Abstract
Objective
Acute exacerbation of chronic obstructive pulmonary disease (AECOPD) is associated with high rates of intensive care unit admission and elevated mortality. While urine output (UO) has been demonstrated to predict short-term adverse outcomes in critically ill patients, there is a paucity of research on its prognostic value for in-hospital mortality in AECOPD patients. This study aims to examine the association between UO and in-hospital mortality in AECOPD patients, and to assess its potential as a non-invasive prognostic indicator.
Methods
A retrospective cohort study included 938 AECOPD patients from the Medical Information Mart for Intensive Care IV database. Patients were grouped by initial 24-h UO: low UO (≤800 mL), moderate UO (800–2500 mL, reference), and high UO (≥2500 mL). Restricted cubic spline regression, receiver operating characteristic curves, Kaplan–Meier (K-M) survival analysis, Cox proportional hazards regression models, subgroup analysis, and mediation analysis evaluated UO's association with mortality.
Results
In-hospital mortality was 16.2%. UO showed a U-shaped non-linear association with mortality (P for nonlinearity < 0.05), with the lowest risk at 1375–4988 mL/24 h (hazard ratio
Conclusion
A U-shaped association exists between initial 24-h UO and short-term mortality in AECOPD. UO is an important but non-independent prognostic marker, functioning as a surrogate for overall disease severity. Its clinical application requires corroboration with other clinical markers.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a heterogeneous respiratory disorder characterized by persistent respiratory symptoms and airflow limitation. 1 Statistically, COPD ranks as the fourth leading cause of mortality worldwide and has emerged as a significant and escalating global health concern, imposing a substantial societal and economic burden. 2 It is projected that by 2060, more than 540,000 individuals will die annually from COPD and its related conditions. 3 Acute exacerbation of COPD (AECOPD) is defined as an acute worsening of respiratory symptoms and airflow limitation triggered by various factors. 3 This exacerbation leads to a marked decline in lung function, impaired quality of life, and an increased risk of mortality. 4 Hospitalized patients with AECOPD typically exhibit high rates of intensive care unit (ICU) admission, ranging from 10% to 25%,5–7 and mortality rates varying between 20% and 40%.8,9 Therefore, the early identification of AECOPD patients admitted to the ICU who are at high risk of mortality is of paramount importance for optimizing clinical management, facilitating timely interventions, and ultimately reducing mortality rates.
To address this critical need for early risk stratification, numerous multifactorial prognostic models have been developed and validated to assess mortality risk in patients with AECOPD.10,11 Among the most established tools is the DECAF score, which has been robustly validated for predicting in-hospital mortality based on clinical and laboratory parameters.12,13 Recognizing the unique challenges of specific patient subgroups, researchers have also refined these tools for high-risk cohorts, such as developing the v-DECAF score for patients requiring invasive mechanical ventilation (MV), 14 a population that continues to be a focus of recent prognostic research. Alongside these scoring systems, international guidelines, notably from the Global Initiative for Chronic Obstructive Lung Disease (GOLD), 1 provide comprehensive management frameworks. However, the clinical utility of these established models can be limited by a reliance on parameters that may not be immediately available upon admission, while broad guidelines offer less specific direction for dynamic, real-time risk assessment in the ICU. This highlights a need for simple, readily available indicators for immediate risk assessment.
24-h urine output (UO), a routine monitoring indicator in intensive care medicine, is uniquely positioned to fill this gap. It is dynamic, continuous, and cost-effective, reflecting systemic tissue perfusion and renal function. It demonstrates significant predictive value for short-term adverse outcomes in various critical illnesses, including sepsis, 15 acute myocardial infarction, 16 acute kidney injury (AKI), 17 acute respiratory distress syndrome, 18 and pulmonary embolism. 19 Studies indicate that reduced UO is positively correlated with increased mortality in critically ill patients; however, its prognostic value in AECOPD has not been systematically evaluated. Existing research has explored the application of various biomarkers in AECOPD prognostic assessment and developed related risk prediction models.20–23 Nevertheless, compared to laboratory-dependent biomarkers, the convenience and real-time nature of UO monitoring render it more feasible in clinical practice.
To date, no studies have systematically evaluated the association between initial 24-h UO and in-hospital mortality in ICU-admitted patients with AECOPD, particularly regarding the prognostic significance of elevated UO. As a key indicator of renal function and circulatory perfusion, UO holds potential value in the early risk assessment of AECOPD. To address this gap, the present study, based on the Medical Information Mart for Intensive Care IV (MIMIC-IV) database, 24 aims to systematically assess the relationship between initial 24-h UO and in-hospital mortality in AECOPD patients, providing evidence to guide clinical decision-making in critically ill COPD patients.
