Abstract
Contrast-induced encephalopathy (CIE) is a rare but potentially serious complication that can occur after endovascular procedures involving iodinated contrast media. This report describes two cases of CIE following elective treatment of intracranial aneurysms. The first case involved patients in their 70s patient with multiple comorbidities who developed neurological symptoms, including right gaze preference, left hemibody incoordination, and diffuse right cerebral hemispheric edema, after receiving a flow-diverting stent. The second case was a patient in their late 70s with a history of subarachnoid hemorrhage who experienced progressive encephalopathy after coil embolization, despite initially normal imaging findings. Both patients were managed with supportive care, including anticonvulsants and aggressive fluid management, and both showed significant neurological improvement within days. These cases highlight the diagnostic challenges of CIE, as imaging findings may vary, and underscore the need for clinicians to maintain a high index of suspicion, particularly in patients with risk factors such as advanced age, hypertension, and prior cerebrovascular events. Early recognition, prompt supportive management, and close monitoring are essential. Importantly, CIE can occur even with the use of modern, low-osmolar contrast agents like Isovue-300, emphasizing the need for continued vigilance and further research into prevention and risk factor identification.
Introduction
Contrast-induced encephalopathy (CIE) is a rare but serious complication that can arise from endovascular procedures. It is characterized by temporary cortical disturbances after the administration of iodinated contrast media. Although the exact incidence of CIE remains unclear, estimates suggest an overall incidence rate of in 0.3%–4%. 1
The pathophysiology of CIE is not fully understood; however, it is believed to result from a temporary disruption of the blood–brain barrier, allowing contrast media to penetrate the cerebral parenchyma. 2 This influx of foreign material triggers various neurological symptoms, including encephalopathy, seizures, cortical blindness, and focal neurological deficits. 3 Although CIE is usually self-limiting and resolves within 24–72 hours, rare cases of persistent neurological deficits have been reported.4,5 CIE is more commonly associated with intra-arterial administration of contrast media, CIE post intravenous (IV) administration has been rarely reported. 6 This difference could be attributed to the higher local concentration of contrast media in the cerebral circulation and direct neurotoxic effects during intra-arterial administration. 7
Previous studies have reported similar instances of CIE following endovascular treatment for intracranial aneurysms. 8 Matsubara et al. detailed a series of cases involving hemodialysis patients, highlighting the increased risk of CIE within this population. 9 Another study by Leong and Fanning documented a case of persistent neurological deficit following CIE after aneurysm coiling, highlighting the potential for long-term sequelae. 4
The diagnosis of CIE primarily relies on clinical presentation and imaging findings. Characteristic imaging features include cortical and subcortical contrast enhancement, cerebral edema, and subarachnoid contrast enhancement observed on computed tomography (CT) or magnetic resonance imaging (MRI). 10 These findings, along with the temporal relationship to contrast administration and the exclusion of other potential causes, are essential for an accurate diagnosis. 11
Although rare, timely identification and proper treatment are essential for improving patient outcomes. This case report discusses two patients who experienced CIE after the intra-arterial use of Isovue-300 in elective endovascular procedures for intracranial aneurysms during admission at Wesley Medical Center in 2025. These cases emphasize the clinical features, diagnostic difficulties, and management approaches for this uncommon yet important complication. The reporting of this study conforms to CARE guidelines. 12
Case presentation
Patient 1
An adult in their early 70s with a history of hyperlipidemia, Sjogren's disease, fibromyalgia, and hypothyroidism presented for elective treatment of a recurrent unruptured right posterior communicating artery aneurysm in November 2024. The patient had a previous history of right ptosis, periorbital pain, and pupillary asymmetry in October 2021, which led to the discovery and initial coiling of a 6.7-mm posterior communicating artery aneurysm. Follow-up imaging in June 2024 revealed a 2.5-mm wide by 2 mm long area of filling within the proximal aspect of the aneurysm.
