Abstract
Objective
This study aimed to investigate the association between nutritional appetite status—assessed during a standardized eight-week hospitalization period using the Simplified Nutritional Appetite Questionnaire (SNAQ)—and discharge outcomes at a one-year follow-up in older hospitalized patients with multiple comorbidities.
Methods
A prospective one-year observational study was conducted in a convalescent geriatric hospital. This study was conducted with 77 patients aged 65 and older with comorbidities who have been on oral intake for more than 8 weeks after hospitalization. Patients were divided into low (n = 40) and normal (n = 37) appetite groups based on a SNAQ score cut-off of 14. Odds ratio (OR) analysis was conducted to compare improved and deteriorated discharge outcomes at the one-year mark according to appetite status, adjusted for Charlson comorbidity index, activities of daily living score, and nutritional intake.
Results
The low appetite group had significantly lower energy (1215.68 ± 68.96 kcal vs. 1495.50 ± 48.03 kcal, P = 0.001) and protein intake (55.66 ± 2.83 g vs. 65.75 ± 1.88 g, P = 0.004). They reported poorer appetite, felt full with fewer mouthfuls, worse food taste, and fewer meals daily (P < 0.05). Adjusted for Charlson comorbidity index, Activities of daily living score, and nutritional intake, the low appetite group had a higher risk of adverse discharge outcomes, such as death or intensive care, with an adjusted OR of 8.524 (95% CI: 1.331–59.739, P = 0.024). Their likelihood of improvement, defined as discharge to a nursing home or home, was reduced, with an adjusted OR of 0.109 (95% CI: 0.013–0.915, P = 0.041).
Conclusions
This study underscores the clinical importance of assessing and promoting appetite to improve discharge outcomes and reduce healthcare costs for long-term hospitalized geriatric patients.
Introduction
With the increase in the aging population, there is a corresponding rise in the number of older patients requiring long-term hospitalization, thereby intensifying the burden on healthcare systems. 1 In South Korea, the average length of stay (LOS) for patients in long-term care hospitals is reported to be 25.4 days for men and 32.2 days for women, 2 which is more than twice the average length of stay compared to Organization for Economic Co-operation and Development (OECD) nations and European standards. 3 Such prolonged morbidity leads to an overall strain on the health system and is consequently associated with significantly higher societal costs. 4
In clinical settings, reduced appetite and a lack of motivation to eat among older patients are directly linked to decreased food intake, 5 which can lead to weight loss and a decline in nutritional status. 6 Also, it is reported that nutritional status during hospitalization is a key factor contributing to worsened outcomes and an extended LOS, regardless of demographic factors, living conditions, and disease severity. 7 Appetite during the initial weeks of hospitalization can serve as an early indicator of nutritional risk, with significant implications for long-term health outcomes, 8 as early malnutrition is linked to adverse outcomes over time, ultimately impacting overall resilience.9,10
Particularly in hospitalized older patients with comorbidities, appetite is a critical indicator directly linked to prognosis. Despite its importance, it is often overlooked in clinical settings for long-term outcomes. Alarmingly, a report indicates that appetite was mentioned in only 13.3% of medical notes, 11 highlighting a significant gap between ideals and reality in patient assessments. While previous research has partially explored the impact of nutritional appetite on disease progression in older adults, studies on its relationship with diverse discharge outcomes in long-term hospitalized patients are limited in mortality risks. Existing literature is limited as it either focuses on healthy geriatric populations, 12 is conducted in community-based settings,13,14 or examines only specific genders 15 or specific diseases,16,17 and lacks longitudinal follow-up data relevant to prognosis. 18
Therefore, the aim of this study was to investigate the association between nutritional appetite status—assessed during a standardized eight-week hospitalization period using the Simplified Nutritional Appetite Questionnaire (SNAQ)—and discharge outcomes at a one-year follow-up in older adults admitted to a long-term care hospital with multiple comorbidities. We hypothesized that lower SNAQ scores during hospitalization would be associated with poorer discharge outcomes, including increased mortality, transfer to higher-acuity care, and a reduced likelihood of being discharged home. This study aims to provide longitudinal evidence on the prognostic value of appetite in institutionalized older patients in long-term care settings.