Data and methods
Data sources
This study utilized the MIMIC-IV (version 3.0) database, a publicly available, single-center critical care database. 24 This database contains data from patients admitted to the Beth Israel Deaconess Medical Center (BIDMC) in Boston, Massachusetts, USA, between 2008 and 2022. All patient identifiers were removed in compliance with health data privacy standards, and the creation of the database received ethical approval from the Institutional Review Boards (IRB) at the Massachusetts Institute of Technology (MIT) and BIDMC. Consequently, the requirement for individual patient informed consent was waived. Researchers were required to complete human research protection training provided by the National Institutes of Health and obtain certification (Certification ID: 13821916) before accessing the database. The conduct of this study adhered strictly to the ethical principles outlined in the Declaration of Helsinki, and this report was prepared in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology statement. 25
Study population
This study screened patients diagnosed with AECOPD according to the International Classification of Diseases, Tenth Revision (ICD-10) codes J44.0 and J44.1 from the MIMIC-IV database. The inclusion criteria were as follows: (1) Age ≥18 years; (2) ICU stay ≥24 h; (3) availability of UO records (mL/24 h) within the first 24 h of admission. Only patients with their first ICU admission were included, and those without UO data within the first 24 h were excluded (Figure 1). Following the application of the inclusion and exclusion criteria, a final cohort of 938 patients was included. As this was a retrospective cohort study using a pre-existing database, no a priori sample size calculation was performed; the sample size was determined by the number of eligible records.

Study design and analysis flowchart. LUO ≤ 800 mL; MUO: 800–2500 mL; HUO ≥ 2500 mL. RCS: restricted cubic spline; ROC: receiver operating characteristic; K-M: Kaplan–Meier.
Data extraction
This study extracted raw data from the MIMIC-IV database using Navicat Premium (version 15.0). The extracted data included demographic information, survival data, vital signs, laboratory tests, comorbidities, scoring systems, UO, medication use, and interventions. Specific variables were as follows: (1) Demographic information: Age, gender, weight, height, ICU and hospital length of stay, 28-day survival time, and outcomes; (2) Vital signs: Heart rate (HR), mean arterial pressure (MAP), systolic blood pressure (SBP), diastolic blood pressure (DBP), respiratory rate (RR), body temperature, and oxygen saturation (SpO2); (3) Laboratory tests: White blood cell count (WBC), white blood cell differential counts (including neutrophils, lymphocytes, and monocytes), red blood cell count (RBC), platelet count (PLT), hemoglobin (HGB), hematocrit (HCT); serum potassium (K), serum sodium (Na), lactate, creatinine (Cr), and blood urea nitrogen (BUN); (4) Comorbidities: congestive heart failure, hypertension, diabetes, myocardial infarction, peptic ulcer disease, respiratory failure, malignant tumors, and AKI; (5) Scoring systems: Sequential Organ Failure Assessment (SOFA) score, Simplified Acute Physiology Score II (SAPSII), Acute Physiology and Chronic Health Evaluation III (APSIII) score, Glasgow Coma Scale (GCS), and Charlson Comorbidity Index (CCI). Additional data included UO, MV, MV hours, invasive MV, loop diuretic (furosemide) use, and continuous renal replacement therapy (CRRT). Initial 24-h UO ≥ 2500 mL was defined as high UO (HUO), 800–2500 mL as the reference group (moderate UO, MUO), and ≤ 800 mL as low UO (LUO).
Outcome definitions
The primary outcome of this study was in-hospital mortality, with secondary outcomes including ICU mortality and 28-day all-cause mortality.
Statistical analysis
The Kolmogorov–Smirnov test was used to assess the normality of data distribution. Normally distributed continuous variables were expressed as mean ± standard deviation (Mean ± SD) and compared using the independent samples t-test. Non-normally distributed variables were presented as median (interquartile range, IQR) and analyzed using the Kruskal–Wallis H test. Categorical variables were reported as percentages and compared using the chi-square test.
Restricted cubic spline (RCS) regression models evaluated the non-linear relationship between UO and clinical outcomes. The predictive ability of UO for mortality risk was assessed using receiver operating characteristic (ROC) curves and the area under the curve (AUC). Kaplan–Meier (K-M) survival analysis estimated the survival rates at different time points, with differences in survival curves compared using the log-rank test. Multivariable Cox proportional hazards (Cox) regression models were applied to assess the association between UO and short-term mortality, with four models constructed: Model 1, unadjusted; Model 2, adjusted for age and gender; Models 3 and 4, progressively adjusted for laboratory parameters, vital signs, and comorbidities to evaluate the independent effect of UO on in-hospital mortality. Model results were reported as hazard ratios (
Additionally, exploratory subgroup analyses were performed to further investigate potential differences between specific populations. Mediation analysis was conducted to evaluate the effects via the mediators, including Cr, BUN, CGS, AKI, and CRRT.
Handling of missing data was performed in two steps. First, any variable with a missing data rate exceeding 20% was excluded from the analysis. For the remaining variables, we employed Multivariate Imputation by Chained Equations to perform multiple imputations. This iterative method was chosen to appropriately account for the uncertainty of missing data while preserving the relationships between variables. Details of the variables with missing data and their respective proportions are provided in Supplemental Table S1. Other data were derived from the mean of multiple measurements within the first 24 h of ICU admission.
Data processing and analysis were performed using R software (version 4.3.3) and Zstats 1.0(www.zstats.net). All statistical tests were conducted using a two-tailed approach, with P < 0.05 considered statistically significant.
