Abstract
Background
The clinical use of transthoracic echocardiography (TTE) in patients with acute respiratory distress syndrome (ARDS) in the intensive care unit (ICU) has dramatically increased, its impact on long-term prognosis in these patients has not been studied. This study aimed to explore the effect of early-TTE on long-term mortality in patients with moderate-to-severe ARDS in ICU.
Methods
A total of 2833 patients with moderate-to-severe ARDS who had or had not received early-TTE were obtained from the Medical Information Mart for Intensive Care (MIMIC-III) database after imputing missing values by a random forest model, patients were divided into early-TTE group and non-early-TTE group according to whether they received TTE examination in ICU. A variety of statistical methods were used to balance 41 covariates and increase the reliability of this study, including propensity score matching, inverse probability of treatment weight, covariate balancing propensity score, multivariable regression, and doubly robust estimation. Chi-Square test and t-tests were used to examine the differences between groups for categorical and continuous data, respectively.
Results
There was a significant improvement in 90-day mortality in the early-TTE group compared to non-early-TTE group (odds ratio = 0.79, 95% CI: 0.64–0.98, p-value = 0.036), revealing a beneficial effect of early-TTE. Net-input was significantly decreased in the early-TTE group on the third day of ICU admission and throughout the ICU stay, compared with non-early-TTE group (838.57 vs. 1181.89 mL, p-value = 0.014; 4542.54 vs. 8025.25 mL, p-value = 0.05). There was a significant difference in the reduction of serum lactate between the two groups, revealing the beneficial effect of early-TTE (0.59 vs. 0.83, p-value = 0.009). Furthermore, the reduction in the proportion of acute kidney injury demonstrated a correlation between early-TTE and kidney protection (33% vs. 40%, p-value < 0.001).
Conclusions
Early application of TTE is beneficial to improve the long-term mortality of patients with moderate-to-severe ARDS.
Introduction
The dyspnea as a manifestation of acute respiratory distress syndrome (ARDS) is characterized by non-cardiogenic pulmonary edema and rapidly progressive hypoxemia. Despite recent advances in medical technology, in-hospital mortality rate in ARDS patients remains high, ranging from 40% to 50%. 1 Therefore, early identification and timely intervention are important measures to improve the prognosis of ARDS.
Lung protective mechanical ventilation and fluid management are widely used to improve survival in ARDS patients by maintaining adequate oxygenation, carbon dioxide removal, and systemic perfusion. 2 Nonetheless, these strategies should be applied with caution, as they may also cause negative impacts on patients. Mechanical ventilation can increase the loading of right ventricle through hemodynamic and respiratory systems, further increasing the risk of cardiovascular dysfunction.3,4 Fluid overload caused by increased alveolar-capillary barrier permeability in ARDS patients may increase hydrostatic pressure and promote the development of pulmonary edema. 5 Therefore, optimal management with adequate oxygen supply is required to minimize the occurrence of lung edema.6–10 Furthermore, acute cor pulmonary heart disease, characterized by severe right ventricular dysfunction, is a frequent and usually lethal complication of ARDS and has been considered as an independent risk factor for mortality in ARDS.11–13
Transthoracic echocardiography (TTE) is a noninvasive technique that is frequently used to collect important hemodynamic data. 14 TTE information can provide valuable diagnostic and prognostic information that support the strategic planning of fluid management, vasopressor, and ventilator use to avoid pulmonary edema, left ventricular failure, shock, and multiple organ dysfunction due to right ventricular dysfunction.15–17 Therefore, early-TTE may be of great clinical benefit in ARDS patients.
A recent study showed that early-TTE can significantly reduce 28-day mortality, suggesting a clinical benefit of early-TTE on short-term survival outcomes in ARDS patients. 18 However, the impact of early-TTE on the long-term outcomes of ARDS patients has not been reported. In fact, long-term outcomes may be more likely to be influenced by multiple factors, such as pulmonary dysfunction, psychological morbidity, neuromuscular weakness, and long-term cognitive impairment.19–21 Moreover, up to 40% of ARDS survivors had at least one post-discharge hospitalization during the 1-year follow-up period,22,23 suggesting that the risk of death in ARDS patients after discharge remains high.24–26 In addition, recent studies have shown that there are large discrepancies between ARDS survivors at discharge and those during long-term follow-up.27–30
Given this discrepancy between short-term and long-term mortality among ARDS survivors, the impact of early-TTE on long-term mortality in ARDS patients should be explored. Therefore, this study investigated whether early-TTE can significantly improve long-term mortality in patients with moderate-to-severe ARDS.
