Abstract
Objective
Soft tissue sarcomas (STSs) are rare, heterogeneous tumors of mesenchymal origin that arise from muscle, fat, fibrous tissue, blood vessels, nerves, and connective tissue. They pose significant diagnostic and therapeutic challenges, and limited data exist on their epidemiology and outcomes in sub-Saharan Africa. This study assessed the clinicopathological characteristics and one-year survival of patients with STSs managed at a tertiary facility in Tanzania.
Methods
A retrospective cohort review was conducted of medical records from January 2019 to April 2023. Data included patient demographics, tumor site, histological subtype, grade, stage at diagnosis, treatment modalities, and follow-up. Survival was estimated using Kaplan–Meier analysis, and Cox proportional hazards regression was applied to identify predictors of one-year survival.
Results
Ninety-six cases were analyzed. Males constituted 54.2% of patients, and the majority (70.8%) were aged 15–64 years. Rhabdomyosarcoma was the most frequent histological type (34.4%), followed by sarcoma not otherwise specified (28.1%). Surgery was the predominant treatment modality. The overall one-year survival rate was 63.8% (95% CI: 48.0–71.7). Female sex (adjusted hazard ratio (HR): 3.36; 95% CI: 1.25–9.03; p = 0.016) and absence of surgery (adjusted HR: 5.57; 95% CI: 1.92–16.14; p = 0.002) were independent predictors of increased mortality. The advanced stage (Stage IV: HR = 9.37; unknown stage: HR 10.30) showed a trend toward poorer outcomes, although not statistically significant. Histological subtype was not clearly associated with mortality, though gastrointestinal stromal tumors (GISTs) trended toward improved survival (HR = 0.06; p = 0.057). Adjuvant chemotherapy was associated with increased mortality (HR = 4.39; p = 0.049), likely due to confounding by indication.
Conclusion
Approximately two-thirds of patients with STSs survive one year post-diagnosis. Prognosis is strongly influenced by sex, stage, and surgical management. These findings highlight the need for early diagnosis, appropriate surgery, and structured multidisciplinary care to improve survival outcomes in Tanzanian tertiary settings.
Keywords
Introduction
Soft tissue sarcomas (STSs) are a highly heterogeneous group of tumors of mesenchymal origin. 1 STSs are classified based on their histological origin. There are more than 70 histological subtypes of STSs, which have different clinical, prognostic, and therapeutic features. 2 Some common types include liposarcoma (originates in fat tissue), leiomyosarcoma (arises from smooth muscle cells), rhabdomyosarcoma (derived from skeletal muscle cells), angiosarcoma (originates from blood or lymphatic vessels), synovial sarcoma (frequently occurs near joints in young adults), and undifferentiated pleomorphic sarcoma (previously known as malignant fibrous histiocytoma, with no clear line of differentiation). 2 STSs are rare, accounting for <1% of adult malignant tumors. The incidence of STS is about 5 cases per 100,000 people annually. 3 The tumors can occur at any age, but certain types, such as rhabdomyosarcoma, are more common in children. 4
The risk factors for STSs are not well established. Genetic syndromes such as Li–Fraumeni syndrome, neurofibromatosis type 1, and familial retinoblastoma have been implicated. Similarly, prior radiation therapy, chronic lymphedema, and exposure to chemical exposures such as vinyl chloride and dioxins have been associated with an increased risk. 1 STS lesions have a wide range of clinical presentations, depending partly on the location and the stage of the disease. 3 Most patients present with a painless mass or swelling, often growing progressively. 5 Symptoms may arise from compression or invasion of nearby structures (e.g. pain and functional impairment). 5 Retroperitoneal sarcomas may present with nonspecific symptoms such as abdominal pain or fullness. 6
The diagnosis of STS can be challenging,6,7,8 and it is usually arrived at with different techniques.6,7 Radiological tests such as magnetic resonance imaging (MRI) are preferred for extremity and trunk lesions, while computed tomography (CT) scans are useful for retroperitoneal or thoracic tumors; positron emission tomography (PET)/CT may help in staging and assessing metastatic disease. 9 Histopathology coupled with immunohistochemistry is essential for the definitive diagnosis. 10 A core needle biopsy is preferred, but an open biopsy may be necessary in some cases. When feasible, molecular testing for specific translocations or mutations is recommended, for example, SYT-SSX in synovial sarcoma. 1 Management of STS is done through a multidisciplinary team approach involving specialized surgeons, clinical and radiation oncologists, nuclear medicine specialists, radiologists, pathologists, rehabilitation medicine specialists, palliative care specialists, oncology nurses, and others.6,11
Surgical resection is the mainstay of treatment, aiming for wide margins to minimize recurrence. 12 Preoperative or postoperative radiotherapy is useful to reduce recurrence risk. Chemotherapy is used in specific subtypes of STS, while targeted therapy is used for tumors with specific genetic alterations. 11 Survival and prognosis usually depend on the tumor's size, grade, histological subtype, location, and extent of metastasis. 5 Five-year survival rates range from 50% to 70% for localized disease but drop significantly with metastases.5,13 Studies about STS in the sub-Saharan region, including Tanzania, are rare. 14 Thus, this study was carried out to address this knowledge gap by assessing the clinicopathological features as well as the survival among patients with STS at a tertiary center in northern Tanzania.
