) assumption was systematically assessed for all included covariates using Schoenfeld residual tests. Evidence of significant violation of the proportional hazards assumption for any covariate indicated the presence of time-dependent effects. In such instances, to ensure robust estimation, the final analysis employed a time-dependent Cox proportional hazards model for fitting. Variables exhibiting a variance inflation factor (VIF) greater than 5 were excluded to mitigate multicollinearity. Kaplan-Meier survival analysis evaluated endpoint incidence across different BSS groups. Restricted cubic splines (RCS) were employed to assess the dose-response relationships. Subgroup analyses were performed for comorbidities. The log-likelihood ratio test was used to assess potential interactions between the BSS and these subgroup factors. To control for false-positive findings due to multiple testing, the resulting interaction p-values were adjusted using the Benjamini-Hochberg (BH) false discovery rate (FDR) correction. All the statistical tests performed as two-sided, and p-value < .05 was considered statistically significant.
Results
Clinical characteristics
Variables with missing rates over 20% (BMI 46.19%, HbA1c 86.36%, albumin 84.20%, lactate 35.91%, pH 27.46%) were excluded. For remaining covariates with lower missingness (<20%), multiple imputation was performed (random forest, m = 5). The imputed variables and their missing rates were: mean blood pressure (MBP, 0.02%), respiratory rate (0.07%), temperature (2.51%). saturation of peripheral oxygen (SpO2, 0.02%), white blood cells (WBC, 15.47%), platelets (15.28%), red blood cells (RBC, 15.51%), blood urea nitrogen (BUN, 10.31%), creatinine (10.24%), potassium (9.93%), serum glucose (10.96%), and international normalized ratio (INR, 14.03%). Full details are provided in Supplementary Table 1.