Materials and methods
Study design and patients
Patients from the Medical Information Mart for Intensive Care (MIMIC-III) database who met the following inclusion criteria were included in the study: (a) age ≥ 16 years old; (b) use of mechanical ventilation on the first day in the intensive care unit (ICU); (c) length of ICU stay ≥7 days; and (d) diagnosis of ARDS according to the Berlin definition. MIMIC-III database lacked chest x-ray results. Thus, to rule out cardiogenic pulmonary edema as a potential etiology of acute hypoxemic respiratory failure, 31 we excluded patients who had heart failure as the admission diagnosis in the MIMIC-III database.
For patients with multiple ICU readmissions, only data from their first admission were obtained and used for analysis. For patients admitted to the ICU for less than 24 h, it is difficult to determine why they left the ICU and whether they developed ARDS within a short period of time. To simplify the process of personnel screening, we have added restrictions on the length of ICU stay.
Patients who underwent TTE within 24 h after admission to the ICU were divided into early-TTE group, and the rest were divided into non-early-TTE group. All ARDS patients in the study cohort were further graded into mild, moderate, and severe ARDS according to the PaO2/FiO2 (P/F) ratio of the Berlin definition, 32 as follows: mild: 200 < P/F ≤ 300 mmHg; moderate: 100 < P/F ≤ 200 mmHg; severe: P/F ≤ 100 mmHg. Patients diagnosed with mild ARDS were excluded from this study.
Data sources
The data used in this study obtained from a public database, the MIMIC-III, which contains information on more than 15,000 patients admitted to ICU. The first measurement data of patients after admission to the ICU were extracted from the MIMIC-III database by using PostgreSQL, including age, sex, weight, Sequential organ failure assessment (SOFA) score, 33 Simplified acute physiology score (SAPS) II score, 34 Elixhauser score, 35 Acute physiology and chronic health evaluation (APACHE) III score, 36 concomitant diseases (chronic obstructive pulmonary disease (COPD), atrial fibrillation (AFIB), chronic kidney disease, congestive heart failure (CHF), liver disease, coronary heart disease (CAD), malignancy), vital signs (mean arterial pressure (MAP), heart rate, temperature, central venous pressure (CVP)), interventions (vasopressors (epinephrine, norepinephrine, dopamine, dobutamine, phenylephrine), and sedative drugs (midazolam, propofol, diazepam, dexmedetomidine, alprazolam)), laboratory values (white blood cell (WBC) count, platelet count, hemoglobin (HB), lactate, chloride, creatinine, creatinine kinase, sodium, N-terminal prohormone of brain natriuretic peptide (NT-proBNP), troponin, pH, partial pressure of oxygen (PO2), P/F ratio, potassium, partial pressure of carbon dioxide (PCO2), bicarbonate, blood urea nitrogen (BUN)), the amount of input (include intravenous fluids, blood products, oral medications, and tube feedings) and output (include urine output, chest tube drainage, wound drainage, and dialysis output) in the first, second, third day after admission to ICU, the total amount of in-hospital input and output.
The primary outcome was 90-day mortality after ICU admission. Secondary outcomes included mechanical ventilation- and vasopressor-free days within 28 days of ICU admission. Total net-input patients received on days one, two, and three in the ICU. The reductions in serum creatinine and serum lactate were calculated as follows: for the early-TTE group, the measured value of serum creatine and serum lactate after TTE minus the measured value before TTE. For the non-early-TTE group, the value measured 48 h after admission to the ICU minus the value measured for the first time in the ICU.
Statistical analysis
R package “missForest” was used to impute missing values through a random forest model. 37 Propensity score matching (PSM) was used to match characteristics of study covariates to reduce bias. 38 A propensity score (PS) was calculated for each individual using logistic regression based on covariates, including age, gender, service unit, weight, SOFA score, SAPS II score, Elixhauser score, APACHE III score, height, use of vasopressor, percentage of severe state, use of sedatives, CHF, AFIB, renal, liver, COPD, CAD, malignancy, temperature, CVP, MAP, heart rate, WBC, HB, platelet, pH, PO2, PCO2, P/F ratio, sodium, potassium, bicarbonate, chloride, BUN, creatinine, creatinine kinase, NT-proBNP, troponin, lactate, and day of lCU admission. Nearest neighbor matching was used to find a patient in the non-early-TTE group whose PS score was closest to the patient in the early-TTE group (maximum caliper = 0.2, 1:1 matched), and then the two patients were no longer involved in subsequent matching (replace = F). To assess the balance of covariate distributions between early-TTE and non-early-TTE groups, standardized mean differences (SMDs) were calculated. In this study, t-test and Chi-Square test were used for comparison analysis of continuous and categorized data, respectively. The power of the significant test was calculated using R package “pwr.”