Materials and methods
This was a hospital-based retrospective cohort study that was conducted between September 2023 and September 2024 at the Kilimanjaro Christian Medical Centre (KCMC), which is a zonal referral hospital in northern Tanzania. KCMC is a teaching hospital for KCMC University. The hospital offers general and specialized medical services, including surgery and oncology, and it serves the population of five regions living in the northern part of the country. Participants in this study included patients with histologically confirmed diagnoses of STS who obtained care at KCMC between January 2019 and April 2023. Diagnosis of STS in this cohort was established primarily through histopathological examination of biopsy or surgical specimens. Radiological imaging, including MRI for extremity and trunk lesions, CT for retroperitoneal or thoracic tumors, and PET/CT for staging where available, was used to support diagnosis and staging. Clinical biomarkers were not routinely used for diagnosis in this setting and, therefore, were not included in the analysis.
Patients with Kaposi sarcoma or those with substantial missing data were excluded. Patients were selected consecutively during the study period based on the stipulated eligibility criteria. Enrolled participants were followed for one year from the time of diagnosis through their attendance at the scheduled clinic visit via the electronic health management system (EHMS), and those not attending were contacted via the phone numbers registered in the EHMS. Study variables included one-year survival post diagnosis of STS, age, gender, histological subtype, and treatment patterns. The KCMC cancer registry was used to identify patients with STS in the study period. EHMS was used to obtain clinical characteristics and treatment patterns. All deaths at one year post diagnosis, regardless of the cause, were recorded from EHMS or as reported by the next of kin reached through mobile phone.
Descriptive statistics were performed using SPSS version 24, and Kaplan–Meier survival curves were used to estimate the survival probabilities of the patients with STS throughout time. Cox regression analysis was conducted to identify predictors of survival. Hazard ratios (HRs) and 95% confidence intervals (CIs) were reported to quantify the relationship between each predictor and survival outcome.
All patient details have been de-identified so that no individual can be identified in any way in accordance with ethical standards. The authors conducted this study in accordance with the Declaration of Helsinki (1975, revised in 2024), and the reporting of this study conforms to EQUATOR guidelines.15,16 This study was approved by the Institutional Review Board at KCMC University (approval no. PG45/2023) on 12 August 2023.
Results
Clinicopathological characteristics of patients with STSs
A total of 96 patients met the inclusion criteria and were included in the final analysis. The median age at diagnosis was 41 years, with an interquartile range of 19 to 52. There was a male predominance. Of those with a known clinical stage at presentation, the majority of them presented with stage IV disease (25.0%). Rhabdomyosarcoma and sarcoma not otherwise specified (NOS) are the most common STSs, accounting for 34.38% and 28.13%, respectively. A total of 68 out of 96 patients with STS are treated at KCMC Hospital. Among the 68 patients who received treatment, 48 (70.58%) underwent surgery and 20 (29.41%) received chemotherapy as the only therapy (Table 1).
Baseline demographic, clinicopathologic, and treatment characteristics of patients with soft tissue sarcoma (n = 96).

IQR: interquartile range; NOS: not otherwise specified; GIST: gastrointestinal stromal tumor.