Table 1 shows the baseline characteristics of selected patients. Data from 20,590 diabetes patients were analyzed, with a mean age of 68.08 years, 41.88% were female. Patients with lower BSSs exhibited higher SOFA scores, APSIII scores, and OASIS scores. They were more likely to require renal replacement therapy, mechanical ventilation, vasopressors and insulin. Statistically significant differences were observed between the two BSS risk groups in terms of cerebrovascular disease, congestive heart failure, malignancy, liver disease, sepsis, malnutrition, heart rate, MBP, respiratory rate, temperature, SpO2, WBC, platelets, RBC, BUN, creatinine, potassium, serum glucose, INR. In terms of outcomes, patients with lower BSS demonstrated poorer prognoses, evidenced by elevated short-term mortality at 30, 60, and 90 days.
Clinical characteristics of critically ill patients with diabetes mellitus.
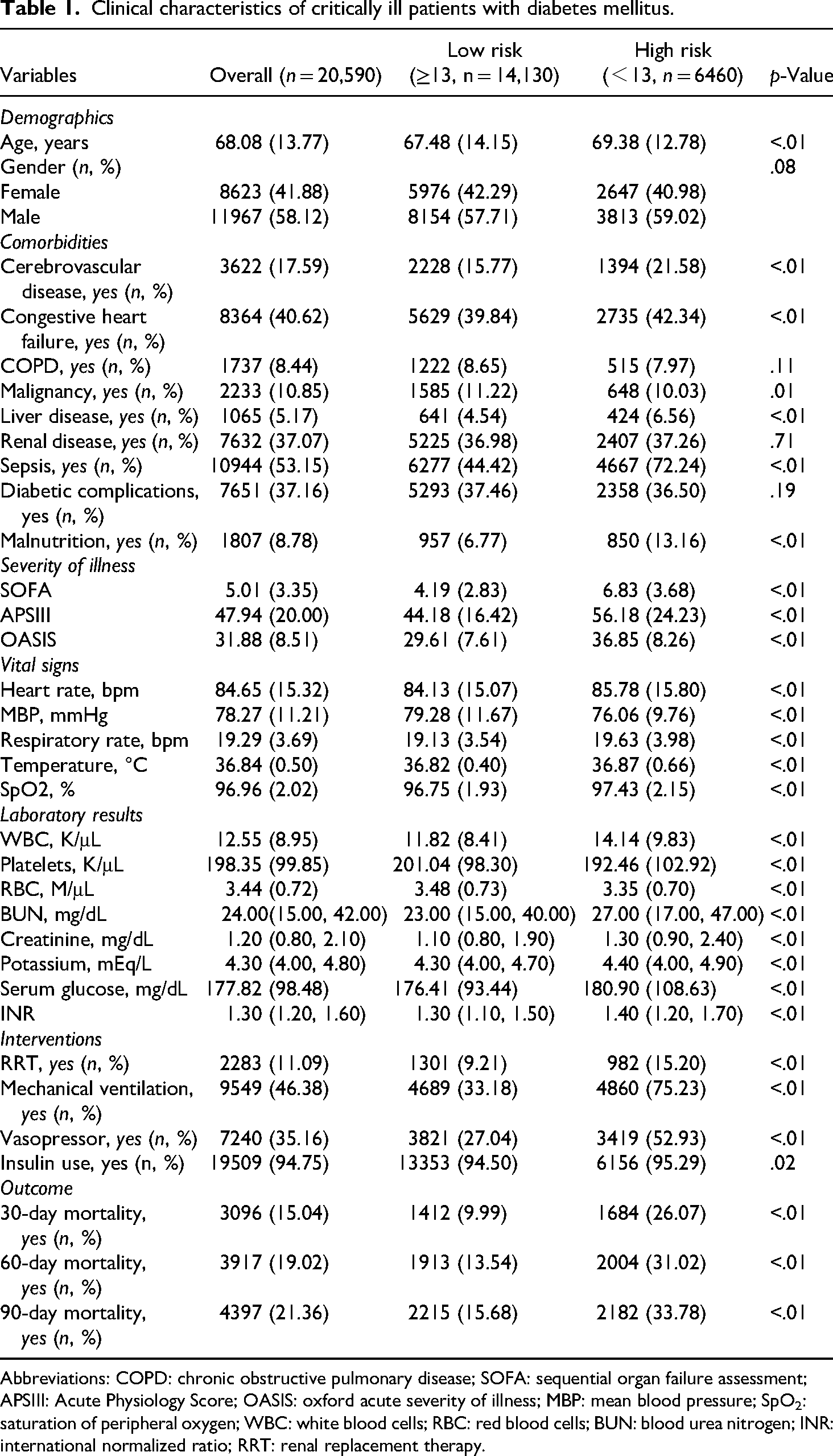
Abbreviations: COPD: chronic obstructive pulmonary disease; SOFA: sequential organ failure assessment; APSIII: Acute Physiology Score; OASIS: oxford acute severity of illness; MBP: mean blood pressure; SpO2: saturation of peripheral oxygen; WBC: white blood cells; RBC: red blood cells; BUN: blood urea nitrogen; INR: international normalized ratio; RRT: renal replacement therapy.
To assess the level of systemic inflammation, this study calculated the systemic inflammation index (SII, a composite inflammatory marker integrating platelet, neutrophil, and lymphocyte counts (SII = platelet × neutrophil/lymphocyte count). 12 In the patient subgroup with available data in this study, the SII was significantly higher in the high-risk group than in the low-risk group. Detailed results are presented in Supplementary Table 2.