Method and materials
Study design and subject recruitment
This study is a prospective observational study utilizing actual clinical data collected from patient observations at a geriatric hospital in Daegu, Republic of Korea. The appetite assessment period spanned 8 weeks, and discharge outcomes were evaluated one year after admission. Patients admitted to the hospital between October 2023 and February 2024 were eligible for inclusion, with the follow-up of the last patient completed in February 2025. When designing this study, we considered the hospital's admission rates, ∼20 patients per month, who met inclusion criteria, and anticipated enrolling around 100 patients for the 5 months. Patients who agreed to participate and met the inclusion criteria were enrolled by the attending physician. The reporting of this study adheres to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) checklist for cohort studies, in line with EQUATOR Network recommendations. 19
Subjects’ inclusion criteria
Patients were consecutively enrolled based on the order of their admissions, and only those who met the predefined inclusion criteria were included in the study. Our study subjects were patients hospitalized for recuperation who were capable of oral intake. The inclusion criteria were as follows: (i) patients older than 65 years admitted for recovery (including cachexia or secondary sarcopenia, chronic kidney disease, and post-stroke sequelae, etc.), (ii) maintained oral diet intake with an expected LOS of more than 8 weeks, (iii) at least two medical conditions, including one primary diagnosis (such as dementia, stroke, cancer, or chronic kidney disease, etc.) and at least one comorbid condition (such as diabetes or hypertension (HTN), etc.), and (iv) chronic disease status with a primary disease onset more than 6 months prior to the start of the study. The exclusion criteria were (i) patients younger than 65 years, (ii) inability to maintain oral intake or transitioning to enteral nutrition, and (iii) initiating treatments aimed at enhancing appetite (such as nutritional counselling, medication, or injections), and (iv) patients with advanced dementia who were unable to communicate meaningfully were excluded at the enrollment stage to ensure the validity of appetite assessments. The expected LOS of more than 8 weeks was determined at the time of admission by the attending physician in consultation with the hospital's care management team. This estimation was based on clinical factors such as the patient's primary diagnosis, degree of functional impairment, number and type of comorbidities, and anticipated rehabilitation or supportive care needs. Patients admitted for short-term acute care or those with discharge plans within a few weeks (e.g. post-surgical recovery or respite care) were excluded from enrollment. This approach ensured the inclusion of patients likely to complete the eight-week appetite assessment period. After enrollment (n = 100), baseline data were collected at admission, appetite assessments were conducted over 8 weeks, and follow-up discharge data were collected at one year. Finally, 77 participants were included in the analysis. The flow diagram of the current study is shown in Figure 1.

Flow diagram of this study.
Sample size calculation
The sample size was estimated based on a priori power analysis using G*Power 3.1.9.7 for a two-tailed chi-square test to compare discharge outcomes (e.g. death, transfer, and home discharge) between two appetite groups (low vs. normal), assuming a medium effect size (w = 0.3), α = 0.05, and 80% power. 20 Based on this, a minimum of 88 participants was required. Accounting for an estimated 15% attrition rate over the one-year follow-up period, the target recruitment size was set at ∼100 patients. This estimation was also supported by the hospital's admission rate (∼20 eligible patients per month), making the target feasible within the five-month recruitment window. A total of 100 patients were enrolled, and 77 participants completed the study and were included in the final analysis.
Study period
In this study, we selected patients who were expected to remain hospitalized for at least 8 weeks. This threshold was intentionally set to capture the mid-term appetite trajectory in older inpatients. According to the Korea Health Insurance Review and Assessment Service (HIRA), the national average hospital stay among older adults in Korea is ∼ 23.7 days, 21 while long-term hospitalization is formally defined as ≥ 90 days in Korea. Therefore, the eight-week period represents a clinically meaningful transitional phase between short-term and long-term care—a pre-long-term stage—during which appetite patterns may begin to diverge depending on chronic disease burden, functional decline, and environmental factors. This fixed time frame was selected to align with the predictive validity range of the SNAQ (which has been validated to predict weight loss over a period of 2 weeks to 3 months) and to minimize confounding effects of subsequent medical or nutritional interventions that may occur later during hospitalization. In practice, extending appetite assessments beyond this period proved difficult due to medical interruptions (such as prescriptions of appetite-stimulating medications or transitions to enteral nutrition) and variability in patients’ hospital stays. Therefore, the eight-week period was the most viable and consistent assessment timeframe for all participants in our study.
Basic information and measurements
Patients’ height and weight were assessed at admission as part of a routine check-up by a nurse. When it wasn't possible to measure height, the most recent measurement or the patient's self-reported height was used instead. To address the primary causes of morbidity, patients were divided into four primary disease groups: dementia, chronic kidney disease, post-stroke, cancer, and another category which included three cases (one each of pressure ulcer, spinal stenosis, and fracture). Additionally, the presence of diabetes mellitus (DM) and HTN was also investigated. Given that many patients were bedridden, registered ward nurses also evaluated the risk of pressure ulcers using the Braden scale. 22 The Braden Scale categorizes pressure ulcer risk into five levels: very high (≤ 9), high (10–12), moderate (13–14), mild (15–16), and no risk (17–23).