Survival estimate of patients with STSs
The overall one-year survival rate is 63.83% (95% CI, 0.5123–0.7399) (Figure 1). Patients with gastrointestinal stromal tumor (GIST), fibrosarcoma, and liposarcoma show a higher survival rate (85.71% (95% CI, 0.3341–0.9786), 83.33% (95% CI, 0.2731–0.9747), and 80% (95% CI, 0.4087–0.9459)) (Figure 2). Patients with stages I, II, and III of STS show a higher survival rate of 88.89% (95% CI, 0.4330–0.9836), 76.55% (95% CI, 0.4842–0.9036), and 75.0% (0.01279–0.9605), respectively (Figure 3).

Kaplan–Meier estimate of overall survival among patients with soft tissue sarcoma.

Kaplan–Meier estimate of survival by tumor histological subtypes among patients with soft tissue sarcoma.

Kaplan–Meier estimate of survival by tumor stages among patients with soft tissue sarcoma.
Predictors of one-year survival
In the survival analysis, female sex and lack of surgical intervention emerged as independent predictors of one-year mortality. Females had more than a threefold higher risk of death compared to males (adjusted HR: 3.36; 95% CI: 1.25–9.03; p = 0.016), while patients who did not undergo surgery had over a fivefold increased risk (adjusted HR: 5.57; 95% CI: 1.92–16.14; p = 0.002). Tumor stage showed trends toward worse survival in advanced (stage IV) and unknown stages, although these did not reach statistical significance (adjusted HR = 9.37; p = 0.077; adjusted HR: 10.30; p = 0.066, respectively). Histological subtype was not significantly associated with survival, though GIST showed a non-significant trend toward improved outcomes (adjusted HR: 0.06; 95% CI: 0.01–1.08; p = 0.057). Adjuvant chemotherapy was associated with increased mortality (adjusted HR: 4.39; 95% CI: 1.00–19.24; p = 0.049), likely reflecting confounding by indication, whereas neoadjuvant chemotherapy and age groups were not significantly associated with survival outcomes (Table 2).
Unadjusted and adjusted HRs for one-year mortality among patients with soft tissue sarcoma.
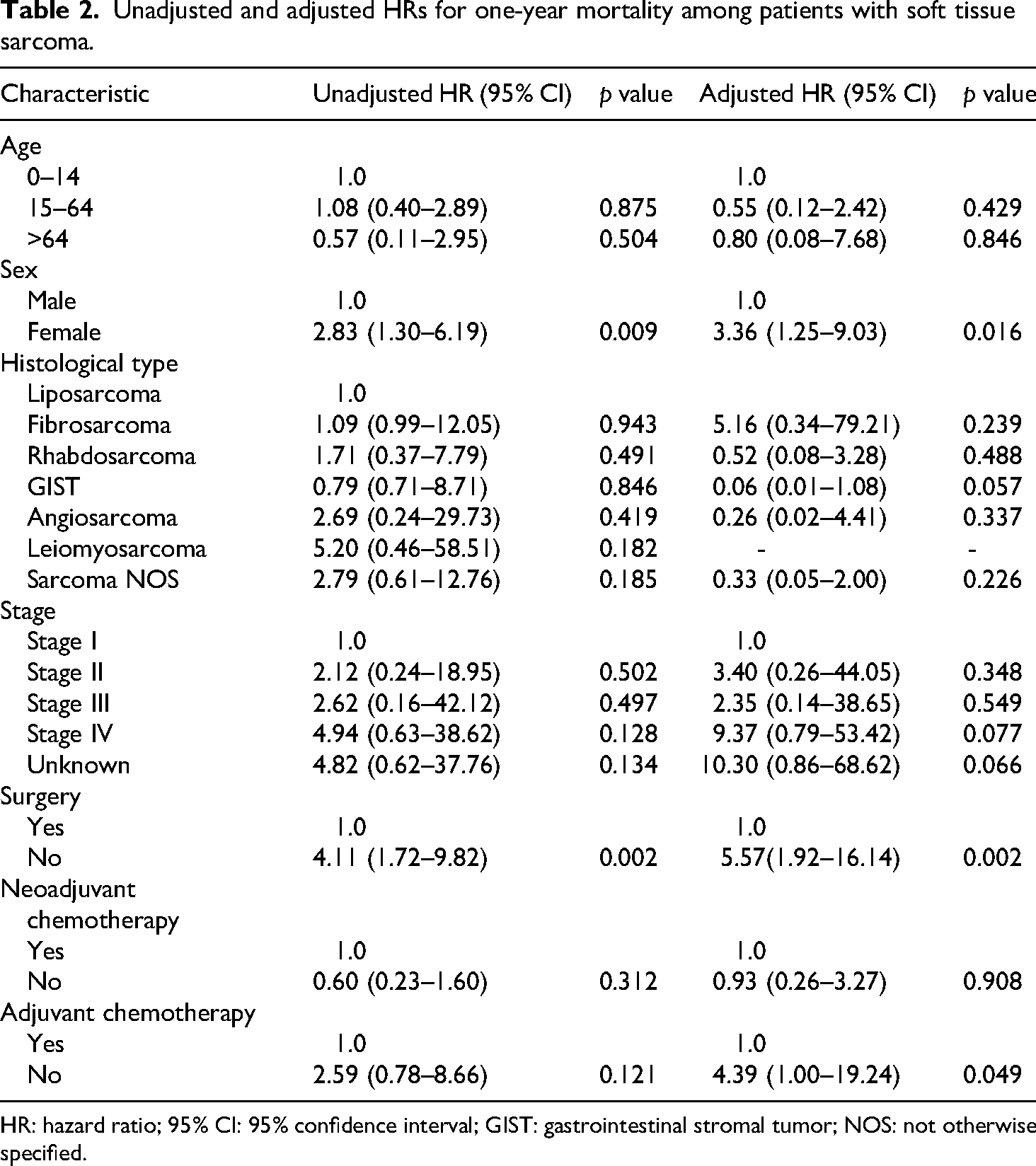
HR: hazard ratio; 95% CI: 95% confidence interval; GIST: gastrointestinal stromal tumor; NOS: not otherwise specified.
Discussion
In this study conducted at a tertiary referral center in Tanzania, we analyzed the clinicopathological characteristics of STSs and assessed one-year survival outcomes along with their predictors. Our findings highlight a relatively low one-year survival rate of 63.8%, consistent with reports from similar low- to middle-income settings. The predominant histological subtype was rhabdomyosarcoma, and the majority of patients presented with high-grade tumors and advanced disease.