Associations between BSS and outcomes
Kaplan-Meier survival analysis curves were employed to assess the incidence of various outcomes between two groups, as depicted in Figure 2. The high-risk group had significantly lower survival rates at 30-day, 60-day, and 90-day intervals for all-cause mortality (all log-rank p < .01). During the 30-day, 60-day, and 90-day follow-up periods, mortality was recorded in 3,096, 3,917, and 4397 patients, respectively. After multivariable adjustment, a time-dependent Cox regression model was employed to account for violations of the PH assumption. Schoenfeld residual tests identified deviations from the PH assumption for several covariates, including heart rate, respiratory rate, SpO2, MBP, SOFA, APSIII, OASIS, malignancy, malnutrition, WBC, BUN, creatinine, potassium, INR, serum glucose, mechanical ventilation, vasopressors, and insulin use (Supplementary Table 3). The time-dependent model revealed that a higher BSS was significantly associated with reduced all-cause mortality in ICU patients with diabetes at 30 days (HR 0.86, 95% CI 0.84–0.87, p < .01), 60 days (HR: 0.86, 95% CI: 0.85–0.88, p < .01), and 90 days (HR: 0.87, 95% CI: 0.86–0.88, p < .01). All variables included in the multivariate analysis demonstrated a VIF below 5, indicating the absence of significant multicollinearity. When BSS was analyzed as a categorical variable, patients in the high-risk group exhibited a significantly higher mortality rate at 30 days (HR: 1.76, 95% CI: 1.62–1.91, p < .01) compared with those in the low-risk group. Similarly, the mortality rates at 60 days (HR: 1.71, 95% CI: 1.60–1.84, p < .01), 90 days (HR: 1.70, 95% CI: 1.58–1.81, p < .01) were also significantly higher in the high-risk group, indicating a notable difference (Table 2).

Kaplan‒Meier all-cause mortality survival analysis curve. (a) Showing comparison of 30-day mortality between Braden scale score groups. (b) Showing comparison of 60-day mortality between Braden scale score groups. (c) Showing comparison of 90-day mortality between Braden scale score groups.
Time-dependent cox proportional hazards model for mortality in patients with diabetes.

Time-dependent cox proportional hazards model included adjustment for all covariates, categorized as time-fixed or time-dependent. Time-fixed covariates: age, gender, cerebrovascular disease, congestive heart failure, COPD, liver disease, renal disease, sepsis, diabetic complications, temperature, platelets, RBC, RRT. Time-dependent covariates: heart rate, respiratory rate, SpO2, MBP, SOFA, APSIII, OASIS, malignancy, malnutrition, WBC, BUN, creatinine, potassium, INR, serum glucose, mechanical ventilation, vasopressor, insulin use. Time-dependent covariates were modeled using time-varying functions, defined as tt(x,t)=x*log(t + 1).
Abbreviations: COPD: chronic obstructive pulmonary disease; SOFA: sequential organ failure assessment; APSIII: Acute Physiology Score; OASIS: oxford acute severity of illness; MBP: mean blood pressure; SpO2: saturation of peripheral oxygen; WBC: white blood cells; RBC: red blood cells; BUN: blood urea nitrogen; INR: international normalized ratio; RRT: renal replacement therapy; HR: hazard ratio; CI: confidence interval.
Figure 3 employs smoothed curve fitting to model the non-linear association between BSS and mortality risk, illustrating a statistically significant dose-response relationship. Restricted cubic spline analysis revealed a non-linear relationship between the BSS and 90-day all-cause mortality (p-nonlinear = .03), with a monotonically decreasing hazard ratio as the BSS increased, though no distinct inflection point was identified. In contrast, the associations with 30-day (p-nonlinear = .63) and 60-day (p-nonlinear = .27) mortality demonstrated negative linear trends.

Restricted cubic spline curves depicting the relationship between BSS and all-cause mortality at (a) 30 days, (b) 60 days, and (c) 90 days.
Subgroup analysis
Table 3 displays the results of stratified analyses examining the correlation between BSS and 30-day all-cause mortality in distinct clinical subgroups. Following adjustment for confounding factors and after applying Benjamini-Hochberg false discovery rate correction to the interaction p-values, significant interaction effects remained for cerebrovascular disease, congestive heart failure, malignancy, liver disease, and sepsis (corrected p for interaction < .05). Among these subgroups, the protective effect of a higher BSS was particularly pronounced in patients with cerebrovascular disease (HR: 0.78, 95% CI: 0.74–0.81, p < .01), compared to their counterparts without these conditions (HR: 0.88, 95% CI: 0.86–0.90, p < .01).