Results
Baseline characteristics of the included participants
A total of 938 AECOPD patients were included. Table 1 presents the baseline characteristics associated with in-hospital mortality. The median age was 72 years (IQR 65–80), with 51.17% male patients. The median initial 24-h UO was 1395 mL/24 h (IQR 896–2250). Compared with the survivor group, the non-survivor group had higher APS III scores (median 57 (IQR 45–72.25) vs 41 (IQR 32–52), P < 0.05). The survivor group had a significantly higher median UO than the non-survivor group (1450 mL vs. 1035 mL, P < 0.001). Several baseline characteristics, including age, HR, RR, body temperature, DBP, HCT, PLT, HGB, pH, BUN, Cr, and AKI, significantly influenced in-hospital mortality (P < 0.05) (Table 1). Non-survivors were more likely to receive invasive MV and CRRT (P < 0.001). Supplemental Table S2 presents the baseline characteristics associated with ICU mortality. Compared with the survivor group, the non-survivor group had higher serum sodium levels (P < 0.05), while other baseline characteristics were similar to those in Table 1. The median UO in the non-survivor group was significantly lower than in the survivor group (P < 0.05) (Supplemental Table S2). Table 2 presents the baseline characteristics of patients stratified by LUO, MUO, and HUO groups. As UO increased, age, BUN, Cr, APS III, SOFA, and SAPS II levels decreased, whereas PCO2, RBC, DBP, and MAP progressively increased. Significant differences were observed among the three groups in the prevalence of comorbidities (AKI, congestive heart failure, diabetes complications) and the use of CRRT and diuretics (P < 0.001).
Baseline characteristics of the survivors and non-survivors groups.

LUO ≤ 800 mL; MUO: 800–2500 mL; HUO ≥ 2500 mL. HR: heart rate; RR: respiratory rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; MBP: mean blood pressure; WBC: white blood cell; RBC: red blood cell; PLT: platelet; HCT: hematocrit; HGB: hemoglobin; PaO2: partial pressure of arterial oxygen; PaCO2: partial pressure of arterial carbon dioxide; pH: potential of hydrogen; BUN: blood urea nitrogen; Cr: creatinine; APS III: acute physiology score III; SOFA: sequential organ failure assessment; SAPSII: simplified acute physiology score II; GCS: glasgow coma scale; CKD: chronic kidney disease; AKI: acute kidney injury; AKI stage: acute kidney injury stage; MV: mechanical ventilation; MV hours: mechanical ventilation hours; Invasive MV: invasive mechanical ventilation; CRRT: continuous renal replacement therapy; LUO: low urine output; MUO: moderate urine output; HUO: high urine output.
Baseline characteristics of patients grouped by urine output.
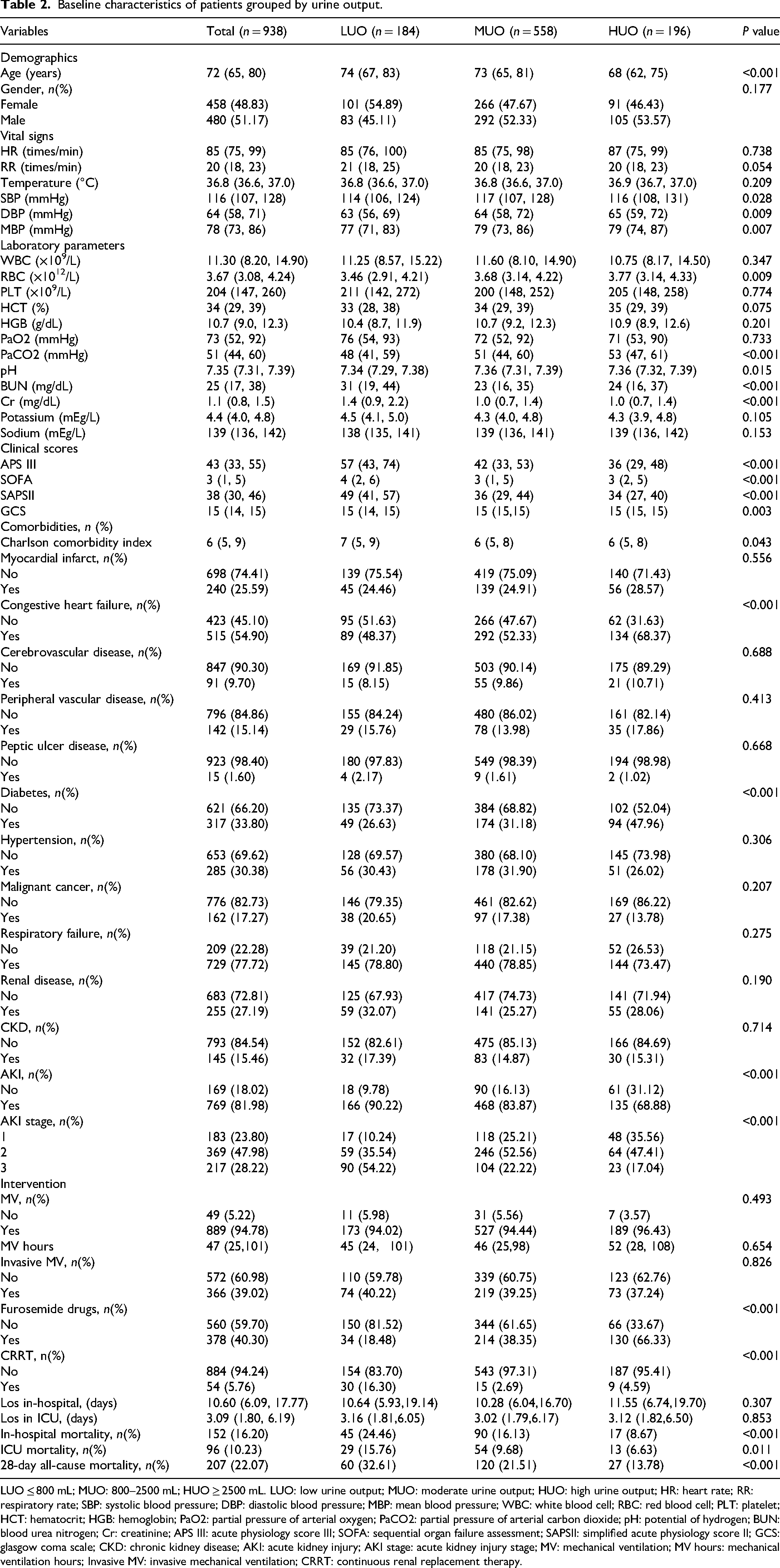