In November 2024, the patient reported a 5-day history of periorbital headaches, lightheadedness, and slight worsening of ptosis. After extensive discussions and imaging studies, including CT head, MRA, and MRI, the decision was made to treat the aneurysm endovascularly. The patient was optimized on dual antiplatelet therapy with Aspirin (Bayer AG, Leverkusen, Germany) 81 mg daily and Brilinta (AstraZeneca, Wilmington, DE, USA) 60 mg twice daily prior to the procedure.
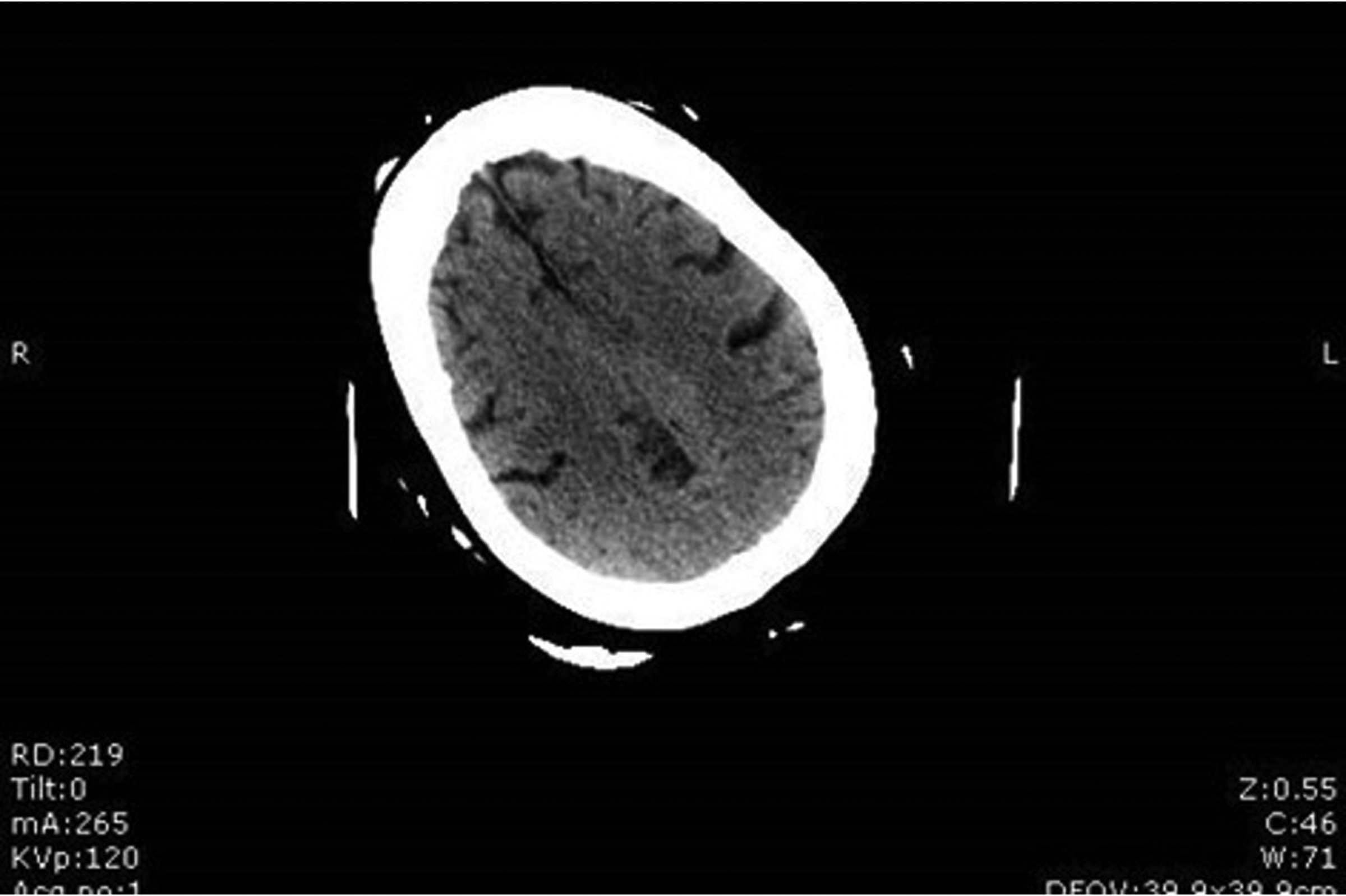
Subsequently, the patient underwent successful treatment with a single FREDX (4 mm × 13 mm) flow-diverting stent construct. A total of 150 cc of Isovue-300 contrast material was utilized during the procedure. The total fluoroscopy time for the procedure lasted 27.8 minutes. Post-procedurally, the patient awoke without new deficits but subsequently developed right gaze preference, left hemibody incoordination, and extinction to bilateral simultaneous touch. Immediate imaging, including CT head without contrast, CTA of the head and neck, and CT cerebral perfusion analysis, revealed diffuse right cerebral hemispheric edema, likely representing contrast-induced encephalopathy (Figure 1). A diagnostic angiogram confirmed patent stent placement without thrombosis or large vessel occlusion. The patient exhibited bilateral lower extremity jerking movements and received anticonvulsant therapy, including 1 mg of Versed (Roche, Basel, Switzerland) and 1 g IV Keppra (UCB Inc., Brussels, Belgium), which was continued at 500 mg twice daily.