Nutritional Appetite Questionnaire (NAQ)
All participants were assessed for appetite using the SNAQ and Council on NAQ (CNAQ) over this standardized eight-week period, starting from the day of admission. The SNAQ was chosen as the primary appetite screening tool because it is concise, easy to administer in clinical settings, and has well-established predictive validity for weight loss in older inpatient populations. 23 Although alternative tools such as the Appetite Loss in Older Adults with and without Cognitive Impairment (ALOC) scale have been developed for patients with significant cognitive decline, 24 the SNAQ was considered more feasible and appropriate for our study context at the time of design due to its widespread use, clear cut-off values, and practical applicability across a mixed population with varying levels of cognitive function. The CNAQ consists of a total of eight questions, each scored on a 1–5-point scale, resulting in a maximum score of 40 points. The SNAQ is derived from four of CNAQ questions, focusing on appetite, feeling of satiety, taste, and the number of meals per day, with a total possible score of 20 points. The SNAQ is known to have an optimal cutoff of 14 for determining the risk of malnutrition in a wide range of older populations,12,25 and it has been reported that lower SNAQ scores are associated with higher mortality rates among older adults. 26 In Korean clinical settings, the SNAQ has been validated for its reliability and validity, 27 and we assessed the NAQ at admission, 4 weeks, and 8 weeks using both the CNAQ and SNAQ.
Appetite assessment and the appetite groups
For patients who were bedridden but cognitively able, the SNAQ was completed through structured interviews conducted by trained staff to ensure comprehension and consistency. Dementia status at admission was confirmed using each patient's electronic medical records, including formal diagnoses and recent Mini-Mental State Examination (MMSE) results when available. Among the total sample, ∼54% (42 out of 77 patients) had mild to moderate dementia at baseline. For these participants, the questionnaire was administered with assistance from caregivers or family members when necessary, and proxy responses were accepted if direct self-reporting was not feasible. This flexible approach ensured consistent and reliable appetite data collection across participants with varying levels of cognitive function. Based on their average SNAQ score over the initial eight-week period (across three assessment points), patients with an average score of 14 or lower were classified as the low appetite group (n = 37), while those with an average score above 14 were classified as the normal appetite group (n = 40).
Discharge outcome at one-year follow-up
At the one-year point from admission, the discharge outcomes of patients were categorized as follows: death, transfer (to an upper-tier hospital for intensive care), continued hospitalization at the same facility, and discharge (residing either at a nursing home or at home). At the one-year follow-up, 53 of the 77 participants remained hospitalized at our facility. For the remaining 24 participants, outcomes were determined through a review of discharge records, telephone interviews, and face-to-face meetings with caregivers. In addition, to assess the relationship between appetite and discharge outcomes, the outcome cases were classified into two categories: deteriorated cases (death or transfer to an upper-tier hospital for intensive care, 13 out of the participants) and improved cases (discharge to a nursing home or home, 11 out of the participants), except those who remained hospitalized at our facility.
Comorbidity and ADL assessments
Older patients do not get admitted to long-term care hospitals solely due to a single disease. Additionally, the independence index is used as an indicator to evaluate prognosis and physical abilities. Therefore, in this study, we used the Charlson comorbidity index (CCI) and Barthel activities of daily living (ADL) index to assess the level of comorbidities and independence of the patients. The CCI was originally developed to evaluate the impact of comorbidities on a patient's survival rate. 28 The CCI assigns scores to 19 conditions, with a total range from 0 to 37, and a score of 5 or more indicates a high comorbidity burden and increased mortality risk. It is used as an indicator for predicting the prognosis of cancer patients 29 or individuals who have undergone surgical procedures. 30 Furthermore, the comorbidity index has been validated as an independent predictor of future costs in relation to various specific conditions. 28 The Barthel ADL index is a tool used to assess a person's ability to perform basic self-care tasks and evaluate their level of independence. 31 In our study, we used a 20-point version of the Barthel index that was developed in Korean. 32 This modified scale measures the individual's capability to perform daily activities, with higher scores indicating greater independence. Comorbidity and ADL assessments were conducted by medical staff upon admission.
Diet types and nutritional intake assessment
Patients received individualized dietary prescriptions based on their medical conditions, and registered dietitians (RDs) assessed their dietary intake on a weekly basis throughout the eight-week study period. Visual estimation of plate waste was used to evaluate the actual intake at each meal, a method widely employed in clinical practice to assess food consumption. 33 Specifically, each meal was inspected before and after consumption, and intake for each meal component (e.g. rice, soup, meat or fish, vegetables, and dairy) was estimated by the RD using an 11-point scale (0 = none consumed, 10 = fully consumed). The visually estimated intake data were then converted into energy and macronutrient values, including carbohydrate, protein, and fat, using CAN-Pro 5.0, a validated nutrient analysis software developed by the Korean Nutrition Society. 34 The average daily energy and protein intake over the eight-week period was calculated and used for analysis. The dietary assessment process is detailed in our previous study. 35
Ethical review
This study was approved by the Institutional Review Board of Jeonju University (jjIRB-230503-HR-2022–1212) and conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from all participants at admission, including consent for the use of clinical data in research. All personal information was de-identified in accordance with ethical and institutional guidelines.