LUO ≤ 800 mL; MUO: 800–2500 mL; HUO ≥ 2500 mL. LUO: low urine output; MUO: moderate urine output; HUO: high urine output; HR: heart rate; RR: respiratory rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; MBP: mean blood pressure; WBC: white blood cell; RBC: red blood cell; PLT: platelet; HCT: hematocrit; HGB: hemoglobin; PaO2: partial pressure of arterial oxygen; PaCO2: partial pressure of arterial carbon dioxide; pH: potential of hydrogen; BUN: blood urea nitrogen; Cr: creatinine; APS III: acute physiology score III; SOFA: sequential organ failure assessment; SAPSII: simplified acute physiology score II; GCS: glasgow coma scale; CKD: chronic kidney disease; AKI: acute kidney injury; AKI stage: acute kidney injury stage; MV: mechanical ventilation; MV hours: mechanical ventilation hours; Invasive MV: invasive mechanical ventilation; CRRT: continuous renal replacement therapy.
Clinical outcomes
Analysis of clinical outcomes in AECOPD patients revealed an in-hospital mortality rate of 16.20%, with ICU mortality and 28-day all-cause mortality rates of 10.23% and 22.07%, respectively (Table 2). The mean length of stay was 10.6 days in the hospital and 3.09 days in the ICU. The key finding was a significant association between UO levels and prognosis: when grouped by increasing UO levels (LUO, MUO, HUO), in-hospital mortality showed a significant gradient decline at 24.46%, 16.13%, and 8.67%, respectively (P < 0.001). This negative correlation trend of decreasing mortality with increasing UO was also evident in ICU mortality and 28-day all-cause mortality (Table 2).
RCS regression modes and ROC curve analysis
RCS regression models analyzed the association between UO and mortality rates. Figure 2(a)–(c) shows a significant nonlinear association (U-shaped curve, P for nonlinearity < 0.05) between UO and in-hospital, ICU, and 28-day all-cause mortality. When UO ranged from 1375 and 4988 mL/24 h, the risk of in-hospital mortality was significantly reduced (

RCS and ROC curves for urine output and mortality in AECOPD patients. (a–c) RCS plots illustrating the non-linear associations between urine output (mL/24 h) and mortality risks (in-hospital, ICU, and 28-day all-cause) in AECOPD patients. (d–f) ROC curves evaluating the predictive performance of urine output for mortality outcomes (in-hospital, ICU, and 28-day all-cause) in AECOPD patients. RCS: restricted cubic spline; ROC: receiver operating characteristic; HR: hazard ratio; CI: confidence interval; AUC: area under the curve; AECOPD: acute exacerbation of chronic obstructive pulmonary disease.
ROC curve analysis was utilized to assess the prognostic value of UO for in-hospital, ICU, and 28-day all-cause mortality. The AUC for UO was 0.62 for in-hospital and 28-day all-cause mortality, and 0.61 for ICU mortality, indicating limited predictive value (Figure 2(d)–(f)). To assess for incremental prognostic value, we integrated UO with the SOFA score; however, this yielded only a marginal improvement in model discrimination (AUC increased to 0.65; Supplemental Figure 1). This limited enhancement is likely attributable to the inherent collinearity between these markers, as the SOFA renal subscore is itself defined by UO. ROC curve analysis identified optimal UO cut-off points of approximately 1123 mL/24 h for in-hospital mortality (sensitivity 32%, specificity 46%) and 1048 mL/24 h for ICU mortality (sensitivity 31%, specificity 32%) (Supplementary Table S3).