Immediate postoperative imaging revealing right cerebral hemispheric edema.
Management included aggressive fluid resuscitation with IV normal saline, 20% mannitol (serum osmolarity goal 320, osmolar gap less than 20), and 3% hypertonic saline (sodium goal 145-155). Dexamethasone was initiated at 4 mg IV every 6 hours. EEG monitoring showed electrographic attenuation within the right cerebral hemisphere but no seizures.
Over the following days, the patient's condition gradually improved. CT head on day 2 showed interval improvement of diffuse edema within the right cerebral hemisphere. MRI revealed a minimal area of punctate infarct within the cerebellum but no diffusion restriction compatible with stroke within the right cerebral hemisphere (Figure 2). Dexamethasone was tapered down to 2 mg IV every 6 hours for 24 hours, then to 1 mg IV every 6 hours for the following 24 hours before discontinuation.
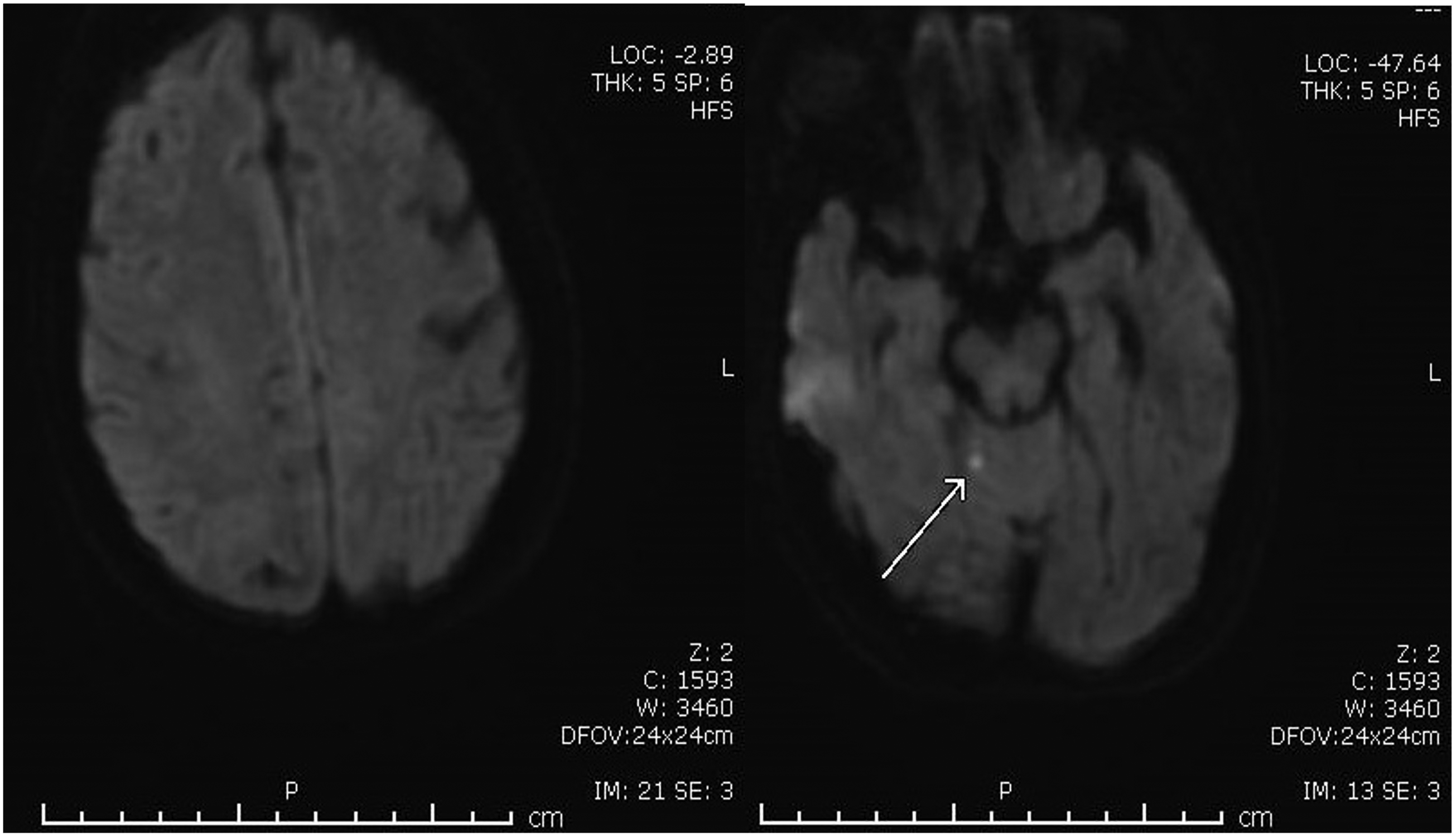
MRI showing a minimal area of punctate infarct within the cerebellum, but no diffusion restriction compatible with stroke.
By day 3, there was near-complete normalization of the neurological exam, and resolution of edema initially seen on CT (Figure 3). The patient was transitioned to oral medications, including Keppra 500 mg twice daily, aspirin 81 mg daily, and Brilinta 60 mg twice daily. The nasogastric tube was removed after the patient passed a swallow evaluation. The patient was discharged home on day 5 with a significantly improved neurological status. Written consent for the publication of this case report was obtained from the patient.