Subgroup analysis for BSSs and risk of 30-day mortality.
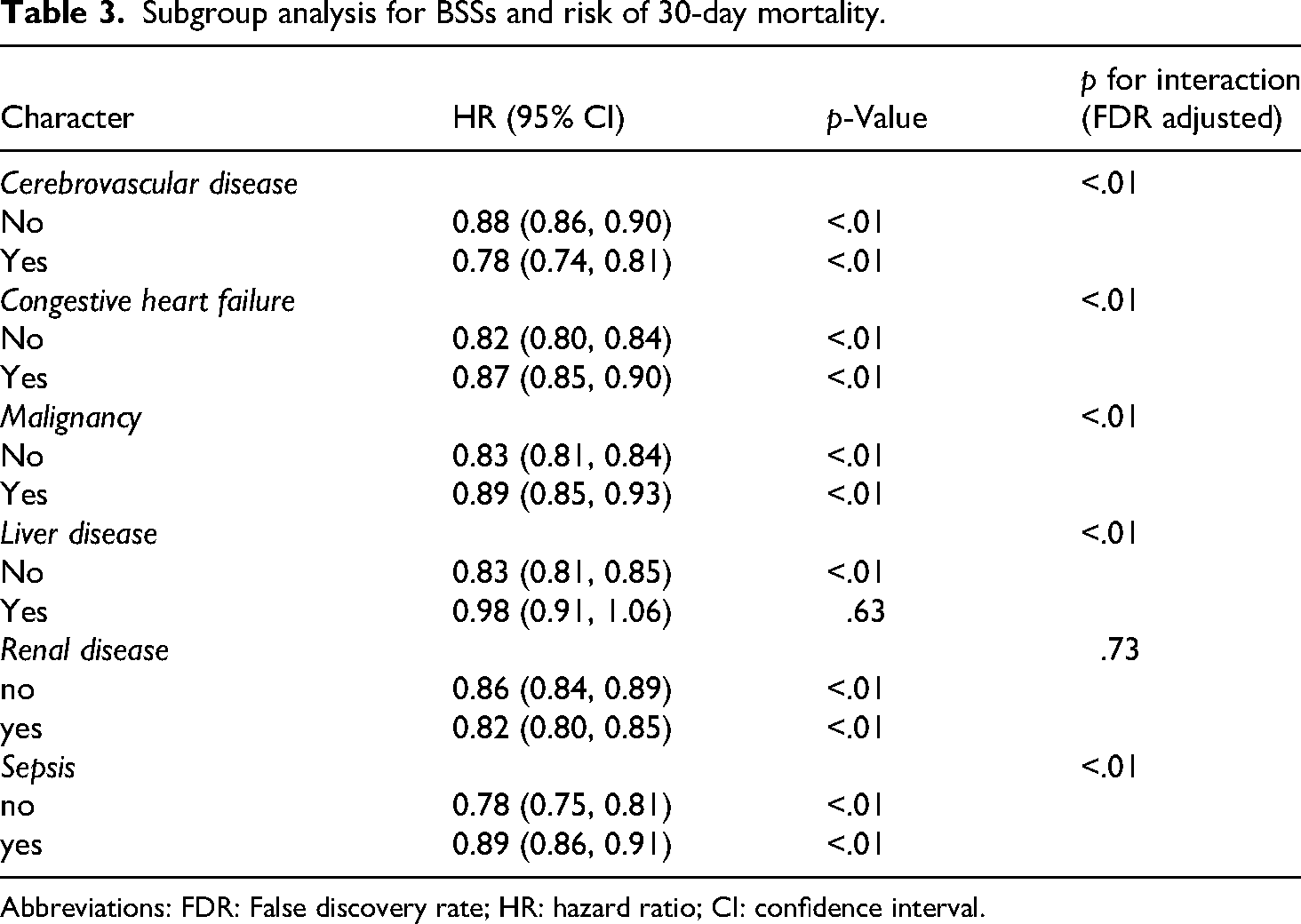
Abbreviations: FDR: False discovery rate; HR: hazard ratio; CI: confidence interval.