To our knowledge, this is one of the first analyses in the country of clinicopathological features and one-year survival and its predictors in patients with STS. In this study, the overall one-year survival rate of patients with STS is 63.8%. The relatively higher survival rates were observed in patients with GIST, fibrosarcoma, and liposarcoma. The use of imatinib may explain the higher survival rates of patients with GIST. However, higher survival rates have been reported in previous studies. For instance, Unal et al. 17 reported a one-year survival rate of 93.4%. The less aggressive nature of fibrosarcoma and liposarcoma could partly be the reason for the higher survival rates observed. 13 The lowest survival rates were found in patients with rhabdomyosarcoma and sarcoma NOS. Rhabdomyosarcomas are well known to be aggressive STSs. 18 As the clinical stage advanced, the one-year survival rate decreased.
As expected, the patients with stage IV disease had the lowest survival rate, in line with previous studies elsewhere.19–22 The lower survival rate was seen in patients with an unknown stage. The reasons for this observation are not clear. There is a possibility that those patients who were not staged could have advanced disease. The TNM staging system for soft tissue tumors applies to all STSs of the extremities and trunk, abdomen, thoracic visceral organs, and retroperitoneum except Kaposi sarcoma, GISTs, fibromatosis (desmoid tumor), and infantile fibrosarcoma. 23 Sarcomas arising within the confines of the dura mater, including the brain, are not optimally staged by this system. There are several factors known to affect the survival rate in patients with STS. Studies have clearly established that positive surgical margins are strongly associated with adverse survival-related outcomes in localized STSs, and local recurrence has a significant impact on overall survival, but not on disease-specific survival. 24 Reference literature regarding STS in developing countries is sparse. 14 Most of the published studies are of individual case reports or case series.6–8 Much of the available literature is from the developed world, making it hard to compare our findings to theirs.25,26 However, it is important to recognize the differences between the two settings. The one-year survival of 87.8% is reported in Europe. 13 This shows that the survival rate is lower in our setting.