Imaging on postoperative day 3, showing resolution of edema seen on initial CT.
Patient 2
A patient in their late 70s with a history of hypertension, type 2 diabetes, hyperlipidemia, and previous subarachnoid hemorrhage due to a ruptured anterior communicating artery aneurysm in December 2021 presented with complaints of ongoing headaches for 3–4 weeks. The patient had a complex medical history, including previous endovascular coil embolization of the ruptured aneurysm, subsequent deep vein thrombosis, and pulmonary embolism, and inferior vena cava filter placement.
Follow-up imaging revealed significant aneurysm recurrence, measuring 7.5 mm long by 5 mm wide, which was treated with stent-assisted coil embolization in January 2022. However, a December 2023 angiogram showed another recurrence measuring 5 mm long by 5 mm wide (Figure 4).

Angiogram showing recurrence.
On November 8, 2024, the patient underwent successful endovascular coil embolization (Figure 5). A total of 160 cc of Isovue 300 contrast material was utilized during the procedure. The total fluoroscopy time for the procedure lasted 29.7 minutes. Post-procedure, she initially followed commands but became progressively encephalopathic over the next few hours. Immediate imaging, including CT head without contrast, CTA of the head and neck, and CT cerebral perfusion analysis, did not reveal intracranial hemorrhage or large vessel occlusion. EEG showed left hemispheric lateralized rhythmic delta activities and generalized periodic discharges, suggesting an increased risk of seizures.