Data collection and bias control
Efforts were made to minimize potential sources of bias throughout the study design and data collection process. To reduce selection bias, only patients who met strict inclusion criteria—being 65 years or older, expected to stay at least 8 weeks, and capable of oral intake—were enrolled consecutively upon admission. This approach minimized arbitrary selection and ensured homogeneity in baseline health status across participants. To reduce measurement bias, all appetite assessments were conducted using the validated SNAQ, administered consistently by trained clinical staff. Information bias was addressed by using predefined clinical criteria and consistent data collection protocols. Data collectors were blinded to the study hypothesis.
Statistical analyses
The study's statistical design comprised following components: (1) identifying the proportion of older patients with comorbidities who maintained oral intake for over 8 weeks and displayed low appetite (SNAQ score below 14), (2) presenting the average and changes in scores for each item in the CNAQ and SNAQ by appetite group (low vs. normal appetite), and (3) analyzing the relationship between discharge outcomes and appetite. Variables and data are presented as numerical values and percentages for categorical variables, and as means ± standard errors (SEs) for continuous variables. P values for basic characteristics, nutritional intake, and NAQ scores based on appetite group were obtained using chi-square tests for categorical variables and ANOVA tests for continuous variables. Logistic regression analyses were employed to calculate the ORs and 95% confidence intervals (CIs) for discharge outcomes, assessing deteriorated risk and improvement likelihood according to appetite, in both crude and adjusted models (accounting for basic characteristics, nutritional intakes, comorbidity index, and ADLs). All analyses were performed using SPSS version 28.0 (IBM/SPSS Corp., Chicago, IL, USA), with a two-tailed P value of < 0.05 considered indicative of statistical significance.
Results
Basic characteristics of patients
Table 1 presents the baseline characteristics of the study participants stratified by appetite status (low vs. normal). The mean age of participants was similar between the low appetite group (80.03 ± 1.98 years) and the normal appetite group (82.18 ± 1.10 years), with no statistically significant difference (P = 0.336). The CCI was significantly higher in the low appetite group (3.35 ± 0.27) compared to the normal appetite group (2.15 ± 0.14, P < 0.001), indicating a greater disease burden among those with poor appetite. In terms of macronutrient intake, the mean daily energy intake was significantly lower in the low appetite group (1215.68 ± 68.96 kcal/day) than in the normal appetite group (1495.50 ± 48.03 kcal/day, P = 0.001). Similarly, protein intake was significantly reduced in the low appetite group (55.66 ± 2.83 g/day vs. 65.75 ± 1.88 g/day, P = 0.004). Other indicators, such as activities of daily living (Barthel index), pressure ulcer risk (Braden score), and prevalence of hypertension or DM, did not differ significantly between the two groups. Detailed descriptive results for the item-level CNAQ and SNAQ scores are provided in Supplemental Table 1.
Basic characteristics of the study participants.

CCI: Charlson comorbidity index; ADL: activities of daily living.
Mean ± standard error (SE) or the number of subjects. P values were derived from chi-square tests (categorical variables) and ANOVA tests (continuous variables) between two groups.
Other disease; fracture, skin ulcer, spinal stenosis. P < 0.05 was considered significant.
NAQ results
Table 2 presents the average scores for each of the eight questions from the NAQ, comparing the low and normal appetite groups. Significant between-group differences were observed in several core appetite-related domains. Participants in the low appetite group reported a significantly lower subjective appetite (3.01 ± 0.14) compared to the normal appetite group (4.38 ± 0.09, P < 0.001), and felt full with fewer mouthfuls (3.21 ± 0.15 vs. 3.98 ± 0.05, P < 0.001). They also rated food taste less favorably (2.72 ± 0.16 vs. 4.28 ± 0.13, P < 0.001), and perceived their appetite compared to younger years as poorer (2.65 ± 0.12 vs. 3.41 ± 0.12, P < 0.001). Furthermore, the low appetite group reported fewer daily meals (3.86 ± 0.07 vs. 4.08 ± 0.05, P = 0.014). No statistically significant differences were observed between groups for items related to hunger frequency, nausea, or general mood, although numerical trends suggested slightly lower scores in the low appetite group.
Response results for each question of the Nutrition Appetite Questionnaire.