Sensitivity analysis
Sensitivity analysis was conducted using the inverse probability of treatment weight (IPTW),39,40 covariate balancing propensity score (CBPS),
41
multivariable regression, and doubly robust estimation.
42
IPTW is a PS-based weighting method. Each individual is given a weight of 1/PS and 1/(1-PS) according to whether it belongs to the treatment group or the control group. A new cohort is obtained according to weight sampling, which can reduce the influence of cofoundering factors. However, a very large or very low PS will cause the individual to have a large weight, thereby increasing the variability of the estimated treatment effect.
43
Therefore, the improved stabilized weights proposed by Cole SR et al. was used for weighting
43
:
The CBPS approach refers to modeling treatment allocation while optimizing covariate balance. This was done by balancing the conditional probabilities of score and treatment assignment using the dual features of PSs as covariates. CBPS was conducted using the R package “CBPS.” 44 Doubly robust estimation is an estimation technique that prevents model misspecification using two models. One for outcomes, and another for exposure. If one of the models is correct (not necessarily both), this will yield an unbiased estimate of the exposure-outcome association. Here, we used R package “fastDR” for doubly robust estimation. 45 In addition, the multivariable regression used in this study was based on logic regression analysis using R package “stats.”
Results
Propensity score matching and other methods
After reviewing 61,532 patients admitted to the ICU from the MIMIC-III database, ARDS patients were initially identified according to the following criteria: (a) patients had a first-time ICU admission and (b) patients had a diagnosis of ARDS according to the Berlin criteria. Finally, a total of 3912 eligible ARDS patients were included in the study cohort, including 1079 mild, 1858 moderate, and 975 severe ARDS patients (Figure 1). The number of patients in the early-TTE group and non-early-TTE was 811 and 2022, respectively. Since patients with moderate and severe ARDS are more likely to develop pulmonary heart disease and may benefit from early-TTE, we hypothesized that early-TTE would be of great clinical value in these patients. Therefore, we focused on the prognostic value of early-TTE in patients with moderate and severe ARDS.

Flow chart illustrating the exclusion and inclusion criteria used to screen 2833 patients as the final study cohort. Abbreviations: MIMIC: Medical Information Mart for Intensive Care; ARDS: Acute respiratory distress syndrome.
Table 1 summarizes the characteristics of patients with moderate-to-severe ARDS who received or did not receive early-TTE. Patients in the early-TTE group had significantly higher disease severity scores than those in the non-early-TTE group, including SAPS II score (23.13 ± 4.74 vs. 21.72 ± 4.64, p-value < 0.001), SOFA score (8.51 ± 3.38 vs. 7.18 ± 3.37, p-value < 0.001), APACHE III score (61.29 ± 23.33 vs. 51.82 ± 21.58, p-value <0.001), and Elixhauser score (4.27 ± 2.25 vs. 3.51 ± 2.09, p-value <0.001). A higher proportion of patients in the early-TTE groups received vasopressor therapy (68.3% vs. 51.5%, p-value <0.001) and use of sedatives (48.5% vs. 42.8%, p-value = 0.007).
Demographic an d characteristics of patients with moderate-to-severe ARDS stratified by early-TTE.