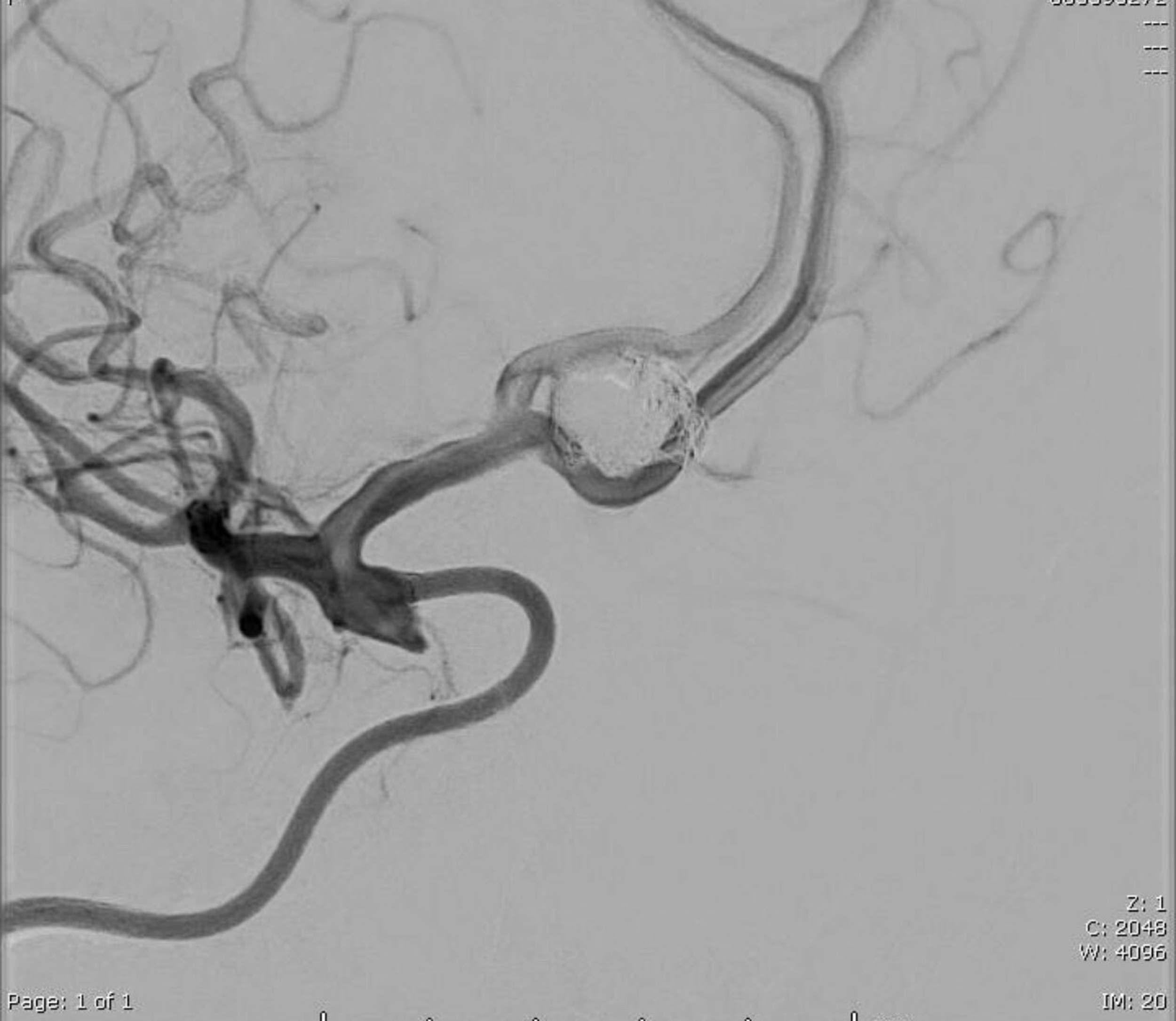
Angiogram showing successful endovascular coil embolization.