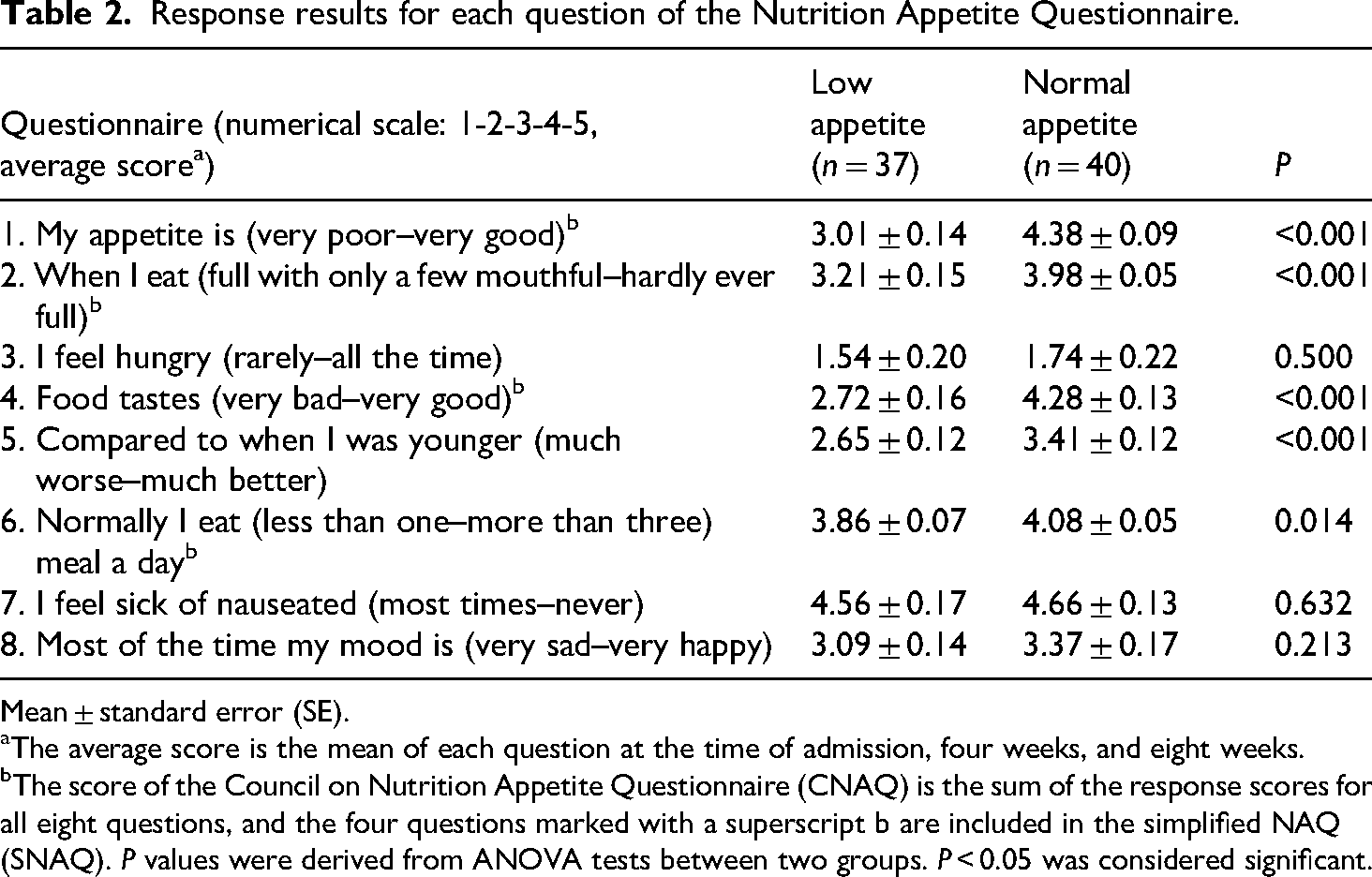
Mean ± standard error (SE).
The average score is the mean of each question at the time of admission, four weeks, and eight weeks.
The score of the Council on Nutrition Appetite Questionnaire (CNAQ) is the sum of the response scores for all eight questions, and the four questions marked with a superscript b are included in the simplified NAQ (SNAQ). P values were derived from ANOVA tests between two groups. P < 0.05 was considered significant.