K-M curves and Cox proportional hazards regression models
K-M curve analysis for in-hospital survival revealed that the HUO group had significantly higher survival rates than the LUO group (P < 0.05) (Figure 3). ICU and 28-day survival curves exhibited the same trend (Figure 4). To further explore the factors associated with UO and in-hospital mortality in AECOPD patients, Cox regression models were constructed (Table 3). Across four models,
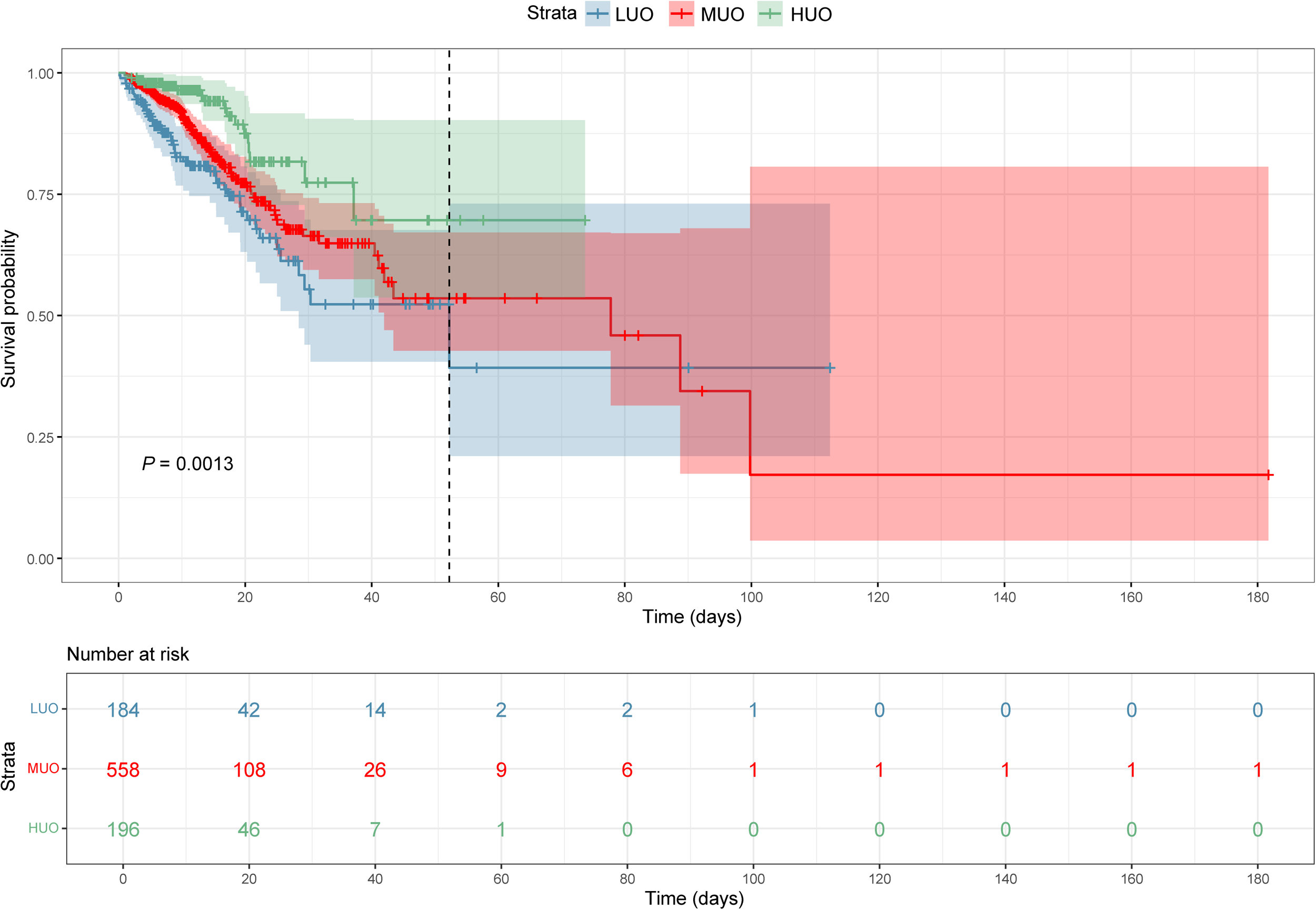
The Kaplan–Meier survival curve for in-hospital mortality. LUO ≤ 800 mL; MUO: 800–2500 mL; HUO ≥ 2500 mL. LUO: low urine output; MUO: moderate urine output; HUO: high urine output.

The Kaplan–Meier survival curve of ICU mortality (a) and 28-day all-cause mortality (b). LUO ≤ 800 mL; MUO: 800–2500 mL; HUO ≥ 2500 mL. LUO: low urine output; MUO: moderate urine output; HUO: high urine output.
Cox proportional hazards models for urine output and mortality outcomes.