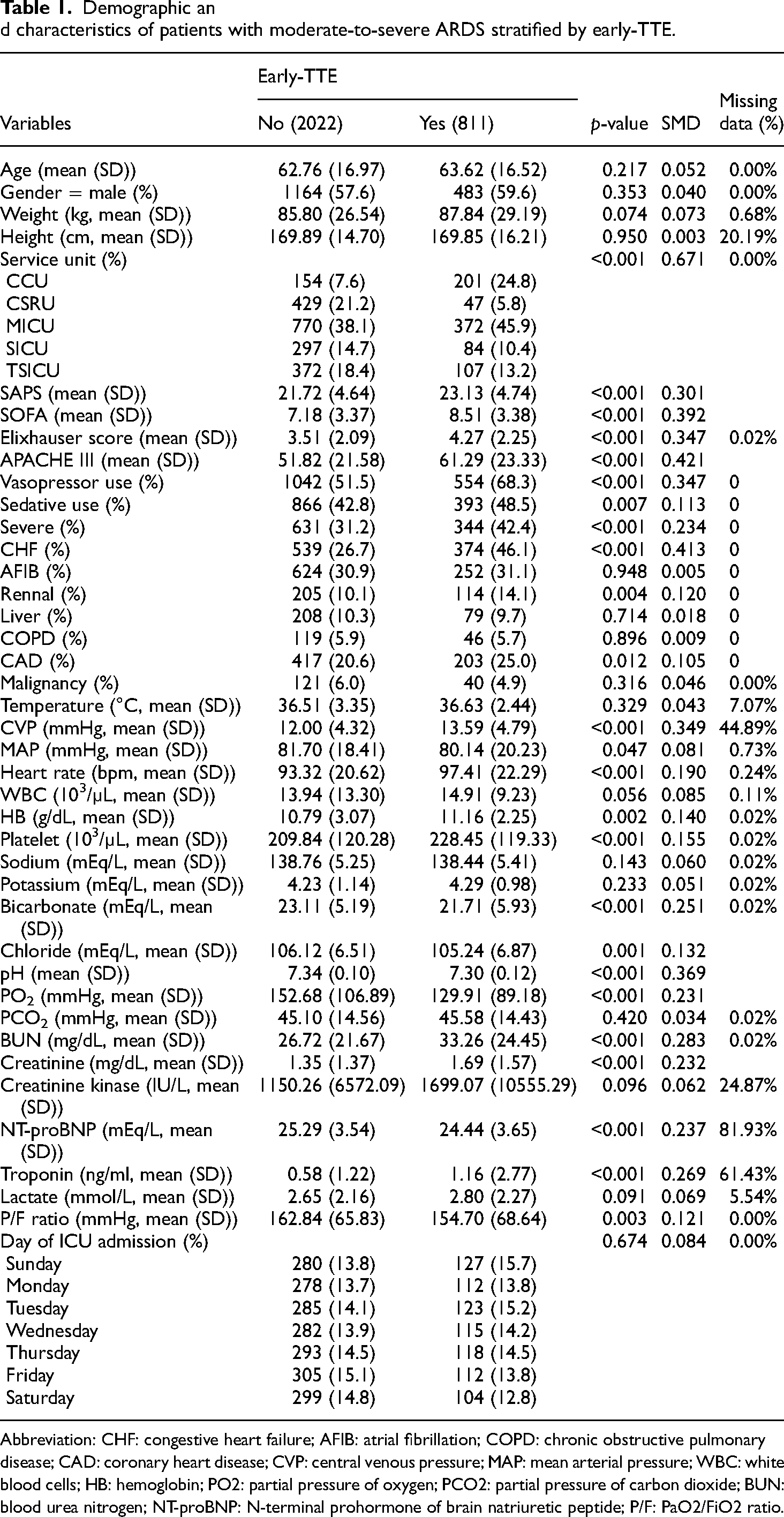
Abbreviation: CHF: congestive heart failure; AFIB: atrial fibrillation; COPD: chronic obstructive pulmonary disease; CAD: coronary heart disease; CVP: central venous pressure; MAP: mean arterial pressure; WBC: white blood cells; HB: hemoglobin; PO2: partial pressure of oxygen; PCO2: partial pressure of carbon dioxide; BUN: blood urea nitrogen; NT-proBNP: N-terminal prohormone of brain natriuretic peptide; P/F: PaO2/FiO2 ratio.
After PSM and adjusting for 41 covariates, baseline characteristics were well balanced, with all covariates having an SMD of less than 0.1 (Table 2). In addition, cohorts obtained by other methods also showed well-balanced covariates (Tables 3 and 4). Figure 2 shows the SMD before and after matching, indicating that these methods worked well.