Discharge outcomes at one-year follow-up
Table 3 presents the one-year discharge outcomes stratified by appetite status. The mortality rate was significantly higher in the low appetite group, with 16.2% (six of 37) of patients deceased at follow-up, compared to only 2.5% (one of 40) in the normal appetite group (P = 0.036). Additionally, the rate of successful discharge to home or nursing home settings was significantly lower in the low appetite group (5.4%) than in the normal appetite group (22.5%, P = 0.032). While not statistically significant, the proportion of patients who were transferred to a higher-acuity hospital for intensive care was higher in the low appetite group (13.5% vs. 2.5%, P = 0.072), suggesting a trend toward clinical deterioration in this group. Continued hospitalization at the same facility remained the most common outcome in both groups (64.9% in low appetite vs. 72.5% in normal appetite, P = 0.470), reflecting the chronic care nature of the study setting.
Discharge outcome at follow-up according to the appetite group.

The number of subjects and percentages. P values were derived from chi-square tests. P < 0.05 was considered significant.
Relationship between appetite status and discharge outcomes
Table 4 summarizes the results of multiple logistic regression analyses examining the association between appetite status and one-year discharge outcomes. Discharge outcomes were categorized into two composite endpoints: improvement (defined as discharge to home or nursing home) and deterioration (defined as death or transfer to intensive care). Patients in the low appetite group were significantly less likely to experience improvement compared to those in the normal appetite group. The crude OR for improvement was 0.197 (95% CI: 0.039–0.981, P = 0.047). After adjusting for relevant covariates—including sex, age, primary diagnosis, Braden ulcer risk score, energy and protein intake, Barthel index for activities of daily living, and the CCI—the adjusted OR further decreased to 0.109 (95% CI: 0.013–0.915, P = 0.041), suggesting that low appetite was independently associated with a 90% reduced chance of favorable discharge. Conversely, low appetite was strongly associated with an increased risk of deterioration. The crude OR for deterioration was 8.038 (95% CI: 1.644–39.303, P = 0.010), which increased to 8.524 (95% CI: 1.331–59.739, P = 0.024) in the adjusted model.
Odds ratios of discharge outcome at one-year follow-up.

The values are odds ratios (ORs) and 95% confidence intervals (CIs). P values were derived from the multiple logistic regression analysis. The normal appetite group was used as a reference (reference: normal appetite = 1.000).
Adjusted model; sex, age, diagnosis, Branden ulcer risk score, energy and protein intake, Barthel index of activities of daily living, and Charlson comorbidity index were included as covariates. P < 0.05 was considered significant.
Discussion
In this study, we demonstrated that low appetite has a greater adverse effect on discharge prognosis in long-term hospitalized older patients. Notably, this research is among the first to examine oral intake appetite over an eight-week period in the initial period of long-term hospitalized older patients and to establish its association with discharge outcomes at one-year follow-up. By considering potential prognostic factors such as the number of comorbidities, intake levels, and ADL levels as covariates, the ORs were further amplified, clarifying the impact of appetite levels on discharge outcomes. These findings offer important implications for the management of nutrition and appetite in hospitalized patients, particularly in societies with increasing numbers of long-term care residents.
Loss of appetite is a prevalent concern among hospitalized older patients in medical facilities, can lead to weight loss, and poor health outcomes, such as higher mortality rates. 36 Therefore, efforts are being made in practices to avoid malnutrition and improve outcomes through the use of the SNAQ index. In the frail older population aged 60 and above, the prevalence of poor appetite according to the SNAQ criteria has been reported to be quite high, even in community settings: 21.2% in Japan (≥ 70 years old 37 ), 24.1% in Malaysia (≥ 60 years old 38 ) and 33.3% in Brazil (≥ 60 years old 39 ). Meanwhile, a previous study in the UK, 15 which assessed appetite levels in hospitalized men over 70 using the same questionnaire as our current study, found a prevalence of poor appetite at 41%. In our study, possibly due to the older age and higher comorbidity index of the participants, we found that low appetite was common, with 48% of patients hospitalized for over 8 weeks and maintaining oral intake being classified into the low appetite group.
Our study further holds significance as it reports significant differences in each individual item of the CNAQ for hospitalized older patients. Although a previous study was conducted online and limited to the four questions of the SNAQ, 40 which is a subset of CNAQ, an examination of each question affecting appetite among 385 individuals aged 65 and over in China showed that; Question 1, poor appetite was associated with a higher malnutrition risk; Question 2, fullness associated with a higher frailty risk; and Question 4, diminished taste associated with a higher frailty risk and higher anxiety risk. This is similar to our findings, where three out of the four SNAQ items (Questions 1, 2, and 4; poor appetite, fullness, and diminished taste) showed differences between the low appetite and normal groups. In a previous meta-analysis, 41 it was reported that feelings of fullness are greater in older adults, which is overall associated with reduced appetite. Specifically, the weighted mean difference (WMD) for fullness after overnight fasting was 6 mm higher in older adults than young adults. Considering the physiological changes associated with aging, such as increased sensitivity to fullness and a slowdown in the appetite-regulating hormones, 42 and combining this with the responses from the SNAQ, factors related to fullness can be identified as risk indicators for appetite-related issues and are associated with frailty, malnutrition, and psychological status.