Discussion
To our knowledge, this represents the first investigation to specifically examine the relationship between BSS at admission and adverse outcomes in critically ill patients with diabetes. Using a large publicly available medical database, we conducted a retrospective cohort study to evaluate this association. After adjusting for various covariates, time-dependent Cox regression results showed that both linear and grouped BSS suggested a monotonic association between lower initial BSS and increased risk of short-term mortality. Subgroup analyses further confirmed the robustness of these conclusions among patients with varying characteristics. These findings underscore the significance of the BSS in clinical nursing practice. It can be utilized not only for assessing the risk of pressure ulcers but also as a tool for evaluating the short-term mortality risk of diabetes patients in the ICU.
The BSS is extensively employed in nursing practice to assess patients’ susceptibility to pressure injury. As part of standard clinical protocol, the majority of critically ill patients receive BSS evaluations shortly after ICU admission, enhancing its feasibility and integration into routine care. Recently, a retrospective cohort study focusing on cardiac intensive care unit (CICU) patients identified the BSS as an independent predictor of hospital and post-discharge mortality. 13 Similarly, a retrospective study involving 28,312 adult ICU patients diagnosed with sepsis found that reduced BSS values were strongly associated with significantly higher 90-day and 180-day mortality rates. 14 Our results are consistent with these earlier findings, further supporting the potential prognostic value of the BSS in critically ill populations. Nevertheless, evidence regarding the predictive utility of the BSS specifically for diabetic patients in the ICU remains limited. This study specifically investigates the critically ill diabetic population within the ICU, precisely evaluating short-term all-cause mortality at 30, 60, and 90 days rather than relying on conventional in-hospital mortality endpoints. Furthermore, the study also revealed a novel finding that the predictive utility of the BSS exhibits substantial variation across different comorbidity subgroups. Such investigations may help refine critical care practices and improve patient outcomes.
This study demonstrates a consistent monotonic inverse relationship between BSSs and short-term mortality risk in critically ill patients with diabetes. Several pathophysiological mechanisms may underlie this clinically significant association. First, the BSS serves as a multidimensional composite marker of frailty. It provides a multidimensional assessment of the patient's potential risk for pressure injuries and indirectly reveals the patient's overall health status, including sensory perception, mobility, and nutritional status.15,16 The score's components directly assess critical domains of patient vulnerability: sensory perception reflects neurological integrity and consciousness level, physical activity indicates mobility and functional status, moisture captures tissue perfusion and incontinence, and nutrition reflects catabolic state and metabolic reserve. A low BSS effectively identifies patients with multiple underlying impairments. Reduced mobility predisposes to adverse events such as hypostatic pneumonia, pressure injuries, deep vein thrombosis, and pulmonary embolism. 17 Poor nutritional status is linked to immune dysfunction and impaired recovery, 18 while skin moisture suggests tissue edema, incontinence, or abnormal perfusion, which collectively represent core determinants of adverse outcomes in critical illness. Consequently, a low BSS effectively identifies patients with exhausted physiological compensatory mechanisms, making them exceptionally vulnerable to progressive organ dysfunction and death. This pathophysiological state explains its powerful prognostic value for mortality beyond mere skin breakdown risk. More importantly, in diabetic patients, the frailty captured by BSS synergistically interacts with diabetes-specific pathophysiology. Diabetic patients are at high risk for pressure injuries and impaired wound healing. Diabetes compromises tissue viability and impairs defense through chronic microcirculatory impairment, immune dysfunction, and a persistent pro-inflammatory state.19–22 A low BSS, indicating critical reductions in mobility and nutritional status, acts as an amplifier of diabetic pathology. Immobility induces and exacerbates insulin resistance, creating a vicious cycle of profound hyperglycemia. 