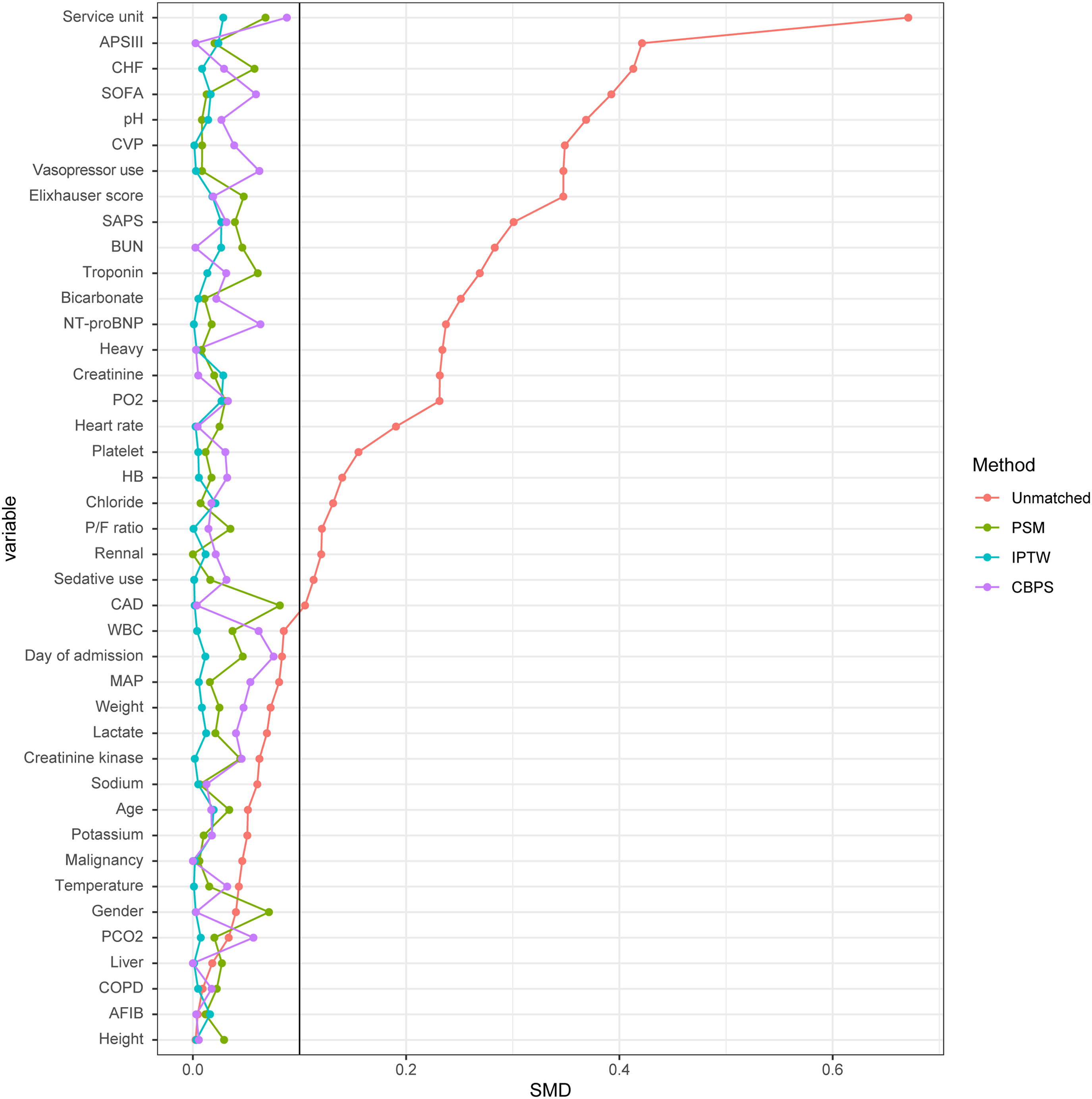
Balancing covariates using the PSM, IPTW, and CBPS models. All models showed that covariates were well balanced. Abbriviations: PSM: propensity score matching; IPTW: inverse probability of treatment weighting; CBPS: covariate balancing propensity score; SMD: standardized mean difference.
The covariates of the matched cohorts were balanced between the groups with and without early-TTE using propensity score matching.

Abbreviation: CHF: congestive heart failure; AFIB: atrial fibrillation; COPD: chronic obstructive pulmonary disease; CAD: coronary heart disease; CVP: central venous pressure; MAP: mean arterial pressure; WBC: white blood cells; HB: hemoglobin; PO2: partial pressure of oxygen; PCO2: partial pressure of carbon dioxide; BUN: blood urea nitrogen; NT-proBNP: N-terminal prohormone of brain natriuretic peptide; P/F: PaO2/FiO2 ratio.