To our knowledge, this study is the first to assess the longitudinal association between initial appetite and discharge outcomes, examining both deteriorating and protective effects, over the time span from hospital admission to one year. Notably, although no prior study has suggested the likelihood of inpatient discharge to home, our research found that the likelihood was reduced by ∼90% in the low appetite group compared to those with better appetite. Previous hospital-based SNAQ studies primarily focused on appetite in relation to a single deteriorated outcome, mortality. Previous study of 296 older individuals admitted to the hospital, poor appetite was reported to be associated with 2.62-fold increased odds of mortality at 6 months. 43 Furthermore, the study indicates that each 1-point drop in the SNAQ score is linked to a 22% increase in the odds of six-month mortality, even after adjusting for length of stay. Additionally, there is a report indicating that among older female patients, the hazard ratio (HR) during acute hospitalization was 2.29. 15 In the case of oncological patients, the relationship between poor appetite, SNAQ score under 14, and survival rates is even more pronounced, with reported HR of 4.7 at 3 months and 3.7 at 6 months. 44 The current study found that the OR for worsened prognosis, including mortality and higher level of intensive care, exceeded 8, which is higher than previous reports, likely due to our older study population (average age 80.59), longer follow-up (one year), and higher mean CCI score (over 3).
Initial appetite might influence long-term discharge outcomes by being associated with various physiological and psychological health factors. First, initial positive appetite can help delay or improve the progression of underlying diseases through preventing undernutrition over time. A high appetite typically indicates adequate nutritional intake, 45 and in our study, the superior nutritional intake was also observed in the normal appetite group. According to the Longitudinal Aging Study Amsterdam (LASA), 46 which prospectively followed the incidence of undernutrition in older adults over 9 years, poor initial appetite was one of only two determinants (the other being difficulties walking stairs) that significantly increased the risk of developing undernutrition in the multivariate model, with a notable cumulative incidence difference between those with normal and poor appetite. A study tracking the two-year prognosis of patients in Japan who underwent Transcatheter Aortic Valve Implantation (TAVI) found that appetite status at the time of discharge was significantly associated with long-term outcomes, with multivariate Cox regression analysis identifying less appetite as a significant predictor of two-year major adverse cardiovascular and cerebrovascular events. These longitudinal studies suggest that appetite influences better nutritional status, thereby improving disease outcomes and providing long-term protective effects.
Second, initial appetite status has a long-term impact on functional capacity and physical performance decline after discharge, which is related to the likelihood of living at home. A multicenter, prospective cohort study of over 400 patients aged 70 and above found that decreased appetite was reported by 51% at hospital admission and 17% three months post-discharge, and this decrease was associated with lower muscle strength, mobility skills, and physical performance. In addition, a study among rehabilitation patients aged 65 and older found that those with normal SNAQ scores were significantly more likely to achieve functional independence measure (FIM ≥ 90; 40.5% vs. 68.4% 47 ). Highlighting that discharge to home is often associated with independence. These longitudinal studies can be said to suggest that appetite influences independence over an extended period.
Third, a strong appetite may offer protective effects against other acute complications and the development of inflammation during the initial hospitalization period. According to a six-month longitudinal observational study, 15 having a low appetite as measured by the SNAQ is associated with an increased risk of hospital-acquired infections. Moreover, Pourhassan et al. 48 found that patients who experienced a decrease in serum C-reactive protein (CRP) levels, indicating reduced inflammation, also reported significant improvements in appetite. This supports the idea that a stable appetite may be associated with a reduction in early warning signs of health deterioration, suggesting that the normal appetite group in the early stages of our study might have been at lower risk for infections and similar complications.
This study has several limitations. First, it is a single-center study with a reduced sample size due to medical interruptions, such as transitions to enteral nutrition, as the study required maintaining oral intake for more than 8 weeks. For instance, the small number of patients who passed away limited our ability to extract receiver operating characteristic (ROC) or HR results. Despite our efforts to recruit a clinically relevant cohort, the number of participants in certain discharge outcome groups at the one-year follow-up was relatively small, which may have limited the statistical power to detect significant between-group differences. Second, although we targeted older individuals with comorbidities and considered the comorbidity index as a covariate, the study included a mix of various conditions that could impact outcomes. Besides the ∼48.6%–60.0% of patients with dementia, the study also included patients with chronic kidney disease and cancer, which could be critical to prognosis. This diversity in conditions should be taken into account when generalizing the study results. Third, although this study tracked appetite over an eight-week period, it did not monitor appetite status over a full year, leaving the appetite condition at the time of death or transfer to a higher-level hospital unknown. This time gap may complicate the determination of causality; thus, caution is needed when interpreting causal relationships. Future studies should aim to explore deeper connections through single-disease, multicenter, large-scale, and long-term research to further validate the findings.