The patient received prompt anticonvulsant therapy, including 2 mg of Ativan (Bausch Health Companies Inc., Laval, Canada), followed by Briviact (UCB, Brussels, Belgium) 50 mg twice daily and Vimpat (UCB Pharmaceutical, Smyrna, GA, USA) 50 mg twice daily. Treatment also included aggressive IV fluid hydration and continuation of aspirin 81 mg daily. Steroids were avoided in this patient due to a prior history of steroid-induced psychosis/confusion. Continuous EEG monitoring was implemented to closely observe seizure activity. MRI of the brain without contrast showed minimal punctate and scattered foci of acute ischemia within the right cerebral hemispheres but no intracranial hemorrhage (Figure 6). The patient also developed atrial fibrillation during her hospital stay.

MRI of the brain without contrast showing minimal punctate and scattered foci of acute ischemia but no intracranial hemorrhage.
By day 3, her neurological examination significantly improved, with the patient becoming more alert, following commands, and responding appropriately to questions. By day 5, she had returned to her baseline neurological status. Anticoagulation with Eliquis was initiated for secondary stroke prevention in the setting of atrial fibrillation. The patient was discharged to her facility on day 7 with a completely normalized neurological exam. Her treatment regimen at discharge included continued anticonvulsant therapy and anticoagulation for atrial fibrillation. Written consent for the publication of this case report was obtained from the patient.
Discussion
The two cases presented in this report highlight the rare but significant occurrence of contrast-induced encephalopathy (CIE) following intra-arterial administration of Isovue-300 during elective endovascular treatment of intracranial aneurysms. These cases contribute to the existing body of literature on CIE and demonstrate the importance of early recognition and management. Despite advancements in contrast agents with improved safety profiles, endovascular procedures are not without risk.
Incidence and etiology
CIE is a rare adverse event linked to the use of iodinated contrast media. The incidence of CIE in cerebral angiography procedures is estimated to range from 0.3% to 4%. 1 Our cases reflect these statistics, highlighting the infrequency of this complication. The pathophysiology of CIE remains partially understood, but it is thought to involve a temporary disruption of the blood-brain barrier, permitting the contrast media to penetrate the cerebral parenchyma. 13 This BBB disruption may be triggered by several factors, including uncontrolled hypertension, ischemic injury, or the chemical properties of the contrast agent itself.10,14,15 Once the BBB is compromised, the contrast medium can leak into the brain tissue and subarachnoid space, leading to a cascade of neurological symptoms such as encephalopathy, seizures, and focal deficits.6,15,16 Additional mechanisms, such as arterial vasospasm and microcirculatory disturbances, may also contribute to the clinical presentation. 6 Notably, CIE can occur with all classes of iodinated contrast agents—including non-ionic, low-osmolar, and iso-osmolar formulations.6,15
Risk factors
Both patients presented with risk factors correlated with an increased likelihood of developing CIE. Current literature recognizes several significant clinical risk factors, including advanced age, hypertension, and a history of cerebrovascular events.6,9Additionally, procedure-specific factors play a crucial role, particularly the administration of large volumes of contrast media and extended procedure durations. 4 The first patient, in their early 70s, had a history of hyperlipidemia and prior aneurysm treatment. Similarly, the second patient, in their late 70s, presented with a history of hypertension, type 2 diabetes, and a previous history of subarachnoid hemorrhage. These clinical factors may have contributed to their susceptibility to CIE, underscoring the importance of careful patient assessment and procedural planning in high-risk individuals. Future research should focus on identifying specific risk factors and developing targeted prevention strategies. Additionally, the role of novel neuroprotective agents in mitigating the effects of CIE warrants investigation. 17
Clinical presentation and imaging findings
The clinical presentations observed in our cases were consistent with those documented in the existing literature. The first patient exhibited a preference for right gaze, left hemibody incoordination, and extinction to bilateral simultaneous touch, whereas the second patient experienced progressive encephalopathy. These manifestations align with the diverse neurological deficits reported in cases of CIE, which can vary from mild confusion to severe neurological deficits that may resemble an acute stroke.6,18