The covariates of the matched cohorts were balanced between the groups with and without early-TTE using inverse probability of treatment weighting.

Abbreviation: CHF: congestive heart failure; AFIB: atrial fibrillation; COPD: chronic obstructive pulmonary disease; CAD: coronary heart disease; CVP: central venous pressure; MAP: mean arterial pressure; WBC: white blood cells; HB: hemoglobin; PO2: partial pressure of oxygen; PCO2: partial pressure of carbon dioxide; BUN: blood urea nitrogen; NT-proBNP: N-terminal prohormone of brain natriuretic peptide; P/F: PaO2/FiO2 ratio.
The covariates of the matched cohorts were balanced between the groups with and without early-TTE using CBPS.

Abbreviation: CHF: congestive heart failure; AFIB: atrial fibrillation; COPD: chronic obstructive pulmonary disease; CAD: coronary heart disease; CVP: central venous pressure; MAP: mean arterial pressure; WBC: white blood cells; HB: hemoglobin; PO2: partial pressure of oxygen; PCO2: partial pressure of carbon dioxide; BUN: blood urea nitrogen; NT-proBNP: N-terminal prohormone of brain natriuretic peptide; P/F: PaO2/FiO2 ratio.
Considering that patients in non-early-TTE group may also receive TTE 24 h after admission to the ICU, we compared the number of TTE used for patients in the non-early-TTE group with the total number of TTE except for the earliest one for patients in the early-TTE group. The result indicated that there was no significant difference between groups (Figure 3).

There were no differences between the number of TTE used for patients in non-early-TTE group and the total number of TTE, except for the earliest one for patients in early-TTE groups.
Primary outcomes
Patients in the early-TTE group had a significantly lower 90-day mortality rate compared with those in non-early-TTE group (33.4% vs. 39.1%, p-value = 0.026). The results of Kaplan–Meier curve analysis revealed that early-TTE significantly improved the survival rate (p-value = 0.031, Figure 4). The adjusted odds ratio was 0.79 (95% CI: 0.64 − 0.98, p-value = 0.036). Sensitivity analysis based on other four different methods also showed similar results (Table 5), confirming the positive effect of early-TTE on improving the 90-day mortality in patients with moderate-to-severe ARDS.

Kaplan–Meier survival analysis of propensity score-matched patients stratified by early-TTE.
Analysis of primary outcome based on five different models.

Legend: Determination of the benefit of early-TTE after balancing covariates using different statistical methods, including ORs of Multivariate, PSM, IPTW, CBPS, and Doubly robust estimation. Abbreviation: PSM: propensity score matching; IPTW: I nverse probability of treatment weighting; CBPS: covariate balancing propensity score.
Secondary outcomes
Since the above results suggest that early-TTE can improve long-term mortality, we sought to investigate the underlying reason for this benefit. We assessed several secondary outcomes. To better illustrate the condition of the patients’ fluid in the circulation, we generated a “net-input,” which is equal to the input minus the output. As shown in Table 6, the duration of vasopressors and mechanical ventilation did not differ significantly between the two groups. However, the amount of net-input in the early-TTE group on the third day of ICU admission was significantly lower than that in the non-early-TTE group (838.57 vs. 1181.89 mL, p-value = 0.014). Similar results were also observed in the total amount of net-input (early-TTE vs. non-early-TTE: 4542.54 vs. 8025.25 mL, p-value = 0.05). Although the decrease in serum creatinine did not differ significantly between the two groups, the proportion of patients with acute kidney injury (AKI) decreased in the early-TTE group (Figure 5, 33% vs 40%, p-value <0.001), which demonstrated the correlation between early-TTE and protection of kidney. AKI were identified by assessing whether serum creatinine increased by 0.3 mg/dL during the first 3 days after ICU admission (non-early-TTE group) or after the first TTE (early-TTE group).46,47 Serum lactate could reflect the tissue hypoxia caused by impaired gas exchange and increased oxygen demand in patients with ARDS,48–51 and also reflect global tissue perfusion and organ dysfunction in critically ill patients.52–55 Therefore, the decrease in serum lactate reduction was significantly higher in the early-TTE than non-early-TTE group (0.83 vs. 0.59 mmol/L, p-value = 0.009), suggesting that early-TTE is associated with improved prognosis of ARDS.