In our cohort, female sex was associated with increased one-year mortality. The reasons for this are not clear, and it is contrary to the previous reports. 27 The finding may reflect case-mix (histology distribution like leiomyosarcomas, site patterns, stage at presentation, or treatment access) rather than a universal sex effect. 28 While sex is not consistently identified as a prognostic factor in STS, prior studies have suggested potential biological and clinical explanations. 29 Hormonal influences, differential tumor biology, and variations in histological subtype distribution may contribute to sex-specific survival outcomes. Furthermore, disparities in referral patterns, stage at presentation, or treatment access could partially explain the observed association. 30 Given the limited and sometimes conflicting evidence in the literature, our findings highlight the need for larger multi-institutional studies to better clarify the role of sex in STS prognosis.
In our study, not being operated on and not receiving neoadjuvant chemotherapy were statistically significant observations as predictors of death in patients with STS. Nearly similar findings were reported in the previous studies. For instance, in their study, Stojadinovic et al. 31 established that high-grade, size at or above 5 cm, and deep location predict distant failure and tumor-related mortality for patients with STS. Complete surgical resection is the recommended treatment approach to achieve local control. 12 Stratification by prognostic factors will facilitate selection of patients with STS for adjuvant systemic therapies. Additionally, patients who underwent surgery were found to have a lower risk of dying at one year post-diagnosis. 26 This is possibly explained by the fact that patients undergoing surgery have localized disease, and it's curative with the intent of removing all tumors, achieving R0 resection. Though not statistically significant, it is of clinical significance to note that higher death risks are observed in patients with stage IV and unknown disease stages.
Based on our results, early surgical intervention should be prioritized in the management of STS to improve overall survival. 5 Patients with advanced or undocumented stages may represent higher-risk groups requiring closer monitoring and potentially more aggressive supportive care. 28 While histological subtype was not independently predictive, trends observed in GIST and adjuvant chemotherapy use highlight the need for individualized treatment planning. Additionally, transitioning from simple data collection to structured follow-up systems will significantly improve the accuracy and reliability of prognostic assessments in disease survival, including STS.
This study is limited by its retrospective, single-center design and relatively small sample size, which may reduce the robustness and generalizability of the findings. Furthermore, clinical biomarkers were not routinely assessed in our cohort, and their absence may have limited the depth of diagnostic information and prognostic evaluation. Another important limitation is the reliance on simple data collection in our setting; transitioning toward structured follow-up systems would enhance the accuracy and reliability of prognostic assessments in future studies. Another notable limitation of our study is the large proportion of patients with unknown stage at diagnosis (36.5%). This has affected the sample size of the subjects included in the final analysis. This primarily reflects the retrospective nature of our data and referral patterns, where some patients presented without a complete staging work-up. Because disease stage is one of the strongest prognostic factors in STS, this missing information may have introduced bias into survival estimates. It is possible that patients with undocumented stages differed systematically in disease burden compared with staged patients, which could either under- or overestimate the observed associations. Thus, future prospective, multi-center studies with larger cohorts and more robust clinical data collection are recommended to validate and expand the current study findings.
Conclusion
In this retrospective cohort of patients with STS, the majority were working-age adults, and rhabdomyosarcoma was the most common histological subtype. Surgery appeared to be associated with improved one-year survival, while female sex and lack of surgical intervention were linked to higher mortality. Tumor stage and histological subtype showed trends toward influencing survival, though not all reached statistical significance. Adjuvant chemotherapy was associated with increased mortality, likely reflecting confounding by indication. These findings may inform clinical decision-making and highlight areas for further research; however, causal conclusions cannot be drawn due to the observational design and limited sample size.
Footnotes
Abbreviations
Acknowledgments
The authors wish to thank the staff in the departments of General Surgery, Pathology, and Oncology, particularly from the Cancer Registry Unit, as well as the KCMC hospital management, for supporting this study. We also declare that an artificial intelligence-based language tool (ChatGPT) was used solely to improve the grammar, clarity, and readability of the manuscript. No AI tools were used for data collection, analysis, or interpretation.
Consent to participate
As this was a retrospective cohort study utilizing existing medical records, it was not possible to obtain informed consent from the study participants. This study was approved by the Institutional Review Board at KCMC University (approval no. PG45/2023) on 12 August 2023, which waived the requirement for informed consent.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science for Africa Foundation for the Developing Excellence in Leadership, Training, and Science in Africa (DELTAS Africa) program (Del-22-008) with support from Wellcome Trust and the UK Foreign, Commonwealth, and Development Office, and is part of the EDCPT2 program supported by the European Union.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