23 This hyperglycemic state further damages the vascular endothelium, worsens microcirculatory flow, and compromises neutrophil function.24,25 Simultaneously, the inadequate nutrition and reduced protein intake inherent in a low score accelerate catabolism, depleting essential metabolic reserves necessary for stress response.26,27 This study found that the SII was higher in the high-risk group than in the low-risk group, revealing an association between BSS and the level of systemic inflammation in patients with diabetes. Elevated SII reflects neutrophilic hyperactivation, lymphocytopenia, and thrombocytosis, indicating a state of systemic inflammatory hyperresponsiveness, immune exhaustion, and hypercoagulability. Activated platelets and neutrophils form aggregates that obstruct capillaries, exacerbating hypoperfusion in tissues, including the skin, which directly corresponds to low BSS. In patients with diabetes, the impairment of skin barrier function in individuals with low BSS increases pathogen translocation and infection risk, aligning with high SII. The immune dysfunction associated with low BSS may be one of the underlying mechanisms contributing to short-term mortality in critically ill patients, although future prospective studies with more comprehensive inflammatory marker data are needed to explore this further. 28 Finally, the combined effect of frailty and diabetic pathology drives the central pathway to mortality through heightened infection susceptibility, impaired wound healing, and multi-organ failure. This explains the particularly strong predictive power of BSS for mortality risk in patients with diabetes.
Despite observing a stable and significant inverse correlation between the BSS and mortality in the overall population, the study hypothesized that this predictive potency might vary across patients with different comorbidities, given the heterogeneity of the ICU patient cohort. To investigate this, this study performed stratified analyses. These analyses revealed that the BSS demonstrated a stronger predictive power and protective effect against mortality in subgroups of patients with cerebrovascular disease. This enhanced association in patients with cerebrovascular disease may be attributed to a synergistic effect between the multidimensional assessment of BSS and the underlying neuropathological state. Due to pre-existing neurological conditions, these patients often exhibit a higher prevalence of impaired consciousness, limb paralysis, sensory impairment, and dysphagia. 29 These factors directly contribute to severely restricted mobility, diminished sensory perception, increased moisture (due to potential incontinence or drooling secondary to dysphagia), and poorer nutritional status, all of which are core components assessed by the BSS. Consequently, a low BSS in this population primarily reflects an accurate quantification of the severity of pre-existing neurological deficits. Beyond merely assessing neurological impairment, the BSS also acts as a sensitive indicator of systemic physiological derangement. Subscales such as Nutrition and Moisture may sensitively reflect the systemic pathophysiological state secondary to neurological injury. For instance, skin moisture may indicate autonomic dysfunction leading to abnormal sweating or tissue edema, while a poor nutrition score may reflect dysphagia, a hypermetabolic state, or feeding intolerance. Thus, a very low BSS may signal a transition from localized neurological injury to a critical phase of combined multi-system decompensation, which is directly linked to extremely high mortality risk. Furthermore, in these vulnerable patients, the marginal benefit derived from proactive nursing interventions, such as regular repositioning, meticulous skin care, and nutritional support, is likely to be substantially greater. Consequently, a higher BSS, indicative of a better baseline condition and responsiveness to care, is associated with a more pronounced survival advantage in cerebrovascular disease population. A higher BSS indirectly reflects adequate nursing care and effective complication management, which translates into a lower mortality rate.