Difference in the proportion of AKI between the two groups. AKI was identified by assessing a 0.3 mg/dL increase in serum creatinine during the first 3 days after ICU admission (non-early-TTE group) or after the first TTE (early-TTE group). AKI: acute kidney injury.
Analysis of secondary outcome in propensity score-matched patients with moderate-to-severe ARDS.

Legend: Net-input was significantly decreased in the early-TTE group on the third day of ICU admission and throughout the hospital stay. The reduction in serum creatinine was significantly different between the two groups, revealing the beneficial effect of early-TTE on renal function.
Discussion
The severity of the illness and long lengths of ICU stay are positively associated with substantial treatment costs, family burden, and the use of healthcare resources. 56 Given that there may be differences between long-term and short-term outcomes in patients with ARDS, short-term outcomes such as in-hospital mortality can not fully reflect long-term outcomes. 57 While a recent study described the benefit of early-TTE on short-term outcomes in ARDS patients, its impact on long-term outcomes remains unclear. 18
To ensure the robustness of our study, we conducted multiple sensitivity analyses. In Tables 1–4, the SMD of 41 covariates was less than 0.1, demonstrating their balance. Although there were some patients from CCU, considering the exclusion of patients with heart failure and the excellent effect of balancing of baseline, we speculate that the bias was minimal. According to the results of analyses using balanced covariates by PSM, IPTW, and CBPS, early-TTE can significantly improve 90-day mortality, supporting the benefit of early-TTE on long-term survival outcomes in ARDS patients. Besides, we also found a significantly difference in infusion volume on day 3, indicating the adjustments in fluid management. Notably, the reduction of serum lactate was higher in early-TTE group compared to no-early-TTE group, which indicated a better prognosis. Also, the decrease in the percentage of AKI between two groups revealed that the early-TTE was correlated to improved renal function. Taken together, the benefit of early-TTE on long-term outcome may be attributable to three aspects: first, early-TTE can help clinicians identify cardiovascular disease and its related comorbidities, and then provide timely treatments. This is because recent studies have found that comorbidities and sequelae in ARDS patients are highly correlated with long-term outcomes.30,58 In particular, cardiac injury is known to be associated with increased long-term morbidity and mortality.59,60 TTE, as a non-invasive cardiac imaging, can provide valuable information about cardiac morphology and function and help clinicians identify potential comorbidities such as right ventricular dysfunction, infective endocarditis, 61 cardiac amyloidosis, 62 and left ventricular hypertrophy. 63 Second, in clinical practice, results of TTE could guide the adjustments to fluid management strategies. Recent studies have noted that fluid overload is highly associated with worse outcomes, such as increased 90-day mortality, prolonged duration of ventilation, and longer ICU and hospital stays,64–66 whereas conservative fluid management is significantly associated with better outcomes, such as shorter duration of ventilation and shorter ICU stays.7,8 In addition, inappropriate fluid management strategies may increase the risk of long-term cognitive impairment, which negatively affect the long-term prognosis of ARDS patients. 67 Recent studies have further revealed the heterogeneity of ARDS: ARDS patients with different phenotypes may respond differently to the same fluid management strategy. 68 In the study by Liu et al., for example, three distinct ARDS clinical phenotypes were identified, each with different clinical features and outcomes. 69 Fluid-conservative strategy was associated with improved mortality in phenotype II ARDS but worse outcome in phenotype III ARDS, supporting the need for customized and optimal fluid management in ARDS patients. TTE is currently widely used to assess fluid responsiveness, which helps to adjust fluid management strategies in a safe and non-invasive manner.70,71 This is consistent with our finding that net-input was significantly different between early-TTE group and non-early-TTE group, supporting the notion that adjustments of fluid administration are associated with improved long-term outcomes in ARDS patients. Also, appropriate fluid management strategies can help improve hypoxia and tissue perfusion which can be revealed by serum lactate.5,72 In our results, the early-TTE group had better serum lactate improvement than the non-early-TTE group, suggesting that TTE may help improve tissue hypoxia and perfusion by helping adjust fluid strategies, thereby improving prognosis. Third, early-TTE is beneficial to the improvement of kidney function in ARDS patients. Severe AKI is known to be significantly associated with short-term outcomes in critically ill patients. 