LUO ≤ 800 mL; MUO: 800–2500 mL; HUO ≥ 2500 mL. Model 1 was unadjusted; Model 2 was adjusted for gender and age; Models 3 was adjusted for gender, age, blood urea nitrogen, creatinine, hematocrit, hemoglobin, potassium, sodium, white blood cell, red blood cell, platelet, heart rate, respiratory rate, temperature, acute physiology score III, sequential organ failure assessment, simplified acute physiology score II, glasgow coma scale. Model 4 was adjusted for gender, invasive mechanical ventilation, myocardial infarct, congestive heart failure, peripheral vascular disease, cerebrovascular disease, peptic ulcer disease, diabetes, renal disease, malignant cancer, hypertension, respiratory failure, chronic kidney disease, Furosemide drugs, age, blood urea nitrogen, creatinine, hematocrit, hemoglobin, potassium, sodium, white blood cell, red blood cell, platelet, heart rate, respiratory rate, temperature, acute physiology score III, sequential organ failure assessment, simplified acute physiology score II, glasgow coma scale. HR: hazard ratio; CI: confidence interval; LUO: low urine output; MUO: moderate urine output; HUO: high urine output; Ref: reference.
Subgroup analysis and mediation effect analysis
A subgroup analysis for UO risk stratification was performed in patients with AECOPD across eight subgroups: age, gender, diabetes, hypertension, AKI, invasive MV, loop diuretic (furosemide) use, and CRRT. The analysis revealed no significant interaction between UO and these subgroups (P > 0.05), indicating the stability of UO's effect (Figure 5). Across most AECOPD subgroups, HUO was associated with lower in-hospital mortality. In the mediation analysis of Cr, BUN, GCS, AKI, and CRRT, only GCS showed a significant mediation effect (P < 0.05), accounting for 8.68% (Figure 6). The other variables did not exhibit a significant effect (Supplementary Table S4).

Subgroup analysis of urine output for in-hospital mortality. LUO ≤ 800 mL; MUO: 800–2500 mL; HUO ≥ 2500 mL. HR: hazard ratio; CI: confidence interval; LUO: low urine output; MUO: moderate urine output; HUO: high urine output; Ref: reference; AKI: acute kidney injury; Invasive MV: invasive mechanical ventilation; CRRT: continuous renal replacement therapy.

Mediation analysis of GCS on the association between urine output and in-hospital mortality. ACME: average causal mediation effect; ADE: average direct effect; GCS: Glasgow coma scale; UO: urine output.
Discussion
This study conducted a retrospective analysis of 938 patients with AECOPD admitted to the ICU using the MIMIC-IV database, aiming to investigate the relationship between initial 24-h UO and short-term prognosis. The results revealed a significant U-shaped non-linear association between UO and in-hospital, ICU, and 28-day all-cause mortality, with UO in the range of 1375–4988 mL associated with the lowest mortality risk. This finding reveals a dual-risk paradigm, where both oliguria (associated with a higher mortality risk) and excessive polyuria (also correlated with an increased risk) represent deviations from the physiological norm. The range of lowest associated risk should therefore be interpreted as a zone reflecting relative physiological homeostasis, not as a therapeutic target. Values below this range may signal traditional hypoperfusion or AKI, whereas excessive output may signify more complex states like the resolution phase of inflammation or iatrogenic fluid overload.26,27 These findings highlight the clinical utility of initial 24-h UO as a simple bedside indicator, where its U-shaped association with mortality provides critical, dual-directional signals for early risk stratification in patients with AECOPD.
COPD is a progressive inflammatory disorder, with a pathogenesis closely tied to inflammation, oxidative stress, and protease-antiprotease imbalance.2,28–30 AECOPD is commonly triggered by respiratory infections, leading to intensified airway and systemic inflammation.4,31,32 Inflammatory mediators (e.g. TNF-α, IL-6) contribute to lung-kidney crosstalk. This crosstalk, when synergized with hypoxemia and hemodynamic instability, can cause renal tubular damage and a reduced glomerular filtration rate, significantly increasing the risk of AKI.33,34 Our study's finding of a U-shaped association between UO and mortality underscores the critical role of both inflammation and renal function in AECOPD prognosis. However, over 50% of data for key inflammatory markers, such as neutrophil counts and C-reactive protein (CRP), were missing in the MIMIC-IV database. This significant limitation precluded a full elucidation of the role of inflammation. Specifically, the absence of these data has three key implications for the interpretation of the UO-mortality relationship. Inflammation, particularly in the context of systemic inflammatory response syndrome (SIRS), acts as a critical initiator in the causal pathway: severe inflammation induces renal hypoperfusion, which can lead to AKI, ultimately manifesting as low UO.35,36 This mechanistic sequence underscores the role of inflammation as a key mediator. At the same time, inflammation also serves as a potent unmeasured confounder, as it can simultaneously cause low UO (via renal hypoperfusion) and increase mortality through multi-organ injury, thereby distorting the observed association between UO and outcomes.37,38 Furthermore, the prognostic value of UO is likely modified by the inflammatory state—its utility as a marker of hemodynamic stability may be diminished in hyper-inflamed patients but more evident in those with milder inflammation.36,39
AKI is a common complication in ICU-admitted AECOPD patients, significantly associated with higher mortality risk.34,35,40 Clinically, the presence of oliguria or anuria at admission may increase the risk of AKI in AECOPD patients. Our study found that the initial 24-h UO was closely related to AKI incidence and in-hospital mortality (P < 0.05). The association with in-hospital mortality remained significant even after adjusting for confounders such as AKI, CRRT use, and diuretic treatment (P < 0.05). This finding aligns with a study on sepsis shock patients, which demonstrated that initial 24-h UO serves as an independent prognostic factor, unaffected by AKI or therapeutic interventions. 15 These results highlight the critical role of UO monitoring in the clinical management of AECOPD patients, facilitating early identification of high-risk individuals and optimization of treatment strategies.