In addition, the potential influence of unmeasured or residual confounding factors should be acknowledged. Although we adjusted for multiple known covariates—such as age, sex, primary diagnosis, CCI, Braden ulcer risk score, Barthel index, and nutritional intake—other relevant factors may not have been fully captured. For example, psychosocial variables such as depression, caregiver availability, cognitive fluctuations, and medication use were not systematically assessed, but may have significantly influenced both appetite and discharge outcomes. Furthermore, disease severity within diagnostic categories (e.g. stage of cancer or CKD) was not stratified, which may have introduced heterogeneity within the covariates. Despite the use of multivariable regression models to mitigate bias, the observational nature of the study limits our ability to completely eliminate confounding. Future studies incorporating more granular clinical and psychosocial data are warranted to confirm the observed associations.
For the benefit of researchers and readers who may pursue further work in this area, we share the following contextual note to support interpretation and future study design. In the Korean healthcare system, long-term hospitalization primarily occurs in long-term care hospitals (LTCHs), which are distinct from acute care facilities. LTCHs provide continuous medical management and supportive care for older adults with chronic conditions, functional decline, or complex comorbidities. Although the Korean HIRA formally defines long-term hospitalization as stays exceeding 90 days, many older patients remain hospitalized for 6 months or longer. This is largely due to multimorbidity, caregiver burden, limited community-based services, and the national long-term care insurance system, which has supported prolonged institutional care since its introduction in 2008. As a result, Korea reports an average annual length of stay in LTCHs of ∼168 days, 49 one of the longest among OECD countries, and has one of the world's fastest aging trends. 50
This structural context helps explain why more than two-thirds of our study participants remained hospitalized at one year and why the probability of discharge to home is inherently low in this population. Unlike many Western countries that have more robust transitional or home-based rehabilitation models, Korea's infrastructure still heavily relies on institutional care for frail older adults. This prolonged trajectory of care should be taken into account when interpreting our discharge outcomes and highlights the need for caution in generalizing these results to settings with different care models. Our findings underscore the urgent need for future prospective, multicenter studies with larger and more diverse cohorts to better capture the complexity of long-term care transitions and to develop stratified pathways that can improve prognosis and discharge planning for older inpatients.
Conclusion
This study demonstrated a strong correlation between poor appetite and worsened discharge outcomes, highlighting the importance of initial appetite assessment in preventing adverse outcomes and reducing healthcare costs. It provides clinical staff in various hospital settings with an understanding of the need to promote better appetite. By elucidating the relationship between appetite status and discharge outcomes, this study lays the groundwork for future research with critical insights into optimizing prognostic strategies for long-term hospitalized older individuals.
Ethics approval and consent to participate
This study was approved by the Institutional Review Board of Jeonju University (jjIRB230503-HR-2022–1212) and was conducted in accordance with the tenets of the Declaration of Helsinki. Written informed consent for data collection was obtained from all subjects at admission, and all collected data were deidentified for research purposes. The study analysis was conducted in accordance with the regulations of the Medical Service Act and Personal Information Protection Act of Korea.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251364949 - Supplemental material for Low appetite, assessed by the Simplified Nutritional Appetite Questionnaire, is associated with a reduced likelihood of residing at home after hospitalization: A prospective one-year study on older patients with comorbidities
Supplemental material, sj-docx-1-sci-10.1177_00368504251364949 for Low appetite, assessed by the Simplified Nutritional Appetite Questionnaire, is associated with a reduced likelihood of residing at home after hospitalization: A prospective one-year study on older patients with comorbidities by Jung Min Cho, Song Woo Ha, Junghee Jung and Hyunji Kim in Science Progress
Footnotes
Authors’ contributions
JM Cho: conceptualization, methodology, validation, formal analysis, investigation, data curation, writing–original draft, writing–review and editing, visualization, supervision, project administration, and funding acquisition. SW Ha: methodology, investigation, resources, data curation, writing–review and editing, and project administration. J Jung and H Kim: investigation, resources, data curation, and writing–review and editing.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
There are ethical and legal restrictions on sharing a deidentified data set because data contain potentially identifying or sensitive patient information, and this data is regulated and imposed by the Medical Service Act and Personal Information Protection Act of Korea. The data requests may be sent to the Institutional Review Board of Jeonju University (irb@jj.ac.kr), the corresponding author, and Central Convalescent Hospital.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