Similarly, imaging findings in CIE can be diverse, ranging from cortical and subcortical contrast enhancement to cerebral edema.3,9 In our first case, CT imaging revealed diffuse right cerebral hemispheric edema, a characteristic finding in CIE. Interestingly, the second patient's initial CT and CTA were negative for intracranial abnormalities, highlighting that the absence of immediate imaging findings does not preclude the diagnosis of CIE. This variability in imaging presentation underscores the importance of maintaining a high index of suspicion for CIE, even in the absence of typical radiological signs. 19
Management and prognosis
The management of CIE typically involves supportive care, including IV hydration, blood pressure control, and anticonvulsants if necessary. 20 Our patients received similar supportive treatments, including fluid resuscitation and antiepileptics. The use of mannitol, hypertonic saline, and dexamethasone in the first patient aligns with more aggressive management strategies reported in some cases. The prognosis of CIE is generally favorable, with most patients experiencing complete resolution of symptoms within 24–72 hours. 3 Our cases support this observation, with the first patient demonstrating near-complete normalization of neurological status by day three and the second returning to baseline by day 5. This recovery timeline is consistent with the typically self-limiting nature of CIE. 21
Contrast agent considerations
It is worth noting that although CIE is more commonly associated with intra-arterial contrast administration, cases following IV administration have also been reported, albeit rarely. 22 Our cases, which involved intra-arterial administration during aneurysm treatment, represent a more typical scenario for the development of CIE. The use of Isovue-300, a low-osmolar, non-ionic contrast agent, in our cases, is significant, as it shows that even modern contrast agents with enhanced safety profiles can still lead to CIE. 23
Isovue-300, also called iopamidol, is a non-ionic, monomeric, water-soluble agent classified as a low-osmolar contrast medium (LOCM). 24 Other commonly used contrast agents include Visipaque 320 (also called iodixanol) and Omnipaque. Visipaque is a nonionic dimeric contrast agent, whereas Omnipaque is a nonionic monomeric contrast agent. Contrast-induced encephalopathy has been described in the literature as being caused by contrast material of the LOCM class. For instance, a retrospective review of a single center neurointerventional database demonstrated contrast induced encephalopathy in 11 out of 1587 patients—9 of which were from Isovue-300 and the other two from Visipaque 320. 25 While early studies suggested iso-osmolar agents might reduce CIE risk, current guidelines (ESUR, ACR) indicate no significant difference in CIE incidence between iso-osmolar and low-osmolar non-ionic agents. However, Isovue's relative hypertonicity to plasma and cerebrospinal fluid may have contributed to BBB disruption, a key mechanism in CIE pathogenesis. While Isovue-300 falls within the broader category of modern non-ionic LOCMs, our case series and broader literature still indicate that LOCMs may carry residual CIE risk, despite improved safety profiles.
Conclusion
These cases highlight the importance of maintaining a high index of suspicion for CIE in patients undergoing endovascular procedures, especially those with multiple risk factors. Early recognition, prompt supportive management, and close monitoring are essential for achieving optimal outcomes. More research is needed to understand better CIE's risk factors and pathophysiology, which may lead to improved prevention strategies and management protocols in neuro-interventional procedures.
Footnotes
Acknowledgments
We gratefully acknowledge the patient's participation and the contributions of the entire care team, which made this case report possible.
Ethical approval statement
Not applicable.
Informed consent
Written consent for the publication of this case report was obtained from the patient.
Authors’ contribution
CRediT: Conceptualization: MH; data curation: KB and MH; formal analysis: KB and MH; funding acquisition, investigation, methodology, and project administration: MH and HJS; resources: MH and HJS; software and supervision: MH and HJS; validation, visualization, and writing—original draft: KB, MH, and HJS; writing—review and editing: KB, MH, and HJS.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Patient data cannot be made publicly available to ensure confidentiality and privacy. Further details may be available from the corresponding author upon reasonable request and in accordance with ethical guidelines.