73 AKI as a critical risk factor for the development of chronic kidney disease (CKD) affects the long-term prognoses of CKD patients, including those with ARDS.74–76 This is also supported by the results of our study, as ARDS patients in the early-TTE group had a lower proportion of AKI than those in the non-early-TTE group. In other words, early-TTE was associated with improved kidney function, which may lead to better long-term outcomes. Nonetheless, our study was retrospective and observational, and therefore cannot establish a causal relationship between early-TTE and improved kidney function. There may be other confounding factors that influenced the outcomes of the patients, such as the severity of illness, the timing and dose of fluid administration, the use of renal replacement therapy, and the quality of care. Further prospective studies are needed to confirm our findings and elucidate the mechanisms by which early-TTE can prevent AKI in ARDS patients. One possible explanation is that adjustments to fluid management due to early-TTE further optimizes renal perfusion and thus prevents AKI. 77 Surprisingly, there were no significant differences between groups in terms of ventilation-free days and vasopressor-free days. We speculate that early-TTE may lead to changes in the parameters of ventilators rather than duration of use.6,78
This study has some limitations. First, the analyzed data was extracted from the MIMIC-III database, which is a real-world dataset from one medical center. While having the advantage of a large sample size, it may still limited the generalizability of our findings. Second, the analyses of this study were based on the clinical data from 2001 to 2012 in the MIMIC-III database. Improvements in the treatment methods and concepts for patients with ARDS may also affect the outcomes. Third, although TTE has the advantages of non-invasiveness and convenience, the reproducibility of echocardiography may be relatively poor due to differences in the proficiency of echocardiography among clinicians. Furthermore, due to the lack of specific echocardiography parameters and outcomes in the MIMIC-III database, further analysis of the detailed mechanisms of this beneficial effect could not be performed. Fourth, the proportion of missing values in some covariates is large, and random forest imputation of missing values may cause bias.
In general, our results revealed that early-TTE can improve long-term outcome in ARDS patients. The underlying mechanism may be that early-TTE reveals patients’ comorbidities, thereby guiding strategies to adjust fluid management, which may lead directly or indirectly to improved serum lactate level and kidney function. Considering the limitations of our study, more large-scale prospective studies are needed to confirm the beneficial effect of early-TTE on the short-term and long-term outcomes of ARDS patients, and to explore the detailed mechanism of this benefit.
Conclusion
Early-TTE is beneficial to improve the long-term mortality of patients with moderate-to-severe ARDS. Although the detailed mechanism remains to be explored, the benefits of early-TTE may achieved through optimization of subsequent treatments, such as fluid management, decrease serum lactate, and improved kidney function.
Footnotes
Acknowledgments
We acknowledge the MIMIC-III database for providing their platforms and contributors for uploading their meaningful datasets. This manuscript has been previously posted as a preprint at Research Square on September 16, 2022.
Authors’ contributions
QL and NW conceptualized and designed this study, performed analysis and literature review. QL and SN extracted data from the MIMIC-III database. YT, WT, ZT, and NW participated in data analysis and interpretation. QL and HW draft the manuscript and revised the manuscript based on comments from other authors. All authors read and approved the final manuscript.
Availability of data and materials
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chongqing Yongchuan District Natural Science Fund, Chongqing medical scientific research project, (grant number 2020nb00503, 2020FYYX006)
Ethics approval and consent to participate
We used data from the MIMIC-III database, which had approval from the Massachusetts Institute of Technology's institutional review board (no. 0403000206). The study did not affect clinical care and de-identified all protected health information, so individual patient consent was not required.
Author Biographies
Qiuyu Liu is a postgraduate in Critical Care Medicine. Her area of research is the management of ARDS.
Yingkui Tang is a PhD student in Biomedical Engineering. His area of research is Biotechnology and Engineering.
Wu Tao is a Professor in Critical Care Medicine. His area of research is the management of ARDS and sepsis.
Ze Tang is a Professor in Critical Care Medicine. His area of research is the management of ARDS and sepsis.
Hongjin Wang holds a Master of Medicine degree. Her area of research is critical disease focusing on ARDS.
Shiyu Nie holds a Master of Medicine degree. Her area of research is critical disease focusing on sepsis.
Nian Wang is a PhD student in Critical Care Medicine. His area of research is the management of critical diseases.