Typically, studies regard oliguria (<400 mL/24 h) as a danger signal, 41 indicating renal hypoperfusion or renal dysfunction, commonly an early manifestation of AKI, 17 and strongly associated with poor prognosis in critically ill patients.16,18,42 Previous studies have confirmed: UO < 0.85 mL/kg/h is negatively correlated with in-hospital mortality in patients with acute pulmonary embolism 18 ; Li et al. found that low UO (<0.5 mL/kg/h) is an independent risk factor for 30-day mortality in CICU-admitted patients 42 ; and Bao et al. noted that initial 24 h UO < 800 mL is a robust predictor of in-hospital and 1-year mortality in patients with acute myocardial infarction. 16 Our findings are consistent with these reports, suggesting that oliguria serves as an important early warning signal for short-term prognosis in AECOPD patients. However, the specific mechanisms linking oliguria to poor outcomes in AECOPD patients remain incompletely elucidated. Existing studies propose potential mechanisms, including hypoxia and hypercapnia-induced renal vasoconstriction, reducing renal blood flow and glomerular filtration rate, thereby decreasing UO 43 ; systemic inflammation, mediated by cytokines (TNF-α, IL-6, CRP), directly damaging renal tubular epithelial cells and glomeruli while activating the renin-angiotensin-aldosterone system to exacerbate renal vasoconstriction44,45; and right heart failure, leading to decreased cardiac output and renal vein congestion, further impairing renal function.44,46
Polyuria (≥2500 mL/24 h) has significant clinical implications with a dual role in intensive care patients.15,16 Our study corroborates this complexity, revealing a U-shaped association between UO and mortality in AECOPD patients: Moderate polyuria was associated with a lower mortality risk, whereas excessive polyuria (>4988 mL/24 h) correlated with an increased risk. The pathophysiology underlying this phenomenon is closely linked to the dynamic evolution of the systemic inflammatory response, which may modulate UO through at least two distinct mechanisms. On the one hand, moderate polyuria can be viewed as a favorable sign of recovery from a state of severe stress to physiological homeostasis. It serves as a clinical manifestation of improved renal perfusion and a positive therapeutic response,47,48 consistent with our team's prior research observing an association between polyuria and clinical improvement. 49 A deeper mechanism may be the resolution of systemic inflammation. During the acute phase of critical illness, inflammatory mediators increase capillary permeability, leading to significant fluid extravasation into the interstitial space. 50 As the inflammatory response attenuates and vascular barrier function is restored, this sequestered fluid is mobilized back into the intravascular compartment, triggering a physiological diuresis.26,51 Therefore, in this clinical context, moderate polyuria is a clinical indicator of restored endothelial function and successful volume resuscitation. On the other hand, excessive polyuria may herald pathological decompensation or iatrogenic complications. In this scenario, inflammation may exert a more direct organ-damaging effect. Research has shown that systemic inflammation can directly disrupt the renal medullary osmotic gradient and interfere with antidiuretic hormone receptor signaling, leading to impaired urinary concentrating capacity and a state of acquired nephrogenic diabetes insipidus. 52 Furthermore, studies in other critically ill cohorts indicate that excessive polyuria, by causing severe fluid and electrolyte imbalances and intravascular volume depletion, serves as an independent risk factor for mortality.42,53 However, in certain circumstances, HUO may act as a protective factor for in-hospital survival. 16 These findings are consistent with our results, indicating that the clinical interpretation of UO is dependent on its dynamic and the clinical context. Moderate polyuria may represent a “window of recovery” for patients with AECOPD, but it can transform into an adverse prognostic signal if it exceeds a certain physiological threshold. Therefore, clinical practice requires dynamic UO monitoring, with fluid management strategies tailored to patient conditions to prevent potential complications. Future research should focus on identifying the optimal UO threshold and elucidating its physiological mechanisms in AECOPD to provide a foundation for personalized treatment strategies.
These findings have direct clinical implications for fluid stewardship in AECOPD. The identified range of lowest associated risk can serve as a non-invasive early clinical signal, where values outside this range should prompt urgent reassessment of volume status rather than dictating a specific intervention. However, as our study is observational, these thresholds are not therapeutic targets. The core role of UO is to guide and inform a comprehensive clinical assessment that integrates daily weights, fluid balance, and hemodynamic data. Whether actively targeting this UO range can improve outcomes must be addressed in future prospective trials.