Subgroup analysis further revealed that among patients with sepsis, congestive heart failure, malignancies, and liver disease, the hazard ratios in those with comorbidities were lower than in those without, and the protective effect of the BSS was weaker compared to patients with neurological diseases. This differential pattern suggests that the predictive performance of BSS fundamentally varies across comorbid conditions. The core dimensions assessed by BSS relate to functional dependency, including mobility, activity, and nutritional status. However, this study found that the prognostic association of BSS was attenuated in patient groups whose mortality risk stems primarily from intrinsic metabolic disturbances or acute physiological decompensation. For instance, mortality in heart failure and renal disease is largely driven by systemic burdens such as electrolyte imbalances, volume overload, and uremic toxin accumulation. In sepsis, outcomes are predominantly determined by the severity of septic shock and multiorgan failure. Mortality in malignancies is often linked to tumor progression, metastasis, and treatment-related complications. These key pathophysiological processes are not directly captured by BSS assessment items, which may lead to an underestimation of true disease severity and mortality risk in these populations. In contrast, BSS demonstrated the strongest prognostic value in patients with neurological diseases. This may be because impairments such as sensorimotor dysfunction, autonomic dysregulation, and prolonged immobility are not only direct risk factors for pressure injuries but are also closely tied to overall functional status and prognosis. In these patients, the BSS likely serves not only as an indicator of skin risk but also as a composite proxy for global neurological deficit and dependency level. Thus, it is proposed that the prognostic significance of BSS as a functional risk assessment tool depends on the underlying disease profile. While BSS is a strong prognostic indicator in neurovascular patients, its utility is more limited in patients whose mortality is driven mainly by metabolic, systemic, or acute organ failure, such as those with heart failure, renal disease, sepsis, or malignancies. In such populations, risk stratification should not rely excessively on BSS alone but should integrate additional markers reflecting specific organ dysfunction and physiological disturbance for comprehensive assessment.
The study has several strengths. First, leveraging the MIMIC-IV database, this study included 20,590 diabetic patients, providing high statistical power and enhancing the generalizability of our findings in the critical care setting. Second, this is the first study to investigate the prognostic value of the BSS specifically in ICU patients with diabetes. Additionally, the identified stronger protective effect of the BSS in subgroups such as patients with cerebrovascular disease offers valuable insights for risk stratification and personalized management. However, several limitations should be acknowledged. First, as a retrospective observational cohort study, it can demonstrate association but not causation between the BSS and short-term mortality risk. Second, despite rigorous adjustment for available covariates, residual confounding cannot be entirely ruled out. The duration of diabetes was unavailable in the database, while other key indicators of diabetes severity and control, including HbA1c and BMI, were largely missing due to inconsistent recording. Their absence or high proportion of missing values may introduce residual confounding. Third, since mortality pathways differ substantially across clinical contexts, the prognostic meaning of the BSS likely varies accordingly. This study's results reflect an aggregate association across a heterogeneous diabetic ICU population, and future studies should examine this relationship within specific diagnostic subgroups. Forth, the use of data from a single-center database may limit generalizability, as differences in ICU practices, particularly nursing care protocols across healthcare systems and countries, could affect the applicability of our results. External validation in multicenter cohorts is therefore warranted.
Conclusions
As a widely adopted and easily applicable assessment tool, the BSS exhibits significant prognostic utility in critically ill diabetic patients, with particularly pronounced predictive performance in subgroups such as individuals with cerebrovascular accidents. Nevertheless, further prospective, multicenter studies are necessary to validate these findings and to explore the underlying mechanisms.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261420612 - Supplemental material for Association between admission Braden skin score and poor prognosis in critically ill patients with diabetes mellitus: A retrospective cohort study
Supplemental material, sj-docx-1-sci-10.1177_00368504261420612 for Association between admission Braden skin score and poor prognosis in critically ill patients with diabetes mellitus: A retrospective cohort study by Wei Deng, Yuanyuan Peng, Zhen Li and Lixia Luo in Science Progress
Footnotes
Acknowledgements
Thanks to the developers who created and maintain the MIMIC database.
Ethics approval statement
This study utilized data from the MIMIC-IV database, which is de-identified and publicly available. Ethical approval for the use of this database was obtained from the Institutional Review Boards of the Massachusetts Institute of Technology and Beth Israel Deaconess Medical Center, and the requirement for individual patient consent was waived. Accordingly, the Ethics Committee of the Chongqing mental health center has confirmed that no ethical approval was required.
Authors’ contributions
WD and LL conceptualized the study. WD and ZL extracted the data. WD, YP and ZL analyzed the data. WD and YP drafted the original manuscript. LL reviewed and revised the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