This study first revealed a U-shaped nonlinear association between UO and short-term prognosis in AECOPD, revealing an independent association between initial 24 h UO for in-hospital and 28-day all-cause mortality. This finding provides new evidence for early identification of high-risk patients and implementation of goal-directed fluid management in clinical practice. However, this study has several important limitations that must be acknowledged. First, as a retrospective, observational study based on a single-center database (MIMIC-IV), our findings indicate statistical associations but cannot establish causality; the single-center nature also limits the external validity and generalizability of our results. Second, our analysis was constrained by the absence of granular data on key clinical variables. For instance, we could not adjust for fluid intake patterns or the precise use of diuretics, which are strong confounders; aggressive fluid resuscitation or high-dose diuretic therapy can produce a HUO that may reflect a more severe underlying state, such as fluid overload or cardiac dysfunction, potentially masking the true association between HUO and adverse outcomes. Moreover, our use of a static initial 24-h UO value does not capture the dynamic trajectory of a patient's clinical course, which may hold greater prognostic significance. Similarly, the lack of complete data for key inflammatory markers, such as CRP, precluded a quantitative assessment of the role of inflammation as a critical unmeasured confounder. Finally, the clinical interpretation of our findings requires caution. The modest discriminatory power of UO as a standalone marker (AUC = 0.62) underscores its limitation as an isolated predictor. This suggests that while UO can provide an early clinical signal, it should be integrated into multimodal assessment models to guide clinical decision-making. Therefore, future multicenter, prospective studies are warranted to validate our findings and to integrate UO with other clinical and biological data into more robust prognostic models.
Conclusion
In conclusion, a key finding of this study is the significant, U-shaped non-linear association between initial 24-h UO and short-term mortality in critically ill patients with AECOPD. This finding establishes UO as a critical and readily accessible indicator for early risk stratification. However, our analysis provides a deeper insight into the true clinical role of this indicator. The predictive performance of UO as a standalone marker was modest (AUC = 0.62). Furthermore, its association with mortality was attenuated after full clinical adjustment. These findings collectively point to a central conclusion: the prognostic value of UO is not independent, but rather stems from its role as a dynamic clinical sign that comprehensively reflects the patient's overall pathophysiological severity. Therefore, UO should be integrated into multimodal assessment models to guide clinical decision-making. Future multicenter, prospective studies are warranted to validate these findings and to develop and validate integrated prognostic models that can optimize the clinical management of patients with AECOPD.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251387235 - Supplemental material for Relationship between urine output and in-hospital mortality among patients with acute exacerbation of chronic obstructive pulmonary disease: A cohort study based on the Medical Information Mart for Intensive Care IV database
Supplemental material, sj-docx-1-sci-10.1177_00368504251387235 for Relationship between urine output and in-hospital mortality among patients with acute exacerbation of chronic obstructive pulmonary disease: A cohort study based on the Medical Information Mart for Intensive Care IV database by Saimei Nie, Bei Wang, Jianhua Yu, Hongjian Yu, Feng Liu, Jie Zhang, Hongli Yang, Wei Liu, Xiaoxi Wang and Chunyan Su in Science Progress
Footnotes
Acknowledgments
The authors express their heartfelt thanks to the MIT and BIDMC for generously providing access to the MIMIC-IV database, which has been instrumental to this research. We also acknowledge the authors of the cited works for their foundational contributions to the field. Additionally, we are grateful to the peer reviewers for their thoughtful and valuable feedback, which has significantly enhanced the quality of this manuscript. The authors acknowledge the use of an AI-assisted language tool (e.g. ChatGPT) to improve the language and clarity of the manuscript.
Ethics approval
The utilization of the MIMIC-IV database for this retrospective study was conducted in accordance with the ethical principles outlined in the Helsinki Declaration of 1975, as revised in 2024. The study protocol received ethical approval from the Institutional Review Boards (IRBs) of both BIDMC and the MIT. Given that the database consists of comprehensively de-identified patient records and the research involves no intervention and poses minimal risk to individuals, the IRBs granted an exemption for this study. Therefore, individual informed consent was waived. All patient identifiers have been removed or anonymized following strict de-identification protocols in compliance with health data privacy standards, ensuring that no individual can be identified in any way. Consequently, signed consent forms were not required for this analysis.
Authors’ contributions
Conceptualization: Bei Wang, Chunyan Su; Funding acquisition: Chunyan Su; Supervision: Jianhua Yu, Hongjian Yu; Formal analysis: Feng Liu, Jie Zhang; Investigation and data curation: Hongli Yang, Wei Liu, Xiaoxi Wang; Ethics application: Saimei Nie; Original manuscript writing: Saimei Nie; Manuscript review and editing: Chunyan Su. All authors have read and approved the published version of this manuscript and agree to be accountable for all aspects of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Key Research and Development Program of China (2021YFC1712901) and the Special Project on Clinical Evidence-based Research for the Construction of High-level Traditional Chinese Medicine Hospitals of Wangjing Hospital, China Academy of Chinese Medical Sciences (WJYY-XZKT-2023-21).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
